

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50385958 CHEMBL2042298
SMILES: CCNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)nc(NCCc3ccccc3)nc12
InChI Key: InChIKey=UQERKZWKUCVXID-QCUYGVNKSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine A2 receptor (Rattus norvegicus-Rattus norvegicus (rat)) | BDBM50385958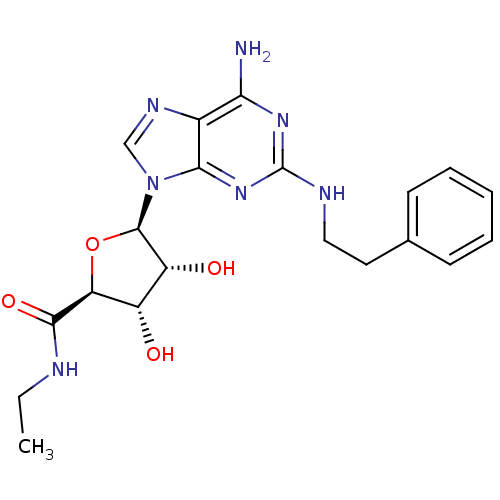 (CHEMBL2042298) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CIBA-GEIGY Corporation Curated by ChEMBL | Assay Description Binding to Adenosine A2 receptor was measured in ADA-pretreated rat striatal membranes using [3H]-CGS- 21680 | J Med Chem 33: 1919-24 (1990) BindingDB Entry DOI: 10.7270/Q2VD701M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50385958 (CHEMBL2042298) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 473 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CIBA-GEIGY Corporation Curated by ChEMBL | Assay Description Adenosine A1 receptor binding was measured in adenosine deaminase (ADA) pretreated rat cortical membranes using [3H]cyclohexyladenosine in the presen... | J Med Chem 33: 1919-24 (1990) BindingDB Entry DOI: 10.7270/Q2VD701M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50385958 (CHEMBL2042298) | NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 145 | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Agonist activity at human adenosine A2B receptor expressed in CHO cells assessed as increase in intracellular cAMP level after 30 mins | J Med Chem 55: 3521-34 (2012) Article DOI: 10.1021/jm300206u BindingDB Entry DOI: 10.7270/Q2FT8N35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50385958 (CHEMBL2042298) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 104 | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Agonist activity at human adenosine A3 receptor expressed in CHO cells assessed as inhibition of forskolin-induced cAMP accumulation | J Med Chem 55: 3521-34 (2012) Article DOI: 10.1021/jm300206u BindingDB Entry DOI: 10.7270/Q2FT8N35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50385958 (CHEMBL2042298) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Agonist activity at human adenosine A2A receptor expressed in CHO cells assessed as increase in intracellular cAMP level after 30 mins | J Med Chem 55: 3521-34 (2012) Article DOI: 10.1021/jm300206u BindingDB Entry DOI: 10.7270/Q2FT8N35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM50385958 (CHEMBL2042298) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 112 | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Agonist activity at human adenosine A1 receptor expressed in CHO cells assessed as inhibition of forskolin-induced cAMP accumulation | J Med Chem 55: 3521-34 (2012) Article DOI: 10.1021/jm300206u BindingDB Entry DOI: 10.7270/Q2FT8N35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||