

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM8888 3-(1H-Inden-2-yl)pyridine::US9271963, 32::indene 1
SMILES: C1C(=Cc2ccccc12)c1cccnc1
InChI Key: InChIKey=NNEAKBWZBQOQDH-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 11B2 (CYP11B2) (Homo sapiens (Human)) | BDBM8888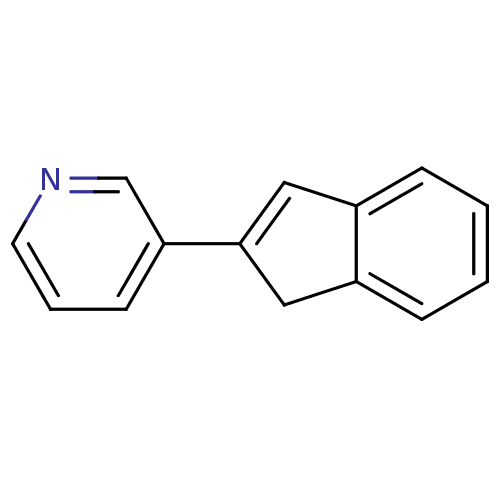 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.40 | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Saarland University | Assay Description The enzyme activity was assayed by monitoring the conversion of deoxycorticosterone to corticosterone in the presence of inhibitor compounds. The pro... | Pharmacol Rev 49: 2222-31 (2006) Article DOI: 10.1021/jm060055x BindingDB Entry DOI: 10.7270/Q2MW2FBS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 11B2 (CYP11B2) (Homo sapiens (Human)) | BDBM8888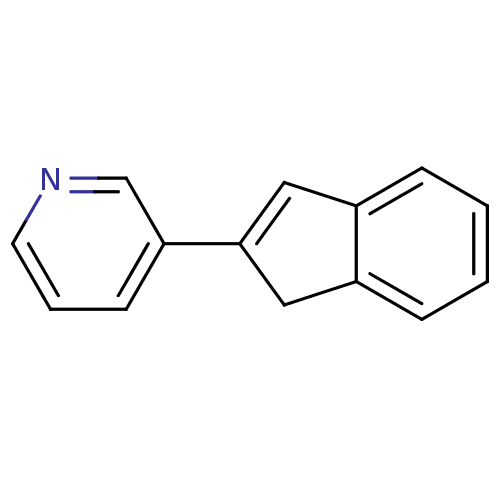 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 13.5 | n/a | n/a | n/a | n/a | n/a | 37 |
UNIVERSITAT DES SAARLANDES US Patent | Assay Description V79 MZh11B1 and V79 MZh 11B2 cells (8˙10^5 cells per well) were grown to confluency on 24-well cell culture plates with 1.9 cm^2 culture area per... | US Patent US9271963 (2016) BindingDB Entry DOI: 10.7270/Q2445KBN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 17A1 (Homo sapiens (Human)) | BDBM8888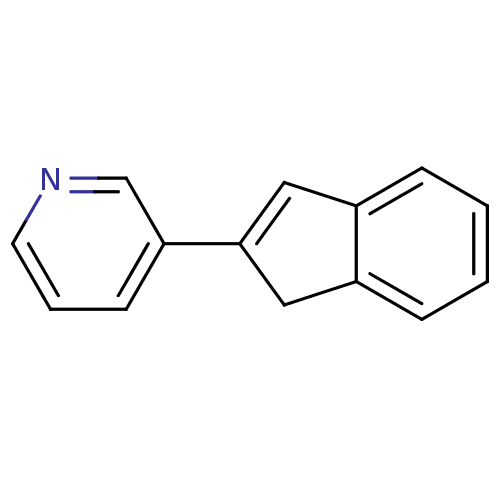 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Saarland University | Assay Description The 17 alpha-hydroxylase activity of CYP 17 was determined by measuring the conversion of progesterone into 17 alpha-hydroxyprogesterone and the bypr... | Pharmacol Rev 49: 2222-31 (2006) Article DOI: 10.1021/jm060055x BindingDB Entry DOI: 10.7270/Q2MW2FBS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 11B1, mitochondrial (Homo sapiens (Human)) | BDBM8888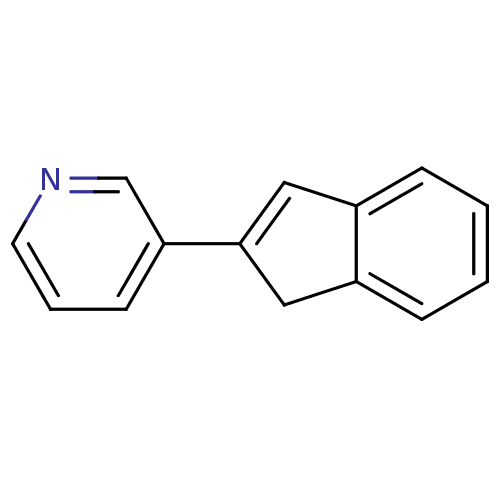 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 862 | n/a | n/a | n/a | n/a | n/a | 37 |
UNIVERSITAT DES SAARLANDES US Patent | Assay Description V79 MZh11B1 and V79 MZh 11B2 cells (8˙10^5 cells per well) were grown to confluency on 24-well cell culture plates with 1.9 cm^2 culture area per... | US Patent US9271963 (2016) BindingDB Entry DOI: 10.7270/Q2445KBN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 11B1, mitochondrial (Homo sapiens (Human)) | BDBM8888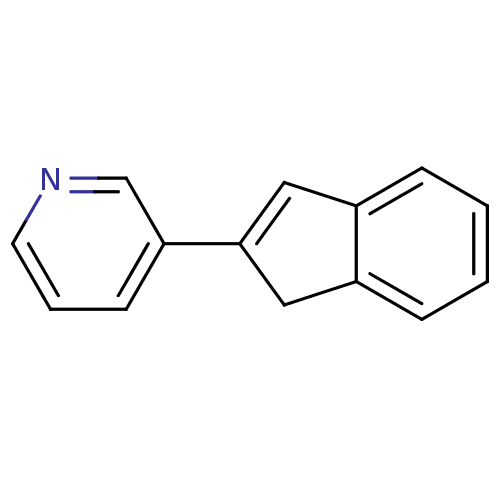 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.39E+3 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Saarland University | Assay Description The enzyme activity was assayed by monitoring the conversion of deoxycorticosterone to corticosterone in the presence of inhibitor compounds. The pro... | Pharmacol Rev 49: 2222-31 (2006) Article DOI: 10.1021/jm060055x BindingDB Entry DOI: 10.7270/Q2MW2FBS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 19A1 (Homo sapiens (Human)) | BDBM8888 (3-(1H-Inden-2-yl)pyridine | US9271963, 32 | indene...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >3.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Saarland University | Assay Description The enzyme activity was assayed by measuring the 3H-labeled H2O formed from 3H-labeled androstenedione during aromatization. After incubation, the re... | Pharmacol Rev 49: 2222-31 (2006) Article DOI: 10.1021/jm060055x BindingDB Entry DOI: 10.7270/Q2MW2FBS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||