 Found 11 hits for monomerid = 8126
Found 11 hits for monomerid = 8126 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM8126
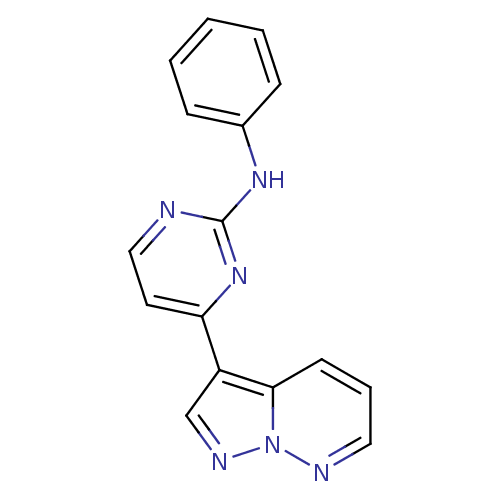
(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D
Curated by ChEMBL
| Assay Description
Inhibition of GSK3beta expressed in Escherichia coli or baculovirus-infected insect cells using ATP as substrate by fluorescence polarization assay |
J Med Chem 54: 5131-43 (2011)
Article DOI: 10.1021/jm200349b
BindingDB Entry DOI: 10.7270/Q2F47PHP |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... |
J Med Chem 47: 4716-30 (2004)
Article DOI: 10.1021/jm040063i
BindingDB Entry DOI: 10.7270/Q2VM49HJ |
More data for this
Ligand-Target Pair | |
Cyclin-Dependent Kinase 4 (CDK4)
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 158 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... |
J Med Chem 47: 4716-30 (2004)
Article DOI: 10.1021/jm040063i
BindingDB Entry DOI: 10.7270/Q2VM49HJ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CDK4 by radioactive glutathione plate-binding assay |
Bioorg Med Chem Lett 18: 5758-62 (2009)
Article DOI: 10.1016/j.bmcl.2008.09.069
BindingDB Entry DOI: 10.7270/Q2J67GZN |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GSK3beta by scintillation proximity assay |
Bioorg Med Chem Lett 18: 5758-62 (2009)
Article DOI: 10.1016/j.bmcl.2008.09.069
BindingDB Entry DOI: 10.7270/Q2J67GZN |
More data for this
Ligand-Target Pair | |
Dual-specificity tyrosine-phosphorylation regulated kinase 1A
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Sussex
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal Hex-tagged DYRK1A (127 to 485 residues) expressed in Escherichia coli BL21 (DE3) incubated for 1.5 hrs by ... |
ACS Med Chem Lett 11: 1620-1626 (2020)
|
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 158 | n/a | n/a | n/a | n/a | n/a | n/a |
Northeastern University
Curated by ChEMBL
| Assay Description
Inhibition of human CDK4 using biotinylated-aminohexyl-Ala-Ala-Ala-Lys-Arg-Arg-Glu-Ile-Leu-Ser-Arg-Arg-Pro-Ser(PO3)-Tyr-Arg-amide as substrate after ... |
J Med Chem 63: 756-783 (2020)
Article DOI: 10.1021/acs.jmedchem.9b01741 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Northeastern University
Curated by ChEMBL
| Assay Description
Inhibition of human GSK3beta using biotinylated-aminohexyl-Ala-Ala-Ala-Lys-Arg-Arg-Glu-Ile-Leu-Ser-Arg-Arg-Pro-Ser(PO3)-Tyr-Arg-amide as substrate af... |
J Med Chem 63: 756-783 (2020)
Article DOI: 10.1021/acs.jmedchem.9b01741 |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Northeastern University
Curated by ChEMBL
| Assay Description
Inhibition of human CDK2 using biotinylated-aminohexyl-Ala-Ala-Ala-Lys-Arg-Arg-Glu-Ile-Leu-Ser-Arg-Arg-Pro-Ser(PO3)-Tyr-Arg-amide as substrate after ... |
J Med Chem 63: 756-783 (2020)
Article DOI: 10.1021/acs.jmedchem.9b01741 |
More data for this
Ligand-Target Pair | |
Cyclin-Dependent Kinase 2 (CDK2)
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline
| Assay Description
The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... |
J Med Chem 47: 4716-30 (2004)
Article DOI: 10.1021/jm040063i
BindingDB Entry DOI: 10.7270/Q2VM49HJ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM8126

(N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...)Show InChI InChI=1S/C16H12N6/c1-2-5-12(6-3-1)20-16-17-10-8-14(21-16)13-11-19-22-15(13)7-4-9-18-22/h1-11H,(H,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human CDK2 by HTRF assay |
Bioorg Med Chem Lett 18: 5758-62 (2009)
Article DOI: 10.1016/j.bmcl.2008.09.069
BindingDB Entry DOI: 10.7270/Q2J67GZN |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data