 Found 767 hits with Last Name = 'appel' and Initial = 'jr'
Found 767 hits with Last Name = 'appel' and Initial = 'jr' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50369460
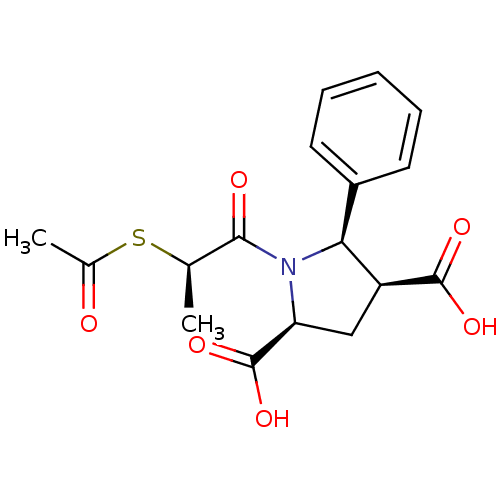
(CHEMBL1788109)Show SMILES C[C@@H](SC(C)=O)C(=O)N1[C@@H](C[C@@H]([C@@H]1c1ccccc1)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C17H19NO6S/c1-9(25-10(2)19)15(20)18-13(17(23)24)8-12(16(21)22)14(18)11-6-4-3-5-7-11/h3-7,9,12-14H,8H2,1-2H3,(H,21,22)(H,23,24)/t9-,12+,13+,14+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Torrey Pines Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Enzyme inhibitory activity towards Angiotensin I converting enzyme |
J Med Chem 42: 3743-78 (1999)
BindingDB Entry DOI: 10.7270/Q22Z167W |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399749
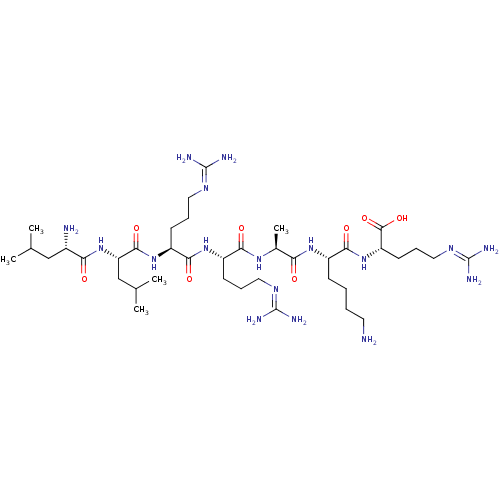
(CHEMBL2179430)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H77N17O8/c1-21(2)19-24(41)31(58)56-29(20-22(3)4)35(62)54-27(13-9-17-49-38(44)45)33(60)53-26(12-8-16-48-37(42)43)32(59)51-23(5)30(57)52-25(11-6-7-15-40)34(61)55-28(36(63)64)14-10-18-50-39(46)47/h21-29H,6-20,40-41H2,1-5H3,(H,51,59)(H,52,57)(H,53,60)(H,54,62)(H,55,61)(H,56,58)(H,63,64)(H4,42,43,48)(H4,44,45,49)(H4,46,47,50)/t23-,24-,25-,26-,27-,28-,29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399753
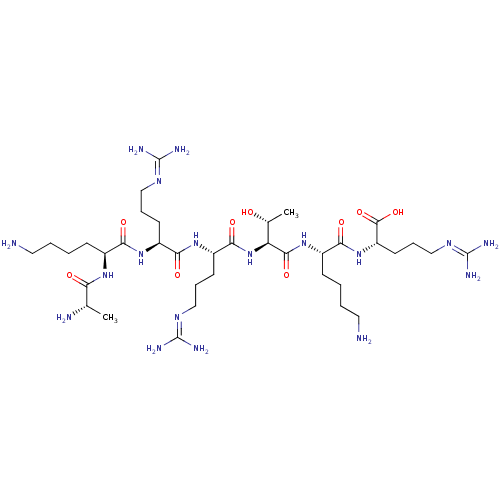
(CHEMBL2179835)Show SMILES [#6]-[#6@@H](-[#8])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C37H74N18O9/c1-20(40)28(57)50-22(10-3-5-15-38)29(58)51-24(12-7-17-47-35(41)42)30(59)52-25(13-8-18-48-36(43)44)32(61)55-27(21(2)56)33(62)53-23(11-4-6-16-39)31(60)54-26(34(63)64)14-9-19-49-37(45)46/h20-27,56H,3-19,38-40H2,1-2H3,(H,50,57)(H,51,58)(H,52,59)(H,53,62)(H,54,60)(H,55,61)(H,63,64)(H4,41,42,47)(H4,43,44,48)(H4,45,46,49)/t20-,21+,22-,23-,24-,25-,26-,27-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50266734
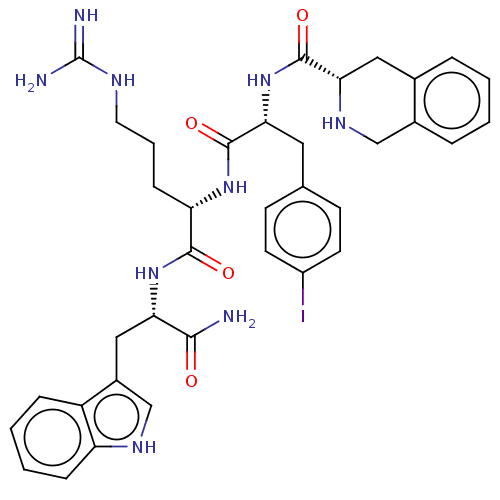
(CHEMBL4096081)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@@H](Cc1ccc(I)cc1)NC(=O)[C@@H]1Cc2ccccc2CN1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r| Show InChI InChI=1S/C36H42IN9O4/c37-25-13-11-21(12-14-25)16-31(46-34(49)30-17-22-6-1-2-7-23(22)19-43-30)35(50)44-28(10-5-15-41-36(39)40)33(48)45-29(32(38)47)18-24-20-42-27-9-4-3-8-26(24)27/h1-4,6-9,11-14,20,28-31,42-43H,5,10,15-19H2,(H2,38,47)(H,44,50)(H,45,48)(H,46,49)(H4,39,40,41)/t28-,29-,30-,31+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Antagonist activity at human MC4R expressed in CHO cells assessed as inhibition of aplha MSH-induced cAMP activation after 45 mins |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399754
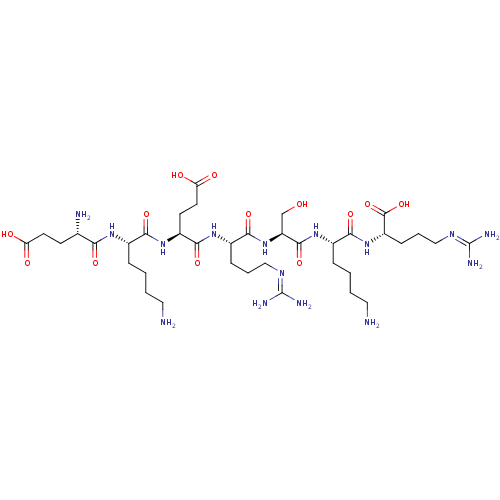
(CHEMBL2179836)Show SMILES [#7]-[#6]-[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C37H69N15O13/c38-15-3-1-7-21(47-29(58)20(40)11-13-27(54)55)30(59)50-24(12-14-28(56)57)33(62)48-23(9-5-17-45-36(41)42)32(61)52-26(19-53)34(63)49-22(8-2-4-16-39)31(60)51-25(35(64)65)10-6-18-46-37(43)44/h20-26,53H,1-19,38-40H2,(H,47,58)(H,48,62)(H,49,63)(H,50,59)(H,51,60)(H,52,61)(H,54,55)(H,56,57)(H,64,65)(H4,41,42,45)(H4,43,44,46)/t20-,21-,22-,23-,24-,25-,26-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399752
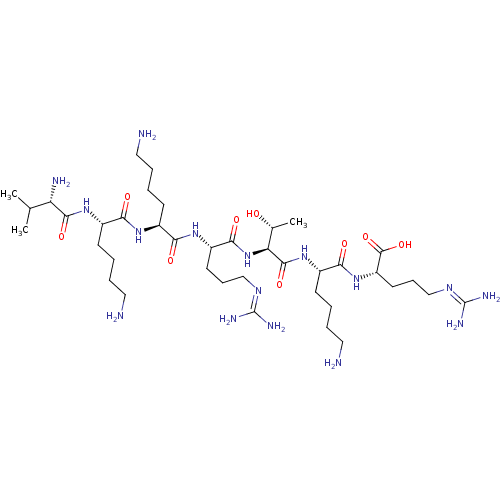
(CHEMBL2179429)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H78N16O9/c1-22(2)29(43)35(61)52-25(13-5-8-18-41)32(58)50-24(12-4-7-17-40)31(57)51-27(15-10-20-48-38(44)45)34(60)55-30(23(3)56)36(62)53-26(14-6-9-19-42)33(59)54-28(37(63)64)16-11-21-49-39(46)47/h22-30,56H,4-21,40-43H2,1-3H3,(H,50,58)(H,51,57)(H,52,61)(H,53,62)(H,54,59)(H,55,60)(H,63,64)(H4,44,45,48)(H4,46,47,49)/t23-,24+,25+,26+,27+,28+,29+,30+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399754

(CHEMBL2179836)Show SMILES [#7]-[#6]-[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C37H69N15O13/c38-15-3-1-7-21(47-29(58)20(40)11-13-27(54)55)30(59)50-24(12-14-28(56)57)33(62)48-23(9-5-17-45-36(41)42)32(61)52-26(19-53)34(63)49-22(8-2-4-16-39)31(60)51-25(35(64)65)10-6-18-46-37(43)44/h20-26,53H,1-19,38-40H2,(H,47,58)(H,48,62)(H,49,63)(H,50,59)(H,51,60)(H,52,61)(H,54,55)(H,56,57)(H,64,65)(H4,41,42,45)(H4,43,44,46)/t20-,21-,22-,23-,24-,25-,26-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399753

(CHEMBL2179835)Show SMILES [#6]-[#6@@H](-[#8])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C37H74N18O9/c1-20(40)28(57)50-22(10-3-5-15-38)29(58)51-24(12-7-17-47-35(41)42)30(59)52-25(13-8-18-48-36(43)44)32(61)55-27(21(2)56)33(62)53-23(11-4-6-16-39)31(60)54-26(34(63)64)14-9-19-49-37(45)46/h20-27,56H,3-19,38-40H2,1-2H3,(H,50,57)(H,51,58)(H,52,59)(H,53,62)(H,54,60)(H,55,61)(H,63,64)(H4,41,42,47)(H4,43,44,48)(H4,45,46,49)/t20-,21+,22-,23-,24-,25-,26-,27-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399751
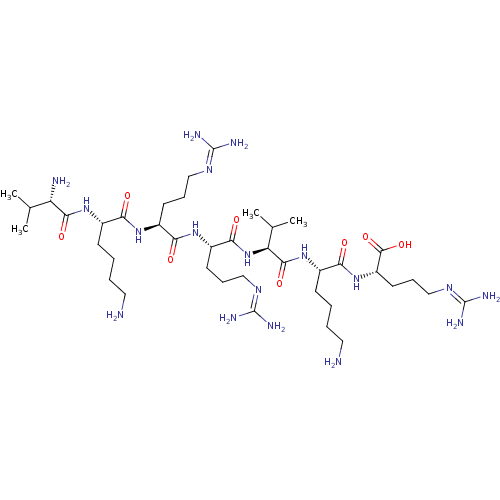
(CHEMBL2179431)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C40H80N18O8/c1-22(2)29(43)35(63)55-24(12-5-7-17-41)31(59)53-26(14-9-19-50-38(44)45)32(60)54-27(15-10-20-51-39(46)47)34(62)58-30(23(3)4)36(64)56-25(13-6-8-18-42)33(61)57-28(37(65)66)16-11-21-52-40(48)49/h22-30H,5-21,41-43H2,1-4H3,(H,53,59)(H,54,60)(H,55,63)(H,56,64)(H,57,61)(H,58,62)(H,65,66)(H4,44,45,50)(H4,46,47,51)(H4,48,49,52)/t24-,25-,26-,27-,28-,29-,30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399752

(CHEMBL2179429)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H78N16O9/c1-22(2)29(43)35(61)52-25(13-5-8-18-41)32(58)50-24(12-4-7-17-40)31(57)51-27(15-10-20-48-38(44)45)34(60)55-30(23(3)56)36(62)53-26(14-6-9-19-42)33(59)54-28(37(63)64)16-11-21-49-39(46)47/h22-30,56H,4-21,40-43H2,1-3H3,(H,50,58)(H,51,57)(H,52,61)(H,53,62)(H,54,59)(H,55,60)(H,63,64)(H4,44,45,48)(H4,46,47,49)/t23-,24+,25+,26+,27+,28+,29+,30+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399751

(CHEMBL2179431)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C40H80N18O8/c1-22(2)29(43)35(63)55-24(12-5-7-17-41)31(59)53-26(14-9-19-50-38(44)45)32(60)54-27(15-10-20-51-39(46)47)34(62)58-30(23(3)4)36(64)56-25(13-6-8-18-42)33(61)57-28(37(65)66)16-11-21-52-40(48)49/h22-30H,5-21,41-43H2,1-4H3,(H,53,59)(H,54,60)(H,55,63)(H,56,64)(H,57,61)(H,58,62)(H,65,66)(H4,44,45,50)(H4,46,47,51)(H4,48,49,52)/t24-,25-,26-,27-,28-,29-,30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399750
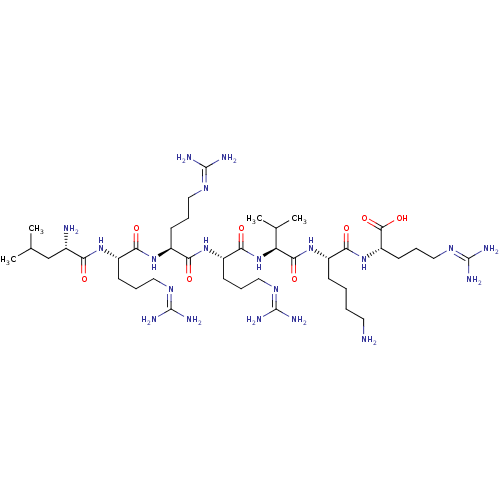
(CHEMBL2179428)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C41H82N20O8/c1-22(2)21-24(43)31(62)56-26(12-7-17-52-38(44)45)32(63)57-27(13-8-18-53-39(46)47)33(64)58-28(14-9-19-54-40(48)49)35(66)61-30(23(3)4)36(67)59-25(11-5-6-16-42)34(65)60-29(37(68)69)15-10-20-55-41(50)51/h22-30H,5-21,42-43H2,1-4H3,(H,56,62)(H,57,63)(H,58,64)(H,59,67)(H,60,65)(H,61,66)(H,68,69)(H4,44,45,52)(H4,46,47,53)(H4,48,49,54)(H4,50,51,55)/t24-,25-,26-,27-,28-,29-,30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399750

(CHEMBL2179428)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C41H82N20O8/c1-22(2)21-24(43)31(62)56-26(12-7-17-52-38(44)45)32(63)57-27(13-8-18-53-39(46)47)33(64)58-28(14-9-19-54-40(48)49)35(66)61-30(23(3)4)36(67)59-25(11-5-6-16-42)34(65)60-29(37(68)69)15-10-20-55-41(50)51/h22-30H,5-21,42-43H2,1-4H3,(H,56,62)(H,57,63)(H,58,64)(H,59,67)(H,60,65)(H,61,66)(H,68,69)(H4,44,45,52)(H4,46,47,53)(H4,48,49,54)(H4,50,51,55)/t24-,25-,26-,27-,28-,29-,30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399749

(CHEMBL2179430)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H77N17O8/c1-21(2)19-24(41)31(58)56-29(20-22(3)4)35(62)54-27(13-9-17-49-38(44)45)33(60)53-26(12-8-16-48-37(42)43)32(59)51-23(5)30(57)52-25(11-6-7-15-40)34(61)55-28(36(63)64)14-10-18-50-39(46)47/h21-29H,6-20,40-41H2,1-5H3,(H,51,59)(H,52,57)(H,53,60)(H,54,62)(H,55,61)(H,56,58)(H,63,64)(H4,42,43,48)(H4,44,45,49)(H4,46,47,50)/t23-,24-,25-,26-,27-,28-,29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12.4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Mus musculus) | BDBM50266704
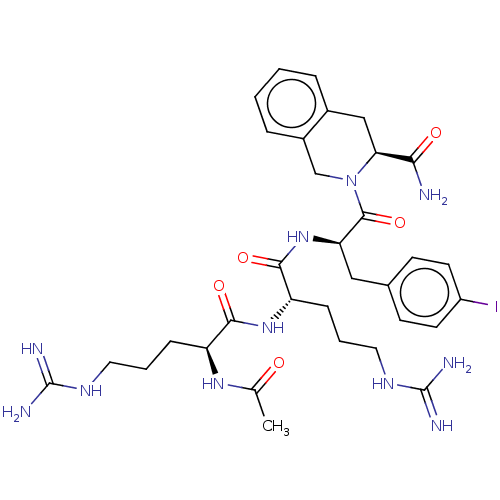
(CHEMBL4083717)Show SMILES CC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](Cc1ccc(I)cc1)C(=O)N1Cc2ccccc2C[C@H]1C(N)=O |r| Show InChI InChI=1S/C33H46IN11O5/c1-19(46)42-24(8-4-14-40-32(36)37)29(48)43-25(9-5-15-41-33(38)39)30(49)44-26(16-20-10-12-23(34)13-11-20)31(50)45-18-22-7-3-2-6-21(22)17-27(45)28(35)47/h2-3,6-7,10-13,24-27H,4-5,8-9,14-18H2,1H3,(H2,35,47)(H,42,46)(H,43,48)(H,44,49)(H4,36,37,40)(H4,38,39,41)/t24-,25-,26+,27-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Antagonist activity at mouse MC4R expressed in HEK293 cells assessed as inhibition of NDP-MSH induced-cAMP accumulation after 2 hrs by alpha screen a... |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399748
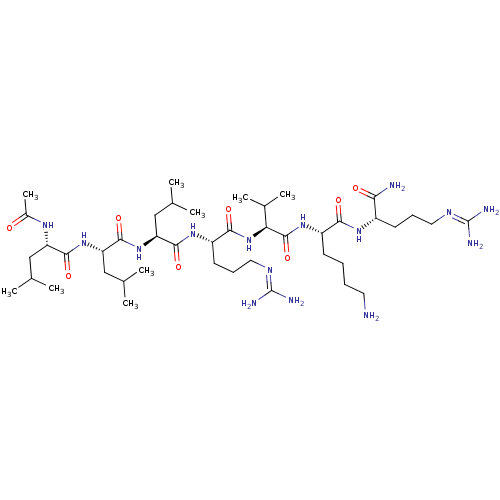
(CHEMBL2179434)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C43H83N15O8/c1-23(2)20-31(52-27(9)59)38(63)56-33(22-25(5)6)40(65)57-32(21-24(3)4)39(64)54-30(16-13-19-51-43(48)49)37(62)58-34(26(7)8)41(66)55-29(14-10-11-17-44)36(61)53-28(35(45)60)15-12-18-50-42(46)47/h23-26,28-34H,10-22,44H2,1-9H3,(H2,45,60)(H,52,59)(H,53,61)(H,54,64)(H,55,66)(H,56,63)(H,57,65)(H,58,62)(H4,46,47,50)(H4,48,49,51)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399747
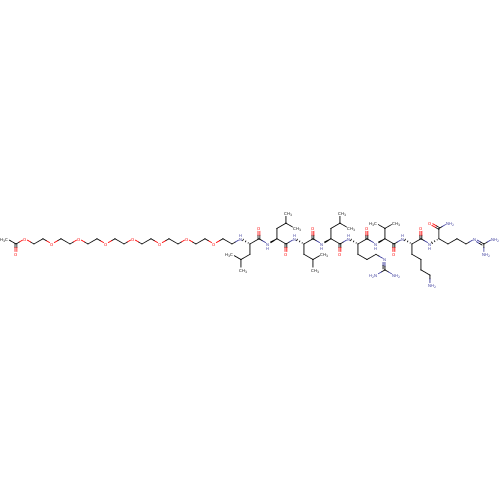
(CHEMBL2179832)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C65H126N16O17/c1-42(2)38-51(72-22-23-91-24-25-92-26-27-93-28-29-94-30-31-95-32-33-96-34-35-97-36-37-98-47(11)82)59(86)78-53(40-44(5)6)61(88)80-54(41-45(7)8)62(89)79-52(39-43(3)4)60(87)76-50(18-15-21-74-65(70)71)58(85)81-55(46(9)10)63(90)77-49(16-12-13-19-66)57(84)75-48(56(67)83)17-14-20-73-64(68)69/h42-46,48-55,72H,12-41,66H2,1-11H3,(H2,67,83)(H,75,84)(H,76,87)(H,77,90)(H,78,86)(H,79,89)(H,80,88)(H,81,85)(H4,68,69,73)(H4,70,71,74)/t48-,49-,50-,51-,52-,53-,54-,55-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399746
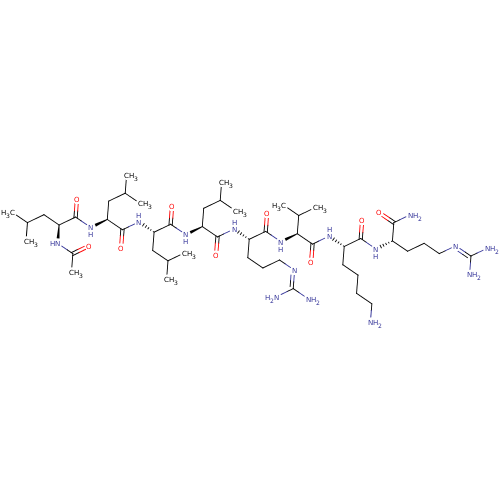
(CHEMBL2179435)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C49H94N16O9/c1-26(2)22-35(58-31(11)66)43(70)62-37(24-28(5)6)45(72)64-38(25-29(7)8)46(73)63-36(23-27(3)4)44(71)60-34(18-15-21-57-49(54)55)42(69)65-39(30(9)10)47(74)61-33(16-12-13-19-50)41(68)59-32(40(51)67)17-14-20-56-48(52)53/h26-30,32-39H,12-25,50H2,1-11H3,(H2,51,67)(H,58,66)(H,59,68)(H,60,71)(H,61,74)(H,62,70)(H,63,73)(H,64,72)(H,65,69)(H4,52,53,56)(H4,54,55,57)/t32-,33-,34-,35-,36-,37-,38-,39-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399761

(CHEMBL2179833)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C57H110N16O9/c1-34(2)30-43(64-27-19-15-13-12-14-16-22-39(11)74)51(78)70-45(32-36(5)6)53(80)72-46(33-37(7)8)54(81)71-44(31-35(3)4)52(79)68-42(25-21-29-66-57(62)63)50(77)73-47(38(9)10)55(82)69-41(23-17-18-26-58)49(76)67-40(48(59)75)24-20-28-65-56(60)61/h34-38,40-47,64H,12-33,58H2,1-11H3,(H2,59,75)(H,67,76)(H,68,79)(H,69,82)(H,70,78)(H,71,81)(H,72,80)(H,73,77)(H4,60,61,65)(H4,62,63,66)/t40-,41-,42-,43-,44-,45-,46-,47-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399759
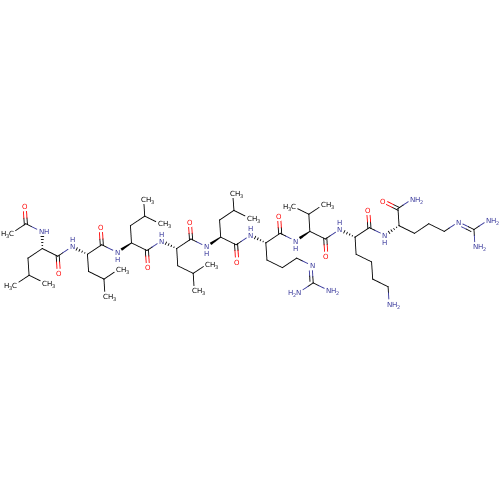
(CHEMBL2179436)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C55H105N17O10/c1-29(2)24-39(64-35(13)73)48(77)68-41(26-31(5)6)50(79)70-43(28-33(9)10)52(81)71-42(27-32(7)8)51(80)69-40(25-30(3)4)49(78)66-38(20-17-23-63-55(60)61)47(76)72-44(34(11)12)53(82)67-37(18-14-15-21-56)46(75)65-36(45(57)74)19-16-22-62-54(58)59/h29-34,36-44H,14-28,56H2,1-13H3,(H2,57,74)(H,64,73)(H,65,75)(H,66,78)(H,67,82)(H,68,77)(H,69,80)(H,70,79)(H,71,81)(H,72,76)(H4,58,59,62)(H4,60,61,63)/t36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Mus musculus) | BDBM50266713
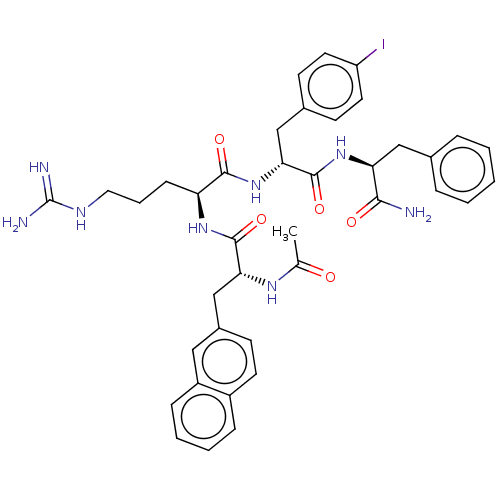
(CHEMBL4089759)Show SMILES CC(=O)N[C@H](Cc1ccc2ccccc2c1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](Cc1ccc(I)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H45IN8O5/c1-24(49)45-33(23-27-13-16-28-10-5-6-11-29(28)20-27)37(52)46-31(12-7-19-44-39(42)43)36(51)48-34(22-26-14-17-30(40)18-15-26)38(53)47-32(35(41)50)21-25-8-3-2-4-9-25/h2-6,8-11,13-18,20,31-34H,7,12,19,21-23H2,1H3,(H2,41,50)(H,45,49)(H,46,52)(H,47,53)(H,48,51)(H4,42,43,44)/t31-,32-,33+,34+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Antagonist activity at mouse MC4R expressed in HEK293 cells assessed as inhibition of NDP-MSH induced-cAMP accumulation after 2 hrs by alpha screen a... |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50387003
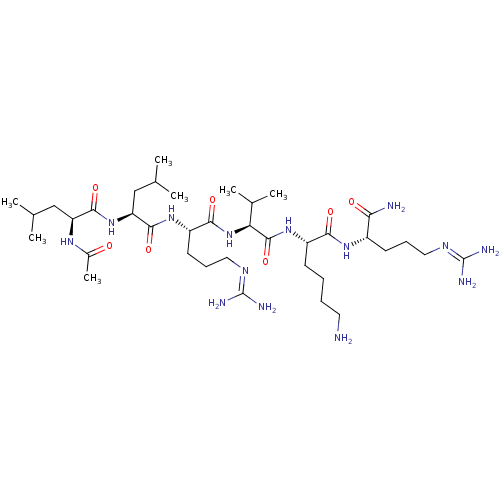
(CHEMBL2049156)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C37H72N14O7/c1-20(2)18-27(46-23(7)52)33(56)50-28(19-21(3)4)34(57)48-26(14-11-17-45-37(42)43)32(55)51-29(22(5)6)35(58)49-25(12-8-9-15-38)31(54)47-24(30(39)53)13-10-16-44-36(40)41/h20-22,24-29H,8-19,38H2,1-7H3,(H2,39,53)(H,46,52)(H,47,54)(H,48,57)(H,49,58)(H,50,56)(H,51,55)(H4,40,41,44)(H4,42,43,45)/t24-,25-,26-,27-,28-,29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399756
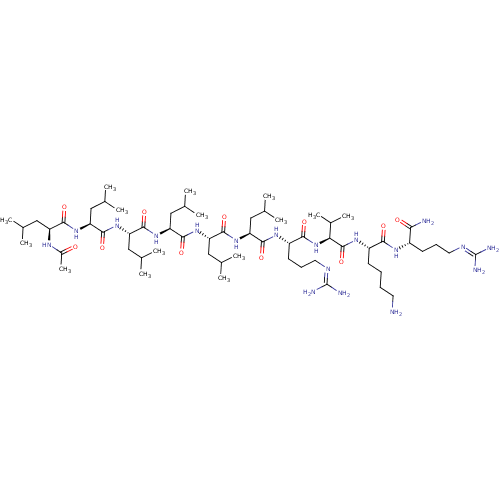
(CHEMBL2179437)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C61H116N18O11/c1-32(2)26-43(70-39(15)80)53(84)74-45(28-34(5)6)55(86)76-47(30-36(9)10)57(88)78-48(31-37(11)12)58(89)77-46(29-35(7)8)56(87)75-44(27-33(3)4)54(85)72-42(22-19-25-69-61(66)67)52(83)79-49(38(13)14)59(90)73-41(20-16-17-23-62)51(82)71-40(50(63)81)21-18-24-68-60(64)65/h32-38,40-49H,16-31,62H2,1-15H3,(H2,63,81)(H,70,80)(H,71,82)(H,72,85)(H,73,90)(H,74,84)(H,75,87)(H,76,86)(H,77,89)(H,78,88)(H,79,83)(H4,64,65,68)(H4,66,67,69)/t40-,41-,42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399748

(CHEMBL2179434)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C43H83N15O8/c1-23(2)20-31(52-27(9)59)38(63)56-33(22-25(5)6)40(65)57-32(21-24(3)4)39(64)54-30(16-13-19-51-43(48)49)37(62)58-34(26(7)8)41(66)55-29(14-10-11-17-44)36(61)53-28(35(45)60)15-12-18-50-42(46)47/h23-26,28-34H,10-22,44H2,1-9H3,(H2,45,60)(H,52,59)(H,53,61)(H,54,64)(H,55,66)(H,56,63)(H,57,65)(H,58,62)(H4,46,47,50)(H4,48,49,51)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399746

(CHEMBL2179435)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C49H94N16O9/c1-26(2)22-35(58-31(11)66)43(70)62-37(24-28(5)6)45(72)64-38(25-29(7)8)46(73)63-36(23-27(3)4)44(71)60-34(18-15-21-57-49(54)55)42(69)65-39(30(9)10)47(74)61-33(16-12-13-19-50)41(68)59-32(40(51)67)17-14-20-56-48(52)53/h26-30,32-39H,12-25,50H2,1-11H3,(H2,51,67)(H,58,66)(H,59,68)(H,60,71)(H,61,74)(H,62,70)(H,63,73)(H,64,72)(H,65,69)(H4,52,53,56)(H4,54,55,57)/t32-,33-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 430 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399747

(CHEMBL2179832)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6]-[#6]-[#8]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C65H126N16O17/c1-42(2)38-51(72-22-23-91-24-25-92-26-27-93-28-29-94-30-31-95-32-33-96-34-35-97-36-37-98-47(11)82)59(86)78-53(40-44(5)6)61(88)80-54(41-45(7)8)62(89)79-52(39-43(3)4)60(87)76-50(18-15-21-74-65(70)71)58(85)81-55(46(9)10)63(90)77-49(16-12-13-19-66)57(84)75-48(56(67)83)17-14-20-73-64(68)69/h42-46,48-55,72H,12-41,66H2,1-11H3,(H2,67,83)(H,75,84)(H,76,87)(H,77,90)(H,78,86)(H,79,89)(H,80,88)(H,81,85)(H4,68,69,73)(H4,70,71,74)/t48-,49-,50-,51-,52-,53-,54-,55-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 480 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399761

(CHEMBL2179833)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C57H110N16O9/c1-34(2)30-43(64-27-19-15-13-12-14-16-22-39(11)74)51(78)70-45(32-36(5)6)53(80)72-46(33-37(7)8)54(81)71-44(31-35(3)4)52(79)68-42(25-21-29-66-57(62)63)50(77)73-47(38(9)10)55(82)69-41(23-17-18-26-58)49(76)67-40(48(59)75)24-20-28-65-56(60)61/h34-38,40-47,64H,12-33,58H2,1-11H3,(H2,59,75)(H,67,76)(H,68,79)(H,69,82)(H,70,78)(H,71,81)(H,72,80)(H,73,77)(H4,60,61,65)(H4,62,63,66)/t40-,41-,42-,43-,44-,45-,46-,47-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50387003

(CHEMBL2049156)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C37H72N14O7/c1-20(2)18-27(46-23(7)52)33(56)50-28(19-21(3)4)34(57)48-26(14-11-17-45-37(42)43)32(55)51-29(22(5)6)35(58)49-25(12-8-9-15-38)31(54)47-24(30(39)53)13-10-16-44-36(40)41/h20-22,24-29H,8-19,38H2,1-7H3,(H2,39,53)(H,46,52)(H,47,54)(H,48,57)(H,49,58)(H,50,56)(H,51,55)(H4,40,41,44)(H4,42,43,45)/t24-,25-,26-,27-,28-,29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399760
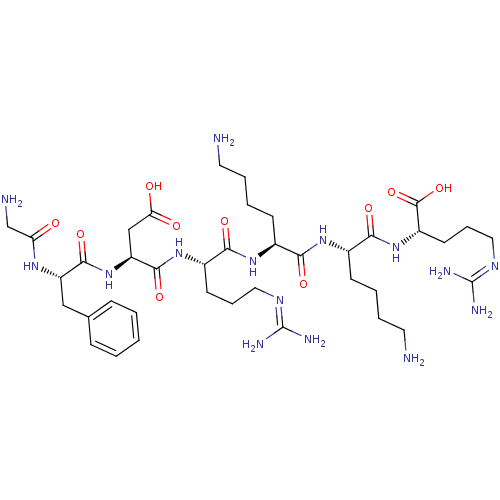
(CHEMBL2179427)Show SMILES [#7]-[#6]-[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H67N15O10/c40-16-6-4-12-24(32(58)51-25(13-5-7-17-41)34(60)53-27(37(63)64)15-9-19-48-39(45)46)50-33(59)26(14-8-18-47-38(43)44)52-36(62)29(21-31(56)57)54-35(61)28(49-30(55)22-42)20-23-10-2-1-3-11-23/h1-3,10-11,24-29H,4-9,12-22,40-42H2,(H,49,55)(H,50,59)(H,51,58)(H,52,62)(H,53,60)(H,54,61)(H,56,57)(H,63,64)(H4,43,44,47)(H4,45,46,48)/t24-,25-,26-,27-,28-,29-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399759

(CHEMBL2179436)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C55H105N17O10/c1-29(2)24-39(64-35(13)73)48(77)68-41(26-31(5)6)50(79)70-43(28-33(9)10)52(81)71-42(27-32(7)8)51(80)69-40(25-30(3)4)49(78)66-38(20-17-23-63-55(60)61)47(76)72-44(34(11)12)53(82)67-37(18-14-15-21-56)46(75)65-36(45(57)74)19-16-22-62-54(58)59/h29-34,36-44H,14-28,56H2,1-13H3,(H2,57,74)(H,64,73)(H,65,75)(H,66,78)(H,67,82)(H,68,77)(H,69,80)(H,70,79)(H,71,81)(H,72,76)(H4,58,59,62)(H4,60,61,63)/t36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399760

(CHEMBL2179427)Show SMILES [#7]-[#6]-[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O |r| Show InChI InChI=1S/C39H67N15O10/c40-16-6-4-12-24(32(58)51-25(13-5-7-17-41)34(60)53-27(37(63)64)15-9-19-48-39(45)46)50-33(59)26(14-8-18-47-38(43)44)52-36(62)29(21-31(56)57)54-35(61)28(49-30(55)22-42)20-23-10-2-1-3-11-23/h1-3,10-11,24-29H,4-9,12-22,40-42H2,(H,49,55)(H,50,59)(H,51,58)(H,52,62)(H,53,60)(H,54,61)(H,56,57)(H,63,64)(H4,43,44,47)(H4,45,46,48)/t24-,25-,26-,27-,28-,29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399757

(CHEMBL2179433)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C31H61N13O6/c1-17(2)16-23(40-19(5)45)28(49)42-22(12-9-15-39-31(36)37)27(48)44-24(18(3)4)29(50)43-21(10-6-7-13-32)26(47)41-20(25(33)46)11-8-14-38-30(34)35/h17-18,20-24H,6-16,32H2,1-5H3,(H2,33,46)(H,40,45)(H,41,47)(H,42,49)(H,43,50)(H,44,48)(H4,34,35,38)(H4,36,37,39)/t20-,21-,22-,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399758
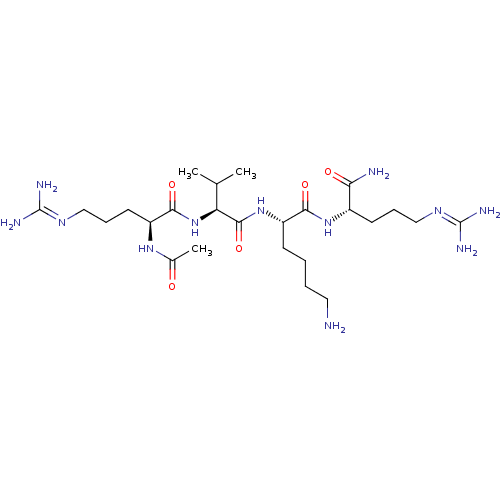
(CHEMBL2179432)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C25H50N12O5/c1-14(2)19(37-22(41)17(34-15(3)38)10-7-13-33-25(30)31)23(42)36-18(8-4-5-11-26)21(40)35-16(20(27)39)9-6-12-32-24(28)29/h14,16-19H,4-13,26H2,1-3H3,(H2,27,39)(H,34,38)(H,35,40)(H,36,42)(H,37,41)(H4,28,29,32)(H4,30,31,33)/t16-,17-,18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399755
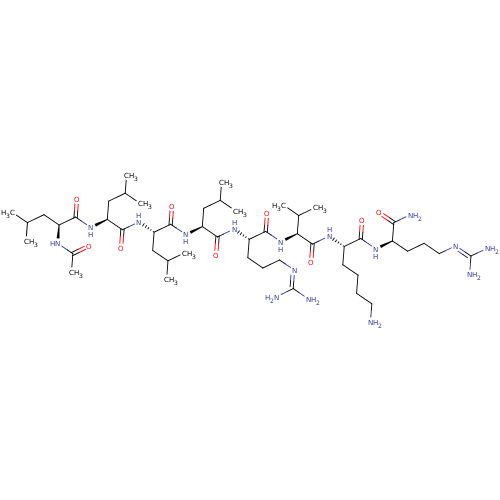
(CHEMBL2179438)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C49H94N16O9/c1-26(2)22-35(58-31(11)66)43(70)62-37(24-28(5)6)45(72)64-38(25-29(7)8)46(73)63-36(23-27(3)4)44(71)60-34(18-15-21-57-49(54)55)42(69)65-39(30(9)10)47(74)61-33(16-12-13-19-50)41(68)59-32(40(51)67)17-14-20-56-48(52)53/h26-30,32-39H,12-25,50H2,1-11H3,(H2,51,67)(H,58,66)(H,59,68)(H,60,71)(H,61,74)(H,62,70)(H,63,73)(H,64,72)(H,65,69)(H4,52,53,56)(H4,54,55,57)/t32-,33+,34+,35+,36+,37+,38+,39+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.38E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399758

(CHEMBL2179432)Show SMILES [#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C25H50N12O5/c1-14(2)19(37-22(41)17(34-15(3)38)10-7-13-33-25(30)31)23(42)36-18(8-4-5-11-26)21(40)35-16(20(27)39)9-6-12-32-24(28)29/h14,16-19H,4-13,26H2,1-3H3,(H2,27,39)(H,34,38)(H,35,40)(H,36,42)(H,37,41)(H4,28,29,32)(H4,30,31,33)/t16-,17-,18-,19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 6
(Homo sapiens (Human)) | BDBM50399757

(CHEMBL2179433)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C31H61N13O6/c1-17(2)16-23(40-19(5)45)28(49)42-22(12-9-15-39-31(36)37)27(48)44-24(18(3)4)29(50)43-21(10-6-7-13-32)26(47)41-20(25(33)46)11-8-14-38-30(34)35/h17-18,20-24H,6-16,32H2,1-5H3,(H2,33,46)(H,40,45)(H,41,47)(H,42,49)(H,43,50)(H,44,48)(H4,34,35,38)(H4,36,37,39)/t20-,21-,22-,23-,24-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399755

(CHEMBL2179438)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C49H94N16O9/c1-26(2)22-35(58-31(11)66)43(70)62-37(24-28(5)6)45(72)64-38(25-29(7)8)46(73)63-36(23-27(3)4)44(71)60-34(18-15-21-57-49(54)55)42(69)65-39(30(9)10)47(74)61-33(16-12-13-19-50)41(68)59-32(40(51)67)17-14-20-56-48(52)53/h26-30,32-39H,12-25,50H2,1-11H3,(H2,51,67)(H,58,66)(H,59,68)(H,60,71)(H,61,74)(H,62,70)(H,63,73)(H,64,72)(H,65,69)(H4,52,53,56)(H4,54,55,57)/t32-,33+,34+,35+,36+,37+,38+,39+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Furin
(Homo sapiens (Human)) | BDBM50399756

(CHEMBL2179437)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C61H116N18O11/c1-32(2)26-43(70-39(15)80)53(84)74-45(28-34(5)6)55(86)76-47(30-36(9)10)57(88)78-48(31-37(11)12)58(89)77-46(29-35(7)8)56(87)75-44(27-33(3)4)54(85)72-42(22-19-25-69-61(66)67)52(83)79-49(38(13)14)59(90)73-41(20-16-17-23-62)51(82)71-40(50(63)81)21-18-24-68-60(64)65/h32-38,40-49H,16-31,62H2,1-15H3,(H2,63,81)(H,70,80)(H,71,82)(H,72,85)(H,73,90)(H,74,84)(H,75,87)(H,76,86)(H,77,89)(H,78,88)(H,79,83)(H4,64,65,68)(H4,66,67,69)/t40-,41-,42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... |
J Med Chem 55: 10501-11 (2012)
Article DOI: 10.1021/jm3011178
BindingDB Entry DOI: 10.7270/Q2NV9KDZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50188149
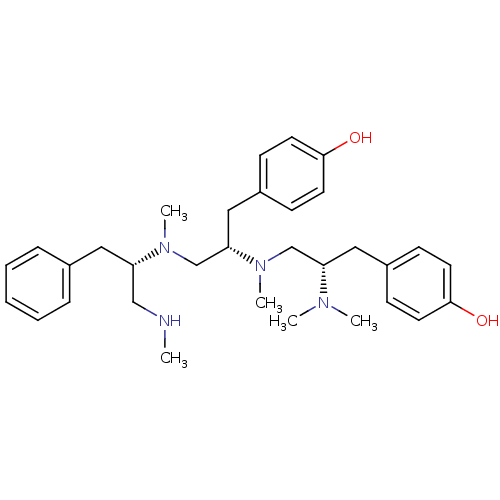
(4-[(2S)-2-(dimethylamino)-3-{[(2S)-1-(4-hydroxyphe...)Show SMILES CNC[C@H](Cc1ccccc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C Show InChI InChI=1S/C32H46N4O2/c1-33-22-28(19-25-9-7-6-8-10-25)35(4)24-30(21-27-13-17-32(38)18-14-27)36(5)23-29(34(2)3)20-26-11-15-31(37)16-12-26/h6-18,28-30,33,37-38H,19-24H2,1-5H3/t28-,29-,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain homogenate |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Mus musculus) | BDBM50017181
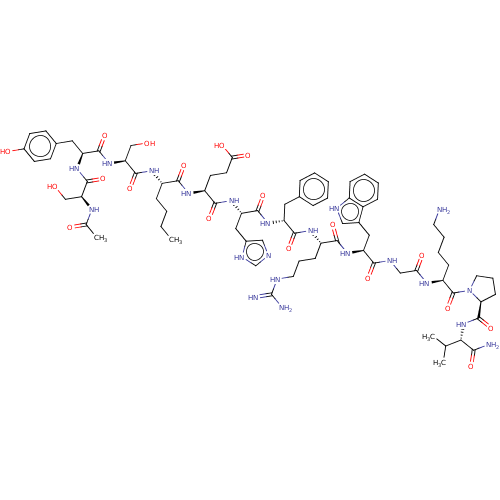
(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Displacement of [125I]-NDP-MSH from mouse MC4R expressed in HEK293 cells after 1 hr by gamma counting |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
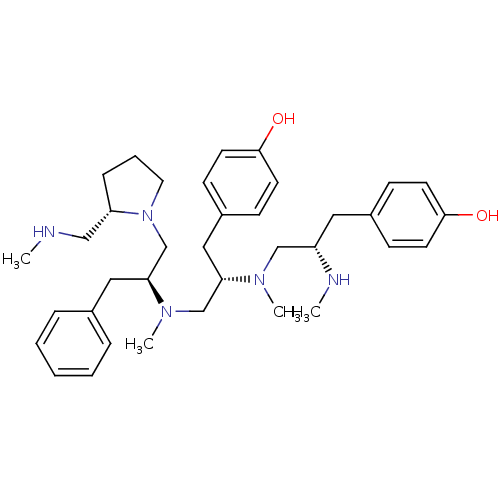
(GUINEA PIG) | BDBM50188156
(4-[(2S)-3-{[(2S)-1-(4-hydroxyphenyl)-3-{methyl[(2S...)Show SMILES CNC[C@@H]1CCCN1C[C@H](Cc1ccccc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C[C@H](Cc1ccc(O)cc1)NC Show InChI InChI=1S/C36H53N5O2/c1-37-24-32-11-8-20-41(32)27-34(22-28-9-6-5-7-10-28)40(4)26-33(23-30-14-18-36(43)19-15-30)39(3)25-31(38-2)21-29-12-16-35(42)17-13-29/h5-7,9-10,12-19,31-34,37-38,42-43H,8,11,20-27H2,1-4H3/t31-,32-,33-,34-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Antagonist activity against mu opioid receptor in guinea-pig ileum assay |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
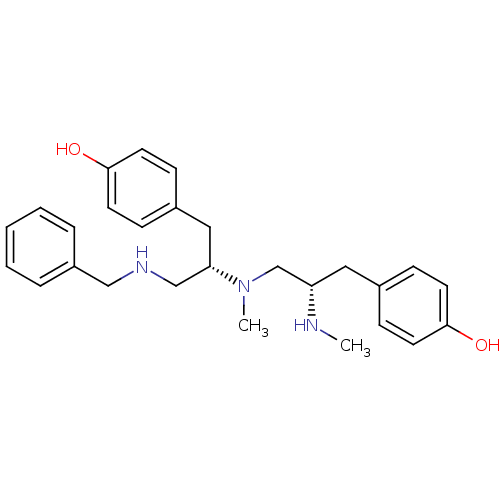
(Rattus norvegicus (rat)) | BDBM50188168
(4-[(2S)-3-{[(2S)-1-(benzylamino)-3-(4-hydroxypheny...)Show SMILES CN[C@H](CN(C)[C@H](CNCc1ccccc1)Cc1ccc(O)cc1)Cc1ccc(O)cc1 Show InChI InChI=1S/C27H35N3O2/c1-28-24(16-21-8-12-26(31)13-9-21)20-30(2)25(17-22-10-14-27(32)15-11-22)19-29-18-23-6-4-3-5-7-23/h3-15,24-25,28-29,31-32H,16-20H2,1-2H3/t24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain homogenate |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 3
(Mus musculus) | BDBM50017181

(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Displacement of [125I]-NDP-MSH from mouse MC3R expressed in HEK293 cells after 1 hr by gamma counting |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
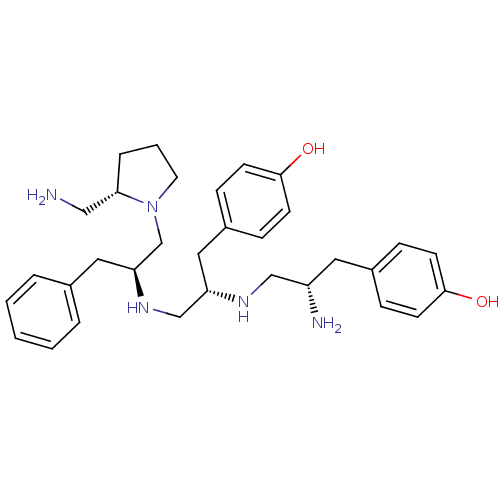
(GUINEA PIG) | BDBM50188167
(4-[(2S)-2-amino-3-{[(2S)-1-{[(2S)-1-[(2S)-2-(amino...)Show SMILES NC[C@@H]1CCCN1C[C@H](Cc1ccccc1)NC[C@H](Cc1ccc(O)cc1)NC[C@@H](N)Cc1ccc(O)cc1 Show InChI InChI=1S/C32H45N5O2/c33-20-30-7-4-16-37(30)23-29(19-24-5-2-1-3-6-24)36-22-28(18-26-10-14-32(39)15-11-26)35-21-27(34)17-25-8-12-31(38)13-9-25/h1-3,5-6,8-15,27-30,35-36,38-39H,4,7,16-23,33-34H2/t27-,28-,29-,30-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Antagonist activity against mu opioid receptor in guinea-pig ileum assay |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50188203
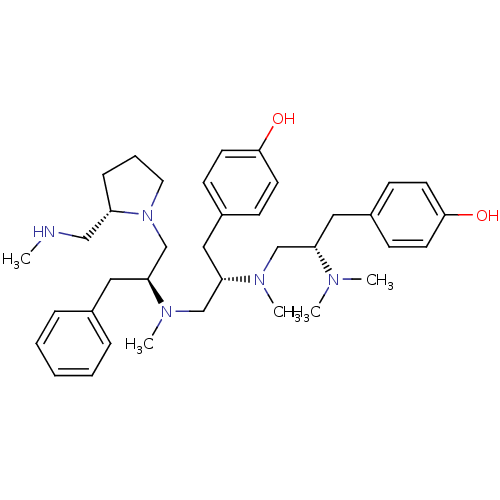
(4-[(2S)-2-(dimethylamino)-3-{[(2S)-1-(4-hydroxyphe...)Show SMILES CNC[C@@H]1CCCN1C[C@H](Cc1ccccc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C Show InChI InChI=1S/C37H55N5O2/c1-38-25-32-12-9-21-42(32)28-35(23-29-10-7-6-8-11-29)41(5)27-34(24-31-15-19-37(44)20-16-31)40(4)26-33(39(2)3)22-30-13-17-36(43)18-14-30/h6-8,10-11,13-20,32-35,38,43-44H,9,12,21-28H2,1-5H3/t32-,33-,34-,35-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Antagonist activity against mu opioid receptor in guinea-pig ileum assay |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
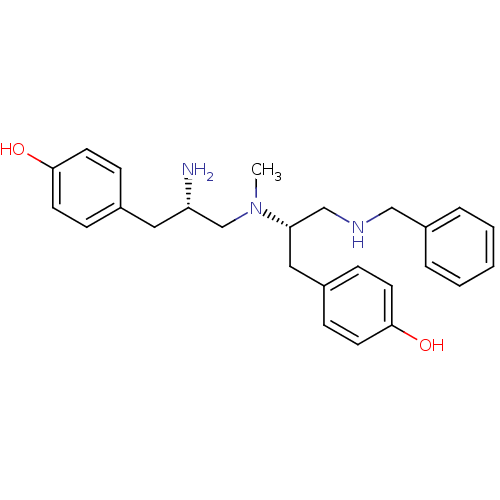
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50188209
(4-[(2S)-2-amino-3-{[(2S)-1-(benzylamino)-3-(4-hydr...)Show SMILES CN(C[C@@H](N)Cc1ccc(O)cc1)[C@H](CNCc1ccccc1)Cc1ccc(O)cc1 Show InChI InChI=1S/C26H33N3O2/c1-29(19-23(27)15-20-7-11-25(30)12-8-20)24(16-21-9-13-26(31)14-10-21)18-28-17-22-5-3-2-4-6-22/h2-14,23-24,28,30-31H,15-19,27H2,1H3/t23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain homogenate |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50188210
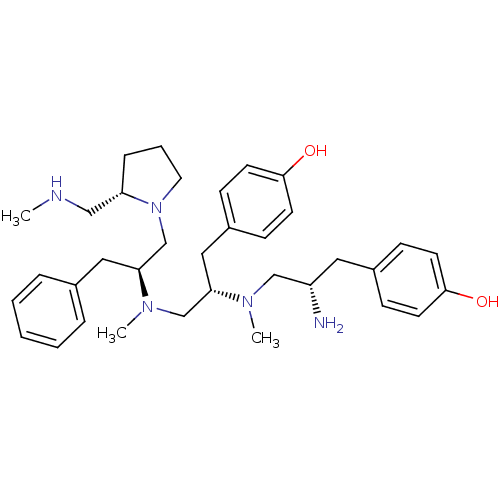
(4-[(2S)-2-amino-3-{[(2S)-1-(4-hydroxyphenyl)-3-{me...)Show SMILES CNC[C@@H]1CCCN1C[C@H](Cc1ccccc1)N(C)C[C@H](Cc1ccc(O)cc1)N(C)C[C@@H](N)Cc1ccc(O)cc1 Show InChI InChI=1S/C35H51N5O2/c1-37-23-31-10-7-19-40(31)26-33(21-27-8-5-4-6-9-27)39(3)25-32(22-29-13-17-35(42)18-14-29)38(2)24-30(36)20-28-11-15-34(41)16-12-28/h4-6,8-9,11-18,30-33,37,41-42H,7,10,19-26,36H2,1-3H3/t30-,31-,32-,33-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Antagonist activity against mu opioid receptor in guinea-pig ileum assay |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50188217
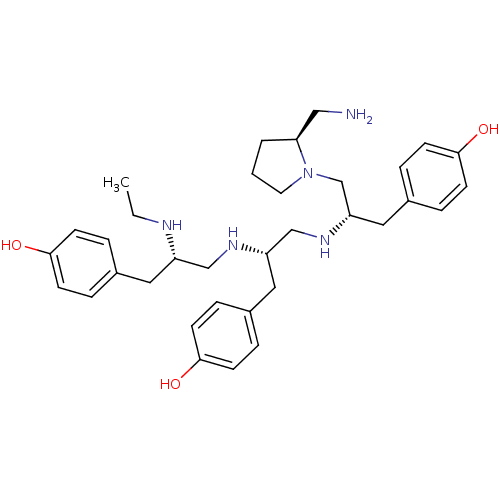
(4-[(2S)-3-{[(2S)-1-{[(2S)-1-[(2S)-2-(aminomethyl)p...)Show SMILES CCN[C@H](CN[C@H](CN[C@H](CN1CCC[C@H]1CN)Cc1ccc(O)cc1)Cc1ccc(O)cc1)Cc1ccc(O)cc1 Show InChI InChI=1S/C34H49N5O3/c1-2-36-28(18-25-5-11-32(40)12-6-25)22-37-29(19-26-7-13-33(41)14-8-26)23-38-30(20-27-9-15-34(42)16-10-27)24-39-17-3-4-31(39)21-35/h5-16,28-31,36-38,40-42H,2-4,17-24,35H2,1H3/t28-,29-,30-,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain homogenate |
Bioorg Med Chem Lett 16: 4331-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.053
BindingDB Entry DOI: 10.7270/Q2028R47 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Mus musculus) | BDBM50266704

(CHEMBL4083717)Show SMILES CC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](Cc1ccc(I)cc1)C(=O)N1Cc2ccccc2C[C@H]1C(N)=O |r| Show InChI InChI=1S/C33H46IN11O5/c1-19(46)42-24(8-4-14-40-32(36)37)29(48)43-25(9-5-15-41-33(38)39)30(49)44-26(16-20-10-12-23(34)13-11-20)31(50)45-18-22-7-3-2-6-21(22)17-27(45)28(35)47/h2-3,6-7,10-13,24-27H,4-5,8-9,14-18H2,1H3,(H2,35,47)(H,42,46)(H,43,48)(H,44,49)(H4,36,37,40)(H4,38,39,41)/t24-,25-,26+,27-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Displacement of [125I]-NDP-MSH from mouse MC4R expressed in HEK293 cells after 1 hr by gamma counting |
J Med Chem 60: 4342-4357 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00301
BindingDB Entry DOI: 10.7270/Q24170J1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50298862
(4-((S)-3-(methylamino)-2-((R)-2-((4-methylcyclohex...)Show SMILES CCC[C@H](CN[C@H](CNC)Cc1ccc(O)cc1)NCC1CCC(C)CC1 |r,wD:3.18,6.5,(27.78,-34.64,;26.45,-35.41,;25.12,-34.65,;23.78,-35.42,;23.79,-36.97,;22.46,-37.74,;22.47,-39.28,;23.81,-40.05,;23.82,-41.59,;25.15,-42.35,;21.14,-40.06,;19.8,-39.3,;19.8,-37.75,;18.47,-37,;17.14,-37.77,;15.79,-37.01,;17.14,-39.32,;18.48,-40.08,;22.45,-34.66,;22.45,-33.11,;21.1,-32.36,;21.08,-30.81,;19.74,-30.06,;18.4,-30.84,;17.07,-30.09,;18.42,-32.39,;19.77,-33.16,)| Show InChI InChI=1S/C23H41N3O/c1-4-5-21(25-15-20-8-6-18(2)7-9-20)17-26-22(16-24-3)14-19-10-12-23(27)13-11-19/h10-13,18,20-22,24-27H,4-9,14-17H2,1-3H3/t18?,20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute for Molecular Studies
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from guinea pig kappa opioid receptor by liquid scintillation counting |
Bioorg Med Chem 17: 5583-97 (2009)
Article DOI: 10.1016/j.bmc.2009.06.026
BindingDB Entry DOI: 10.7270/Q2DZ08C7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

















































