

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM97610 CHEMBL1630715::US8470841, 35
SMILES: CC(C)Oc1ccc(Oc2ccc(CC[C@H](C)NC(C)=O)cc2)cn1
InChI Key: InChIKey=CHLNWUAMDPEDES-HNNXBMFYSA-N
Data: 7 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetyl-CoA carboxylase 2 (ACC2) (Homo sapiens (Human)) | BDBM97610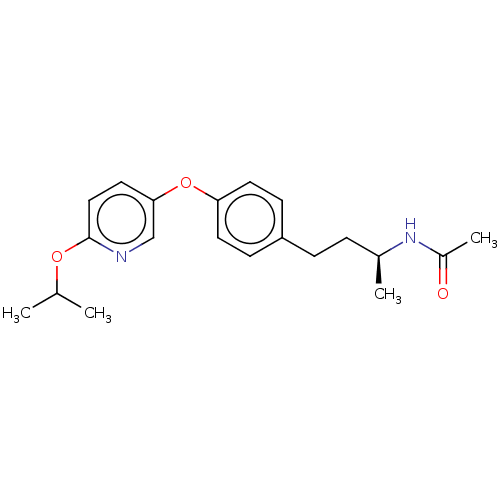 (CHEMBL1630715 | US8470841, 35) | PDB MMDB B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 30 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Sanofi US Patent | Assay Description the enzymatic activity of ACC and its inhibition by test substances were determined by a luminometric assay. | US Patent US8470841 (2013) BindingDB Entry DOI: 10.7270/Q2183537 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 1 (ACC1) (Homo sapiens (Human)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 190 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Sanofi US Patent | Assay Description the enzymatic activity of ACC and its inhibition by test substances were determined by a luminometric assay. | US Patent US8470841 (2013) BindingDB Entry DOI: 10.7270/Q2183537 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 1 (ACC1) (Rattus norvegicus (Rat)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 170 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Sanofi US Patent | Assay Description the enzymatic activity of ACC and its inhibition by test substances were determined by a luminometric assay. | US Patent US8470841 (2013) BindingDB Entry DOI: 10.7270/Q2183537 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 2 (ACC2) (Homo sapiens (Human)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Deutschland GmbH Curated by ChEMBL | Assay Description Inhibition of N-terminal His-tagged human recombinant ACC2 expressed in high five insect cells by ATP consumption assay | J Med Chem 53: 8679-87 (2010) Article DOI: 10.1021/jm101179e BindingDB Entry DOI: 10.7270/Q28K79BT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 1 (ACC1) (Rattus norvegicus (Rat)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Deutschland GmbH Curated by ChEMBL | Assay Description Inhibition of N-terminal His-tagged rat recombinant ACC1 expressed in high five insect cells by ATP consumption assay | J Med Chem 53: 8679-87 (2010) Article DOI: 10.1021/jm101179e BindingDB Entry DOI: 10.7270/Q28K79BT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 1 (ACC1) (Homo sapiens (Human)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-Aventis Deutschland GmbH Curated by ChEMBL | Assay Description Inhibition of N-terminal His-tagged human recombinant ACC1 expressed in high five insect cells by ATP consumption assay | J Med Chem 53: 8679-87 (2010) Article DOI: 10.1021/jm101179e BindingDB Entry DOI: 10.7270/Q28K79BT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetyl-CoA carboxylase 2 (ACC2) (Rattus norvegicus (Rat)) | BDBM97610 (CHEMBL1630715 | US8470841, 35) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 400 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Sanofi US Patent | Assay Description the enzymatic activity of ACC and its inhibition by test substances were determined by a luminometric assay. | US Patent US8470841 (2013) BindingDB Entry DOI: 10.7270/Q2183537 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||