

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50007674 ((+)-erythro 4-[2-(4-Benzyl-piperidin-1-yl)-1-hydro...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398926 (CHEMBL2178786) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398918 (CHEMBL2178778) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398928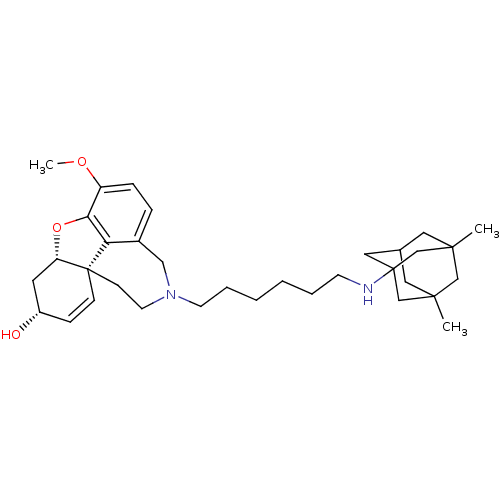 (CHEMBL2178784) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398924 (CHEMBL2178788) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398915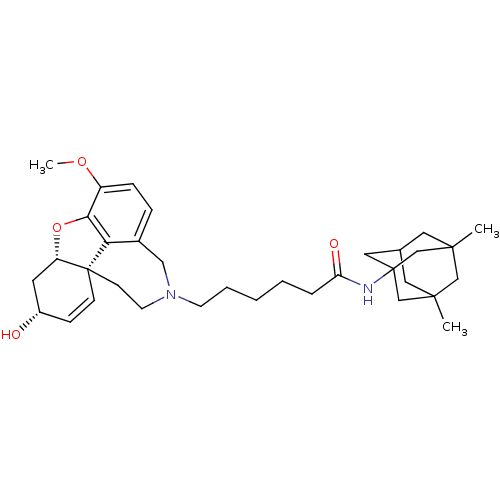 (CHEMBL2178781) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.76E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398927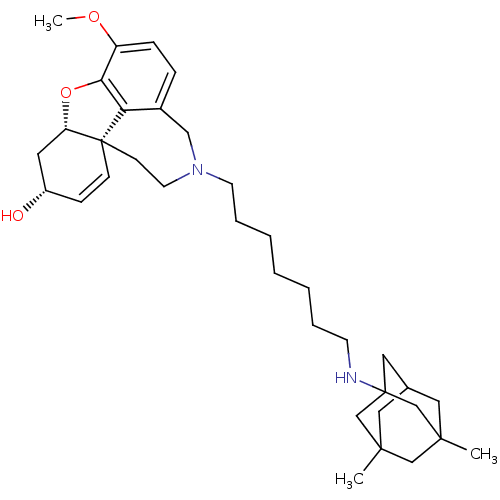 (CHEMBL2178785) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398929 (CHEMBL2178783) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.09E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398922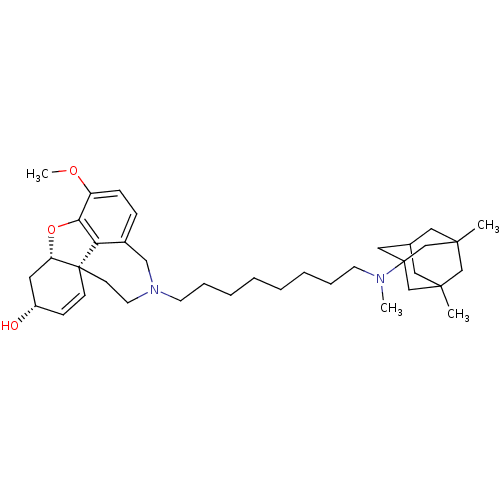 (CHEMBL2178790) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398923 (CHEMBL2178789) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398917 (CHEMBL2178779) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398925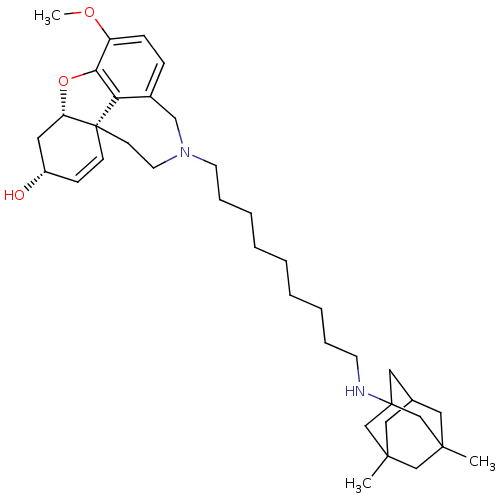 (CHEMBL2178787) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398916 (CHEMBL2178780) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398919 (CHEMBL2178777) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398920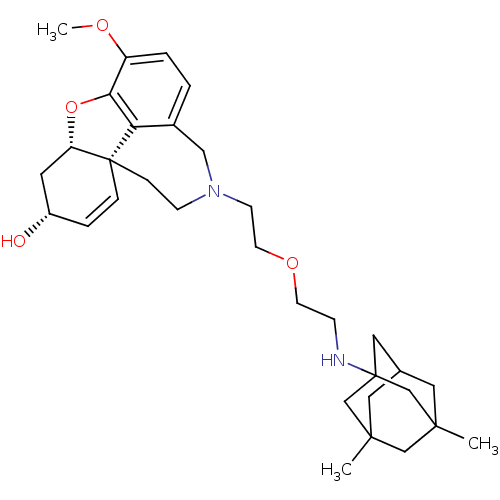 (CHEMBL2178776) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398921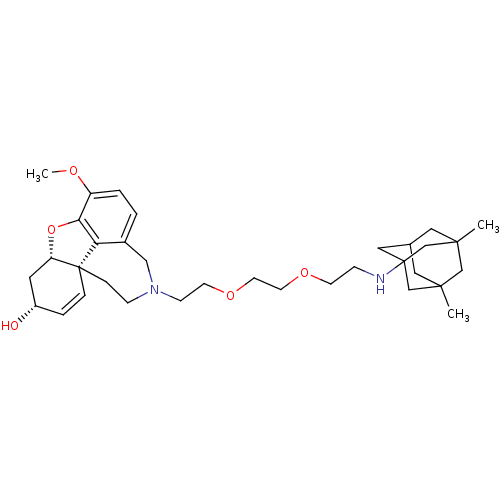 (CHEMBL2178791) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50398930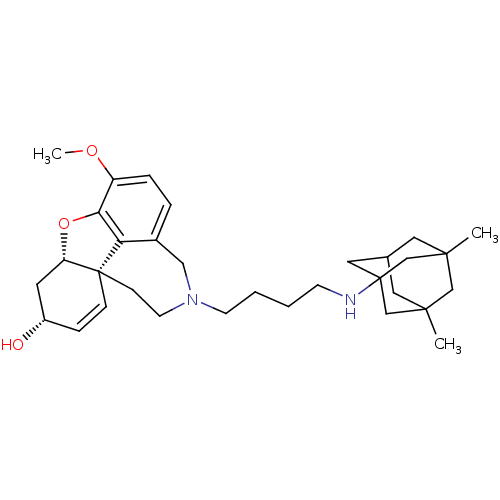 (CHEMBL2178782) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50062599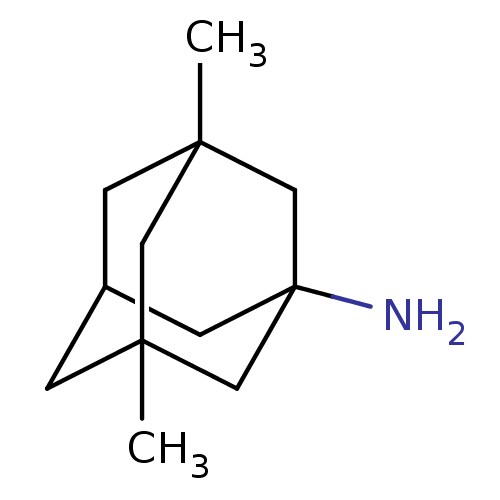 (3,5-Dimethyl-adamantan-1-ylamine | CHEMBL807 | EN3...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM10404 ((1S,12S,14R)-9-methoxy-4-methyl-11-oxa-4-azatetrac...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate receptor ionotropic, NMDA 2B (Rattus norvegicus (Rat)) | BDBM50030386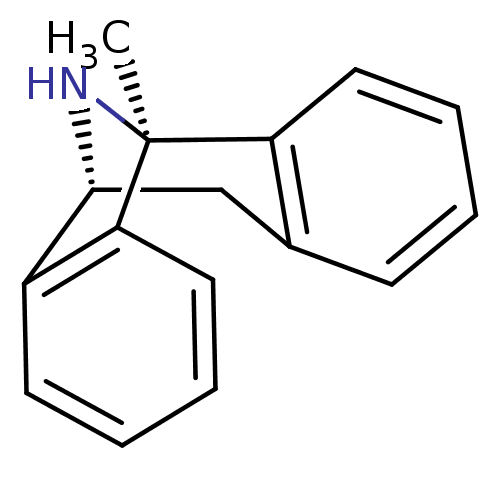 ((1S)-1-methyl-16-azatetracyclo[7.6.1.0^{2,7}.0^{10...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | PDB Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Displacement of [3H]Ifenprodil from NMDAR-2B in Sprague-Dawley rat frontal cortex homogenates after 2 hrs by liquid scintillation counting | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acid ceramidase (Homo sapiens (Human)) | BDBM29080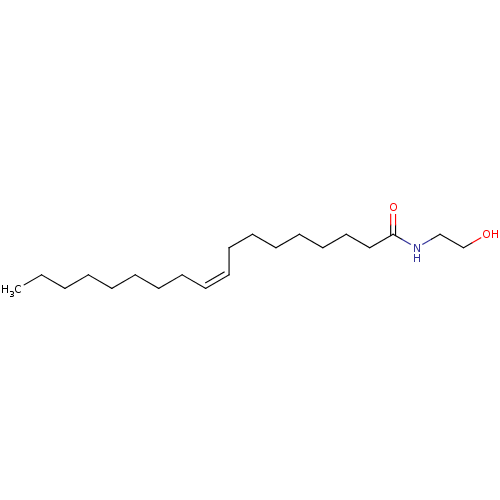 (CHEMBL280065 | N-oleoylethanolamine | Oleamide MEA...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fondazione Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of acid ceramidase (unknown origin) | J Med Chem 56: 3518-30 (2013) Article DOI: 10.1021/jm301879g BindingDB Entry DOI: 10.7270/Q27D2WH5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236331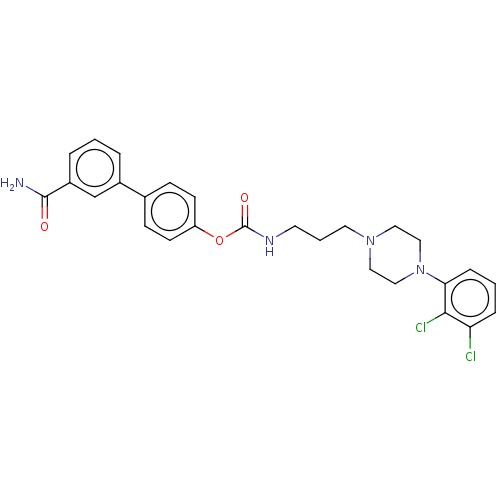 (CHEMBL4091498) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398922 (CHEMBL2178790) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.520 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236343 (CHEMBL4086944) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236330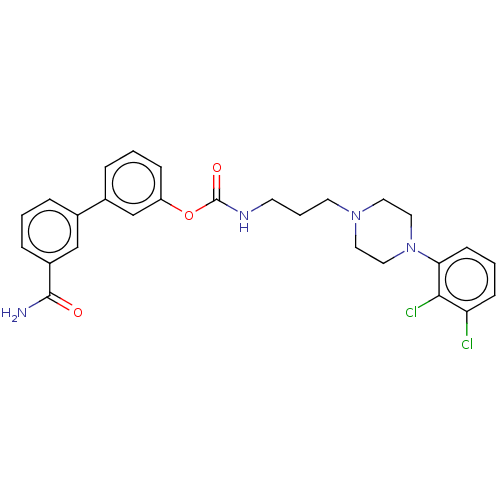 (CHEMBL4070196) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236333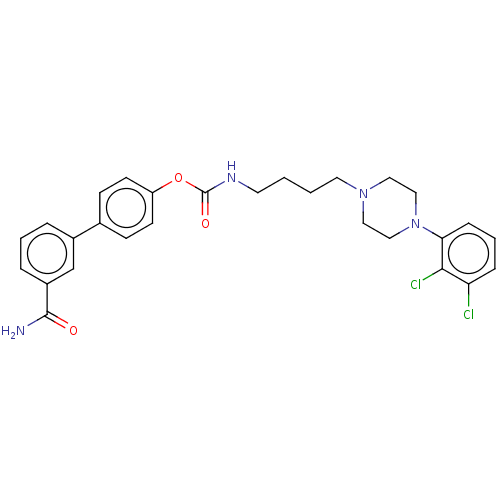 (CHEMBL4092052) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236345 (CHEMBL4079093) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236340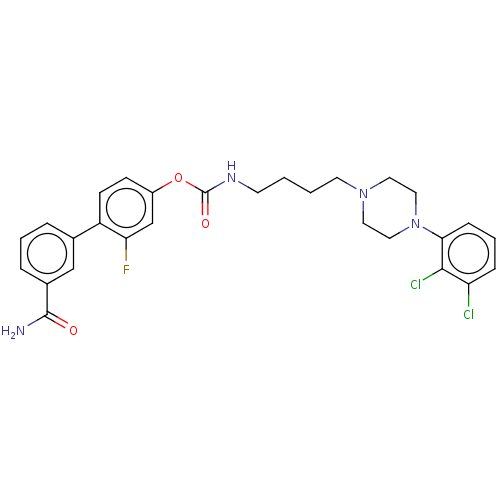 (CHEMBL4069565) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398924 (CHEMBL2178788) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.03 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236326 (CHEMBL4073966) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50245875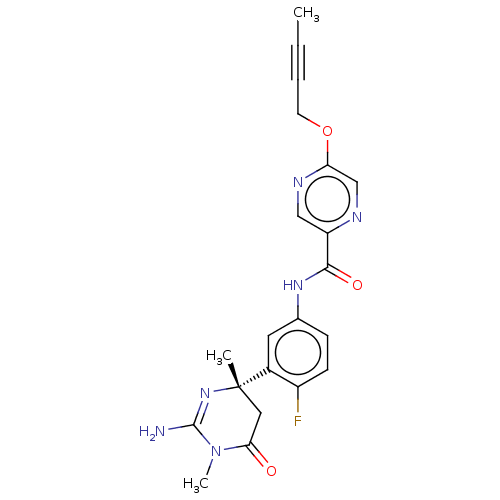 (CHEMBL4086961) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Dundee Curated by ChEMBL | Assay Description Inhibition of BACE-1 (unknown origin) using APP derived polypeptide harboring Lys-Met/Asn-Leu mutation as substrate by FRET assay | J Med Chem 61: 619-637 (2018) Article DOI: 10.1021/acs.jmedchem.7b00393 BindingDB Entry DOI: 10.7270/Q2MP55P1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398928 (CHEMBL2178784) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.16 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236342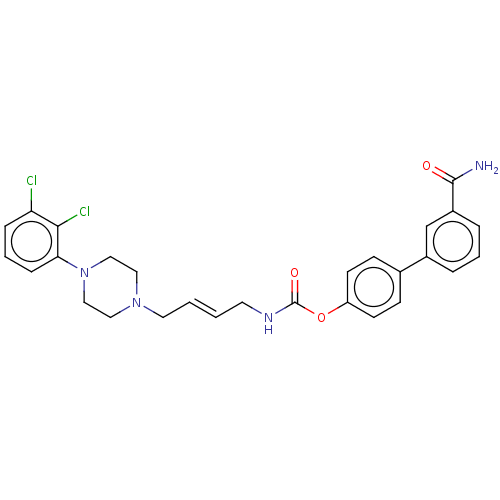 (CHEMBL4065510) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236327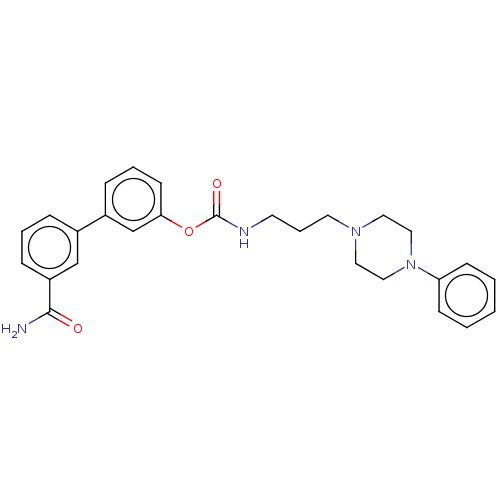 (CHEMBL4100735) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Binding affinity against human Alpha-1a adrenergic receptor | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398923 (CHEMBL2178789) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.33 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50231951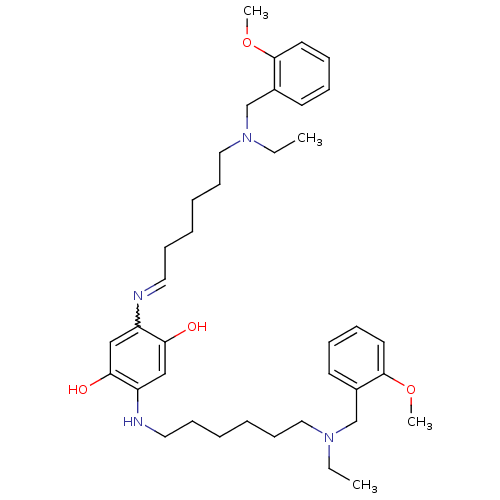 (2,5-bis(6-((2-methoxybenzyl)(ethyl)amino)hexylamin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna Curated by ChEMBL | Assay Description Inhibition of human recombinant AChE using acetylthiocholine iodide as substrate after 20 mins preincubation by Ellman method | J Med Chem 54: 8299-304 (2011) Article DOI: 10.1021/jm200691d BindingDB Entry DOI: 10.7270/Q2V40VNM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236334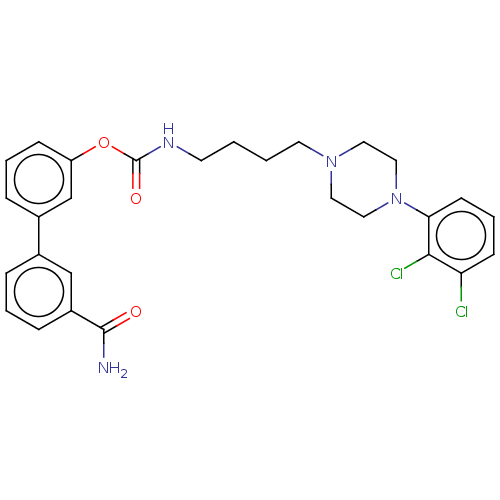 (CHEMBL4071240) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398927 (CHEMBL2178785) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.79 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236329 (CHEMBL4081780) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236328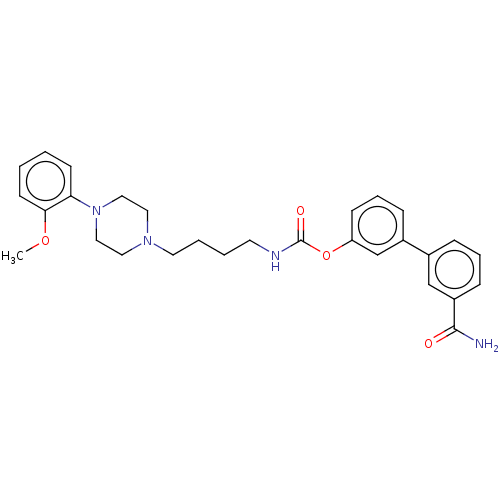 (CHEMBL4099798) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398925 (CHEMBL2178787) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.32 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236356 (CHEMBL4095588) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236357 (CHEMBL4078351) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236353 (CHEMBL4060896) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236332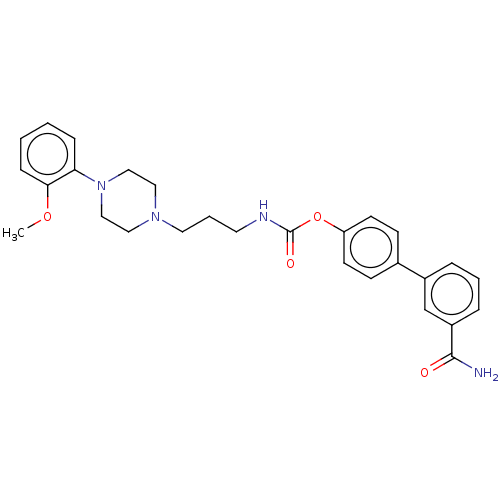 (CHEMBL4072282) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236339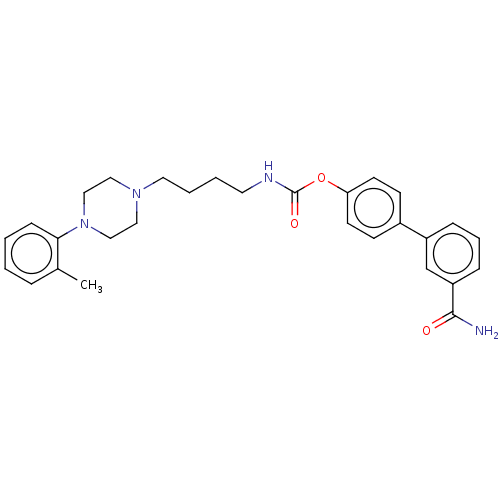 (CHEMBL4094794) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236344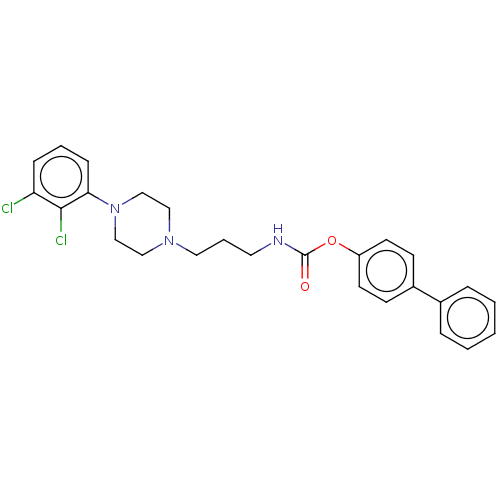 (CHEMBL4103339) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236352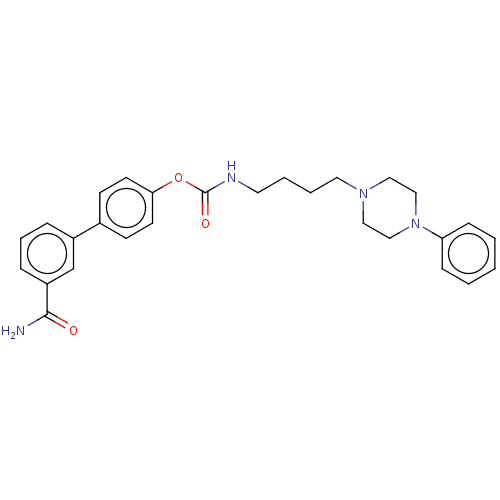 (CHEMBL4090581) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acid ceramidase (Rattus norvegicus (Rat)) | BDBM50431244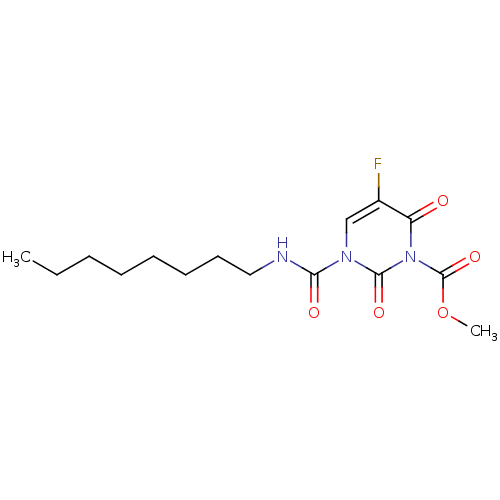 (CHEMBL2333064) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fondazione Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of rat recombinant acid ceramidase expressed in human HEK293 cells using N-lauroylceramide as substrate incubated for 30 mins prior to sub... | J Med Chem 56: 3518-30 (2013) Article DOI: 10.1021/jm301879g BindingDB Entry DOI: 10.7270/Q27D2WH5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50236358 (CHEMBL4063681) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Bologna Curated by ChEMBL | Assay Description Inhibition of human FAAH1 expressed in HEK293 cell membrane-enriched lysate using AMC arachidonyl amide as substrate preincubated for 50 mins followe... | J Med Chem 60: 2287-2304 (2017) Article DOI: 10.1021/acs.jmedchem.6b01578 BindingDB Entry DOI: 10.7270/Q29S1T9P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 449 total ) | Next | Last >> |