

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM893 4-(2-{4-[(2R)-2-[(2S,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-4-phenylbutyl]-2-{[(1S,2R)-2-hydroxy-2,3-dihydro-1H-inden-1-yl]carbamoyl}ethyl]phenoxy}ethyl)-4-hydroxymorpholin-4-ium::L-685,434 deriv. 41::N-(2(R)-Hydroxy-1(S)-indanyl)-5(S)-[(tert-butyloxycarbonyl)amino]-4(S)-hydroxy-6-phenyl-2(R)-[[4-[2-(4-oxo-4-morpholinyl)ethoxy]phenyl]methyl]hexanamide
SMILES: CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccc(OCC[N+]2(O)CCOCC2)cc1)C(=O)N[C@@H]1[C@H](O)Cc2ccccc12
InChI Key: InChIKey=VHGKRXYOWQSVDY-AIIVFDHXSA-O
Data: 1 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIV-1 Protease (Human immunodeficiency virus type 1) | BDBM893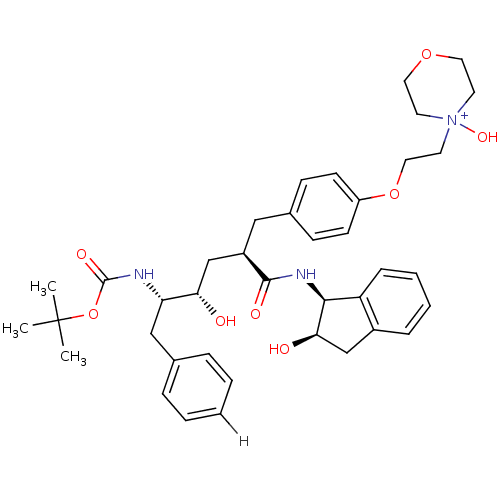 (4-(2-{4-[(2R)-2-[(2S,3S)-3-{[(tert-butoxy)carbonyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 5.5 | 30 |
Merck Research Laboratories | Assay Description Sensitivity of HIV-1 protease activity to protease inhibitors was determined by a peptide substrate cleavage assay. Protease products were analyzed o... | J Med Chem 35: 1685-701 (1992) Article DOI: 10.1021/jm00088a003 BindingDB Entry DOI: 10.7270/Q2JH3JCF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||