

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50430726 CHEMBL2333930
SMILES: C[C@H](Cc1ccc(OCCCCCCCCNc2c3CCCCc3nc3cc(Cl)ccc23)cc1)N(C)CC#C
InChI Key: InChIKey=NXNSSEKZKUPRMX-AREMUKBSSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amine oxidase [flavin-containing] B (Homo sapiens (Human)) | BDBM50430726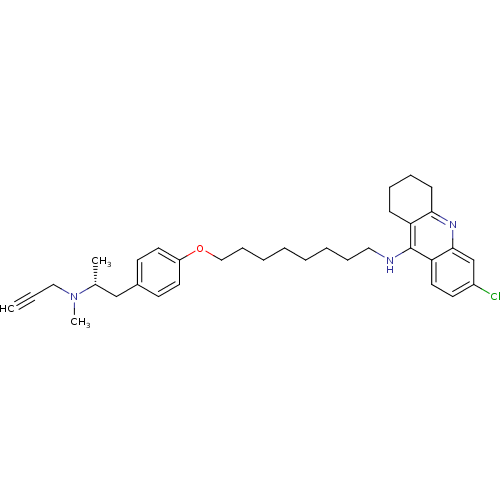 (CHEMBL2333930) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza Universit£ di Roma Curated by ChEMBL | Assay Description Inhibition of human MAOB | J Med Chem 58: 6717-32 (2015) BindingDB Entry DOI: 10.7270/Q2VT1TWM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50430726 (CHEMBL2333930) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University Curated by ChEMBL | Assay Description Inhibition of electric eel AChE using acetylthiocholine chloride as substrate after 15 mins by Ellman's method | Eur J Med Chem 62: 745-53 (2013) Article DOI: 10.1016/j.ejmech.2013.01.039 BindingDB Entry DOI: 10.7270/Q2C82BN6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Butyrylcholinesterase (BuChE) (Equus caballus (Horse)) | BDBM50430726 (CHEMBL2333930) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University Curated by ChEMBL | Assay Description Inhibition of horse serum BChE using butylthiocholine chloride as substrate after 15 mins by Ellman's method | Eur J Med Chem 62: 745-53 (2013) Article DOI: 10.1016/j.ejmech.2013.01.039 BindingDB Entry DOI: 10.7270/Q2C82BN6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase [flavin-containing] B (Homo sapiens (Human)) | BDBM50430726 (CHEMBL2333930) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 129 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University Curated by ChEMBL | Assay Description Inhibition of recombinant human MAO-B using p-benzylamine substrate preincubated for 15 mins before substrate addition measured after 20 mins by fluo... | Eur J Med Chem 62: 745-53 (2013) Article DOI: 10.1016/j.ejmech.2013.01.039 BindingDB Entry DOI: 10.7270/Q2C82BN6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase (flavin-containing) A (Homo sapiens (Human)) | BDBM50430726 (CHEMBL2333930) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 193 | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University Curated by ChEMBL | Assay Description Inhibition of recombinant human MAO-A using p-tyramine substrate preincubated for 15 mins before substrate addition measured after 20 mins by fluores... | Eur J Med Chem 62: 745-53 (2013) Article DOI: 10.1016/j.ejmech.2013.01.039 BindingDB Entry DOI: 10.7270/Q2C82BN6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||