 Found 71 hits with Last Name = 'ajamie' and Initial = 'r'
Found 71 hits with Last Name = 'ajamie' and Initial = 'r' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129720
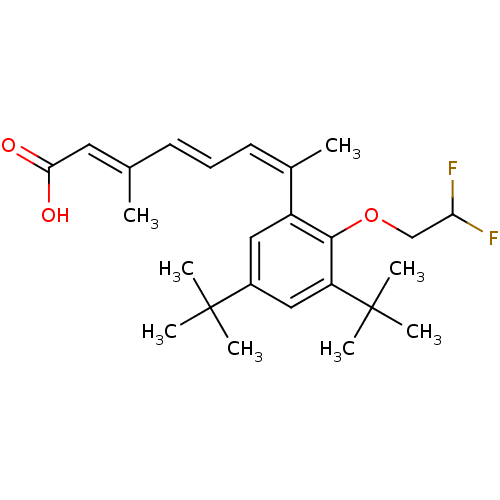
((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H34F2O3/c1-16(12-22(28)29)10-9-11-17(2)19-13-18(24(3,4)5)14-20(25(6,7)8)23(19)30-15-21(26)27/h9-14,21H,15H2,1-8H3,(H,28,29)/b10-9+,16-12+,17-11- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132579

((2E,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C27H40O3/c1-10-11-15-30-25-22(20(3)14-12-13-19(2)16-24(28)29)17-21(26(4,5)6)18-23(25)27(7,8)9/h12-14,16-18H,10-11,15H2,1-9H3,(H,28,29)/b13-12+,19-16+,20-14- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132585
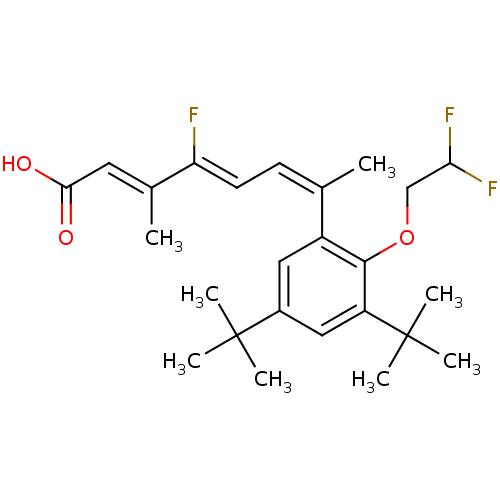
((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C(\F)=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(9-10-20(26)16(2)11-22(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-9-,16-11+,20-10- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132580
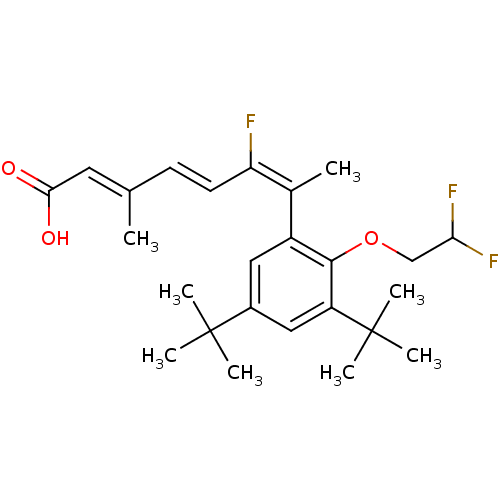
((2E,4E,6E)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C(\F)=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(11-22(29)30)9-10-20(26)16(2)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b10-9+,15-11+,20-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132581
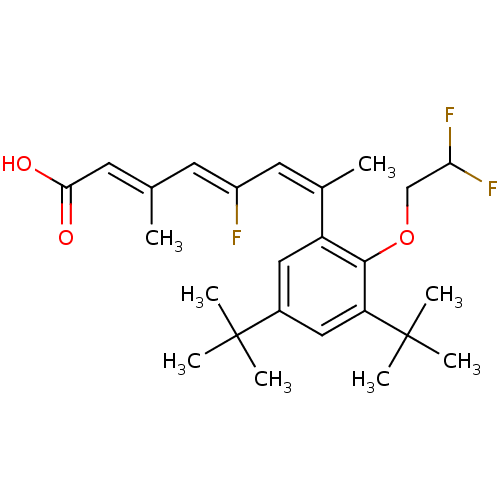
((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C=C(/F)\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(10-22(29)30)9-18(26)11-16(2)19-12-17(24(3,4)5)13-20(25(6,7)8)23(19)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-10+,16-11-,18-9- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129728

((2E,4E,6Z)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-3...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C25H36O3/c1-10-28-23-20(18(3)13-11-12-17(2)14-22(26)27)15-19(24(4,5)6)16-21(23)25(7,8)9/h11-16H,10H2,1-9H3,(H,26,27)/b12-11+,17-14+,18-13- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132584
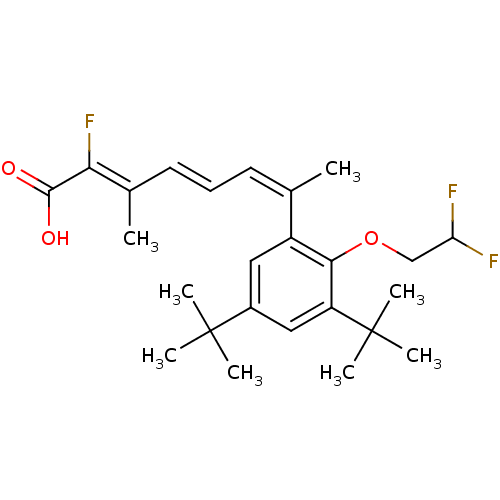
((2Z,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C(\F)C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(10-9-11-16(2)21(28)23(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)22(18)31-14-20(26)27/h9-13,20H,14H2,1-8H3,(H,29,30)/b11-9+,15-10-,21-16- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Antagonist activity against Retinoic acid receptor RXR-alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132583
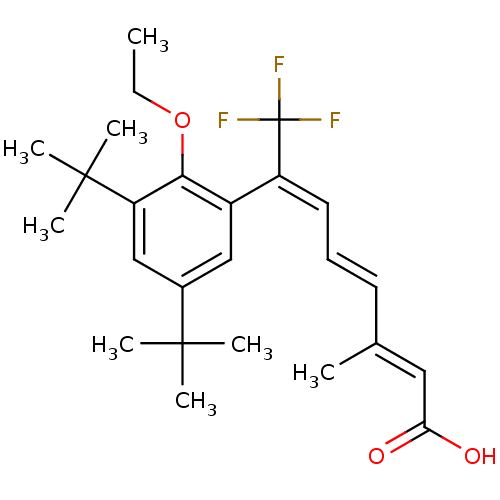
((2E,4E,6E)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-8...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(=C/C=C/C(/C)=C/C(O)=O)\C(F)(F)F Show InChI InChI=1S/C25H33F3O3/c1-9-31-22-18(14-17(23(3,4)5)15-20(22)24(6,7)8)19(25(26,27)28)12-10-11-16(2)13-21(29)30/h10-15H,9H2,1-8H3,(H,29,30)/b11-10+,16-13+,19-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132582
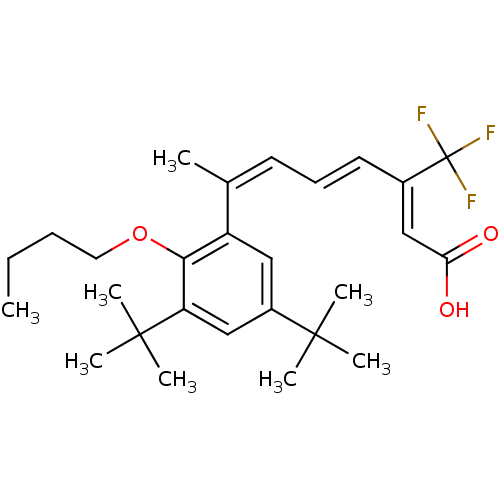
((2Z,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(=C/C(O)=O)/C(F)(F)F Show InChI InChI=1S/C27H37F3O3/c1-9-10-14-33-24-21(15-20(25(3,4)5)16-22(24)26(6,7)8)18(2)12-11-13-19(17-23(31)32)27(28,29)30/h11-13,15-17H,9-10,14H2,1-8H3,(H,31,32)/b13-11+,18-12-,19-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.21E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50129728

((2E,4E,6Z)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-3...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C25H36O3/c1-10-28-23-20(18(3)13-11-12-17(2)14-22(26)27)15-19(24(4,5)6)16-21(23)25(7,8)9/h11-16H,10H2,1-9H3,(H,26,27)/b12-11+,17-14+,18-13- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8.57E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132585

((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C(\F)=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(9-10-20(26)16(2)11-22(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-9-,16-11+,20-10- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132584

((2Z,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C(\F)C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(10-9-11-16(2)21(28)23(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)22(18)31-14-20(26)27/h9-13,20H,14H2,1-8H3,(H,29,30)/b11-9+,15-10-,21-16- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132582

((2Z,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(=C/C(O)=O)/C(F)(F)F Show InChI InChI=1S/C27H37F3O3/c1-9-10-14-33-24-21(15-20(25(3,4)5)16-22(24)26(6,7)8)18(2)12-11-13-19(17-23(31)32)27(28,29)30/h11-13,15-17H,9-10,14H2,1-8H3,(H,31,32)/b13-11+,18-12-,19-17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132580

((2E,4E,6E)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C(\F)=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(11-22(29)30)9-10-20(26)16(2)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b10-9+,15-11+,20-16+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132579

((2E,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C27H40O3/c1-10-11-15-30-25-22(20(3)14-12-13-19(2)16-24(28)29)17-21(26(4,5)6)18-23(25)27(7,8)9/h12-14,16-18H,10-11,15H2,1-9H3,(H,28,29)/b13-12+,19-16+,20-14- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132583

((2E,4E,6E)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-8...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(=C/C=C/C(/C)=C/C(O)=O)\C(F)(F)F Show InChI InChI=1S/C25H33F3O3/c1-9-31-22-18(14-17(23(3,4)5)15-20(22)24(6,7)8)19(25(26,27)28)12-10-11-16(2)13-21(29)30/h10-15H,9H2,1-8H3,(H,29,30)/b11-10+,16-13+,19-12+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50132581

((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C=C(/F)\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(10-22(29)30)9-18(26)11-16(2)19-12-17(24(3,4)5)13-20(25(6,7)8)23(19)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-10+,16-11-,18-9- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50129720

((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H34F2O3/c1-16(12-22(28)29)10-9-11-17(2)19-13-18(24(3,4)5)14-20(25(6,7)8)23(19)30-15-21(26)27/h9-14,21H,15H2,1-8H3,(H,28,29)/b10-9+,16-12+,17-11- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]ATRA binding to retinoid acid receptor gamma expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195706
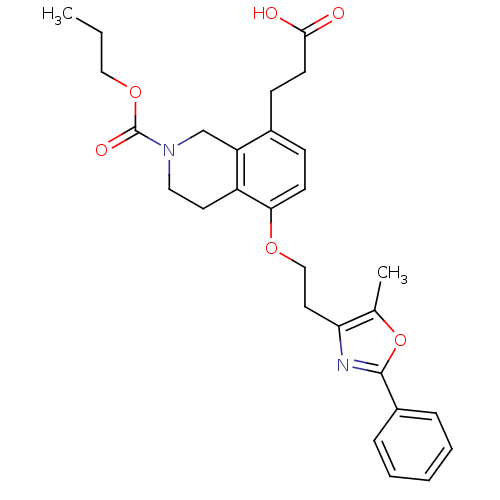
(3-(5-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)-2-(p...)Show SMILES CCCOC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C28H32N2O6/c1-3-16-35-28(33)30-15-13-22-23(18-30)20(10-12-26(31)32)9-11-25(22)34-17-14-24-19(2)36-27(29-24)21-7-5-4-6-8-21/h4-9,11H,3,10,12-18H2,1-2H3,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195704
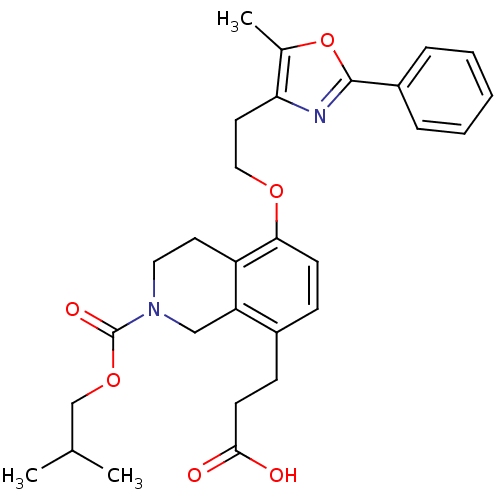
(3-(2-(isobutoxycarbonyl)-5-(2-(5-methyl-2-phenylox...)Show SMILES CC(C)COC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C29H34N2O6/c1-19(2)18-36-29(34)31-15-13-23-24(17-31)21(10-12-27(32)33)9-11-26(23)35-16-14-25-20(3)37-28(30-25)22-7-5-4-6-8-22/h4-9,11,19H,10,12-18H2,1-3H3,(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195699

(5-[2-(2-biphenyl-4-yl-5-methyl-oxazol-4-yl)-ethoxy...)Show SMILES CCCOC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccc(cc3)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C34H36N2O6/c1-3-20-41-34(39)36-19-17-28-29(22-36)26(14-16-32(37)38)13-15-31(28)40-21-18-30-23(2)42-33(35-30)27-11-9-25(10-12-27)24-7-5-4-6-8-24/h4-13,15H,3,14,16-22H2,1-2H3,(H,37,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195703
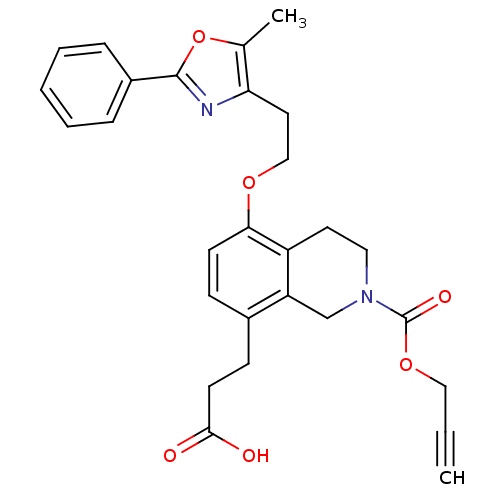
(3-(5-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)-2-((...)Show SMILES Cc1oc(nc1CCOc1ccc(CCC(O)=O)c2CN(CCc12)C(=O)OCC#C)-c1ccccc1 Show InChI InChI=1S/C28H28N2O6/c1-3-16-35-28(33)30-15-13-22-23(18-30)20(10-12-26(31)32)9-11-25(22)34-17-14-24-19(2)36-27(29-24)21-7-5-4-6-8-21/h1,4-9,11H,10,12-18H2,2H3,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085044
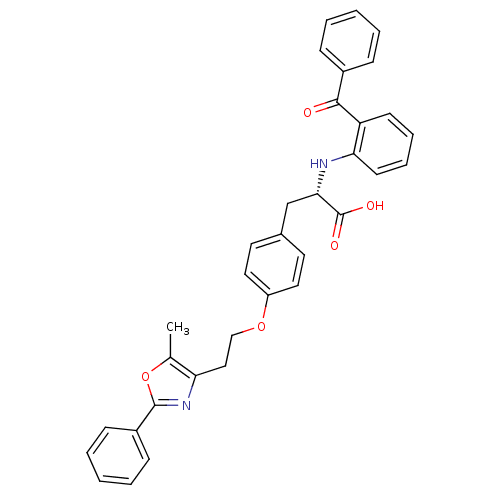
((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...)Show SMILES Cc1oc(nc1CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)-c1ccccc1 Show InChI InChI=1S/C34H30N2O5/c1-23-29(36-33(41-23)26-12-6-3-7-13-26)20-21-40-27-18-16-24(17-19-27)22-31(34(38)39)35-30-15-9-8-14-28(30)32(37)25-10-4-2-5-11-25/h2-19,31,35H,20-22H2,1H3,(H,38,39)/t31-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195705

(5-[2-(2-biphenyl-4-yl-5-methyl-oxazol-4-yl)-ethoxy...)Show SMILES Cc1oc(nc1CCOc1ccc(CCC(O)=O)c2CN(CCc12)C(=O)OCC(C)(C)C)-c1ccc(cc1)-c1ccccc1 Show InChI InChI=1S/C36H40N2O6/c1-24-31(37-34(44-24)28-12-10-26(11-13-28)25-8-6-5-7-9-25)19-21-42-32-16-14-27(15-17-33(39)40)30-22-38(20-18-29(30)32)35(41)43-23-36(2,3)4/h5-14,16H,15,17-23H2,1-4H3,(H,39,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195702
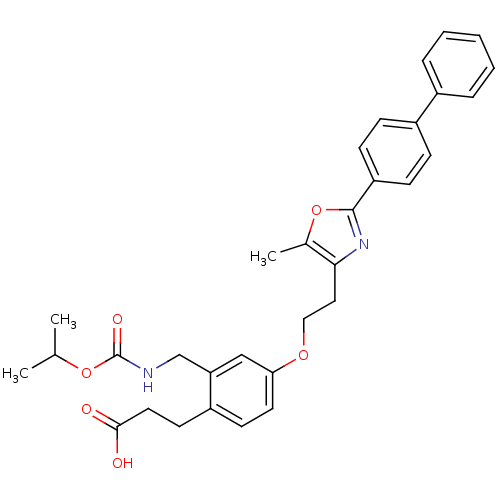
(3-[4-[2-(2-biphenyl-4-yl-5-methyl-oxazol-4-yl)-eth...)Show SMILES CC(C)OC(=O)NCc1cc(OCCc2nc(oc2C)-c2ccc(cc2)-c2ccccc2)ccc1CCC(O)=O Show InChI InChI=1S/C32H34N2O6/c1-21(2)39-32(37)33-20-27-19-28(15-13-25(27)14-16-30(35)36)38-18-17-29-22(3)40-31(34-29)26-11-9-24(10-12-26)23-7-5-4-6-8-23/h4-13,15,19,21H,14,16-18,20H2,1-3H3,(H,33,37)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM28681
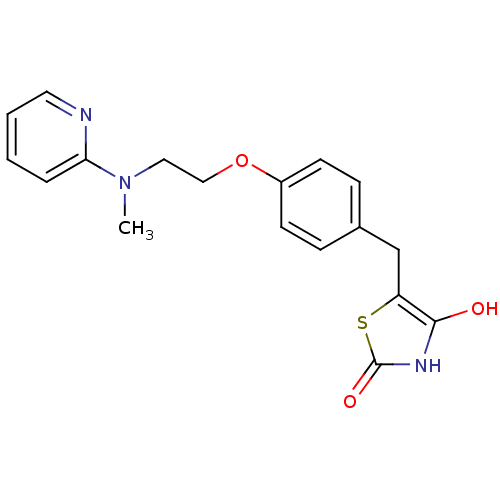
(5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...)Show InChI InChI=1S/C18H19N3O3S/c1-21(16-4-2-3-9-19-16)10-11-24-14-7-5-13(6-8-14)12-15-17(22)20-18(23)25-15/h2-9,22H,10-12H2,1H3,(H,20,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50085044

((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...)Show SMILES Cc1oc(nc1CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)-c1ccccc1 Show InChI InChI=1S/C34H30N2O5/c1-23-29(36-33(41-23)26-12-6-3-7-13-26)20-21-40-27-18-16-24(17-19-27)22-31(34(38)39)35-30-15-9-8-14-28(30)32(37)25-10-4-2-5-11-25/h2-19,31,35H,20-22H2,1H3,(H,38,39)/t31-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085045
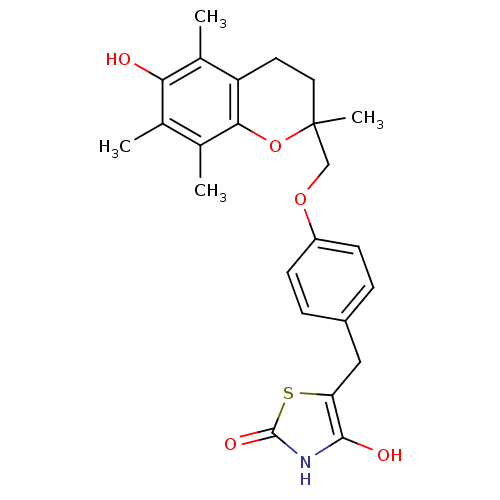
(5-((4-((6-hydroxy-2,5,7,8-tetramethyl-3,4-dihydro-...)Show SMILES Cc1c(C)c2OC(C)(COc3ccc(Cc4sc(=O)[nH]c4O)cc3)CCc2c(C)c1O Show InChI InChI=1S/C24H27NO5S/c1-13-14(2)21-18(15(3)20(13)26)9-10-24(4,30-21)12-29-17-7-5-16(6-8-17)11-19-22(27)25-23(28)31-19/h5-8,26-27H,9-12H2,1-4H3,(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.29E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50195704

(3-(2-(isobutoxycarbonyl)-5-(2-(5-methyl-2-phenylox...)Show SMILES CC(C)COC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C29H34N2O6/c1-19(2)18-36-29(34)31-15-13-23-24(17-31)21(10-12-27(32)33)9-11-26(23)35-16-14-25-20(3)37-28(30-25)22-7-5-4-6-8-22/h4-9,11,19H,10,12-18H2,1-3H3,(H,32,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.42E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50195702

(3-[4-[2-(2-biphenyl-4-yl-5-methyl-oxazol-4-yl)-eth...)Show SMILES CC(C)OC(=O)NCc1cc(OCCc2nc(oc2C)-c2ccc(cc2)-c2ccccc2)ccc1CCC(O)=O Show InChI InChI=1S/C32H34N2O6/c1-21(2)39-32(37)33-20-27-19-28(15-13-25(27)14-16-30(35)36)38-18-17-29-22(3)40-31(34-29)26-11-9-24(10-12-26)23-7-5-4-6-8-23/h4-13,15,19,21H,14,16-18,20H2,1-3H3,(H,33,37)(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.49E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50195706

(3-(5-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)-2-(p...)Show SMILES CCCOC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C28H32N2O6/c1-3-16-35-28(33)30-15-13-22-23(18-30)20(10-12-26(31)32)9-11-25(22)34-17-14-24-19(2)36-27(29-24)21-7-5-4-6-8-21/h4-9,11H,3,10,12-18H2,1-2H3,(H,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50195703

(3-(5-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)-2-((...)Show SMILES Cc1oc(nc1CCOc1ccc(CCC(O)=O)c2CN(CCc12)C(=O)OCC#C)-c1ccccc1 Show InChI InChI=1S/C28H28N2O6/c1-3-16-35-28(33)30-15-13-22-23(18-30)20(10-12-26(31)32)9-11-25(22)34-17-14-24-19(2)36-27(29-24)21-7-5-4-6-8-21/h1,4-9,11H,10,12-18H2,2H3,(H,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM28681

(5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...)Show InChI InChI=1S/C18H19N3O3S/c1-21(16-4-2-3-9-19-16)10-11-24-14-7-5-13(6-8-14)12-15-17(22)20-18(23)25-15/h2-9,22H,10-12H2,1H3,(H,20,23) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM28700
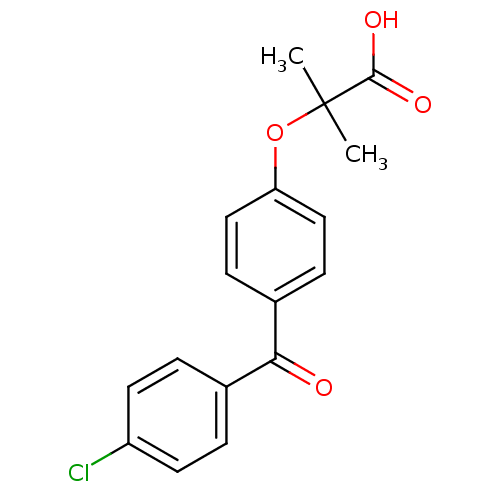
(2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...)Show InChI InChI=1S/C17H15ClO4/c1-17(2,16(20)21)22-14-9-5-12(6-10-14)15(19)11-3-7-13(18)8-4-11/h3-10H,1-2H3,(H,20,21) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM28700

(2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropionic a...)Show InChI InChI=1S/C17H15ClO4/c1-17(2,16(20)21)22-14-9-5-12(6-10-14)15(19)11-3-7-13(18)8-4-11/h3-10H,1-2H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129728

((2E,4E,6Z)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-3...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C25H36O3/c1-10-28-23-20(18(3)13-11-12-17(2)14-22(26)27)15-19(24(4,5)6)16-21(23)25(7,8)9/h11-16H,10H2,1-9H3,(H,26,27)/b12-11+,17-14+,18-13- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Antagonist activity against Retinoic acid receptor RXR-alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132583

((2E,4E,6E)-7-(3,5-Di-tert-butyl-2-ethoxy-phenyl)-8...)Show SMILES CCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(=C/C=C/C(/C)=C/C(O)=O)\C(F)(F)F Show InChI InChI=1S/C25H33F3O3/c1-9-31-22-18(14-17(23(3,4)5)15-20(22)24(6,7)8)19(25(26,27)28)12-10-11-16(2)13-21(29)30/h10-15H,9H2,1-8H3,(H,29,30)/b11-10+,16-13+,19-12+ | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 167 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132585

((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C(\F)=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(9-10-20(26)16(2)11-22(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-9-,16-11+,20-10- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 143 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132581

((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C=C(/F)\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(10-22(29)30)9-18(26)11-16(2)19-12-17(24(3,4)5)13-20(25(6,7)8)23(19)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-10+,16-11-,18-9- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 249 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132582

((2Z,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(=C/C(O)=O)/C(F)(F)F Show InChI InChI=1S/C27H37F3O3/c1-9-10-14-33-24-21(15-20(25(3,4)5)16-22(24)26(6,7)8)18(2)12-11-13-19(17-23(31)32)27(28,29)30/h11-13,15-17H,9-10,14H2,1-8H3,(H,31,32)/b13-11+,18-12-,19-17- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 359 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Antagonist activity against Retinoic acid receptor RXR-alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132580

((2E,4E,6E)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C(\F)=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H33F3O3/c1-15(11-22(29)30)9-10-20(26)16(2)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b10-9+,15-11+,20-16+ | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 6.40 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129720

((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C25H34F2O3/c1-16(12-22(28)29)10-9-11-17(2)19-13-18(24(3,4)5)14-20(25(6,7)8)23(19)30-15-21(26)27/h9-14,21H,15H2,1-8H3,(H,28,29)/b10-9+,16-12+,17-11- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 3.10 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132585

((2E,4Z,6Z)-7-[3,5-Di-tert-butyl-2-(2,2-difluoro-et...)Show SMILES C\C(=C/C(O)=O)\C(\F)=C\C=C(\C)c1cc(cc(c1OCC(F)F)C(C)(C)C)C(C)(C)C Show InChI InChI=1S/C25H33F3O3/c1-15(9-10-20(26)16(2)11-22(29)30)18-12-17(24(3,4)5)13-19(25(6,7)8)23(18)31-14-21(27)28/h9-13,21H,14H2,1-8H3,(H,29,30)/b15-9-,16-11+,20-10- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 4.84E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132582

((2Z,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(=C/C(O)=O)/C(F)(F)F Show InChI InChI=1S/C27H37F3O3/c1-9-10-14-33-24-21(15-20(25(3,4)5)16-22(24)26(6,7)8)18(2)12-11-13-19(17-23(31)32)27(28,29)30/h11-13,15-17H,9-10,14H2,1-8H3,(H,31,32)/b13-11+,18-12-,19-17- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 806 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of 3[H]-9-cis-retinoic acid binding to Retinoic acid receptor RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50132579

((2E,4E,6Z)-7-(2-Butoxy-3,5-di-tert-butyl-phenyl)-3...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)C(\C)=C/C=C/C(/C)=C/C(O)=O Show InChI InChI=1S/C27H40O3/c1-10-11-15-30-25-22(20(3)14-12-13-19(2)16-24(28)29)17-21(26(4,5)6)18-23(25)27(7,8)9/h12-14,16-18H,10-11,15H2,1-9H3,(H,28,29)/b13-12+,19-16+,20-14- | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 9.20 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Synergistic activation of BRL49653 mediated PPAR gamma and retinoid X receptor alpha expressed in CV-1 cell transcriptional activation assay |
Bioorg Med Chem Lett 13: 3191-5 (2003)
BindingDB Entry DOI: 10.7270/Q2736Q9T |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM50195699

(5-[2-(2-biphenyl-4-yl-5-methyl-oxazol-4-yl)-ethoxy...)Show SMILES CCCOC(=O)N1CCc2c(OCCc3nc(oc3C)-c3ccc(cc3)-c3ccccc3)ccc(CCC(O)=O)c2C1 Show InChI InChI=1S/C34H36N2O6/c1-3-20-41-34(39)36-19-17-28-29(22-36)26(14-16-32(37)38)13-15-31(28)40-21-18-30-23(2)42-33(35-30)27-11-9-25(10-12-27)24-7-5-4-6-8-24/h4-13,15H,3,14,16-22H2,1-2H3,(H,37,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.75E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Activity at PPARdelta |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50085044

((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...)Show SMILES Cc1oc(nc1CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)-c1ccccc1 Show InChI InChI=1S/C34H30N2O5/c1-23-29(36-33(41-23)26-12-6-3-7-13-26)20-21-40-27-18-16-24(17-19-27)22-31(34(38)39)35-30-15-9-8-14-28(30)32(37)25-10-4-2-5-11-25/h2-19,31,35H,20-22H2,1H3,(H,38,39)/t31-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 345 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARalpha by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085045

(5-((4-((6-hydroxy-2,5,7,8-tetramethyl-3,4-dihydro-...)Show SMILES Cc1c(C)c2OC(C)(COc3ccc(Cc4sc(=O)[nH]c4O)cc3)CCc2c(C)c1O Show InChI InChI=1S/C24H27NO5S/c1-13-14(2)21-18(15(3)20(13)26)9-10-24(4,30-21)12-29-17-7-5-16(6-8-17)11-19-22(27)25-23(28)31-19/h5-8,26-27H,9-12H2,1-4H3,(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.24E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50195703

(3-(5-(2-(5-methyl-2-phenyloxazol-4-yl)ethoxy)-2-((...)Show SMILES Cc1oc(nc1CCOc1ccc(CCC(O)=O)c2CN(CCc12)C(=O)OCC#C)-c1ccccc1 Show InChI InChI=1S/C28H28N2O6/c1-3-16-35-28(33)30-15-13-22-23(18-30)20(10-12-26(31)32)9-11-25(22)34-17-14-24-19(2)36-27(29-24)21-7-5-4-6-8-21/h1,4-9,11H,10,12-18H2,2H3,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 77 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085044

((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...)Show SMILES Cc1oc(nc1CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)-c1ccccc1 Show InChI InChI=1S/C34H30N2O5/c1-23-29(36-33(41-23)26-12-6-3-7-13-26)20-21-40-27-18-16-24(17-19-27)22-31(34(38)39)35-30-15-9-8-14-28(30)32(37)25-10-4-2-5-11-25/h2-19,31,35H,20-22H2,1H3,(H,38,39)/t31-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of tritium labeled ligand from human PPARgamma by SPA assay |
Bioorg Med Chem Lett 16: 6293-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.028
BindingDB Entry DOI: 10.7270/Q23R0TNK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data