

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50399639 CHEMBL2181197
SMILES: [#6]-[#6]-[#6]-[#6]-[#6@H](-[#7](-[#6])-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1c(-[#6])cc(-[#8])cc1-[#6])-[#6](-[#7])=O
InChI Key: InChIKey=IEAIMVXZKTUXNA-PUHABZHSSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50399639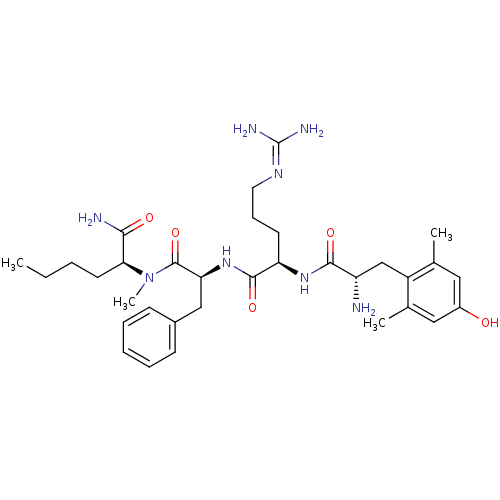 (CHEMBL2181197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs | J Med Chem 55: 9549-61 (2012) Article DOI: 10.1021/jm3008079 BindingDB Entry DOI: 10.7270/Q2TM7C82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50399639 (CHEMBL2181197) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 14.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel Curated by ChEMBL | Assay Description Displacement of [3H]U69,593 from kappa opioid receptor in guinea pig ileum membranes after 2 hrs | J Med Chem 55: 9549-61 (2012) Article DOI: 10.1021/jm3008079 BindingDB Entry DOI: 10.7270/Q2TM7C82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Opioid receptors; mu & delta (Rattus norvegicus (rat)) | BDBM50399639 (CHEMBL2181197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel Curated by ChEMBL | Assay Description Displacement of [3H]DSLET from delta opioid receptor in rat brain membranes after 2 hrs | J Med Chem 55: 9549-61 (2012) Article DOI: 10.1021/jm3008079 BindingDB Entry DOI: 10.7270/Q2TM7C82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50399639 (CHEMBL2181197) | UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.35 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum | J Med Chem 55: 9549-61 (2012) Article DOI: 10.1021/jm3008079 BindingDB Entry DOI: 10.7270/Q2TM7C82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50399639 (CHEMBL2181197) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens | J Med Chem 55: 9549-61 (2012) Article DOI: 10.1021/jm3008079 BindingDB Entry DOI: 10.7270/Q2TM7C82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||