 Found 465 hits with Last Name = 'bechle' and Initial = 'bm'
Found 465 hits with Last Name = 'bechle' and Initial = 'bm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50432208
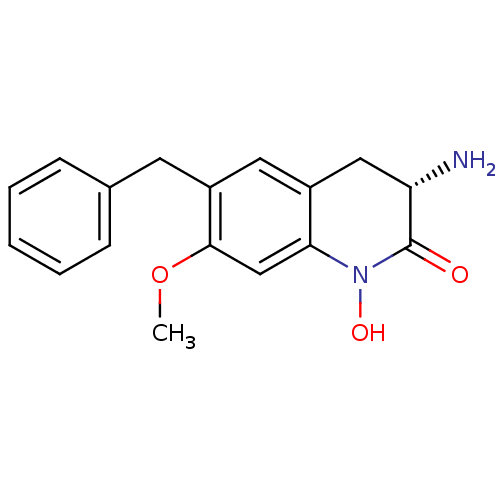
(CHEMBL2347110)Show SMILES COc1cc2N(O)C(=O)[C@@H](N)Cc2cc1Cc1ccccc1 |r| Show InChI InChI=1S/C17H18N2O3/c1-22-16-10-15-12(9-14(18)17(20)19(15)21)8-13(16)7-11-5-3-2-4-6-11/h2-6,8,10,14,21H,7,9,18H2,1H3/t14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50426340
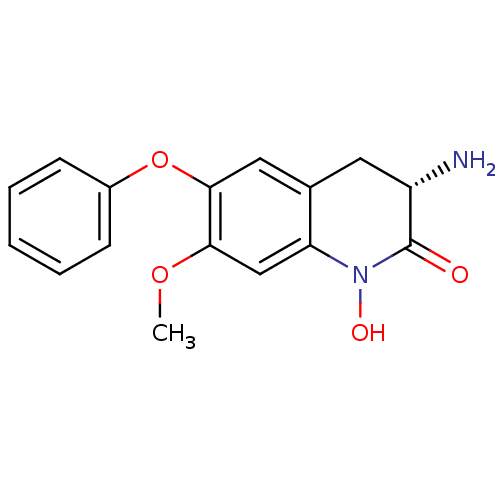
(CHEMBL2321943)Show SMILES COc1cc2N(O)C(=O)[C@@H](N)Cc2cc1Oc1ccccc1 |r| Show InChI InChI=1S/C16H16N2O4/c1-21-14-9-13-10(7-12(17)16(19)18(13)20)8-15(14)22-11-5-3-2-4-6-11/h2-6,8-9,12,20H,7,17H2,1H3/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Irreversible inhibition of human KAT2 using L-kynurenine as substrate measured every 5 mins over 16 hrs by SpectraMax plate reader analysis |
ACS Med Chem Lett 4: 37-40 (2013)
Article DOI: 10.1021/ml300237v
BindingDB Entry DOI: 10.7270/Q2KH0PNV |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107730
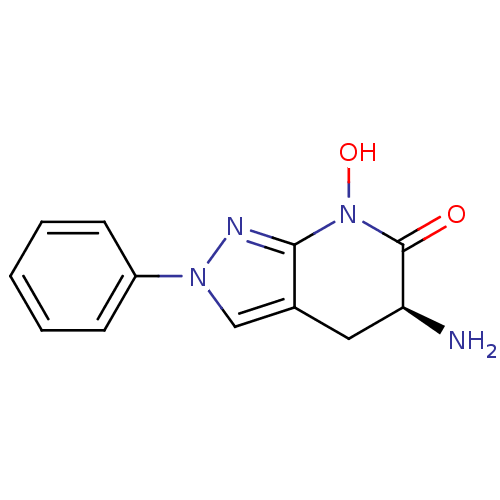
(CHEMBL2347108 | US8933095, 14)Show InChI InChI=1S/C12H12N4O2/c13-10-6-8-7-15(9-4-2-1-3-5-9)14-11(8)16(18)12(10)17/h1-5,7,10,18H,6,13H2/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50426341
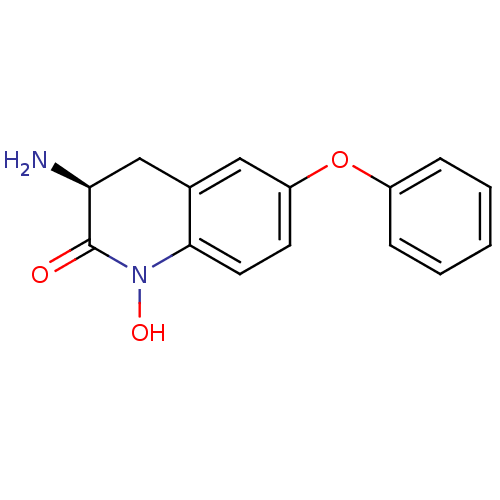
(CHEMBL2321944)Show InChI InChI=1S/C15H14N2O3/c16-13-9-10-8-12(20-11-4-2-1-3-5-11)6-7-14(10)17(19)15(13)18/h1-8,13,19H,9,16H2/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Irreversible inhibition of human KAT2 using L-kynurenine as substrate measured every 5 mins over 16 hrs by SpectraMax plate reader analysis |
ACS Med Chem Lett 4: 37-40 (2013)
Article DOI: 10.1021/ml300237v
BindingDB Entry DOI: 10.7270/Q2KH0PNV |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50386310
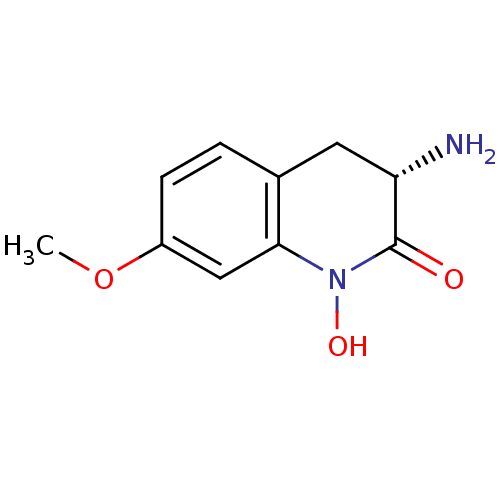
(CHEMBL2049092)Show InChI InChI=1S/C10H12N2O3/c1-15-7-3-2-6-4-8(11)10(13)12(14)9(6)5-7/h2-3,5,8,14H,4,11H2,1H3/t8-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Irreversible inhibition of human KAT2 using L-kynurenine as substrate measured every 5 mins over 16 hrs by SpectraMax plate reader analysis |
ACS Med Chem Lett 4: 37-40 (2013)
Article DOI: 10.1021/ml300237v
BindingDB Entry DOI: 10.7270/Q2KH0PNV |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107747
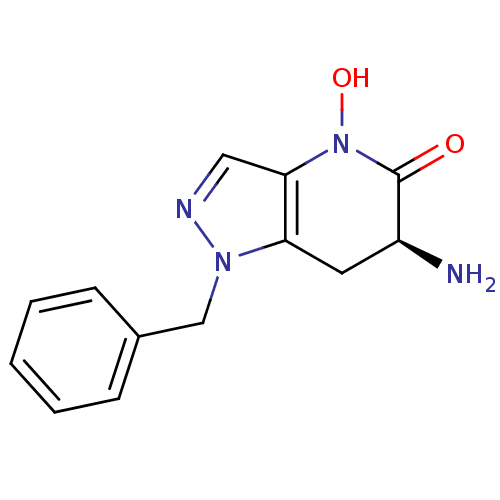
(CHEMBL2347115 | US8933095, 4)Show InChI InChI=1S/C13H14N4O2/c14-10-6-11-12(17(19)13(10)18)7-15-16(11)8-9-4-2-1-3-5-9/h1-5,7,10,19H,6,8,14H2/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50386292
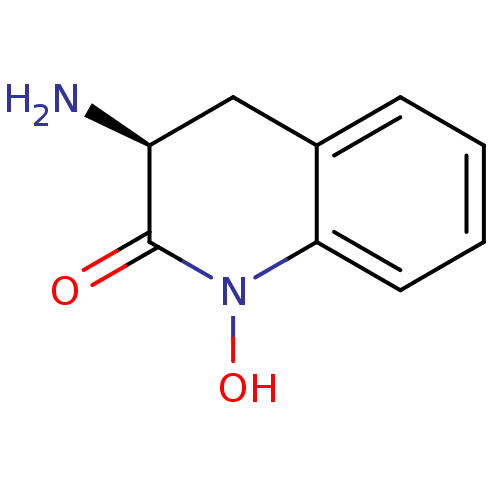
(CHEMBL2047851)Show InChI InChI=1S/C9H10N2O2/c10-7-5-6-3-1-2-4-8(6)11(13)9(7)12/h1-4,7,13H,5,10H2/t7-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM50386292

(CHEMBL2047851)Show InChI InChI=1S/C9H10N2O2/c10-7-5-6-3-1-2-4-8(6)11(13)9(7)12/h1-4,7,13H,5,10H2/t7-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Irreversible inhibition of human KAT2 using L-kynurenine as substrate measured every 5 mins over 16 hrs by SpectraMax plate reader analysis |
ACS Med Chem Lett 4: 37-40 (2013)
Article DOI: 10.1021/ml300237v
BindingDB Entry DOI: 10.7270/Q2KH0PNV |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107738
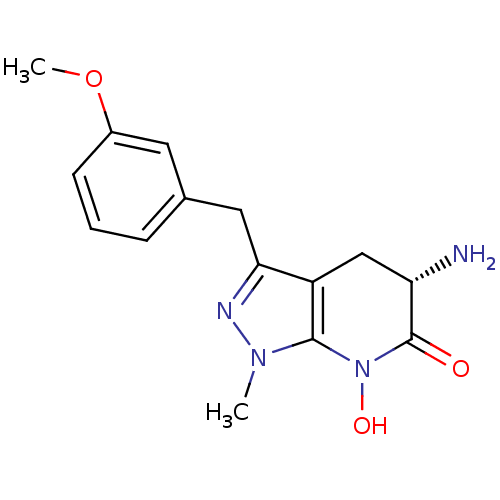
(CHEMBL2347113 | US8933095, 22)Show SMILES COc1cccc(Cc2nn(C)c3N(O)C(=O)[C@@H](N)Cc23)c1 |r| Show InChI InChI=1S/C15H18N4O3/c1-18-14-11(8-12(16)15(20)19(14)21)13(17-18)7-9-4-3-5-10(6-9)22-2/h3-6,12,21H,7-8,16H2,1-2H3/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107746
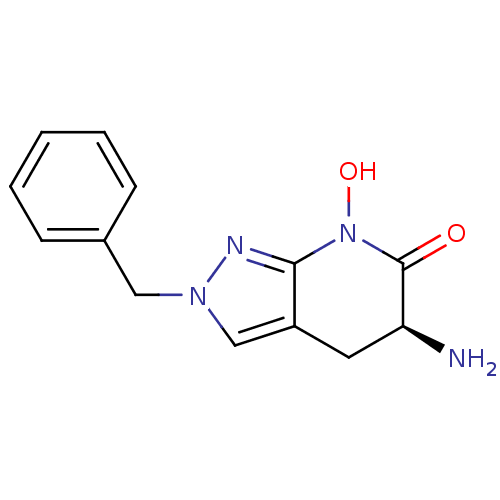
(CHEMBL2347107 | US8933095, 1)Show InChI InChI=1S/C13H14N4O2/c14-11-6-10-8-16(7-9-4-2-1-3-5-9)15-12(10)17(19)13(11)18/h1-5,8,11,19H,6-7,14H2/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107720
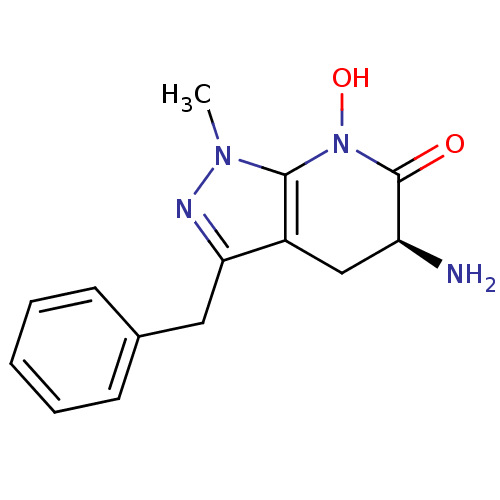
(CHEMBL2347112 | US8598200, 2)Show SMILES Cn1nc(Cc2ccccc2)c2C[C@H](N)C(=O)N(O)c12 |r| Show InChI InChI=1S/C14H16N4O2/c1-17-13-10(8-11(15)14(19)18(13)20)12(16-17)7-9-5-3-2-4-6-9/h2-6,11,20H,7-8,15H2,1H3/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
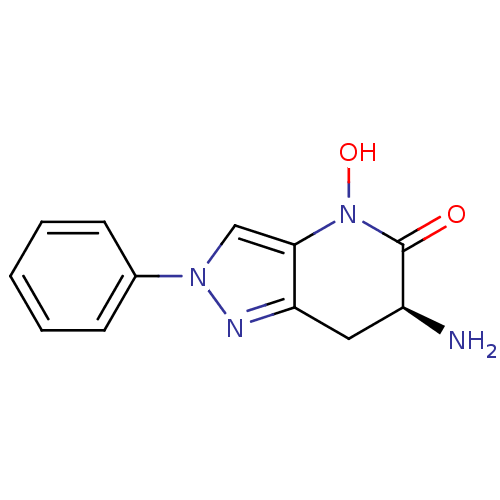
(Homo sapiens (Human)) | BDBM50432200
(CHEMBL2347109 | US8933095, 16)Show InChI InChI=1S/C12H12N4O2/c13-9-6-10-11(16(18)12(9)17)7-15(14-10)8-4-2-1-3-5-8/h1-5,7,9,18H,6,13H2/t9-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial
(Homo sapiens (Human)) | BDBM107722
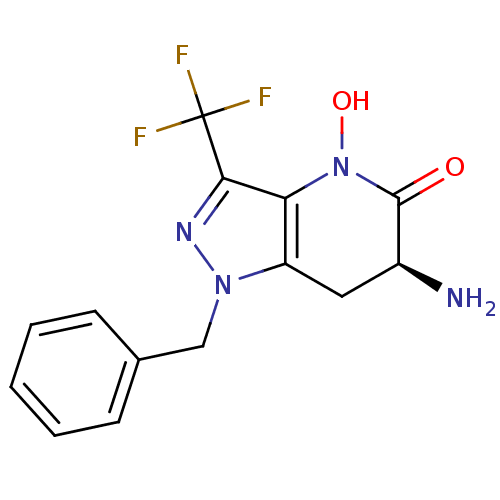
(CHEMBL2347114 | US8933095, 5)Show SMILES N[C@H]1Cc2c(N(O)C1=O)c(nn2Cc1ccccc1)C(F)(F)F |r| Show InChI InChI=1S/C14H13F3N4O2/c15-14(16,17)12-11-10(6-9(18)13(22)21(11)23)20(19-12)7-8-4-2-1-3-5-8/h1-5,9,23H,6-7,18H2/t9-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human KAT2 using L-kynurenine as substrate after 15 to 20 hrs by UV-visible spectra analysis |
Bioorg Med Chem Lett 23: 1961-6 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.039
BindingDB Entry DOI: 10.7270/Q2N87C48 |
More data for this
Ligand-Target Pair | |
High affinity choline transporter 1
(Homo sapiens (Human)) | BDBM50451447
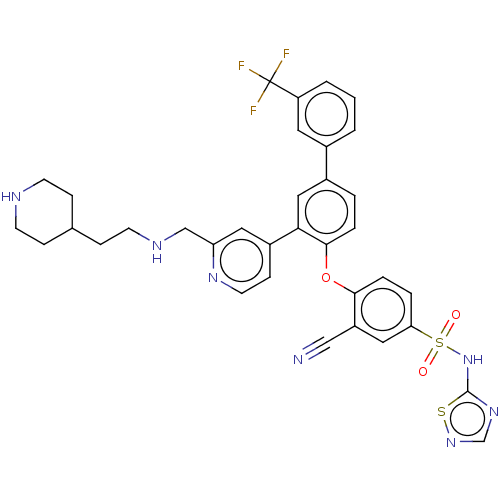
(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]hemicholinium-3 from recombinant human choline transporter after 60 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 1.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]Dofetilide from recombinant human ERG after 60 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium-dependent dopamine transporter
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 2.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]BTCP from recombinant human dopamine transporter after 120 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from recombinant human norepinephrine transporter after 120 min by scintillation counting method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451457

(CHEMBL4210135)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCN(CC2)C(=O)CCc2ccc(cc2)C(=O)c2ccc(OCCC#C)cc2)c1 Show InChI InChI=1S/C55H48F3N7O6S2/c1-2-3-29-70-47-15-12-40(13-16-47)53(67)39-10-7-37(8-11-39)9-20-52(66)65-27-23-38(24-28-65)21-25-60-35-46-31-43(22-26-61-46)49-33-42(41-5-4-6-45(30-41)55(56,57)58)14-18-51(49)71-50-19-17-48(32-44(50)34-59)73(68,69)64-54-62-36-63-72-54/h1,4-8,10-19,22,26,30-33,36,38,60H,3,9,20-21,23-25,27-29,35H2,(H,62,63,64) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451458
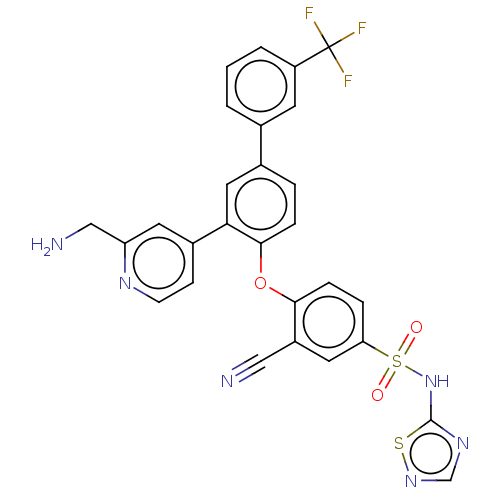
(CHEMBL4205773)Show SMILES NCc1cc(ccn1)-c1cc(ccc1Oc1ccc(cc1C#N)S(=O)(=O)Nc1ncns1)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C28H19F3N6O3S2/c29-28(30,31)21-3-1-2-17(10-21)18-4-6-26(24(13-18)19-8-9-34-22(11-19)15-33)40-25-7-5-23(12-20(25)14-32)42(38,39)37-27-35-16-36-41-27/h1-13,16H,15,33H2,(H,35,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Mus musculus) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of mouse Nav1.7 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451456
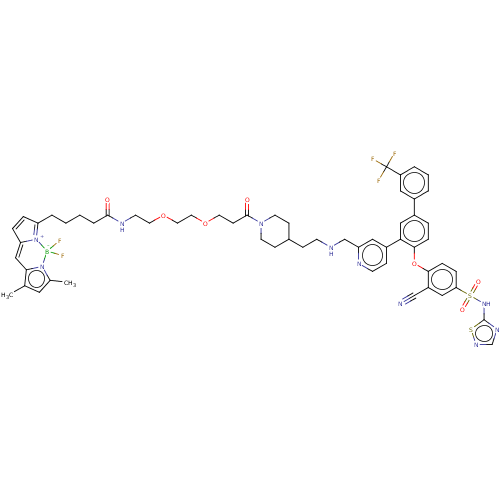
(CHEMBL4215058)Show SMILES Cc1cc(C)n2c1C=C1C=CC(CCCCC(=O)NCCOCCOCCC(=O)N3CCC(CCNCc4cc(ccn4)-c4cc(ccc4Oc4ccc(cc4C#N)S(=O)(=O)Nc4ncns4)-c4cccc(c4)C(F)(F)F)CC3)=[N+]1[B-]2(F)F |c:10,85,t:8| Show InChI InChI=1S/C58H62BF5N10O7S2/c1-39-30-40(2)73-52(39)35-49-12-11-48(74(49)59(73,63)64)8-3-4-9-55(75)68-23-27-80-29-28-79-26-20-56(76)72-24-18-41(19-25-72)16-21-66-37-47-32-44(17-22-67-47)51-34-43(42-6-5-7-46(31-42)58(60,61)62)10-14-54(51)81-53-15-13-50(33-45(53)36-65)83(77,78)71-57-69-38-70-82-57/h5-7,10-15,17,22,30-35,38,41,66H,3-4,8-9,16,18-21,23-29,37H2,1-2H3,(H,68,75)(H,69,70,71) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451458

(CHEMBL4205773)Show SMILES NCc1cc(ccn1)-c1cc(ccc1Oc1ccc(cc1C#N)S(=O)(=O)Nc1ncns1)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C28H19F3N6O3S2/c29-28(30,31)21-3-1-2-17(10-21)18-4-6-26(24(13-18)19-8-9-34-22(11-19)15-33)40-25-7-5-23(12-20(25)14-32)42(38,39)37-27-35-16-36-41-27/h1-13,16H,15,33H2,(H,35,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451454
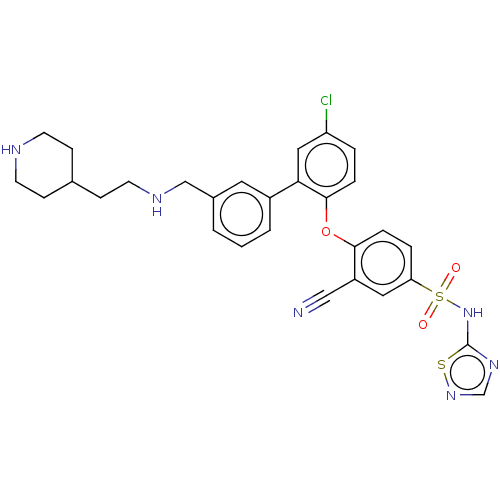
(CHEMBL4207534)Show SMILES Clc1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1cccc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C29H29ClN6O3S2/c30-24-4-6-28(39-27-7-5-25(15-23(27)17-31)41(37,38)36-29-34-19-35-40-29)26(16-24)22-3-1-2-21(14-22)18-33-13-10-20-8-11-32-12-9-20/h1-7,14-16,19-20,32-33H,8-13,18H2,(H,34,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
High affinity nerve growth factor receptor
(Homo sapiens (Human)) | BDBM50457826

(CHEMBL4216500)Show SMILES Cn1cnc(c1)-c1cnc(N)c(O[C@H]2CN(CC2(F)F)C(=O)Cc2ccc(OC(F)(F)F)cc2)c1 |r| Show InChI InChI=1S/C22H20F5N5O3/c1-31-9-16(30-12-31)14-7-17(20(28)29-8-14)34-18-10-32(11-21(18,23)24)19(33)6-13-2-4-15(5-3-13)35-22(25,26)27/h2-5,7-9,12,18H,6,10-11H2,1H3,(H2,28,29)/t18-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkA in human U2OS cells assessed as inhibition of beta-NGF-induced receptor phosphorylation by measuring reduc... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457839

(CHEMBL4211921)Show SMILES Cn1cc(cn1)-c1ccc(O[C@@H]2CCN(C2)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(n1)C(N)=O |r| Show InChI InChI=1S/C23H22F3N5O4/c1-30-12-15(11-28-30)18-6-7-19(21(29-18)22(27)33)34-17-8-9-31(13-17)20(32)10-14-2-4-16(5-3-14)35-23(24,25)26/h2-7,11-12,17H,8-10,13H2,1H3,(H2,27,33)/t17-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457833
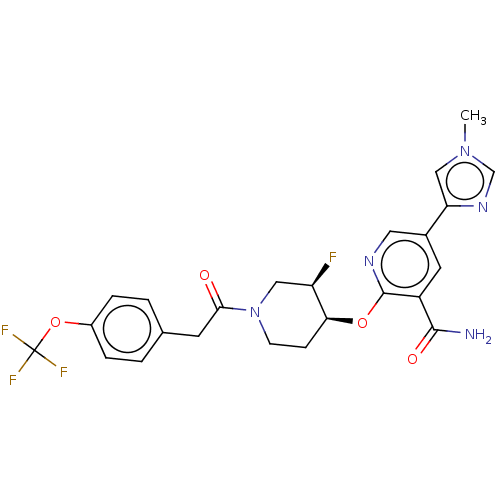
(CHEMBL4210892)Show SMILES Cn1cnc(c1)-c1cnc(O[C@H]2CCN(C[C@H]2F)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O |r| Show InChI InChI=1S/C24H23F4N5O4/c1-32-12-19(31-13-32)15-9-17(22(29)35)23(30-10-15)36-20-6-7-33(11-18(20)25)21(34)8-14-2-4-16(5-3-14)37-24(26,27)28/h2-5,9-10,12-13,18,20H,6-8,11H2,1H3,(H2,29,35)/t18-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451459
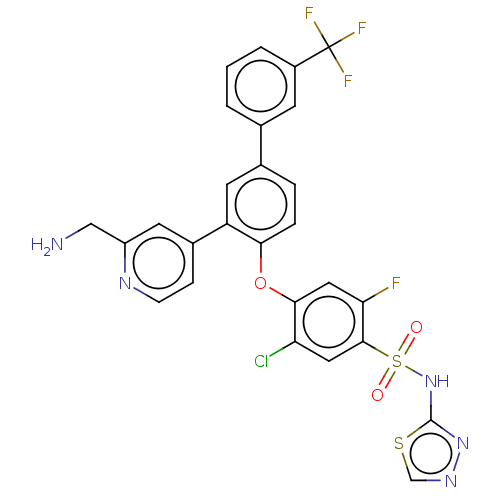
(CHEMBL4208664)Show SMILES NCc1cc(ccn1)-c1cc(ccc1Oc1cc(F)c(cc1Cl)S(=O)(=O)Nc1nncs1)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C27H18ClF4N5O3S2/c28-21-11-25(42(38,39)37-26-36-35-14-41-26)22(29)12-24(21)40-23-5-4-16(15-2-1-3-18(8-15)27(30,31)32)10-20(23)17-6-7-34-19(9-17)13-33/h1-12,14H,13,33H2,(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451453
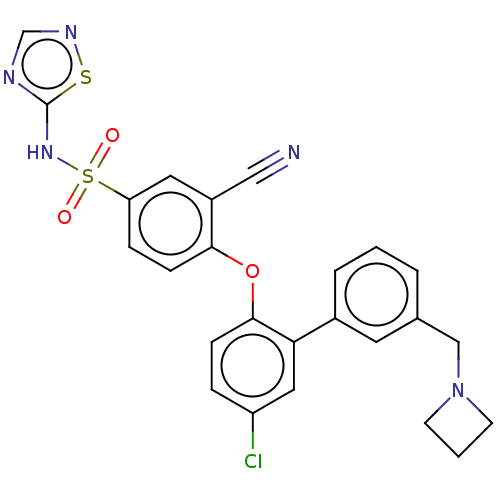
(CHEMBL2325603)Show SMILES Clc1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1cccc(CN2CCC2)c1 Show InChI InChI=1S/C25H20ClN5O3S2/c26-20-5-7-24(22(13-20)18-4-1-3-17(11-18)15-31-9-2-10-31)34-23-8-6-21(12-19(23)14-27)36(32,33)30-25-28-16-29-35-25/h1,3-8,11-13,16H,2,9-10,15H2,(H,28,29,30) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50257167
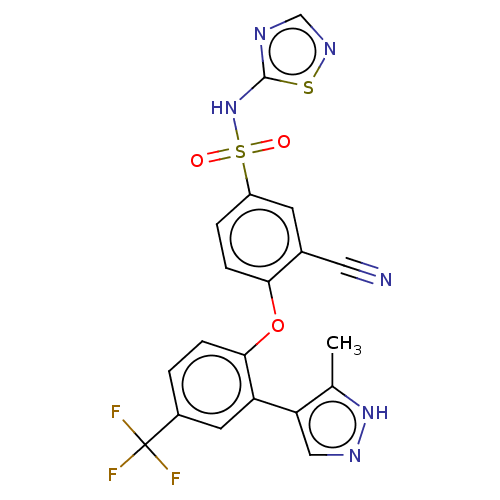
(CHEMBL2325619)Show SMILES Cc1[nH]ncc1-c1cc(ccc1Oc1ccc(cc1C#N)S(=O)(=O)Nc1ncns1)C(F)(F)F Show InChI InChI=1S/C20H13F3N6O3S2/c1-11-16(9-26-28-11)15-7-13(20(21,22)23)2-4-18(15)32-17-5-3-14(6-12(17)8-24)34(30,31)29-19-25-10-27-33-19/h2-7,9-10H,1H3,(H,26,28)(H,25,27,29) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Icagen Inc. , 4222 Emperor Blvd no. 350, Durham, North Carolina 27703, United States.
Curated by ChEMBL
| Assay Description
Inhibition of human NaV1.7 expressed in HEK cells assessed as half inactivation potential at -120 mV holding potential by automated patch clamp metho... |
J Med Chem 60: 7029-7042 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00598
BindingDB Entry DOI: 10.7270/Q21G0PQF |
More data for this
Ligand-Target Pair | |
Free fatty acid receptor 1
(Homo sapiens (Human)) | BDBM50277775
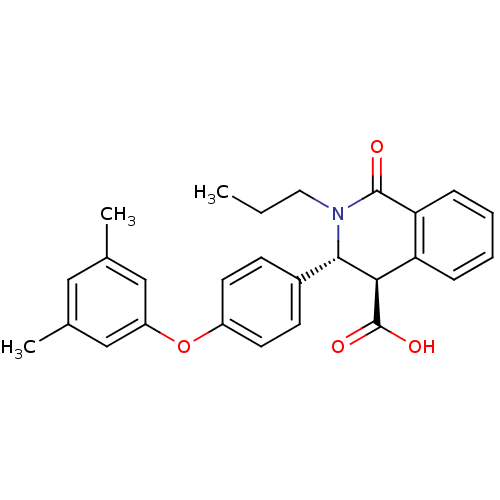
((3R,4R)-3-(4-(3,5-dimethylphenoxy)phenyl)-1-oxo-2-...)Show SMILES CCCN1[C@H]([C@H](C(O)=O)c2ccccc2C1=O)c1ccc(Oc2cc(C)cc(C)c2)cc1 |r| Show InChI InChI=1S/C27H27NO4/c1-4-13-28-25(24(27(30)31)22-7-5-6-8-23(22)26(28)29)19-9-11-20(12-10-19)32-21-15-17(2)14-18(3)16-21/h5-12,14-16,24-25H,4,13H2,1-3H3,(H,30,31)/t24-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global R&D
Curated by ChEMBL
| Assay Description
Antagonist activity at human GPR40 expressed HEK293 cells assessed as effect on intracellular calcium concentration by FLIPR assay |
Bioorg Med Chem Lett 19: 2400-3 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.082
BindingDB Entry DOI: 10.7270/Q23N238P |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457825
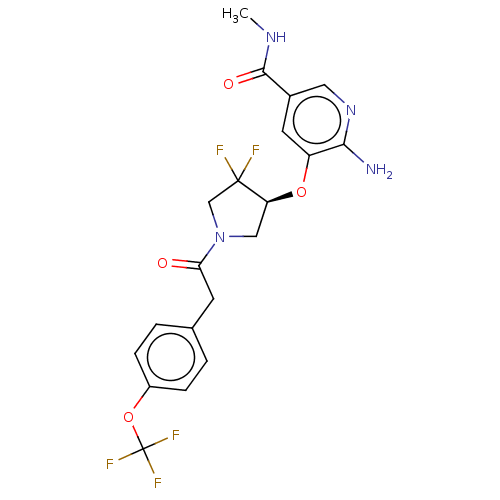
(CHEMBL4207042)Show SMILES CNC(=O)c1cnc(N)c(O[C@H]2CN(CC2(F)F)C(=O)Cc2ccc(OC(F)(F)F)cc2)c1 |r| Show InChI InChI=1S/C20H19F5N4O4/c1-27-18(31)12-7-14(17(26)28-8-12)32-15-9-29(10-19(15,21)22)16(30)6-11-2-4-13(5-3-11)33-20(23,24)25/h2-5,7-8,15H,6,9-10H2,1H3,(H2,26,28)(H,27,31)/t15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457837

(CHEMBL4215846)Show SMILES Cn1cnc(c1)-c1ccc(OC2CCN(CC2)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O Show InChI InChI=1S/C25H25F3N4O4/c1-31-14-21(30-15-31)17-4-7-22(20(13-17)24(29)34)35-18-8-10-32(11-9-18)23(33)12-16-2-5-19(6-3-16)36-25(26,27)28/h2-7,13-15,18H,8-12H2,1H3,(H2,29,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 2 subunit alpha
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.2 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50457833

(CHEMBL4210892)Show SMILES Cn1cnc(c1)-c1cnc(O[C@H]2CCN(C[C@H]2F)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O |r| Show InChI InChI=1S/C24H23F4N5O4/c1-32-12-19(31-13-32)15-9-17(22(29)35)23(30-10-15)36-20-6-7-33(11-18(20)25)21(34)8-14-2-4-16(5-3-14)37-24(26,27)28/h2-5,9-10,12-13,18,20H,6-8,11H2,1H3,(H2,29,35)/t18-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkB in human U2OS cells assessed as inhibition of BDNF-induced receptor phosphorylation by measuring reduction... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50457839

(CHEMBL4211921)Show SMILES Cn1cc(cn1)-c1ccc(O[C@@H]2CCN(C2)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(n1)C(N)=O |r| Show InChI InChI=1S/C23H22F3N5O4/c1-30-12-15(11-28-30)18-6-7-19(21(29-18)22(27)33)34-17-8-9-31(13-17)20(32)10-14-2-4-16(5-3-14)35-23(24,25)26/h2-7,11-12,17H,8-10,13H2,1H3,(H2,27,33)/t17-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkB in human U2OS cells assessed as inhibition of BDNF-induced receptor phosphorylation by measuring reduction... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50257179
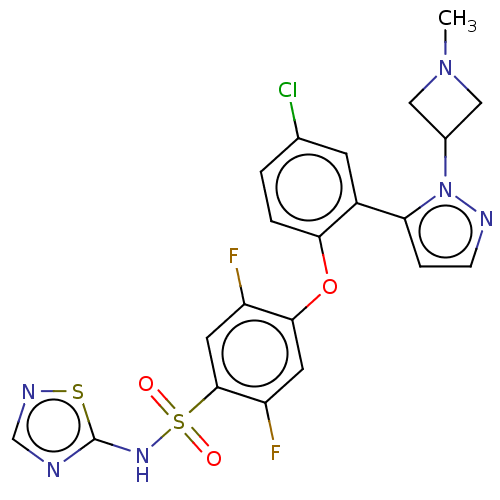
(CHEMBL2325622)Show SMILES CN1CC(C1)n1nccc1-c1cc(Cl)ccc1Oc1cc(F)c(cc1F)S(=O)(=O)Nc1ncns1 Show InChI InChI=1S/C21H17ClF2N6O3S2/c1-29-9-13(10-29)30-17(4-5-26-30)14-6-12(22)2-3-18(14)33-19-7-16(24)20(8-15(19)23)35(31,32)28-21-25-11-27-34-21/h2-8,11,13H,9-10H2,1H3,(H,25,27,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Icagen Inc. , 4222 Emperor Blvd no. 350, Durham, North Carolina 27703, United States.
Curated by ChEMBL
| Assay Description
Inhibition of human NaV1.7 expressed in HEK cells assessed as half inactivation potential at -120 mV holding potential by automated patch clamp metho... |
J Med Chem 60: 7029-7042 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00598
BindingDB Entry DOI: 10.7270/Q21G0PQF |
More data for this
Ligand-Target Pair | |
Free fatty acid receptor 1
(Homo sapiens (Human)) | BDBM50277776
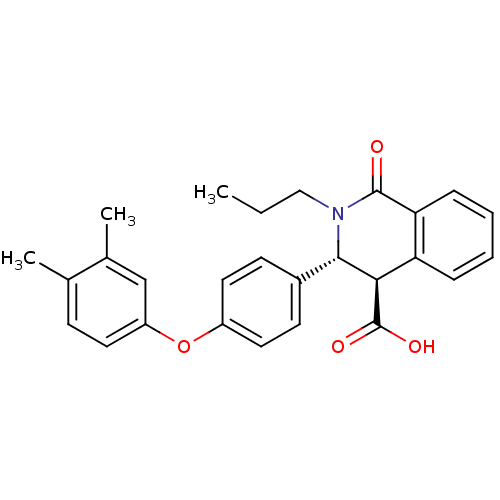
((3R,4R)-3-(4-(3,4-dimethylphenoxy)phenyl)-1-oxo-2-...)Show SMILES CCCN1[C@H]([C@H](C(O)=O)c2ccccc2C1=O)c1ccc(Oc2ccc(C)c(C)c2)cc1 |r| Show InChI InChI=1S/C27H27NO4/c1-4-15-28-25(24(27(30)31)22-7-5-6-8-23(22)26(28)29)19-10-13-20(14-11-19)32-21-12-9-17(2)18(3)16-21/h5-14,16,24-25H,4,15H2,1-3H3,(H,30,31)/t24-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global R&D
Curated by ChEMBL
| Assay Description
Antagonist activity at human GPR40 expressed HEK293 cells assessed as effect on intracellular calcium concentration by FLIPR assay |
Bioorg Med Chem Lett 19: 2400-3 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.082
BindingDB Entry DOI: 10.7270/Q23N238P |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457827
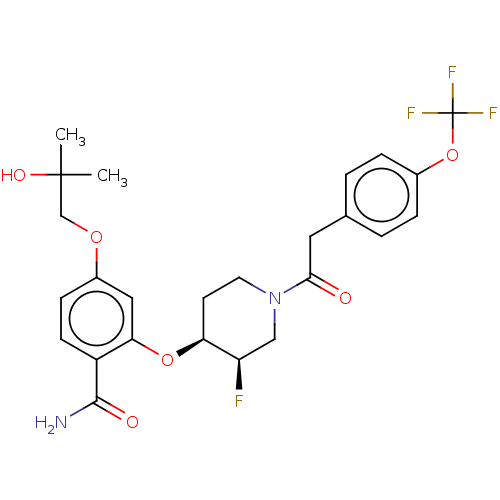
(CHEMBL4203759)Show SMILES CC(C)(O)COc1ccc(C(N)=O)c(O[C@H]2CCN(C[C@H]2F)C(=O)Cc2ccc(OC(F)(F)F)cc2)c1 |r| Show InChI InChI=1S/C25H28F4N2O6/c1-24(2,34)14-35-17-7-8-18(23(30)33)21(12-17)36-20-9-10-31(13-19(20)26)22(32)11-15-3-5-16(6-4-15)37-25(27,28)29/h3-8,12,19-20,34H,9-11,13-14H2,1-2H3,(H2,30,33)/t19-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451448
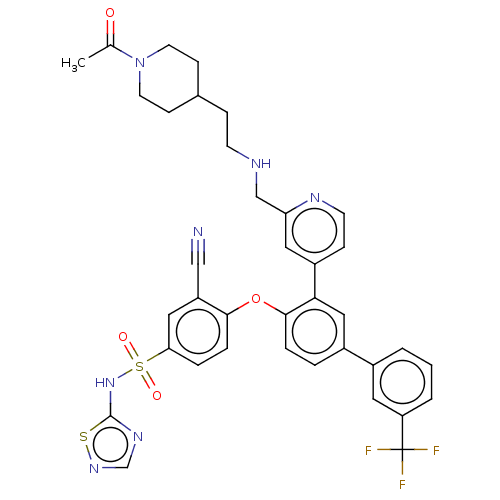
(CHEMBL4213464)Show SMILES CC(=O)N1CCC(CCNCc2cc(ccn2)-c2cc(ccc2Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)-c2cccc(c2)C(F)(F)F)CC1 Show InChI InChI=1S/C37H34F3N7O4S2/c1-24(48)47-15-11-25(12-16-47)9-13-42-22-31-18-28(10-14-43-31)33-20-27(26-3-2-4-30(17-26)37(38,39)40)5-7-35(33)51-34-8-6-32(19-29(34)21-41)53(49,50)46-36-44-23-45-52-36/h2-8,10,14,17-20,23,25,42H,9,11-13,15-16,22H2,1H3,(H,44,45,46) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50257166
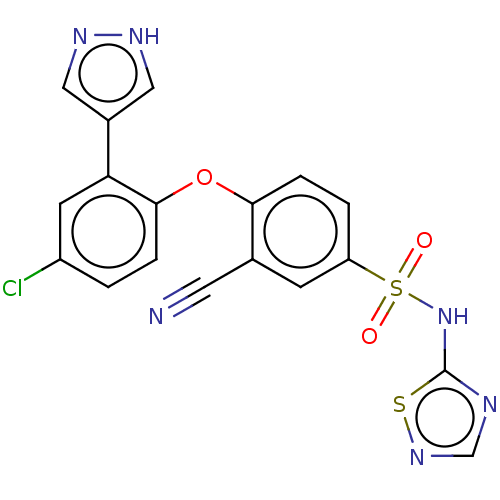
(CHEMBL2325330)Show SMILES Clc1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1cn[nH]c1 Show InChI InChI=1S/C18H11ClN6O3S2/c19-13-1-3-17(15(6-13)12-8-22-23-9-12)28-16-4-2-14(5-11(16)7-20)30(26,27)25-18-21-10-24-29-18/h1-6,8-10H,(H,22,23)(H,21,24,25) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Icagen Inc. , 4222 Emperor Blvd no. 350, Durham, North Carolina 27703, United States.
Curated by ChEMBL
| Assay Description
Inhibition of human NaV1.7 expressed in HEK cells assessed as half inactivation potential at -120 mV holding potential by automated patch clamp metho... |
J Med Chem 60: 7029-7042 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00598
BindingDB Entry DOI: 10.7270/Q21G0PQF |
More data for this
Ligand-Target Pair | |
Free fatty acid receptor 1
(Homo sapiens (Human)) | BDBM50277814
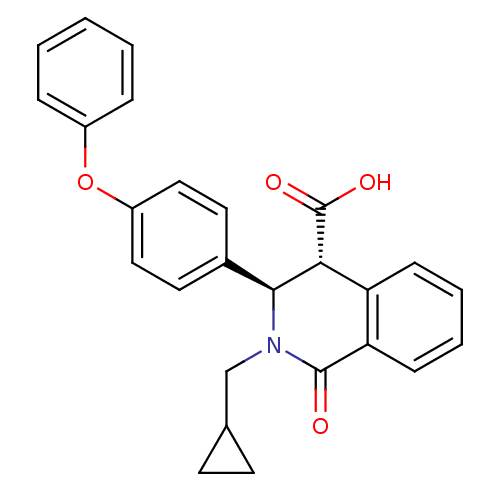
((3R,4R)-2-(cyclopropylmethyl)-1-oxo-3-(4-phenoxyph...)Show SMILES OC(=O)[C@H]1[C@@H](N(CC2CC2)C(=O)c2ccccc12)c1ccc(Oc2ccccc2)cc1 |r| Show InChI InChI=1S/C26H23NO4/c28-25-22-9-5-4-8-21(22)23(26(29)30)24(27(25)16-17-10-11-17)18-12-14-20(15-13-18)31-19-6-2-1-3-7-19/h1-9,12-15,17,23-24H,10-11,16H2,(H,29,30)/t23-,24+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global R&D
Curated by ChEMBL
| Assay Description
Antagonist activity at human GPR40 expressed HEK293 cells assessed as effect on intracellular calcium concentration by FLIPR assay |
Bioorg Med Chem Lett 19: 2400-3 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.082
BindingDB Entry DOI: 10.7270/Q23N238P |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50457824

(CHEMBL4204174)Show SMILES Cn1cc(cn1)-c1ccc(OC2CCN(CC2)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O Show InChI InChI=1S/C25H25F3N4O4/c1-31-15-18(14-30-31)17-4-7-22(21(13-17)24(29)34)35-19-8-10-32(11-9-19)23(33)12-16-2-5-20(6-3-16)36-25(26,27)28/h2-7,13-15,19H,8-12H2,1H3,(H2,29,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkB in human U2OS cells assessed as inhibition of BDNF-induced receptor phosphorylation by measuring reduction... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50451460

(CHEMBL4215917)Show SMILES Fc1cc(Oc2ccc(Cl)cc2-c2ccnc(CN3CCC3)c2)c(Cl)cc1S(=O)(=O)Nc1nncs1 Show InChI InChI=1S/C23H18Cl2FN5O3S2/c24-15-2-3-20(17(9-15)14-4-5-27-16(8-14)12-31-6-1-7-31)34-21-11-19(26)22(10-18(21)25)36(32,33)30-23-29-28-13-35-23/h2-5,8-11,13H,1,6-7,12H2,(H,29,30) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.7 expressed in HEK cells by automated patchXpress electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
NT-3 growth factor receptor
(Homo sapiens (Human)) | BDBM50457828

(CHEMBL4218103)Show SMILES Cn1cc(cn1)-c1ccc(OC2CCN(CC2)C(=O)Nc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O Show InChI InChI=1S/C24H24F3N5O4/c1-31-14-16(13-29-31)15-2-7-21(20(12-15)22(28)33)35-18-8-10-32(11-9-18)23(34)30-17-3-5-19(6-4-17)36-24(25,26)27/h2-7,12-14,18H,8-11H2,1H3,(H2,28,33)(H,30,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkC in human U2OS cells assessed as inhibition of NT3-induced receptor phosphorylation by measuring reduction ... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 9 subunit alpha
(Homo sapiens (Human)) | BDBM50257216
(CHEMBL2325350)Show SMILES Cn1nccc1-c1cc(Cl)ccc1Oc1cc(F)c(cc1F)S(=O)(=O)Nc1ncns1 Show InChI InChI=1S/C18H12ClF2N5O3S2/c1-26-14(4-5-23-26)11-6-10(19)2-3-15(11)29-16-7-13(21)17(8-12(16)20)31(27,28)25-18-22-9-24-30-18/h2-9H,1H3,(H,22,24,25) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Icagen Inc. , 4222 Emperor Blvd no. 350, Durham, North Carolina 27703, United States.
Curated by ChEMBL
| Assay Description
Inhibition of human NaV1.7 expressed in HEK cells assessed as half inactivation potential at -120 mV holding potential by automated patch clamp metho... |
J Med Chem 60: 7029-7042 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00598
BindingDB Entry DOI: 10.7270/Q21G0PQF |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 8 subunit alpha
(Rattus norvegicus) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of rat Nav1.6 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 8 subunit alpha
(Homo sapiens (Human)) | BDBM50451447

(CHEMBL4217988)Show SMILES FC(F)(F)c1cccc(c1)-c1ccc(Oc2ccc(cc2C#N)S(=O)(=O)Nc2ncns2)c(c1)-c1ccnc(CNCCC2CCNCC2)c1 Show InChI InChI=1S/C35H32F3N7O3S2/c36-35(37,38)28-3-1-2-24(16-28)25-4-6-33(48-32-7-5-30(18-27(32)20-39)50(46,47)45-34-43-22-44-49-34)31(19-25)26-11-15-42-29(17-26)21-41-14-10-23-8-12-40-13-9-23/h1-7,11,15-19,22-23,40-41H,8-10,12-14,21H2,(H,43,44,45) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human Nav1.6 expressed in HEK cells by patch clamp electrophysiology method |
Bioorg Med Chem Lett 27: 4805-4811 (2017)
Article DOI: 10.1016/j.bmcl.2017.09.056
BindingDB Entry DOI: 10.7270/Q25Q4ZN1 |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50457837

(CHEMBL4215846)Show SMILES Cn1cnc(c1)-c1ccc(OC2CCN(CC2)C(=O)Cc2ccc(OC(F)(F)F)cc2)c(c1)C(N)=O Show InChI InChI=1S/C25H25F3N4O4/c1-31-14-21(30-15-31)17-4-7-22(20(13-17)24(29)34)35-18-8-10-32(11-9-18)23(33)12-16-2-5-19(6-3-16)36-25(26,27)28/h2-7,13-15,18H,8-12H2,1H3,(H2,29,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at prolink-tagged TrkB in human U2OS cells assessed as inhibition of BDNF-induced receptor phosphorylation by measuring reduction... |
J Med Chem 61: 6779-6800 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00633
BindingDB Entry DOI: 10.7270/Q2K64MPW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data