 Found 136 hits with Last Name = 'ali' and Initial = 'mr'
Found 136 hits with Last Name = 'ali' and Initial = 'mr' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dual specificity protein phosphatase 5 [180-384]
(Homo sapiens (Human)) | BDBM50336799
(5,5',5''-[1,3,6-naphthalenetriyltris(sulfonylimino...)Show SMILES Cc1ccc(cc1NC(=O)c1cccc(NC(=O)Nc2cccc(c2)C(=O)Nc2cc(ccc2C)C(=O)Nc2ccc(c3cc(cc(c23)S(O)(=O)=O)S(O)(=O)=O)S(O)(=O)=O)c1)C(=O)Nc1ccc(c2cc(cc(c12)S(O)(=O)=O)S(O)(=O)=O)S(O)(=O)=O Show InChI InChI=1S/C51H40N6O23S6/c1-25-9-11-29(49(60)54-37-13-15-41(83(69,70)71)35-21-33(81(63,64)65)23-43(45(35)37)85(75,76)77)19-39(25)56-47(58)27-5-3-7-31(17-27)52-51(62)53-32-8-4-6-28(18-32)48(59)57-40-20-30(12-10-26(40)2)50(61)55-38-14-16-42(84(72,73)74)36-22-34(82(66,67)68)24-44(46(36)38)86(78,79)80/h3-24H,1-2H3,(H,54,60)(H,55,61)(H,56,58)(H,57,59)(H2,52,53,62)(H,63,64,65)(H,66,67,68)(H,69,70,71)(H,72,73,74)(H,75,76,77)(H,78,79,80) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 25 | -43.4 | 4.40E+4 | n/a | n/a | n/a | n/a | n/a | 25 |
Texas Wesleyan University
| Assay Description
For the 96-well plate validation assay, sodium orthovanadate (Sigma Aldrich) was utilized as a positive control for inhibition [Swarup et al., Bioche... |
BMC Biochem 16: 19 (2015)
Article DOI: 10.1186/s12858-015-0048-3
BindingDB Entry DOI: 10.7270/Q26972FW |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50406864

(CHEMBL4161244)Show SMILES COc1ccc(cc1OC)-c1cc2ccc(OCCCn3cc(CNC(=O)CCCCC4CCSS4)nn3)cc2oc1=O Show InChI InChI=1S/C31H36N4O6S2/c1-38-27-11-9-21(17-29(27)39-2)26-16-22-8-10-24(18-28(22)41-31(26)37)40-14-5-13-35-20-23(33-34-35)19-32-30(36)7-4-3-6-25-12-15-42-43-25/h8-11,16-18,20,25H,3-7,12-15,19H2,1-2H3,(H,32,36) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.91E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of electric eel AChE using varying levels of acetylthiocholine iodide as substrate pretreated for 5 mins followed by subst... |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50065646

(CHEMBL3087181)Show SMILES O=C(N1CCC(CC1)C(c1ccc2OCOc2c1)c1ccc2OCOc2c1)n1cncn1 Show InChI InChI=1S/C23H22N4O5/c28-23(27-12-24-11-25-27)26-7-5-15(6-8-26)22(16-1-3-18-20(9-16)31-13-29-18)17-2-4-19-21(10-17)32-14-30-19/h1-4,9-12,15,22H,5-8,13-14H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50242634

(5-((biphenyl-4-yl)methyl)-N,N-dimethyl-2H-tetrazol...)Show InChI InChI=1S/C17H17N5O/c1-21(2)17(23)22-19-16(18-20-22)12-13-8-10-15(11-9-13)14-6-4-3-5-7-14/h3-11H,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of MAGL (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM179929

(US9133148, 1a)Show SMILES OC(C1CCN(CC1)C(=O)OC(C(F)(F)F)C(F)(F)F)(c1ccc2OCOc2c1)c1ccc2OCOc2c1 Show InChI InChI=1S/C24H21F6NO7/c25-23(26,27)20(24(28,29)30)38-21(32)31-7-5-13(6-8-31)22(33,14-1-3-16-18(9-14)36-11-34-16)15-2-4-17-19(10-15)37-12-35-17/h1-4,9-10,13,20,33H,5-8,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL transfected in human HEK293T cells assessed as reduction in ABPP binding by competitive binding assay |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM60622
(BDBM50300355 | US11753371, Compound JZL-184 | US91...)Show SMILES OC(C1CCN(CC1)C(=O)Oc1ccc(cc1)[N+]([O-])=O)(c1ccc2OCOc2c1)c1ccc2OCOc2c1 Show InChI InChI=1S/C27H24N2O9/c30-26(38-21-5-3-20(4-6-21)29(32)33)28-11-9-17(10-12-28)27(31,18-1-7-22-24(13-18)36-15-34-22)19-2-8-23-25(14-19)37-16-35-23/h1-8,13-14,17,31H,9-12,15-16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of MAGL (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563791
(CHEMBL4778803)Show SMILES Fc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1Cl | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563794

(CHEMBL4793668)Show SMILES Fc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563792
(CHEMBL4788586)Show SMILES Clc1cccc(Cl)c1NCC(=O)Nc1ccc(cc1)-c1nc2ccccc2s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563793

(CHEMBL4793997)Show SMILES Clc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563790

(CHEMBL4780294)Show SMILES Brc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563801
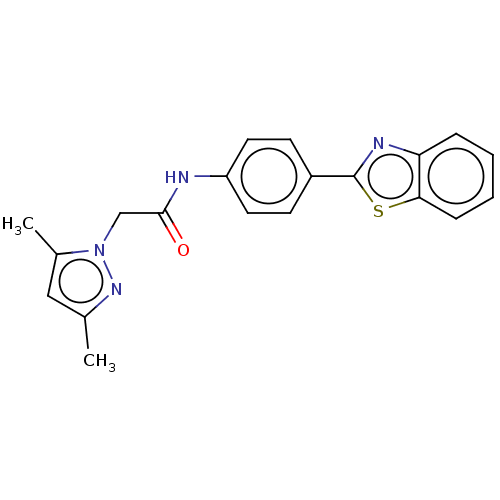
(CHEMBL4779678)Show SMILES Cc1cc(C)n(CC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563802
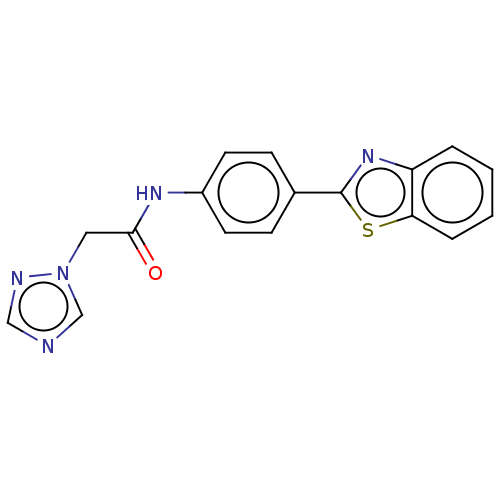
(CHEMBL4793265) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of electric eel AChE using acetylthiocholine iodide as substrate pretreated for 5 mins followed by substrate addition by Ellman's method |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50414921
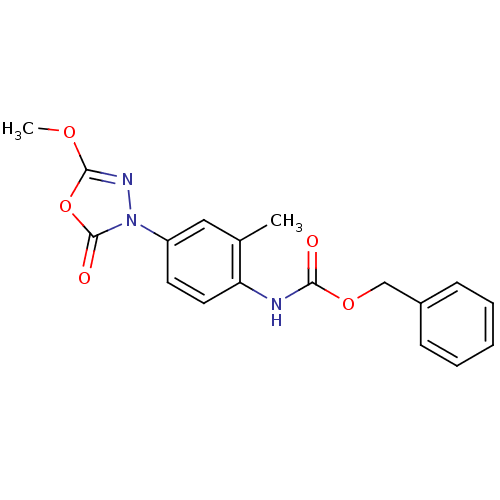
(CHEMBL570812)Show SMILES COc1nn(-c2ccc(NC(=O)OCc3ccccc3)c(C)c2)c(=O)o1 Show InChI InChI=1S/C18H17N3O5/c1-12-10-14(21-18(23)26-17(20-21)24-2)8-9-15(12)19-16(22)25-11-13-6-4-3-5-7-13/h3-10H,11H2,1-2H3,(H,19,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of MAGL (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563803
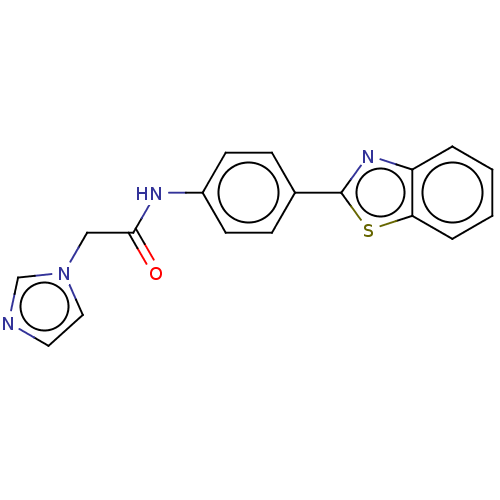
(CHEMBL4797565) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM50320491
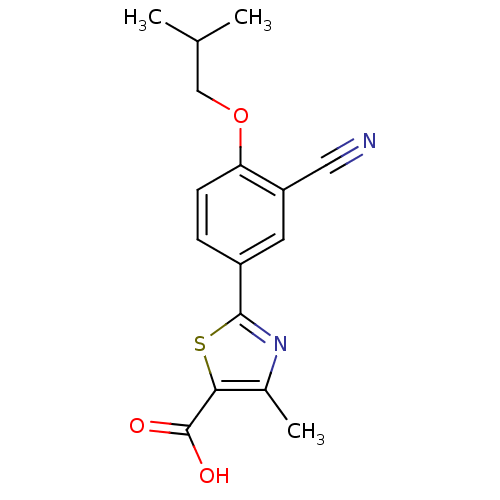
(2-(3-CYANO-4-ISOBUTOXY-PHENYL)-4-METHYL-5-THIAZOLE...)Show InChI InChI=1S/C16H16N2O3S/c1-9(2)8-21-13-5-4-11(6-12(13)7-17)15-18-10(3)14(22-15)16(19)20/h4-6,9H,8H2,1-3H3,(H,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| MMDB
PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563795
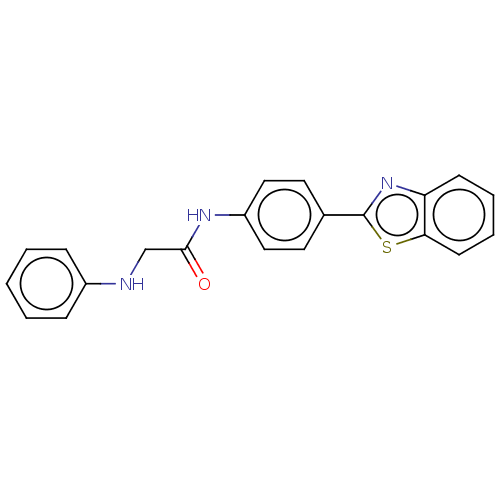
(CHEMBL4786239) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563785

(CHEMBL4792393)Show SMILES NS(=O)(=O)c1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50432829

(CHEMBL2171700)Show InChI InChI=1S/C15H12N2O4/c1-19-14-16-17(15(18)21-14)11-7-9-13(10-8-11)20-12-5-3-2-4-6-12/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of MAGL (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563809

(CHEMBL4794817)Show SMILES O=C(Cn1cnc2ccccc12)Nc1ccc(cc1)-c1nc2ccccc2s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 117 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563797
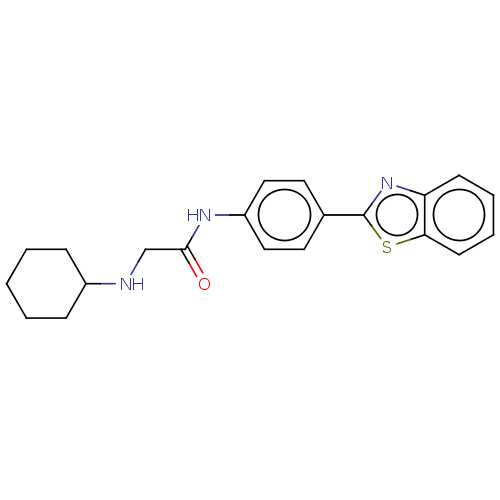
(CHEMBL4777523) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 151 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563808
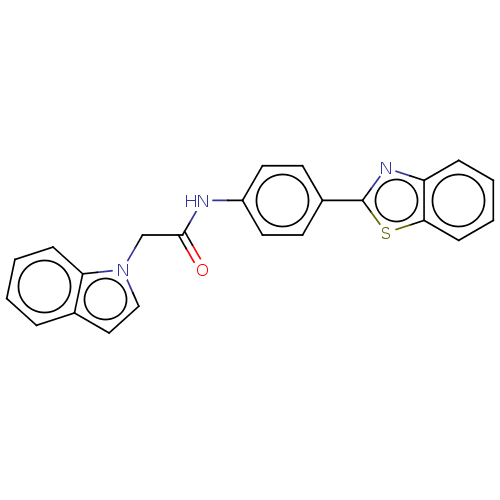
(CHEMBL4793077)Show SMILES O=C(Cn1ccc2ccccc12)Nc1ccc(cc1)-c1nc2ccccc2s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 196 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563796

(CHEMBL4780477) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 242 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563800

(CHEMBL4776276) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 282 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50414921

(CHEMBL570812)Show SMILES COc1nn(-c2ccc(NC(=O)OCc3ccccc3)c(C)c2)c(=O)o1 Show InChI InChI=1S/C18H17N3O5/c1-12-10-14(21-18(23)26-17(20-21)24-2)8-9-15(12)19-16(22)25-11-13-6-4-3-5-7-13/h3-10H,11H2,1-2H3,(H,19,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 424 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563799
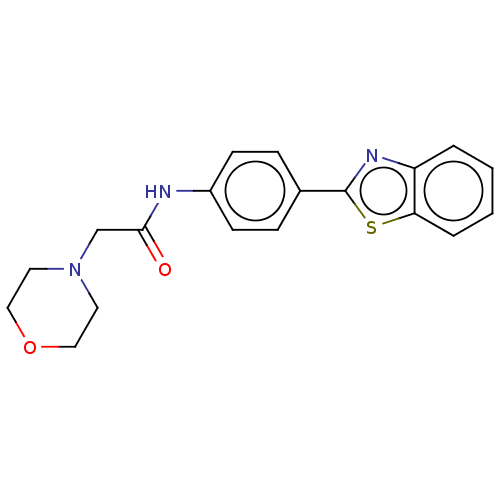
(CHEMBL4785038) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 780 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563798

(CHEMBL4800406) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 815 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM231697
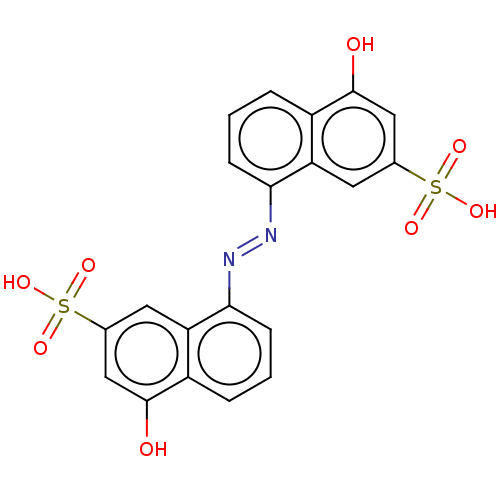
(RR601)Show SMILES Oc1cc(cc2c(cccc12)\N=N\c1cccc2c(O)cc(cc12)S(O)(=O)=O)S(O)(=O)=O Show InChI InChI=1S/C20H14N2O8S2/c23-19-9-11(31(25,26)27)7-15-13(19)3-1-5-17(15)21-22-18-6-2-4-14-16(18)8-12(10-20(14)24)32(28,29)30/h1-10,23-24H,(H,25,26,27)(H,28,29,30)/b22-21+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | 7.0 | 25 |
Concordia University of Wisconsin
| Assay Description
Assays with and without inhibitor were performed in Corning 96-well clear bottom plates having a nonbinding surface, with a total assay volume of 200... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Concordia University of Wisconsin
| Assay Description
This assay was done with full-length protein (containing both domains), andusing pERK as substrate. |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM231697

(RR601)Show SMILES Oc1cc(cc2c(cccc12)\N=N\c1cccc2c(O)cc(cc12)S(O)(=O)=O)S(O)(=O)=O Show InChI InChI=1S/C20H14N2O8S2/c23-19-9-11(31(25,26)27)7-15-13(19)3-1-5-17(15)21-22-18-6-2-4-14-16(18)8-12(10-20(14)24)32(28,29)30/h1-10,23-24H,(H,25,26,27)(H,28,29,30)/b22-21+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | 7.0 | 25 |
Concordia University of Wisconsin
| Assay Description
Assays with and without inhibitor were performed in Corning 96-well clear bottom plates having a nonbinding surface, with a total assay volume of 200... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563788
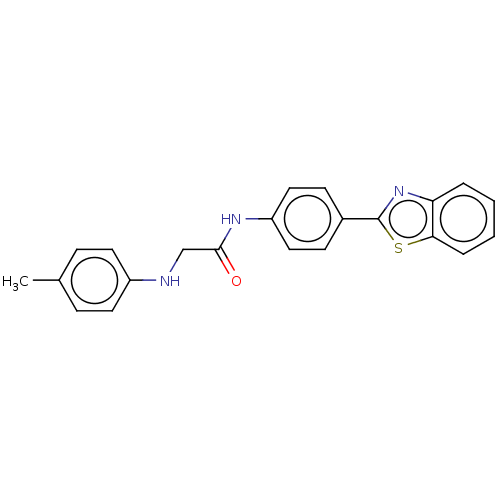
(CHEMBL4798413)Show SMILES Cc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Concordia University of Wisconsin
| Assay Description
Upon the addition of 4 μL of these respective inhibitor stock solutions to the assay buffer, the resulting range in assay inhibitor concentratio... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE using butyrylthiocholine iodide as substrate pretreated for 5 mins followed by substrate addition by Ellman's method |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM178040

(4-methyl-2-{[(2-methylphenyl)methyl]amino}-1,3-thi...)Show InChI InChI=1S/C13H14N2O2S/c1-8-5-3-4-6-10(8)7-14-13-15-9(2)11(18-13)12(16)17/h3-6H,7H2,1-2H3,(H,14,15)(H,16,17) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231697

(RR601)Show SMILES Oc1cc(cc2c(cccc12)\N=N\c1cccc2c(O)cc(cc12)S(O)(=O)=O)S(O)(=O)=O Show InChI InChI=1S/C20H14N2O8S2/c23-19-9-11(31(25,26)27)7-15-13(19)3-1-5-17(15)21-22-18-6-2-4-14-16(18)8-12(10-20(14)24)32(28,29)30/h1-10,23-24H,(H,25,26,27)(H,28,29,30)/b22-21+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Concordia University of Wisconsin
| Assay Description
This assay was done with full-length protein (containing both domains), andusing pERK as substrate. |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.90E+3 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Concordia University of Wisconsin
| Assay Description
Upon the addition of 4 μL of these respective inhibitor stock solutions to the assay buffer, the resulting range in assay inhibitor concentratio... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Concordia University of Wisconsin
| Assay Description
Upon the addition of 4 μL of these respective inhibitor stock solutions to the assay buffer, the resulting range in assay inhibitor concentratio... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563787

(CHEMBL4788417)Show SMILES COc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50406848

(CHEMBL4160630)Show SMILES Clc1ccc(cc1Cl)-c1cc2ccc(OCCCCCn3cc(CNC(=O)CCCCC4CCSS4)nn3)cc2oc1=O Show InChI InChI=1S/C31H34Cl2N4O4S2/c32-27-11-9-21(17-28(27)33)26-16-22-8-10-24(18-29(22)41-31(26)39)40-14-5-1-4-13-37-20-23(35-36-37)19-34-30(38)7-3-2-6-25-12-15-42-43-25/h8-11,16-18,20,25H,1-7,12-15,19H2,(H,34,38) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE using butyrylthiocholine iodide as substrate pretreated for 5 mins followed by substrate addition by Ellman's method |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM178041
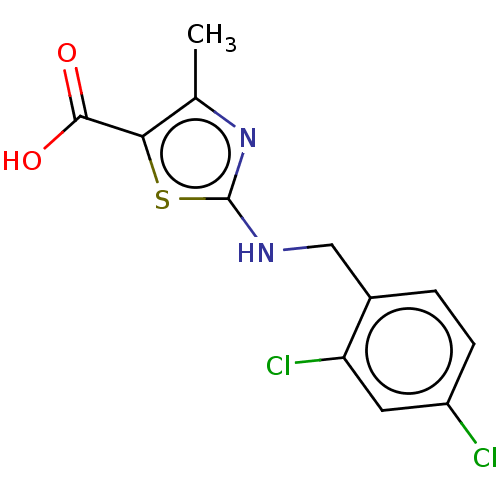
(2-{[(2,4-dichlorophenyl)methyl]amino}-4-methyl-1,3...)Show InChI InChI=1S/C12H10Cl2N2O2S/c1-6-10(11(17)18)19-12(16-6)15-5-7-2-3-8(13)4-9(7)14/h2-4H,5H2,1H3,(H,15,16)(H,17,18) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.10E+3 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM178039
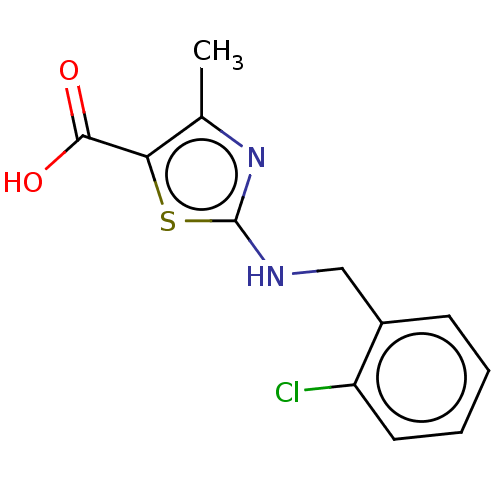
(2-{[(2-chlorophenyl)methyl]amino}-4-methyl-1,3-thi...)Show InChI InChI=1S/C12H11ClN2O2S/c1-7-10(11(16)17)18-12(15-7)14-6-8-4-2-3-5-9(8)13/h2-5H,6H2,1H3,(H,14,15)(H,16,17) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50406860
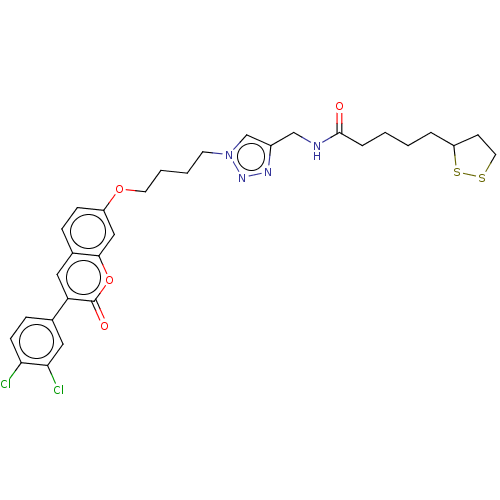
(CHEMBL4159985)Show SMILES Clc1ccc(cc1Cl)-c1cc2ccc(OCCCCn3cc(CNC(=O)CCCCC4CCSS4)nn3)cc2oc1=O Show InChI InChI=1S/C30H32Cl2N4O4S2/c31-26-10-8-20(16-27(26)32)25-15-21-7-9-23(17-28(21)40-30(25)38)39-13-4-3-12-36-19-22(34-35-36)18-33-29(37)6-2-1-5-24-11-14-41-42-24/h7-10,15-17,19,24H,1-6,11-14,18H2,(H,33,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.03E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE using butyrylthiocholine iodide as substrate pretreated for 5 mins followed by substrate addition by Ellman's method |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563786

(CHEMBL4783652)Show SMILES Oc1ccc(NCC(=O)Nc2ccc(cc2)-c2nc3ccccc3s2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM178043
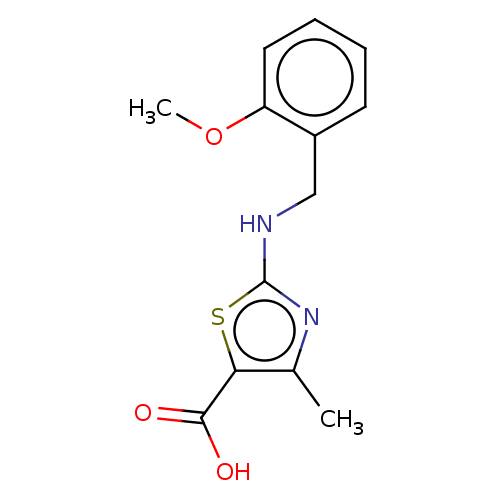
(2-{[(2-methoxyphenyl)methyl]amino}-4-methyl-1,3-th...)Show InChI InChI=1S/C13H14N2O3S/c1-8-11(12(16)17)19-13(15-8)14-7-9-5-3-4-6-10(9)18-2/h3-6H,7H2,1-2H3,(H,14,15)(H,16,17) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.47E+4 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50406864

(CHEMBL4161244)Show SMILES COc1ccc(cc1OC)-c1cc2ccc(OCCCn3cc(CNC(=O)CCCCC4CCSS4)nn3)cc2oc1=O Show InChI InChI=1S/C31H36N4O6S2/c1-38-27-11-9-21(17-29(27)39-2)26-16-22-8-10-24(18-28(22)41-31(26)37)40-14-5-13-35-20-23(33-34-35)19-32-30(36)7-4-3-6-25-12-15-42-43-25/h8-11,16-18,20,25H,3-7,12-15,19H2,1-2H3,(H,32,36) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences
Curated by ChEMBL
| Assay Description
Inhibition of electric eel AChE using acetylthiocholine iodide as substrate pretreated for 5 mins followed by substrate addition by Ellman's method |
Eur J Med Chem 152: 600-614 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.058
BindingDB Entry DOI: 10.7270/Q2WW7M8B |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.65E+4 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Concordia University of Wisconsin
| Assay Description
Upon the addition of 4 μL of these respective inhibitor stock solutions to the assay buffer, the resulting range in assay inhibitor concentratio... |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Xanthine dehydrogenase/oxidase
(Bos taurus (Bovine)) | BDBM178036
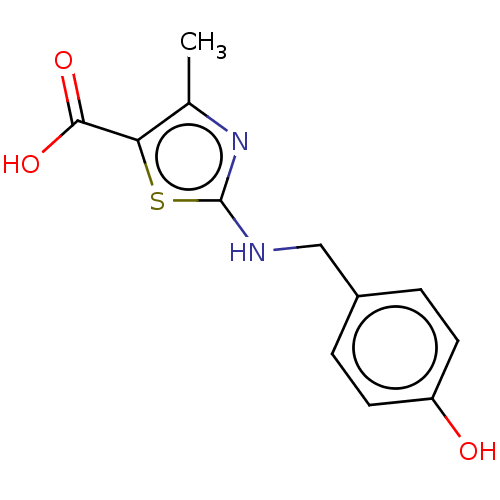
(2-{[(4-hydroxyphenyl)methyl]amino}-4-methyl-1,3-th...)Show InChI InChI=1S/C12H12N2O3S/c1-7-10(11(16)17)18-12(14-7)13-6-8-2-4-9(15)5-3-8/h2-5,15H,6H2,1H3,(H,13,14)(H,16,17) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.15E+4 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Jamia Hamdard
| Assay Description
Xanthine oxidase (XO) assay of 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylic acid derivatives 5a-p was evaluated using Bovine milk XO (grade 1, ... |
Chem Biol Drug Des 87: 508-16 (2016)
Article DOI: 10.1111/cbdd.12686
BindingDB Entry DOI: 10.7270/Q24F1PHQ |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 5
(Homo sapiens (Human)) | BDBM231694

(1-Amin-5-Naphthol-7-Sulfonic acid | NCI2602 | RR53...)Show InChI InChI=1S/C10H9NO4S/c11-9-3-1-2-7-8(9)4-6(5-10(7)12)16(13,14)15/h1-5,12H,11H2,(H,13,14,15) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.28E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Concordia University of Wisconsin
| Assay Description
This assay was done with full-length protein (containing both domains), andusing pERK as substrate. |
BMC Biochem 18: 10 (2017)
Article DOI: 10.1186/s12858-017-0083-3
BindingDB Entry DOI: 10.7270/Q2JW8CSF |
More data for this
Ligand-Target Pair | |
Monoglyceride lipase
(Homo sapiens (Human)) | BDBM50563782
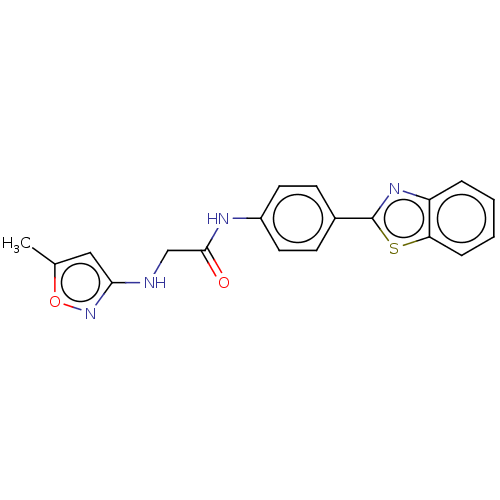
(CHEMBL4786879) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human MAGL using 4-Nitrophenylacetate as substrate |
Citation and Details
Article DOI: 10.1016/j.ejmech.2016.05.038
BindingDB Entry DOI: 10.7270/Q28056BZ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data