

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CN(CCCCCCN)CCCCCCCCN(C)CCCCCCNCC(=O)N1c2ccccc2C(=O)Nc2cccnc12
InChI Key: InChIKey=CVYMNORESIPOLC-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M1 (RAT) | BDBM50407222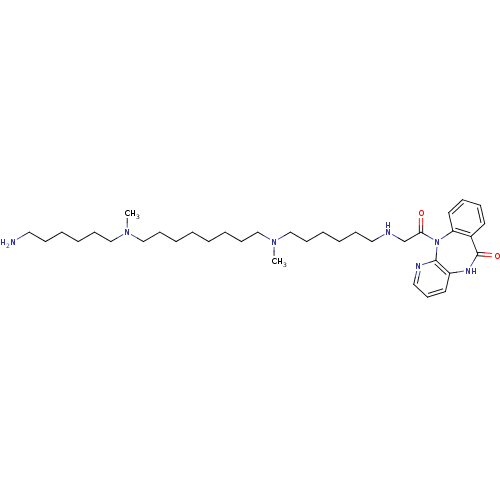 (CHEMBL112244) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Binding affinity for rat cortex Muscarinic acetylcholine receptor M1 | J Med Chem 41: 4150-60 (1998) Checked by Author Article DOI: 10.1021/jm981038d BindingDB Entry DOI: 10.7270/Q2KH0PJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (RAT) | BDBM50407222 (CHEMBL112244) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to rat heart Muscarinic acetylcholine receptor M2 | J Med Chem 41: 4150-60 (1998) Checked by Author Article DOI: 10.1021/jm981038d BindingDB Entry DOI: 10.7270/Q2KH0PJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (RAT) | BDBM50407222 (CHEMBL112244) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Inhibition of NG 108-15 binding to Muscarinic acetylcholine receptor M4 | J Med Chem 41: 4150-60 (1998) Checked by Author Article DOI: 10.1021/jm981038d BindingDB Entry DOI: 10.7270/Q2KH0PJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (RAT) | BDBM50407222 (CHEMBL112244) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 331 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Inhibition of [3H]NMS binding to rat submaxillary gland Muscarinic acetylcholine receptor M3 | J Med Chem 41: 4150-60 (1998) Checked by Author Article DOI: 10.1021/jm981038d BindingDB Entry DOI: 10.7270/Q2KH0PJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50407222 (CHEMBL112244) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | 8.13 | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Antagonistic activity against M2 muscarinic receptor in guinea pig left atrium derived by plotting log(DR - 1) vs log[antagonist] | J Med Chem 37: 3363-72 (1994) BindingDB Entry DOI: 10.7270/Q21V5G5G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor (Cavia porcellus) | BDBM50407222 (CHEMBL112244) | MMDB Reactome pathway KEGG UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | 661 | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Antagonistic activity against M3 muscarinic receptor in guinea pig left ileum derived by plotting log(DR - 1) vs log[antagonist] | J Med Chem 37: 3363-72 (1994) BindingDB Entry DOI: 10.7270/Q21V5G5G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||