

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50443351 CHEMBL3086037::US8669252, 15
SMILES: Clc1ccc(cc1)C(NC(=O)[C@@H]1CC[C@H](C[C@H]1c1ccc(Br)cc1)N1CCOCC1)c1ccncc1
InChI Key: InChIKey=GHTSQNCNYKPRQH-WRENHGGMSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prolylcarboxypeptidase (PRCP) (Homo sapiens (Human)) | BDBM50443351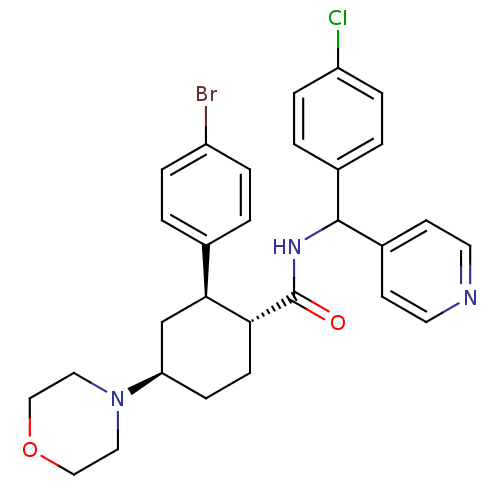 (CHEMBL3086037 | US8669252, 15) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description The potency of compounds of formula I against PrCP was determined by a fluorescence intensity kinetic assay measuring the IC50 values of PrCP inhibit... | US Patent US8669252 (2014) BindingDB Entry DOI: 10.7270/Q2J9652X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prolylcarboxypeptidase (PRCP) (Homo sapiens (Human)) | BDBM50443351 (CHEMBL3086037 | US8669252, 15) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of recombinant human PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay | Bioorg Med Chem Lett 23: 6228-33 (2013) Article DOI: 10.1016/j.bmcl.2013.09.094 BindingDB Entry DOI: 10.7270/Q2DZ09Q4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal Pro-X carboxypeptidase (Mus musculus) | BDBM50443351 (CHEMBL3086037 | US8669252, 15) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.740 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay | Bioorg Med Chem Lett 23: 6228-33 (2013) Article DOI: 10.1016/j.bmcl.2013.09.094 BindingDB Entry DOI: 10.7270/Q2DZ09Q4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||