

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CCCc1nnc(C[C@H](C[C@H](O)CN2CCN(C[C@H]2C(=O)NCC(F)(F)F)C(C)(C)c2ncc(o2)-c2ccc(Cl)cc2)C(=O)N[C@@H]2[C@H](O)COc3ccccc23)o1
InChI Key: InChIKey=LQJQQYDNVOMBJD-YMDWUBKOSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimer of Gag-Pol polyprotein [489-587,Q496K,L499I,M525I,N526D,M535I,R546K,L552P,A560V,G562S,I573V,L579M,I582L] (Human immunodeficiency virus type 1) | BDBM9111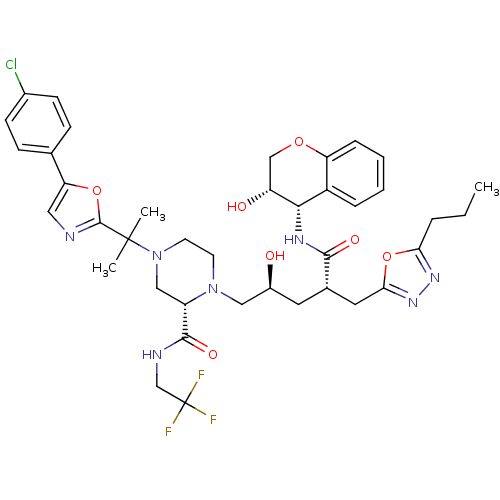 ((2S)-4-{2-[5-(4-chlorophenyl)-1,3-oxazol-2-yl]prop...) | MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories | Assay Description Assay of HIV protease inhibition was performed by peptide cleavage using the substrate Val-Ser-Gln-Asn-beta-naphthylalanine*Pro-Ile-Val. Products of ... | Bioorg Med Chem Lett 14: 4651-4 (2004) Article DOI: 10.1016/j.bmcl.2004.06.092 BindingDB Entry DOI: 10.7270/Q22Z13QX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [489-587,Q496K] (Human immunodeficiency virus type 1) | BDBM9111 ((2S)-4-{2-[5-(4-chlorophenyl)-1,3-oxazol-2-yl]prop...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | <0.0150 | n/a | n/a | n/a | n/a | 5.5 | 30 |
Merck Research Laboratories | Assay Description Assay of HIV protease inhibition was performed by peptide cleavage using the substrate Val-Ser-Gln-Asn-beta-naphthylalanine*Pro-Ile-Val. Products of ... | Bioorg Med Chem Lett 14: 4651-4 (2004) Article DOI: 10.1016/j.bmcl.2004.06.092 BindingDB Entry DOI: 10.7270/Q22Z13QX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||