 Found 4 hits for monomerid = 50433038
Found 4 hits for monomerid = 50433038 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform (PI3K alpha)
(Mus musculus (Mouse)) | BDBM50433038
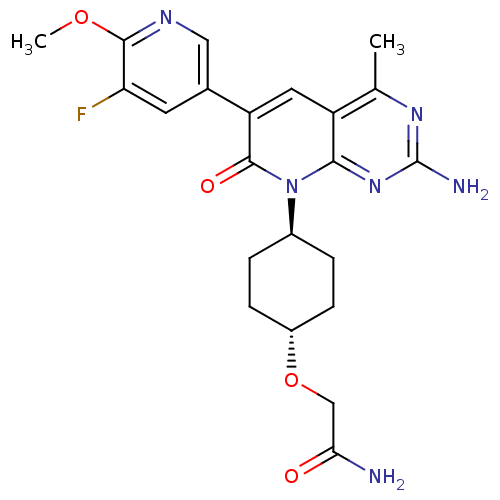
(CHEMBL2375957 | US8633204, 299)Show SMILES COc1ncc(cc1F)-c1cc2c(C)nc(N)nc2n([C@H]2CC[C@@H](CC2)OCC(N)=O)c1=O |r,wU:23.28,wD:20.21,(27.28,-44.28,;25.95,-43.51,;24.61,-44.28,;24.61,-45.82,;23.27,-46.58,;21.94,-45.81,;21.94,-44.27,;23.27,-43.5,;23.27,-41.96,;20.6,-46.56,;19.26,-45.78,;17.92,-46.56,;16.58,-45.8,;16.58,-44.26,;15.25,-46.57,;15.25,-48.11,;13.92,-48.88,;16.58,-48.88,;17.91,-48.12,;19.25,-48.89,;19.24,-50.43,;17.9,-51.19,;17.9,-52.72,;19.22,-53.51,;20.56,-52.74,;20.57,-51.2,;19.21,-55.04,;17.88,-55.81,;16.55,-55.03,;15.21,-55.79,;16.56,-53.49,;20.6,-48.12,;21.93,-48.89,)| Show InChI InChI=1S/C22H25FN6O4/c1-11-15-8-16(12-7-17(23)20(32-2)26-9-12)21(31)29(19(15)28-22(25)27-11)13-3-5-14(6-4-13)33-10-18(24)30/h7-9,13-14H,3-6,10H2,1-2H3,(H2,24,30)(H2,25,27,28)/t13-,14- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.312 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of mouse PI3Kalpha |
Bioorg Med Chem Lett 23: 2787-92 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.020
BindingDB Entry DOI: 10.7270/Q2NZ890X |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50433038

(CHEMBL2375957 | US8633204, 299)Show SMILES COc1ncc(cc1F)-c1cc2c(C)nc(N)nc2n([C@H]2CC[C@@H](CC2)OCC(N)=O)c1=O |r,wU:23.28,wD:20.21,(27.28,-44.28,;25.95,-43.51,;24.61,-44.28,;24.61,-45.82,;23.27,-46.58,;21.94,-45.81,;21.94,-44.27,;23.27,-43.5,;23.27,-41.96,;20.6,-46.56,;19.26,-45.78,;17.92,-46.56,;16.58,-45.8,;16.58,-44.26,;15.25,-46.57,;15.25,-48.11,;13.92,-48.88,;16.58,-48.88,;17.91,-48.12,;19.25,-48.89,;19.24,-50.43,;17.9,-51.19,;17.9,-52.72,;19.22,-53.51,;20.56,-52.74,;20.57,-51.2,;19.21,-55.04,;17.88,-55.81,;16.55,-55.03,;15.21,-55.79,;16.56,-53.49,;20.6,-48.12,;21.93,-48.89,)| Show InChI InChI=1S/C22H25FN6O4/c1-11-15-8-16(12-7-17(23)20(32-2)26-9-12)21(31)29(19(15)28-22(25)27-11)13-3-5-14(6-4-13)33-10-18(24)30/h7-9,13-14H,3-6,10H2,1-2H3,(H2,24,30)(H2,25,27,28)/t13-,14- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of mTOR (unknown origin) |
Bioorg Med Chem Lett 23: 2787-92 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.020
BindingDB Entry DOI: 10.7270/Q2NZ890X |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50433038

(CHEMBL2375957 | US8633204, 299)Show SMILES COc1ncc(cc1F)-c1cc2c(C)nc(N)nc2n([C@H]2CC[C@@H](CC2)OCC(N)=O)c1=O |r,wU:23.28,wD:20.21,(27.28,-44.28,;25.95,-43.51,;24.61,-44.28,;24.61,-45.82,;23.27,-46.58,;21.94,-45.81,;21.94,-44.27,;23.27,-43.5,;23.27,-41.96,;20.6,-46.56,;19.26,-45.78,;17.92,-46.56,;16.58,-45.8,;16.58,-44.26,;15.25,-46.57,;15.25,-48.11,;13.92,-48.88,;16.58,-48.88,;17.91,-48.12,;19.25,-48.89,;19.24,-50.43,;17.9,-51.19,;17.9,-52.72,;19.22,-53.51,;20.56,-52.74,;20.57,-51.2,;19.21,-55.04,;17.88,-55.81,;16.55,-55.03,;15.21,-55.79,;16.56,-53.49,;20.6,-48.12,;21.93,-48.89,)| Show InChI InChI=1S/C22H25FN6O4/c1-11-15-8-16(12-7-17(23)20(32-2)26-9-12)21(31)29(19(15)28-22(25)27-11)13-3-5-14(6-4-13)33-10-18(24)30/h7-9,13-14H,3-6,10H2,1-2H3,(H2,24,30)(H2,25,27,28)/t13-,14- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.686 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Pfizer Inc.
US Patent
| Assay Description
Compounds of the present invention were evaluated for potency against PI3-Kα using an in vitro kinase assay. PI3-Kα activity is measured in... |
US Patent US8633204 (2014)
BindingDB Entry DOI: 10.7270/Q2Q81BQT |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50433038

(CHEMBL2375957 | US8633204, 299)Show SMILES COc1ncc(cc1F)-c1cc2c(C)nc(N)nc2n([C@H]2CC[C@@H](CC2)OCC(N)=O)c1=O |r,wU:23.28,wD:20.21,(27.28,-44.28,;25.95,-43.51,;24.61,-44.28,;24.61,-45.82,;23.27,-46.58,;21.94,-45.81,;21.94,-44.27,;23.27,-43.5,;23.27,-41.96,;20.6,-46.56,;19.26,-45.78,;17.92,-46.56,;16.58,-45.8,;16.58,-44.26,;15.25,-46.57,;15.25,-48.11,;13.92,-48.88,;16.58,-48.88,;17.91,-48.12,;19.25,-48.89,;19.24,-50.43,;17.9,-51.19,;17.9,-52.72,;19.22,-53.51,;20.56,-52.74,;20.57,-51.2,;19.21,-55.04,;17.88,-55.81,;16.55,-55.03,;15.21,-55.79,;16.56,-53.49,;20.6,-48.12,;21.93,-48.89,)| Show InChI InChI=1S/C22H25FN6O4/c1-11-15-8-16(12-7-17(23)20(32-2)26-9-12)21(31)29(19(15)28-22(25)27-11)13-3-5-14(6-4-13)33-10-18(24)30/h7-9,13-14H,3-6,10H2,1-2H3,(H2,24,30)(H2,25,27,28)/t13-,14- | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of AKT phosphorylation at Ser 473 in human BT20 cells |
Bioorg Med Chem Lett 23: 2787-92 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.020
BindingDB Entry DOI: 10.7270/Q2NZ890X |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data