

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM9981 2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-6-en-14-one::3-deoxy steroid 1::CHEMBL71715
SMILES: CC12CCC3C(CCC4=CCCCC34C)C1CCC2=O
InChI Key: InChIKey=MNOAOFLIFDRILH-UHFFFAOYSA-N
PDB links: 3 PDB IDs contain this monomer as substructures. 3 PDB IDs contain inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 19A1 (Homo sapiens (Human)) | BDBM9981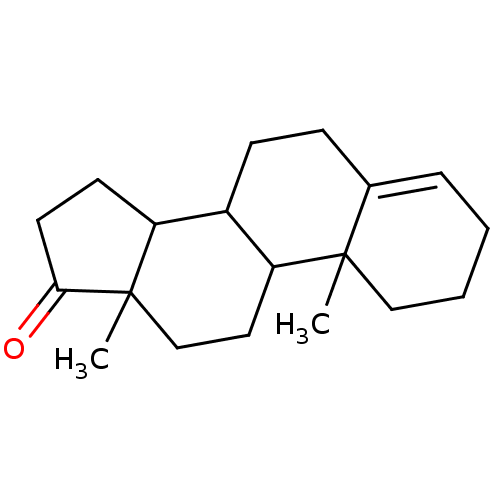 (2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]he...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.80 | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku College of Pharmacy | Assay Description The enzyme activity was assayed by measuring the 3H-labeled H2O formed from [1beta-3H] Androstenedione during aromatization. After incubation, the re... | J Med Chem 39: 2245-52 (1996) Article DOI: 10.1021/jm960047o BindingDB Entry DOI: 10.7270/Q2X34VPV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||