

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50093599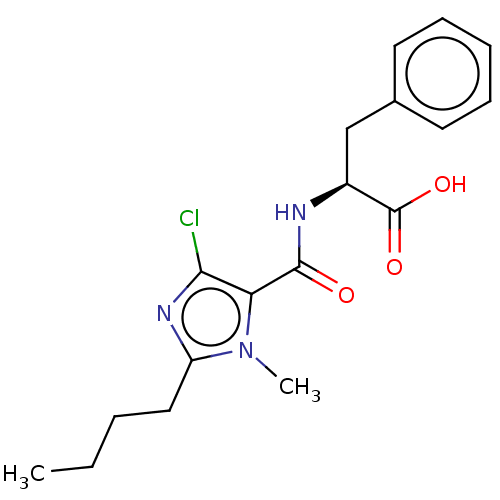 (CHEMBL3585803) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50021153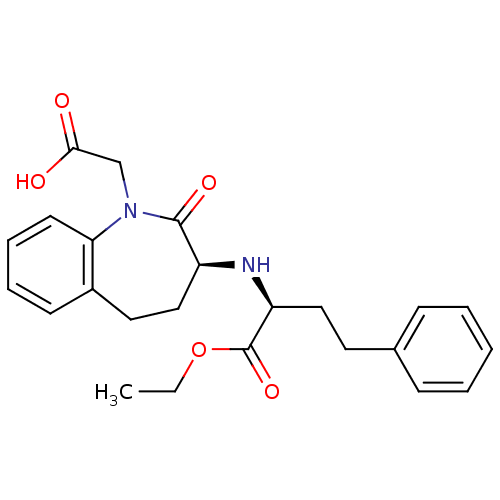 (1H-1-Benzazepine-1-acetic acid, 3-((1-(ethoxycarbo...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50368166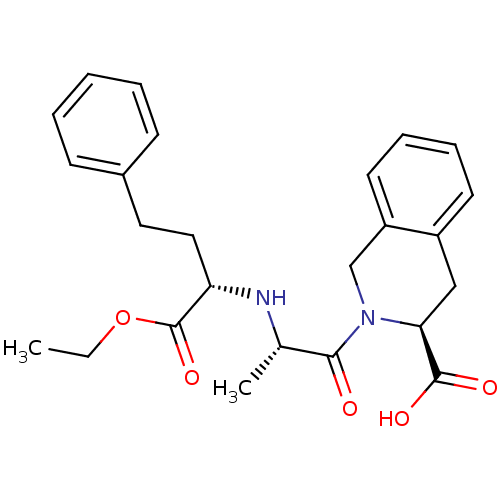 (Accupril | QUINAPRIL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50017129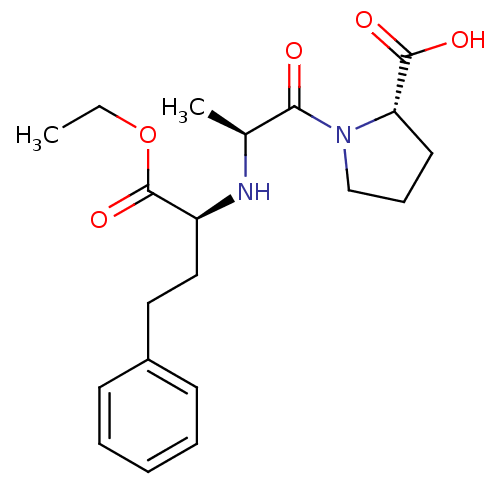 ((S)-1-((S)-2-((R)-1-ethoxy-1-oxo-4-phenylbutan-2-y...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 123 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50021153 (1H-1-Benzazepine-1-acetic acid, 3-((1-(ethoxycarbo...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 212 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50084681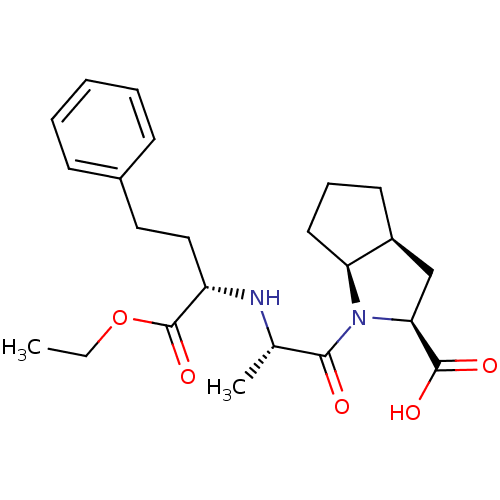 (1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-propi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 253 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50084681 (1-[2-(1-Ethoxycarbonyl-3-phenyl-propylamino)-propi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 253 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50367879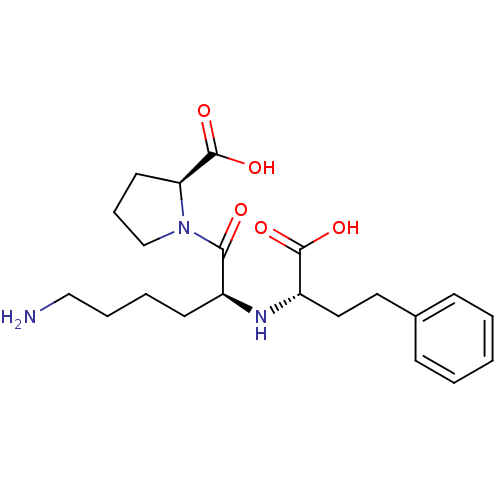 (LISINOPRIL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 262 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50438379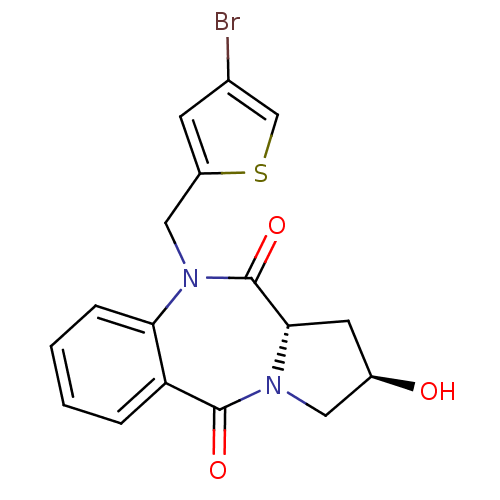 (CHEMBL2413588) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 272 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50367879 (LISINOPRIL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 318 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50438380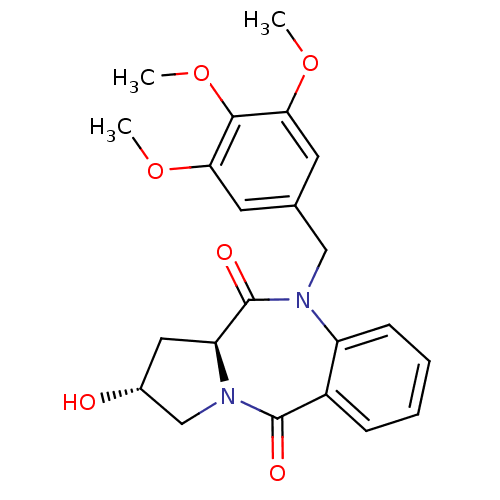 (CHEMBL2413608) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 376 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50438382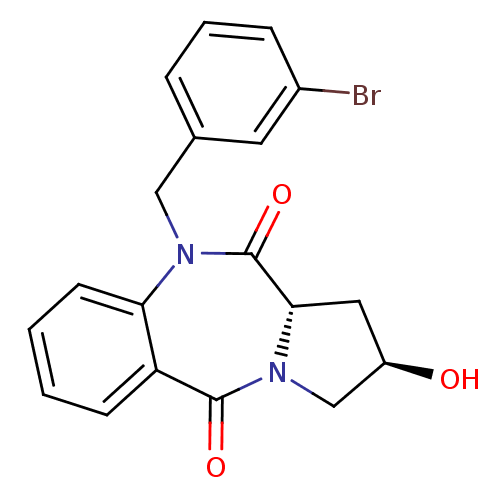 (CHEMBL2413596) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 418 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50093606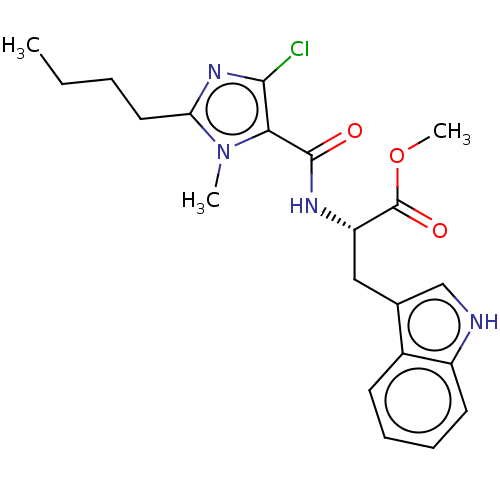 (CHEMBL3585794) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 531 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50093607 (CHEMBL3585792) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 647 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50093601 (CHEMBL3585802) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 657 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50438381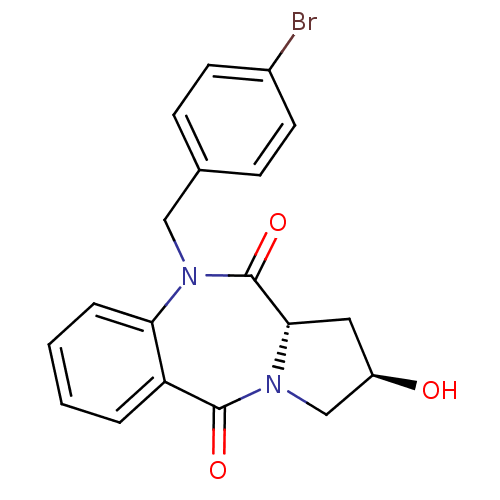 (CHEMBL2413597) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 658 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hydrolysis of hippuryl-histidyl-leucine to hippuric acid and histidyl-leucine after 30 mins | Bioorg Med Chem 21: 4485-93 (2013) Article DOI: 10.1016/j.bmc.2013.05.031 BindingDB Entry DOI: 10.7270/Q2P55PXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50093604 (CHEMBL3585799) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Indian Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as hippuryl-histidyl-leucine hydrolysis after 30 mins by colorimetric method | Bioorg Med Chem 23: 3526-33 (2015) Article DOI: 10.1016/j.bmc.2015.04.024 BindingDB Entry DOI: 10.7270/Q2DB83M4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350679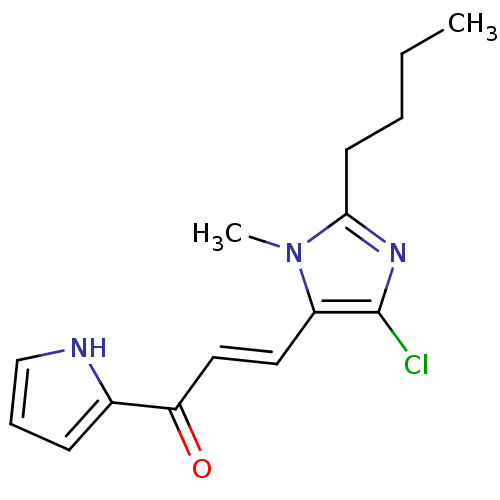 (CHEMBL1813751) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350678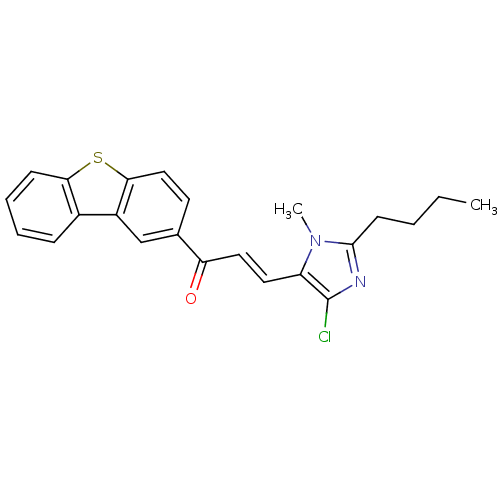 (CHEMBL1813746) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350671 (CHEMBL1813754) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50242280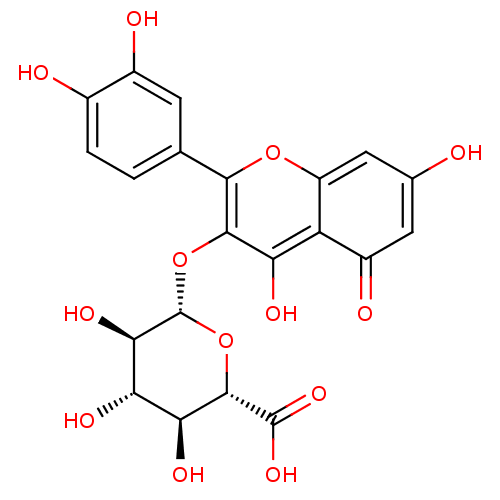 ((2S,3S,4S,5R,6S)-6-(2-(3,4-dihydroxyphenyl)-5,7-di...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50313760 (1-methyl-3-(3',4',5'-trimethoxyphenyl)-5-(4''-meth...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.13E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50159318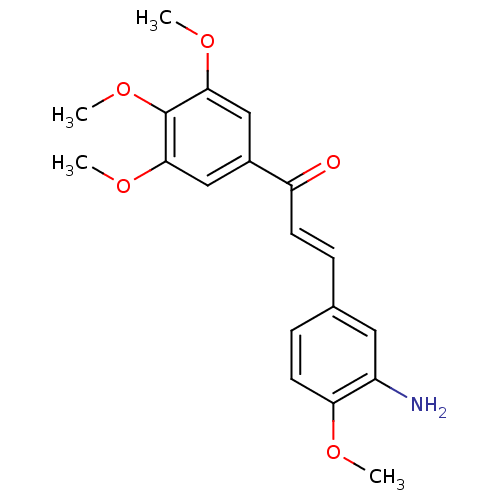 ((E)-3-(3''-Amino-4''-methoxyphenyl)-1-(3',4',5'-tr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.19E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50313764 (5,7-dihydroxy-2-(4-hydroxyphenyl)-3-((2S,3R,4S,5R,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM7458 (5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM7459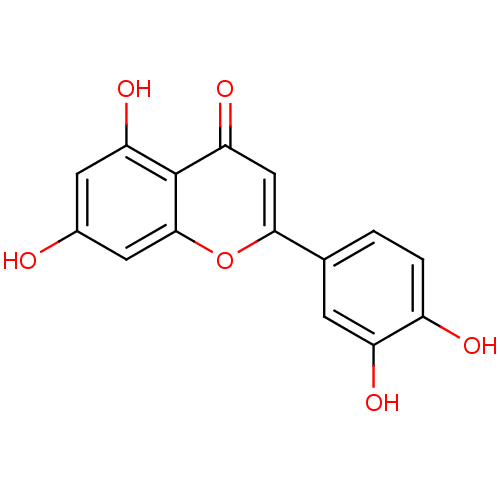 (2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4H-chromen-4...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50042949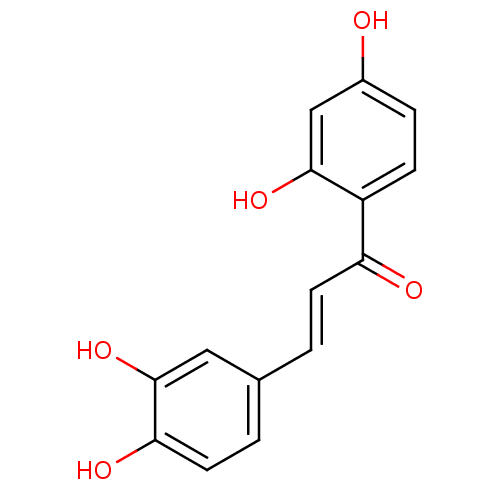 ((E)-1-(2,4-dihydroxyphenyl)-3-(3,4-dihydroxyphenyl...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 7.30E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350675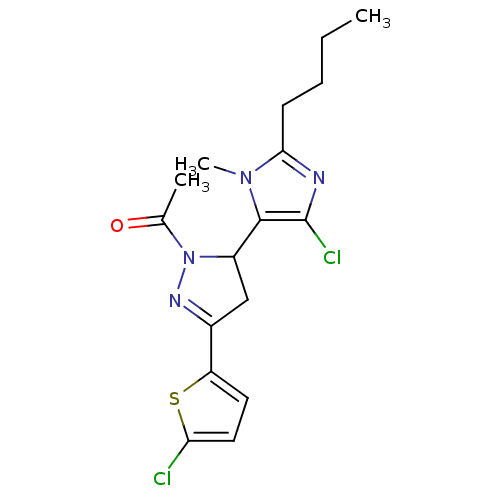 (CHEMBL1813740) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350677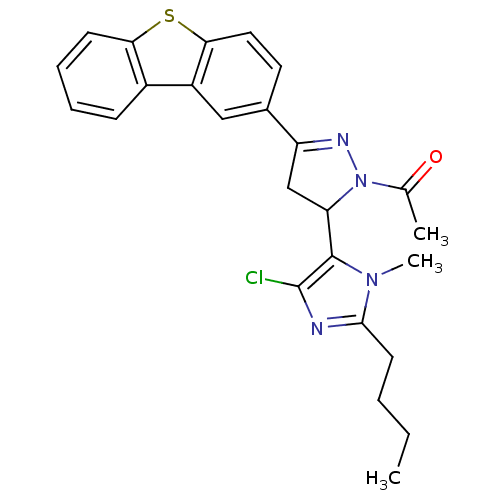 (CHEMBL1813763) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.01E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350674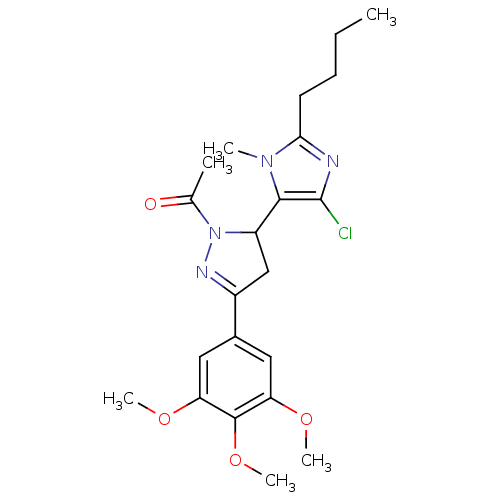 (CHEMBL1813742) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.12E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350673 (CHEMBL1813743) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.22E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350672 (CHEMBL1813744) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.33E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Oryctolagus cuniculus) | BDBM50350676 (CHEMBL1813739) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.35E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
India Institute of Chemical Technology Curated by ChEMBL | Assay Description Inhibition of rabbit lung ACE assessed as reduction in hippuryl-histidyl-leucine substrate by colorimetric assay | Bioorg Med Chem 19: 4772-81 (2011) Article DOI: 10.1016/j.bmc.2011.06.085 BindingDB Entry DOI: 10.7270/Q27M089B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||