

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM78304 2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphenol::2-(2-oxidanyl-5-prop-2-enyl-phenyl)-4-prop-2-enyl-phenol::4-allyl-2-(5-allyl-2-hydroxy-phenyl)phenol::MLS001048917::SMR000387108::cid_72300
SMILES: Oc1ccc(CC=C)cc1-c1cc(CC=C)ccc1O
InChI Key: InChIKey=VVOAZFWZEDHOOU-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM78304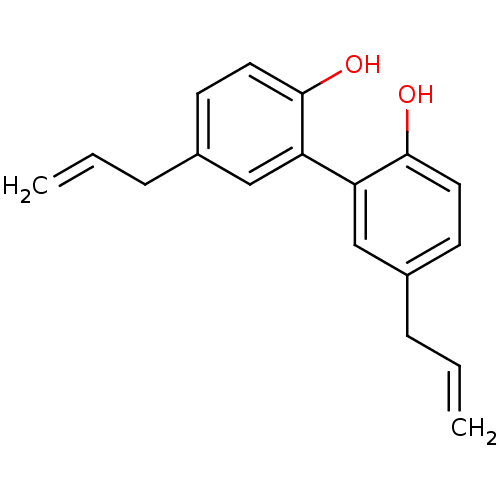 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 1.44E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Displacement of [3H]CP55,940 from recombinant human CB2 receptor expressed in CHO cells after 2 hrs by liquid scintillation counting | ACS Med Chem Lett 4: 41-5 (2013) Article DOI: 10.1021/ml300235q BindingDB Entry DOI: 10.7270/Q2SF2XHW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bern | Assay Description Binding assay with hCB1 and hCB2 receptors. | Chem Biol 18: 1053-64 (2011) Article DOI: 10.1016/j.chembiol.2011.05.012 BindingDB Entry DOI: 10.7270/Q27W69PQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 3.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Displacement of [3H]CP55,940 from recombinant human CB1 receptor expressed in CHO cells after 2 hrs by liquid scintillation counting | ACS Med Chem Lett 4: 41-5 (2013) Article DOI: 10.1021/ml300235q BindingDB Entry DOI: 10.7270/Q2SF2XHW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| GABA-A receptor; GABA-A site (alpha1/beta2 interface) (Homo sapiens (Human)) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.68E+4 | n/a | n/a | n/a | n/a |
University of Vienna Curated by ChEMBL | Assay Description Modulation of GABA Aalpha1beta2 receptor expressed in Xenopus laevis oocytes assessed as potentiation of GABA-induced chloride current at holding pot... | J Med Chem 54: 5349-61 (2011) Article DOI: 10.1021/jm200186n BindingDB Entry DOI: 10.7270/Q2QR4XGP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.28E+3 | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Partial agonist activity at recombinant human CB2 receptor expressed in CHO cells assessed as inhibition of forskolin-induced cAMP accumulation at 10... | ACS Med Chem Lett 4: 41-5 (2013) Article DOI: 10.1021/ml300235q BindingDB Entry DOI: 10.7270/Q2SF2XHW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.83E+4 | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Partial agonist activity at recombinant human CB1 receptor expressed in CHO cells assessed as inhibition of forskolin-induced cAMP accumulation at 10... | ACS Med Chem Lett 4: 41-5 (2013) Article DOI: 10.1021/ml300235q BindingDB Entry DOI: 10.7270/Q2SF2XHW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| TPA: Essential protein of the mitochondrial intermembrane space (Saccharomyces cerevisiae S288c) | BDBM78304 (2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphe...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | PCBioAssay | n/a | n/a | 6.83E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | Assay Description Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute(SBMRI, San Diego, CA... | PubChem Bioassay (2010) BindingDB Entry DOI: 10.7270/Q2F18X6W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||