

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556952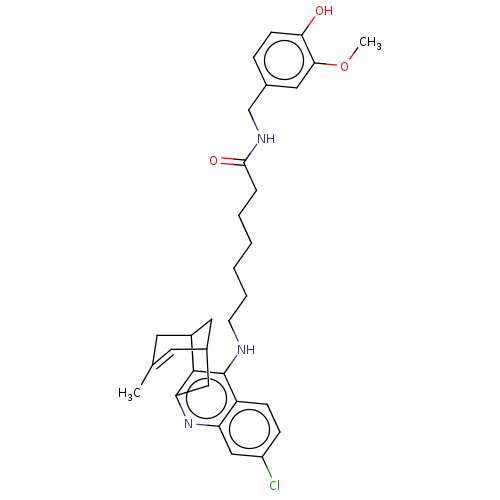 (CHEMBL4754487) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Mixed-type inhibition of recombinant human AChE assessed as inhibition constant using acetylthiocholine iodide as substrate by Cornish-Bowden plot an... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556952 (CHEMBL4754487) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Mixed-type inhibition of recombinant human AChE assessed as dissociation constant for protein-substrate-compound complex using acetylthiocholine iodi... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271142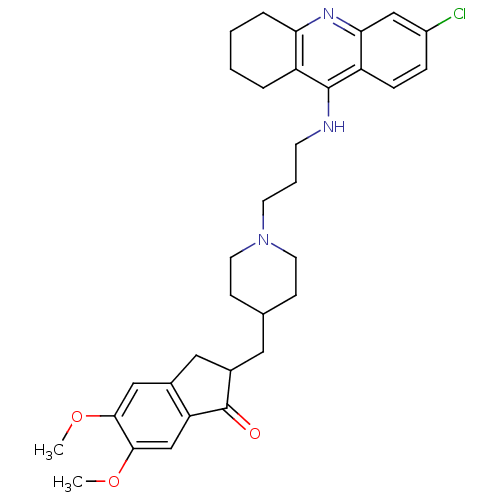 (6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271142 (6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271140 (9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]pi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271141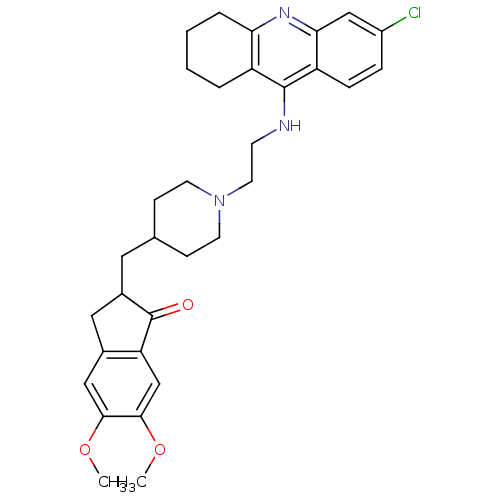 (6-Chloro-9-[(2-{4-[(5,6-dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM9063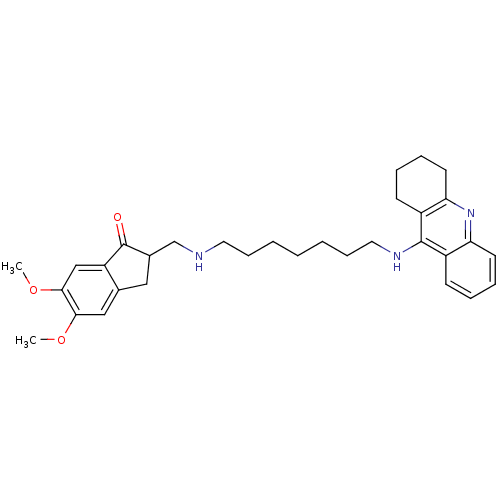 (5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271141 (6-Chloro-9-[(2-{4-[(5,6-dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.670 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556952 (CHEMBL4754487) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 0.770 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271190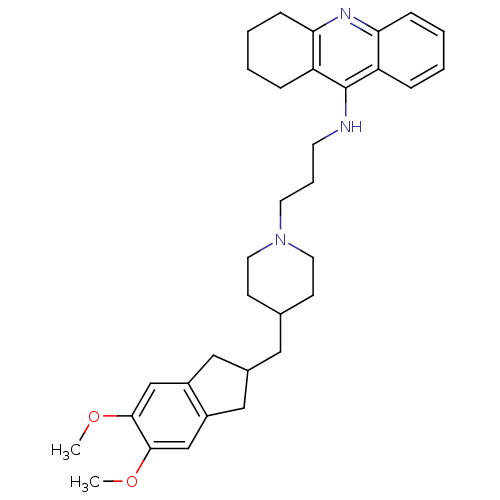 (9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271192 (6-Chloro-9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271140 (9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]pi...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.880 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271192 (6-Chloro-9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.06 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556958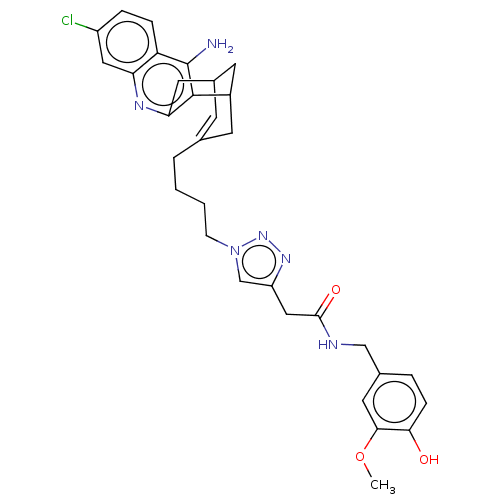 (CHEMBL4744528) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50377921 (SODIUM NITROPRUSSIDE) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.74 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271191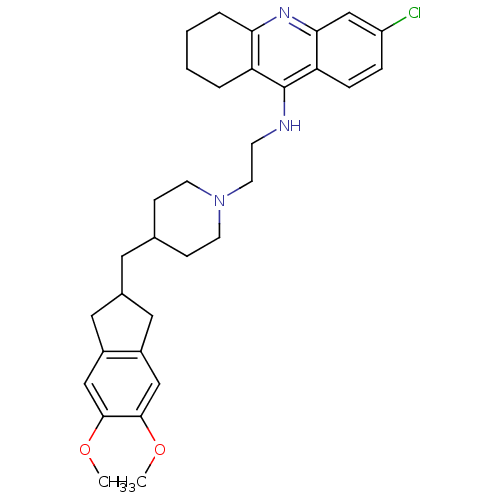 (6-Chloro-9-[(2-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.86 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556953 (CHEMBL4748114) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271190 (9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.16 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271189 (9-[(2-{4-[(5,6-Dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.28 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271191 (6-Chloro-9-[(2-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556954 (CHEMBL4782921) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50377921 (SODIUM NITROPRUSSIDE) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.04 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556951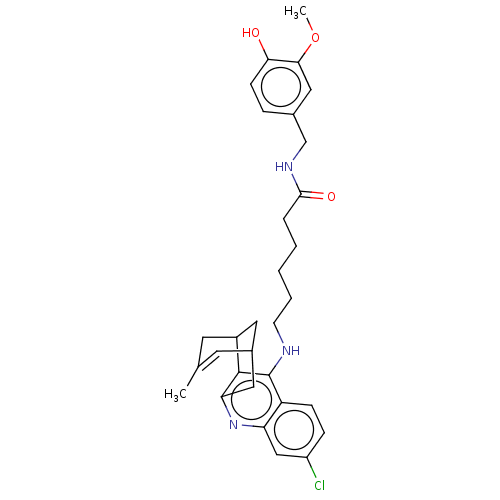 (CHEMBL4788434) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556956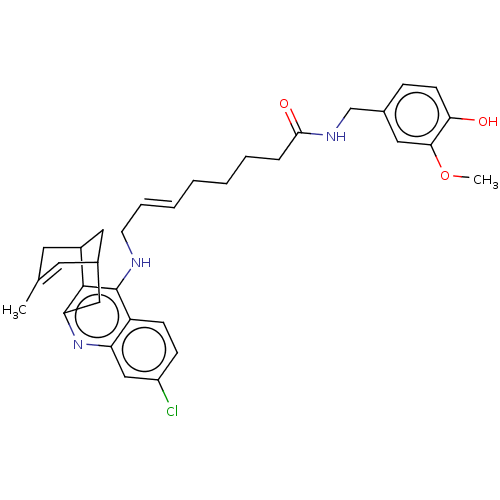 (CHEMBL4741162) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50271189 (9-[(2-{4-[(5,6-Dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.13 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556950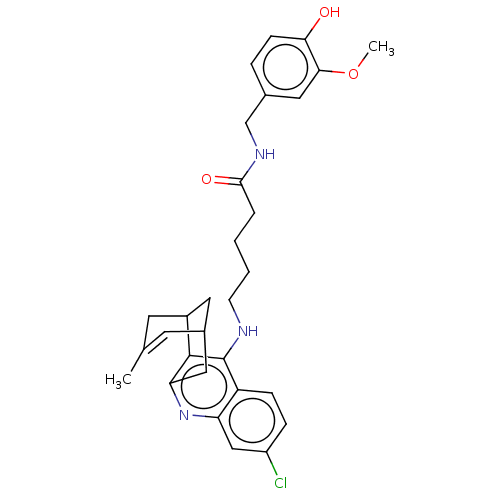 (CHEMBL4743210) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556957 (CHEMBL4794769) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of rat cortex AChE | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM8987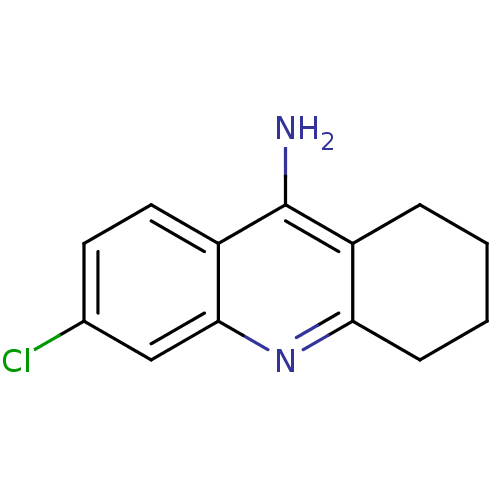 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5.73 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8979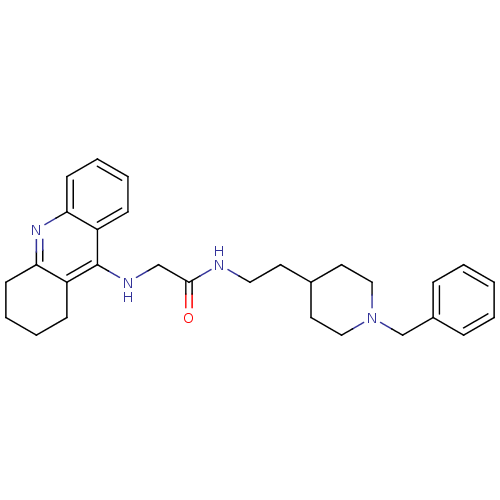 (CHEMBL486901 | N-[2-(1-benzylpiperidin-4-yl)ethyl]...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of rat cortex AChE | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50271190 (9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556958 (CHEMBL4744528) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50271189 (9-[(2-{4-[(5,6-Dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.06 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 8.12 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8987 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.32 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 11.6 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50271140 (9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]pi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12.3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50377921 (SODIUM NITROPRUSSIDE) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 12.4 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM8960 ((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50556955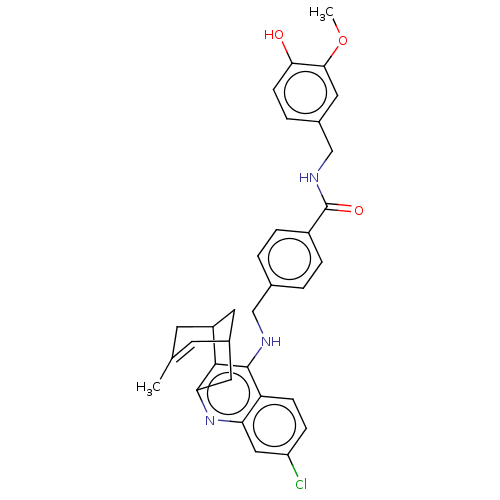 (CHEMBL4782267) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9063 (5,6-Dimethoxy-2-{[7-(1,2,3,4-tetrahydro-acridin-9-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556952 (CHEMBL4754487) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556954 (CHEMBL4782921) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 43.9 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556950 (CHEMBL4743210) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556953 (CHEMBL4748114) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50556957 (CHEMBL4794769) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01775 BindingDB Entry DOI: 10.7270/Q2K077XJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50271142 (6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 66.3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of human serum BChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 72 total ) | Next | Last >> |