

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM6644 6‐phenylpteridine‐2,4,7‐triamine (5)::6-phenylpteridine-2,4,7-triamine::Pteridine deriv. 11::TRIAMTERENE
SMILES: Nc1nc(N)c2nc(c(N)nc2n1)-c1ccccc1
InChI Key: InChIKey=FNYLWPVRPXGIIP-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pteridine reductase 1 (Leishmania major) | BDBM6644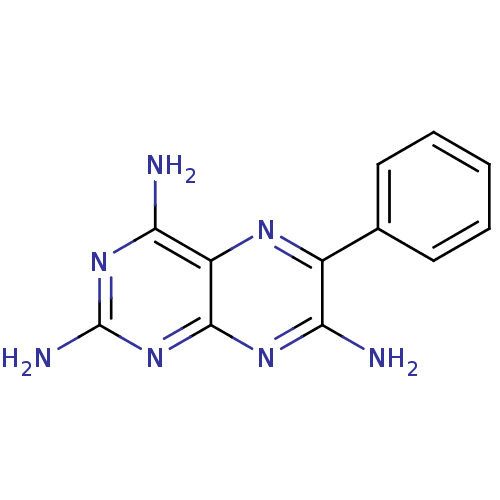 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Dundee Curated by ChEMBL | Assay Description Inhibition of Leishmania major recombinant PTR1 | J Med Chem 53: 221-9 (2010) Article DOI: 10.1021/jm901059x BindingDB Entry DOI: 10.7270/Q2H132XP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase JAK2 (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PCBioAssay | n/a | n/a | 4.61E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Molecular Screening Center Curated by PubChem BioAssay | Assay Description Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Center Affiliation: The Scripps Research Institute (TS... | PubChem Bioassay (2009) BindingDB Entry DOI: 10.7270/Q20863Q9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| dTDP-4-dehydrorhamnose 3,5-epimerase RmlC (Mycobacterium tuberculosis H37Rv) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PCBioAssay | n/a | n/a | 4.37E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
PCMD Curated by PubChem BioAssay | Assay Description Molecular Library Screening Center Network (MLSCN) Penn Center for Molecular Discovery (PCMD) Assay Provider: Michael McNeil, Colorado State Universi... | PubChem Bioassay (2009) BindingDB Entry DOI: 10.7270/Q2TD9VRT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hsf1 protein (Mus musculus) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PCBioAssay | n/a | n/a | n/a | n/a | >1.95E+5 | n/a | n/a | n/a | n/a |
Broad Institute Curated by PubChem BioAssay | Assay Description Keywords: Heat Shock Factor-1 (HSF-1), Stress Response, MG132, NIH3T3, Luminescence Assay Overview: Modified NIH3T3, transformed to express firefly... | PubChem Bioassay (2010) BindingDB Entry DOI: 10.7270/Q2MW2FKX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Casein kinase I isoform alpha (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.68E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Max Planck Institute of Molecular Physiology | Assay Description Single-point measurements analyses and subsequent dose-dependent investigations of selected pteridine derivatives for inhibition of CK1α were pe... | Cell Chem Biol 23: 494-507 (2016) Article DOI: 10.1016/j.chembiol.2016.02.015 BindingDB Entry DOI: 10.7270/Q2GQ6WJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Casein kinase I isoform delta (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.64E+4 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Max Planck Institute of Molecular Physiology | Assay Description A stock solution of 5 % DMSO (BioReagent for molecular biology, Sigma Aldrich) in water was prepared. The CK1 substrate peptide RRKDLHDDEEDEAMSITA (J... | Cell Chem Biol 23: 494-507 (2016) Article DOI: 10.1016/j.chembiol.2016.02.015 BindingDB Entry DOI: 10.7270/Q2GQ6WJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Canalicular multispecific organic anion transporter 1 (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc Curated by ChEMBL | Assay Description Inhibition of human MRP2 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and... | Toxicol Sci 136: 216-41 (2013) BindingDB Entry DOI: 10.7270/Q2JM2D2D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >1.35E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc. Curated by ChEMBL | Assay Description Inhibition of human BSEP expressed in fall armyworm sf9 cell plasma membrane vesicles assessed as reduction in vesicle-associated [3H]-taurocholate t... | Toxicol Sci 118: 485-500 (2010) Article DOI: 10.1093/toxsci/kfq269 BindingDB Entry DOI: 10.7270/Q26Q20JN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Multidrug resistance-associated protein 4 (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc Curated by ChEMBL | Assay Description Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and... | Toxicol Sci 136: 216-41 (2013) BindingDB Entry DOI: 10.7270/Q2JM2D2D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc Curated by ChEMBL | Assay Description Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ... | Toxicol Sci 136: 216-41 (2013) BindingDB Entry DOI: 10.7270/Q2JM2D2D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Canalicular multispecific organic anion transporter 2 (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc Curated by ChEMBL | Assay Description Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ... | Toxicol Sci 136: 216-41 (2013) BindingDB Entry DOI: 10.7270/Q2JM2D2D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-Dependent Kinase 4 (CDK4) (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.10E+5 | n/a | n/a | n/a | n/a | 7.4 | 30 |
Banyu Tsukuba Research Institute | Assay Description In vitro kinase assays using synthetic peptides and purified enzymes were incubated at 30°C for 45 min in buffer that contained 50 uM ATP, and d... | J Med Chem 44: 4615-27 (2001) Article DOI: 10.1021/jm0103256 BindingDB Entry DOI: 10.7270/Q2736P3Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Casein kinase I isoform epsilon (Homo sapiens (Human)) | BDBM6644 (6‐phenylpteridine‐2,4,7‐triamine...) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | Purchase DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.68E+4 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Max Planck Institute of Molecular Physiology | Assay Description A stock solution of 5 % DMSO (BioReagent for molecular biology, Sigma Aldrich) in water was prepared. The CK1 substrate peptide RRKDLHDDEEDEAMSITA (J... | Cell Chem Biol 23: 494-507 (2016) Article DOI: 10.1016/j.chembiol.2016.02.015 BindingDB Entry DOI: 10.7270/Q2GQ6WJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||