 Found 1721 hits with Last Name = 'colwell' and Initial = 'l'
Found 1721 hits with Last Name = 'colwell' and Initial = 'l' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137257
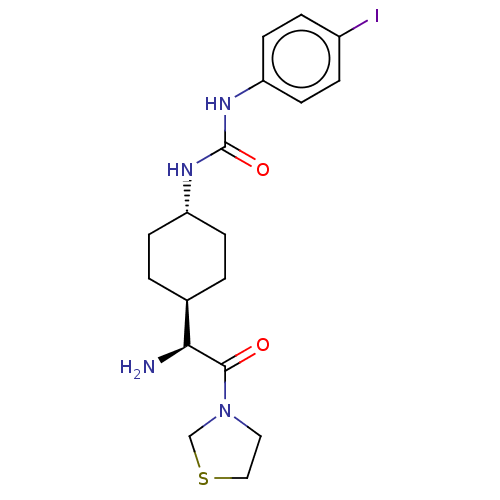
(1-[4-((S)-1-Amino-2-oxo-2-thiazolidin-3-yl-ethyl)-...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NC(=O)Nc1ccc(I)cc1)[C@H](N)C(=O)N1CCSC1 |wU:4.7,1.0,wD:18.20,(1.73,2.3,;3.06,1.54,;1.75,.76,;1.75,-.78,;3.06,-1.54,;4.39,-.78,;4.39,.76,;3.06,-3.08,;4.41,-3.85,;5.74,-3.08,;4.41,-5.39,;5.74,-6.16,;5.74,-7.7,;7.07,-8.47,;8.41,-7.7,;9.74,-8.47,;8.4,-6.14,;7.07,-5.39,;3.06,3.08,;1.73,3.85,;4.41,3.87,;4.41,5.41,;5.74,3.08,;7.15,3.71,;8.17,2.57,;7.4,1.23,;5.9,1.56,)| Show InChI InChI=1S/C18H25IN4O2S/c19-13-3-7-15(8-4-13)22-18(25)21-14-5-1-12(2-6-14)16(20)17(24)23-9-10-26-11-23/h3-4,7-8,12,14,16H,1-2,5-6,9-11,20H2,(H2,21,22,25)/t12-,14-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137261
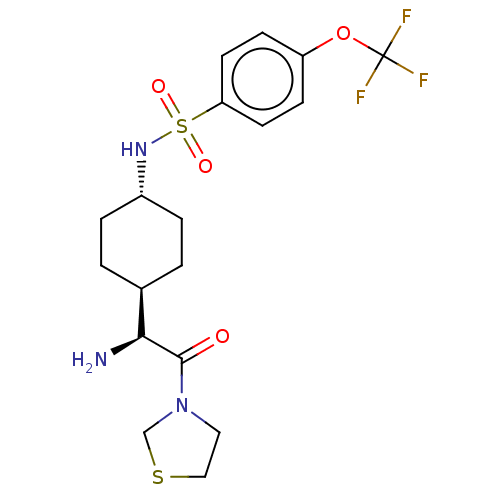
(CHEMBL25211 | N-[4-((S)-1-Amino-2-oxo-2-thiazolidi...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NS(=O)(=O)c1ccc(OC(F)(F)F)cc1)[C@H](N)C(=O)N1CCSC1 |wU:22.24,4.7,1.0,(4.31,-7.01,;4.31,-5.47,;4.31,-3.92,;2.98,-3.15,;1.65,-3.92,;1.65,-5.47,;2.98,-6.24,;.31,-3.13,;-1.02,-3.9,;-.25,-5.25,;-2.37,-4.68,;-1.79,-2.56,;-1.02,-1.23,;-1.79,.11,;-3.34,.11,;-4.12,1.44,;-5.67,1.44,;-5.67,-.1,;-5.67,2.97,;-7.21,1.44,;-4.11,-1.23,;-3.34,-2.56,;5.64,-6.24,;5.64,-7.78,;6.97,-5.47,;6.97,-3.92,;8.3,-6.24,;9.64,-5.47,;10.79,-6.5,;10.15,-7.9,;8.62,-7.74,)| Show InChI InChI=1S/C18H24F3N3O4S2/c19-18(20,21)28-14-5-7-15(8-6-14)30(26,27)23-13-3-1-12(2-4-13)16(22)17(25)24-9-10-29-11-24/h5-8,12-13,16,23H,1-4,9-11,22H2/t12-,13-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137271
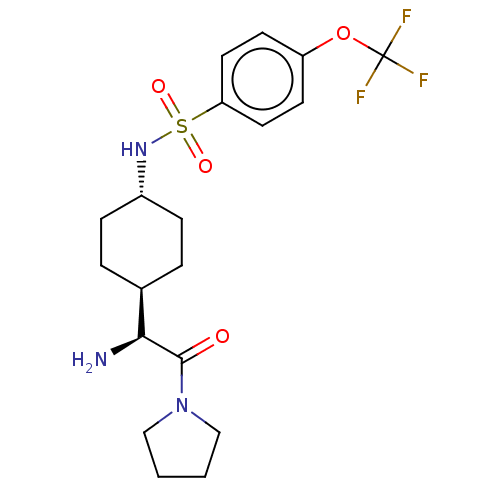
(CHEMBL23979 | N-[4-((S)-1-Amino-2-oxo-2-pyrrolidin...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NS(=O)(=O)c1ccc(OC(F)(F)F)cc1)[C@H](N)C(=O)N1CCCC1 |wU:22.24,4.7,1.0,(4.31,-7.01,;4.31,-5.47,;4.31,-3.92,;2.98,-3.15,;1.65,-3.92,;1.65,-5.47,;2.98,-6.24,;.31,-3.13,;-1.02,-3.9,;-.25,-5.25,;-2.37,-4.68,;-1.79,-2.56,;-1.02,-1.23,;-1.79,.11,;-3.34,.11,;-4.12,1.44,;-5.67,1.44,;-5.67,-.1,;-5.67,2.97,;-7.21,1.44,;-4.11,-1.23,;-3.34,-2.56,;5.64,-6.24,;5.64,-7.78,;6.97,-5.47,;6.97,-3.92,;8.3,-6.24,;8.62,-7.74,;10.15,-7.9,;10.79,-6.5,;9.64,-5.47,)| Show InChI InChI=1S/C19H26F3N3O4S/c20-19(21,22)29-15-7-9-16(10-8-15)30(27,28)24-14-5-3-13(4-6-14)17(23)18(26)25-11-1-2-12-25/h7-10,13-14,17,24H,1-6,11-12,23H2/t13-,14-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137253
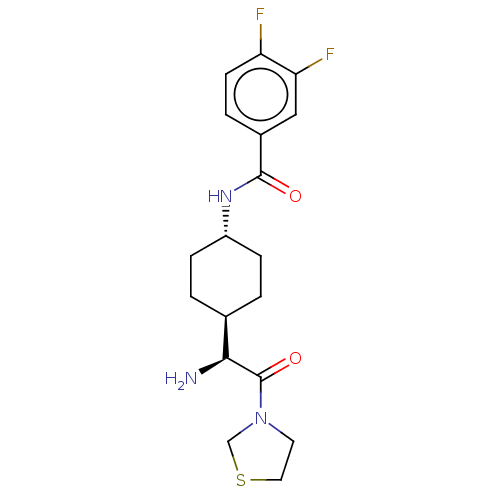
(CHEMBL22359 | N-[4-((S)-1-Amino-2-oxo-2-thiazolidi...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NC(=O)c1ccc(F)c(F)c1)[C@H](N)C(=O)N1CCSC1 |wU:4.7,18.20,1.0,(2.09,-2.17,;2.09,-.63,;2.09,.91,;.75,1.67,;-.57,.91,;-.57,-.63,;.75,-1.4,;-1.9,1.69,;-3.23,.93,;-3.23,-.61,;-4.57,1.69,;-5.89,.93,;-7.24,1.69,;-7.24,3.24,;-8.57,4.01,;-5.89,4.01,;-5.87,5.54,;-4.57,3.24,;3.41,-1.4,;3.41,-2.93,;4.74,-.63,;4.74,.91,;6.07,-1.4,;7.39,-.63,;8.54,-1.67,;7.9,-3.05,;6.39,-2.9,)| Show InChI InChI=1S/C18H23F2N3O2S/c19-14-6-3-12(9-15(14)20)17(24)22-13-4-1-11(2-5-13)16(21)18(25)23-7-8-26-10-23/h3,6,9,11,13,16H,1-2,4-5,7-8,10,21H2,(H,22,24)/t11-,13-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137272
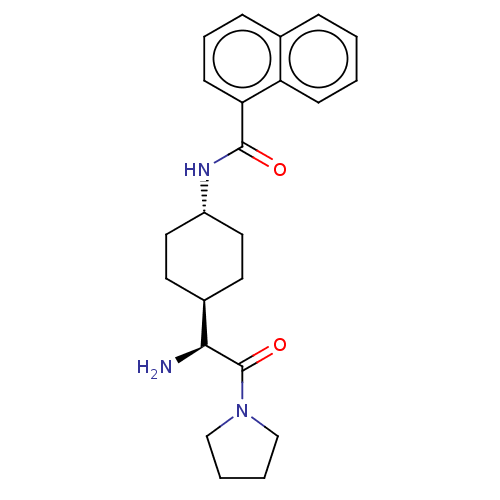
(CHEMBL3084944 | Naphthalene-1-carboxylic acid [4-(...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NC(=O)c1cccc2ccccc12)[C@H](N)C(=O)N1CCCC1 |wU:4.7,1.0,wD:20.23,(12.92,.92,;11.58,.16,;10.25,-.63,;10.25,-2.17,;11.58,-2.92,;12.91,-2.17,;12.91,-.63,;11.58,-4.46,;12.91,-5.23,;14.24,-4.46,;12.91,-6.77,;11.58,-7.54,;11.58,-9.08,;12.91,-9.85,;14.24,-9.06,;15.57,-9.83,;16.89,-9.06,;16.88,-7.52,;15.55,-6.77,;14.24,-7.54,;11.58,1.7,;10.25,2.47,;12.91,2.47,;12.91,4.01,;14.24,1.7,;15.66,2.33,;16.69,1.18,;15.92,-.16,;14.4,.16,)| Show InChI InChI=1S/C23H29N3O2/c24-21(23(28)26-14-3-4-15-26)17-10-12-18(13-11-17)25-22(27)20-9-5-7-16-6-1-2-8-19(16)20/h1-2,5-9,17-18,21H,3-4,10-15,24H2,(H,25,27)/t17-,18-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137264
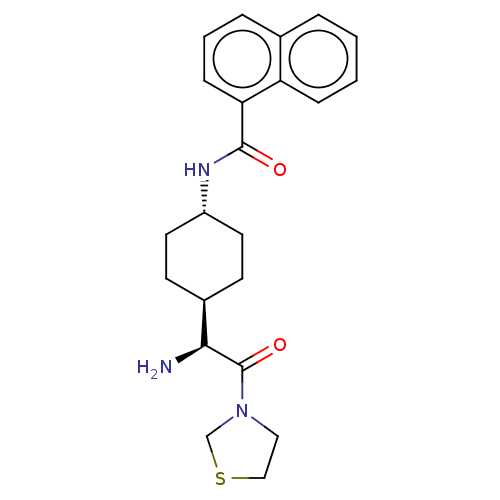
(CHEMBL3084949 | Naphthalene-1-carboxylic acid [4-(...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NC(=O)c1cccc2ccccc12)[C@H](N)C(=O)N1CCSC1 |wU:4.7,1.0,wD:20.23,(1.73,2.3,;3.06,1.54,;1.75,.76,;1.75,-.78,;3.06,-1.54,;4.39,-.78,;4.39,.76,;3.06,-3.08,;4.41,-3.85,;5.74,-3.08,;4.41,-5.39,;5.72,-6.14,;5.74,-7.68,;4.39,-8.45,;3.06,-7.68,;1.75,-8.45,;.42,-7.68,;.42,-6.14,;1.75,-5.39,;3.06,-6.14,;3.06,3.08,;1.73,3.85,;4.41,3.87,;4.41,5.41,;5.74,3.08,;7.15,3.71,;8.17,2.57,;7.4,1.23,;5.9,1.56,)| Show InChI InChI=1S/C22H27N3O2S/c23-20(22(27)25-12-13-28-14-25)16-8-10-17(11-9-16)24-21(26)19-7-3-5-15-4-1-2-6-18(15)19/h1-7,16-17,20H,8-14,23H2,(H,24,26)/t16-,17-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50140521
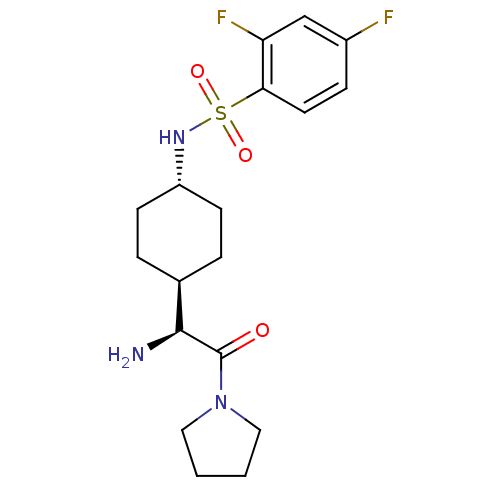
(CHEMBL25437 | N-[4-((S)-1-Amino-2-oxo-2-pyrrolidin...)Show SMILES N[C@@H]([C@H]1CC[C@@H](CC1)NS(=O)(=O)c1ccc(F)cc1F)C(=O)N1CCCC1 |wU:1.0,5.8,wD:2.7,(5.83,-7.06,;5.83,-5.5,;4.5,-4.73,;4.5,-3.19,;3.16,-2.42,;1.83,-3.19,;1.83,-4.73,;3.16,-5.5,;.48,-2.39,;-.86,-3.16,;-.07,-4.51,;-2.2,-3.94,;-1.63,-1.81,;-.86,-.48,;-1.63,.86,;-3.18,.86,;-3.97,2.2,;-3.95,-.48,;-3.18,-1.81,;-3.97,-3.14,;7.16,-4.73,;7.16,-3.19,;8.51,-5.5,;9.85,-4.73,;11,-5.78,;10.37,-7.18,;8.84,-7.02,)| Show InChI InChI=1S/C18H25F2N3O3S/c19-13-5-8-16(15(20)11-13)27(25,26)22-14-6-3-12(4-7-14)17(21)18(24)23-9-1-2-10-23/h5,8,11-12,14,17,22H,1-4,6-7,9-10,21H2/t12-,14-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137260

(CHEMBL22679 | [4-((S)-1-Amino-2-oxo-2-pyrrolidin-1...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NC(=O)OCc1ccccc1)[C@H](N)C(=O)N1CCCC1 |wU:4.7,18.20,1.0,(2.84,-3.3,;2.84,-1.76,;2.84,-.21,;1.5,.56,;.17,-.21,;.17,-1.76,;1.5,-2.53,;-1.18,.58,;-2.52,-.17,;-2.52,-1.74,;-3.85,.58,;-5.19,-.17,;-6.54,.58,;-7.87,-.17,;-9.21,.58,;-9.21,2.14,;-7.87,2.91,;-6.54,2.14,;4.17,-2.53,;4.17,-4.08,;5.5,-1.76,;5.5,-.21,;6.85,-2.53,;8.19,-1.76,;9.34,-2.81,;8.71,-4.19,;7.18,-4.05,)| Show InChI InChI=1S/C20H29N3O3/c21-18(19(24)23-12-4-5-13-23)16-8-10-17(11-9-16)22-20(25)26-14-15-6-2-1-3-7-15/h1-3,6-7,16-18H,4-5,8-14,21H2,(H,22,25)/t16-,17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50265158

(CHEMBL497992 | N-((trans)-4-((S)-1-amino-2-oxo-2-(...)Show SMILES N[C@@H]([C@H]1CC[C@@H](CC1)NS(=O)(=O)c1ccc(NS(=O)(=O)CC(F)(F)F)cc1)C(=O)N1CCCC1 |r,wU:5.8,wD:2.1,1.0,(13.27,5.92,;14.6,5.15,;14.6,3.61,;13.27,2.83,;13.27,1.28,;14.6,.51,;15.95,1.28,;15.95,2.83,;14.6,-1.03,;13.27,-1.79,;14.03,-3.13,;12.5,-.46,;11.93,-2.55,;10.6,-1.76,;9.26,-2.51,;9.25,-4.05,;7.91,-4.81,;7.89,-6.35,;6.35,-6.34,;9.43,-6.37,;7.88,-7.89,;6.54,-8.65,;5.2,-9.4,;7.3,-9.99,;5.78,-7.31,;10.58,-4.84,;11.91,-4.08,;15.93,5.92,;15.93,7.46,;17.27,5.15,;17.43,3.63,;18.93,3.31,;19.7,4.64,;18.67,5.79,)| Show InChI InChI=1S/C20H29F3N4O5S2/c21-20(22,23)13-33(29,30)25-15-7-9-17(10-8-15)34(31,32)26-16-5-3-14(4-6-16)18(24)19(28)27-11-1-2-12-27/h7-10,14,16,18,25-26H,1-6,11-13,24H2/t14-,16-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50137270

(CHEMBL3084938 | N-[4-((S)-1-Amino-2-oxo-2-pyrrolid...)Show SMILES [H][C@@]1(CC[C@@H](CC1)NS(=O)(=O)c1ccc(NC(=O)N(C)C)cc1)[C@H](N)C(=O)N1CCCC1 |wU:4.7,1.0,wD:23.25,(10.79,7.46,;12.14,6.7,;10.81,5.91,;10.81,4.36,;12.14,3.6,;13.47,4.36,;13.47,5.91,;12.14,2.06,;13.48,1.29,;13.48,-.26,;14.81,2.06,;14.81,.51,;16.12,1.29,;17.45,.53,;17.45,-1.03,;18.8,-1.8,;20.13,-1.03,;20.13,.51,;21.47,-1.8,;21.46,-3.34,;22.81,-1.03,;16.12,-1.78,;14.79,-1.01,;12.14,8.23,;10.8,9,;13.48,9.01,;13.48,10.55,;14.81,8.24,;14.97,6.7,;16.48,6.38,;17.26,7.72,;16.21,8.85,)| Show InChI InChI=1S/C21H33N5O4S/c1-25(2)21(28)23-16-9-11-18(12-10-16)31(29,30)24-17-7-5-15(6-8-17)19(22)20(27)26-13-3-4-14-26/h9-12,15,17,19,24H,3-8,13-14,22H2,1-2H3,(H,23,28)/t15-,17-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Potassium channel HERG Kv11.1 |
Bioorg Med Chem Lett 14: 43-6 (2003)
BindingDB Entry DOI: 10.7270/Q26972ZM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50151798
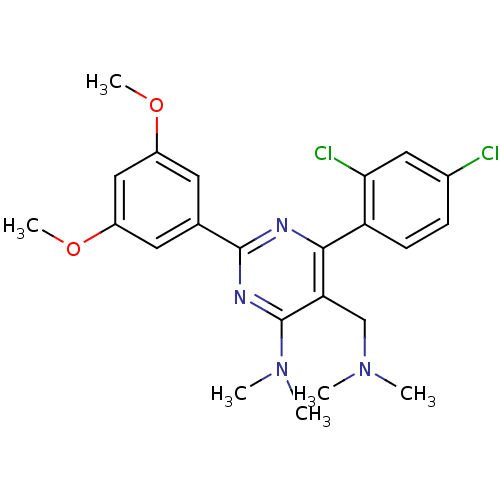
(CHEMBL186877 | [6-(2,4-Dichloro-phenyl)-2-(3,5-dim...)Show SMILES COc1cc(OC)cc(c1)-c1nc(N(C)C)c(CN(C)C)c(n1)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C23H26Cl2N4O2/c1-28(2)13-19-21(18-8-7-15(24)11-20(18)25)26-22(27-23(19)29(3)4)14-9-16(30-5)12-17(10-14)31-6/h7-12H,13H2,1-6H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Dipeptidyl peptidase IV |
Bioorg Med Chem Lett 14: 4759-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.099
BindingDB Entry DOI: 10.7270/Q2XK8F1Q |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50187939
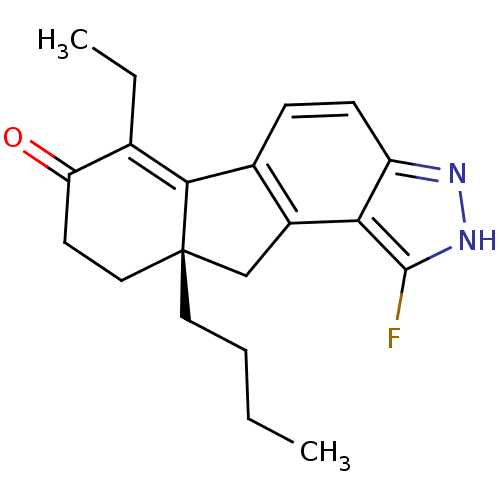
((S)-9a-butyl-6-ethyl-1-fluoro-8,9,9a,10-tetrahydro...)Show SMILES CCCC[C@]12Cc3c(ccc4n[nH]c(F)c34)C1=C(CC)C(=O)CC2 |t:19| Show InChI InChI=1S/C20H23FN2O/c1-3-5-9-20-10-8-16(24)12(4-2)18(20)13-6-7-15-17(14(13)11-20)19(21)23-22-15/h6-7H,3-5,8-11H2,1-2H3,(H,22,23)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERbeta |
Bioorg Med Chem Lett 16: 3896-901 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.036
BindingDB Entry DOI: 10.7270/Q24T6J01 |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50187954
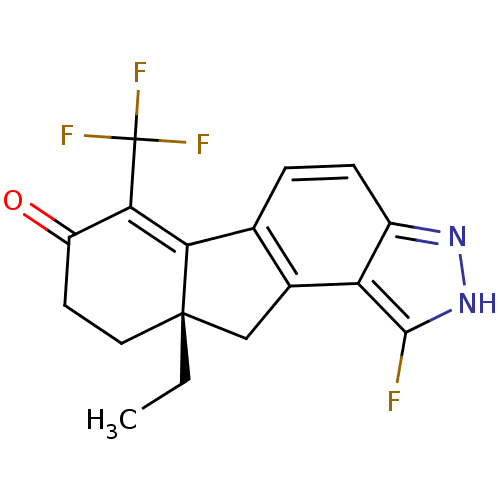
((S)-9a-ethyl-1-fluoro-6-(trifluoromethyl)-8,9,9a,1...)Show SMILES CC[C@]12Cc3c(ccc4n[nH]c(F)c34)C1=C(C(=O)CC2)C(F)(F)F |c:17| Show InChI InChI=1S/C17H14F4N2O/c1-2-16-6-5-11(24)14(17(19,20)21)13(16)8-3-4-10-12(9(8)7-16)15(18)23-22-10/h3-4H,2,5-7H2,1H3,(H,22,23)/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERbeta |
Bioorg Med Chem Lett 16: 3896-901 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.036
BindingDB Entry DOI: 10.7270/Q24T6J01 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50153406
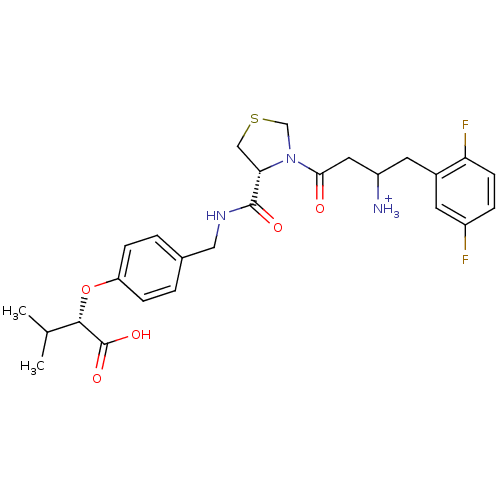
(CHEMBL366290 | Trifluoro-acetate3-{(R)-4-[4-((S)-1...)Show SMILES CC(C)[C@H](Oc1ccc(CNC(=O)[C@@H]2CSCN2C(=O)CC([NH3+])Cc2cc(F)ccc2F)cc1)C(O)=O Show InChI InChI=1S/C26H31F2N3O5S/c1-15(2)24(26(34)35)36-20-6-3-16(4-7-20)12-30-25(33)22-13-37-14-31(22)23(32)11-19(29)10-17-9-18(27)5-8-21(17)28/h3-9,15,19,22,24H,10-14,29H2,1-2H3,(H,30,33)(H,34,35)/p+1/t19?,22-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant Dipeptidylpeptidase IV |
Bioorg Med Chem Lett 14: 5151-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.056
BindingDB Entry DOI: 10.7270/Q2QR4WM2 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162799
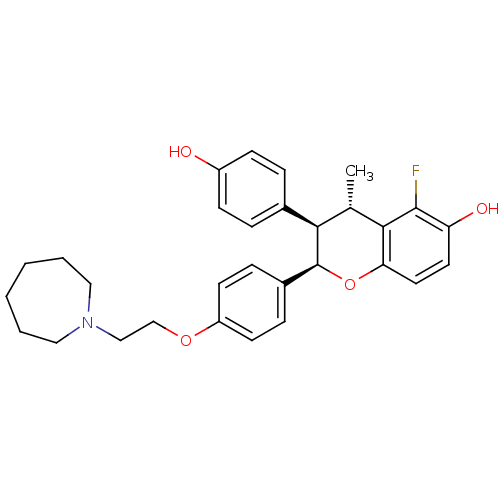
((2R,3R,4S)-2-[4-(2-Azepan-1-yl-ethoxy)-phenyl]-5-f...)Show SMILES C[C@H]1[C@@H]([C@@H](Oc2ccc(O)c(F)c12)c1ccc(OCCN2CCCCCC2)cc1)c1ccc(O)cc1 Show InChI InChI=1S/C30H34FNO4/c1-20-27(21-6-10-23(33)11-7-21)30(36-26-15-14-25(34)29(31)28(20)26)22-8-12-24(13-9-22)35-19-18-32-16-4-2-3-5-17-32/h6-15,20,27,30,33-34H,2-5,16-19H2,1H3/t20-,27+,30-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50153384
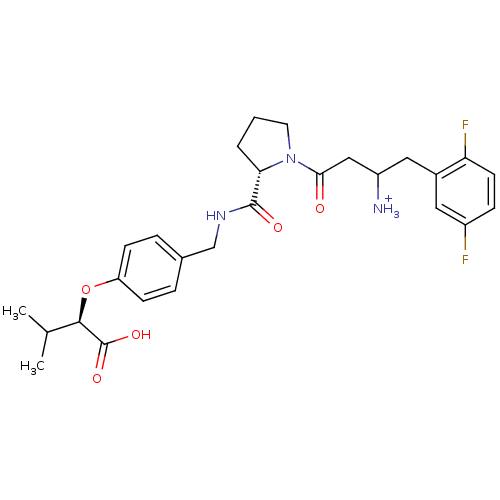
(CHEMBL364102 | Trifluoro-acetate3-{(S)-2-[4-((R)-1...)Show SMILES CC(C)[C@@H](Oc1ccc(CNC(=O)[C@@H]2CCCN2C(=O)CC([NH3+])Cc2cc(F)ccc2F)cc1)C(O)=O Show InChI InChI=1S/C27H33F2N3O5/c1-16(2)25(27(35)36)37-21-8-5-17(6-9-21)15-31-26(34)23-4-3-11-32(23)24(33)14-20(30)13-18-12-19(28)7-10-22(18)29/h5-10,12,16,20,23,25H,3-4,11,13-15,30H2,1-2H3,(H,31,34)(H,35,36)/p+1/t20?,23-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant Dipeptidylpeptidase IV |
Bioorg Med Chem Lett 14: 5151-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.056
BindingDB Entry DOI: 10.7270/Q2QR4WM2 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146277
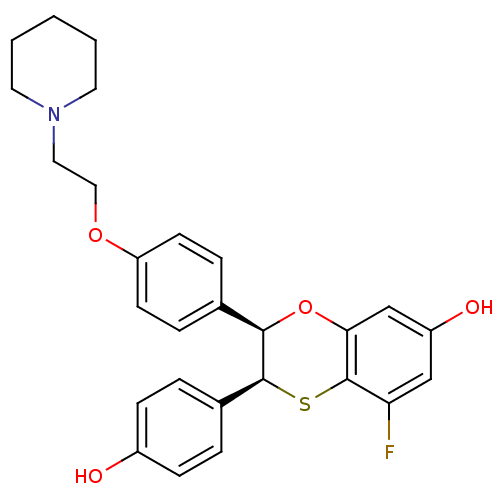
((2R,3S)-5-Fluoro-3-(4-hydroxy-phenyl)-2-[4-(2-pipe...)Show SMILES Oc1ccc(cc1)[C@@H]1Sc2c(F)cc(O)cc2O[C@@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H28FNO4S/c28-23-16-21(31)17-24-27(23)34-26(19-4-8-20(30)9-5-19)25(33-24)18-6-10-22(11-7-18)32-15-14-29-12-2-1-3-13-29/h4-11,16-17,25-26,30-31H,1-3,12-15H2/t25-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor beta |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162804
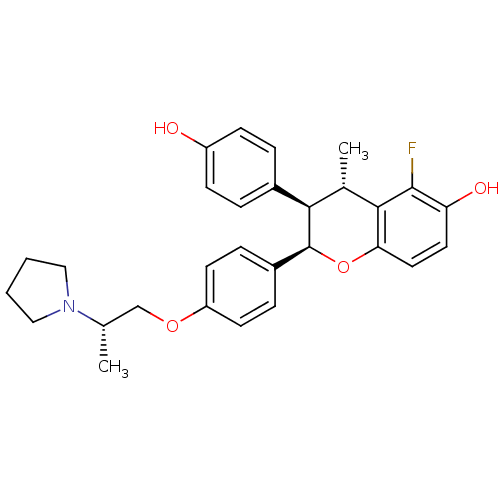
((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@@H](COc1ccc(cc1)[C@@H]1Oc2ccc(O)c(F)c2[C@@H](C)[C@@H]1c1ccc(O)cc1)N1CCCC1 Show InChI InChI=1S/C29H32FNO4/c1-18(31-15-3-4-16-31)17-34-23-11-7-21(8-12-23)29-26(20-5-9-22(32)10-6-20)19(2)27-25(35-29)14-13-24(33)28(27)30/h5-14,18-19,26,29,32-33H,3-4,15-17H2,1-2H3/t18-,19-,26+,29-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146269
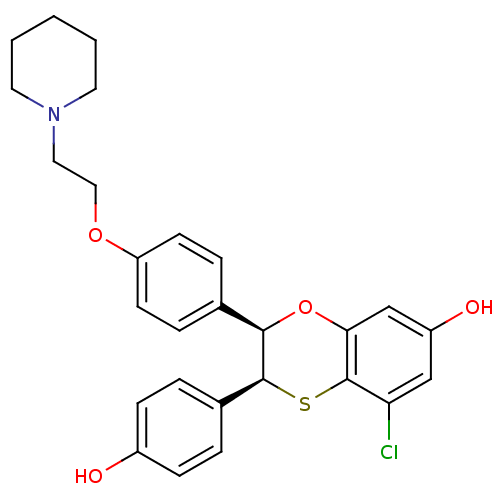
((2R,3S)-5-Chloro-3-(4-hydroxy-phenyl)-2-[4-(2-pipe...)Show SMILES Oc1ccc(cc1)[C@@H]1Sc2c(Cl)cc(O)cc2O[C@@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H28ClNO4S/c28-23-16-21(31)17-24-27(23)34-26(19-4-8-20(30)9-5-19)25(33-24)18-6-10-22(11-7-18)32-15-14-29-12-2-1-3-13-29/h4-11,16-17,25-26,30-31H,1-3,12-15H2/t25-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.510 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor alpha |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162794

((2R,3R,4S)-3-(4-Hydroxy-phenyl)-4-methyl-2-[4-(2-p...)Show SMILES C[C@H]1[C@@H]([C@@H](Oc2cc(O)ccc12)c1ccc(OCCN2CCCCC2)cc1)c1ccc(O)cc1 Show InChI InChI=1S/C29H33NO4/c1-20-26-14-11-24(32)19-27(26)34-29(28(20)21-5-9-23(31)10-6-21)22-7-12-25(13-8-22)33-18-17-30-15-3-2-4-16-30/h5-14,19-20,28-29,31-32H,2-4,15-18H2,1H3/t20-,28-,29+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.660 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162807
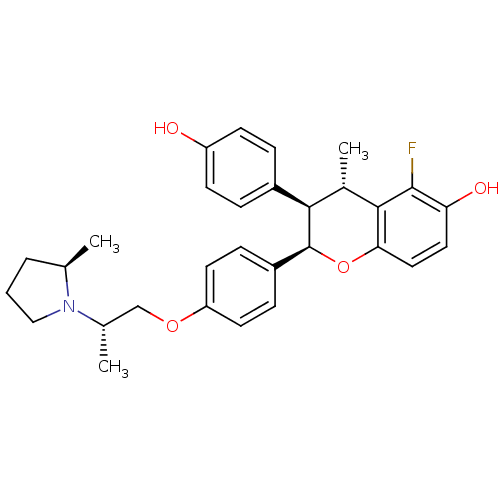
((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@@H](COc1ccc(cc1)[C@@H]1Oc2ccc(O)c(F)c2[C@@H](C)[C@@H]1c1ccc(O)cc1)N1CCC[C@H]1C Show InChI InChI=1S/C30H34FNO4/c1-18-5-4-16-32(18)19(2)17-35-24-12-8-22(9-13-24)30-27(21-6-10-23(33)11-7-21)20(3)28-26(36-30)15-14-25(34)29(28)31/h6-15,18-20,27,30,33-34H,4-5,16-17H2,1-3H3/t18-,19+,20+,27-,30+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162812

((2R,3R,4S)-5-FLUORO-3-(4-HYDROXYPHENYL)-4-METHYL-2...)Show SMILES C[C@H]1[C@@H]([C@@H](Oc2ccc(O)c(F)c12)c1ccc(OCCN2CCCCC2)cc1)c1ccc(O)cc1 Show InChI InChI=1S/C29H32FNO4/c1-19-26(20-5-9-22(32)10-6-20)29(35-25-14-13-24(33)28(30)27(19)25)21-7-11-23(12-8-21)34-18-17-31-15-3-2-4-16-31/h5-14,19,26,29,32-33H,2-4,15-18H2,1H3/t19-,26+,29-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162809
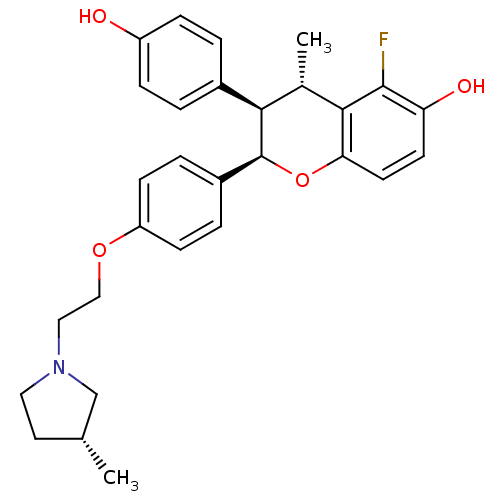
((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@@H]1CCN(CCOc2ccc(cc2)[C@@H]2Oc3ccc(O)c(F)c3[C@@H](C)[C@@H]2c2ccc(O)cc2)C1 Show InChI InChI=1S/C29H32FNO4/c1-18-13-14-31(17-18)15-16-34-23-9-5-21(6-10-23)29-26(20-3-7-22(32)8-4-20)19(2)27-25(35-29)12-11-24(33)28(27)30/h3-12,18-19,26,29,32-33H,13-17H2,1-2H3/t18-,19+,26-,29+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50144849
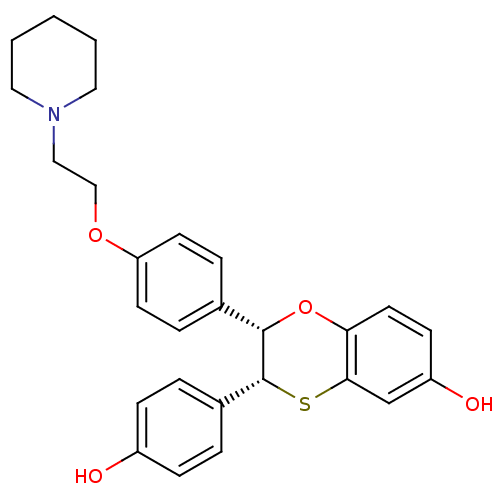
((2S,3R)-2-(4-(2-(PIPERIDIN-1-YL)ETHOXY)PHENYL)-2,3...)Show SMILES Oc1ccc(cc1)[C@H]1Sc2cc(O)ccc2O[C@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H29NO4S/c29-21-8-4-20(5-9-21)27-26(32-24-13-10-22(30)18-25(24)33-27)19-6-11-23(12-7-19)31-17-16-28-14-2-1-3-15-28/h4-13,18,26-27,29-30H,1-3,14-17H2/t26-,27+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146262
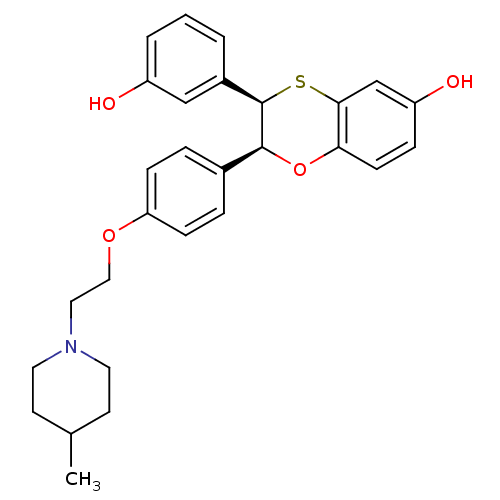
((2S,3R)-3-(3-Hydroxy-phenyl)-2-{4-[2-(4-methyl-pip...)Show SMILES CC1CCN(CCOc2ccc(cc2)[C@@H]2Oc3ccc(O)cc3S[C@@H]2c2cccc(O)c2)CC1 Show InChI InChI=1S/C28H31NO4S/c1-19-11-13-29(14-12-19)15-16-32-24-8-5-20(6-9-24)27-28(21-3-2-4-22(30)17-21)34-26-18-23(31)7-10-25(26)33-27/h2-10,17-19,27-28,30-31H,11-16H2,1H3/t27-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor alpha |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162801
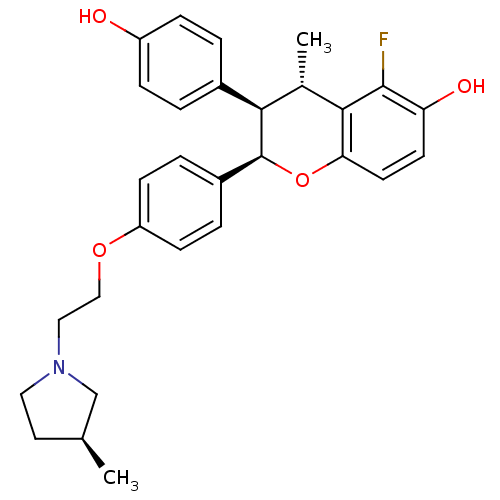
((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@H]1CCN(CCOc2ccc(cc2)[C@@H]2Oc3ccc(O)c(F)c3[C@@H](C)[C@@H]2c2ccc(O)cc2)C1 Show InChI InChI=1S/C29H32FNO4/c1-18-13-14-31(17-18)15-16-34-23-9-5-21(6-10-23)29-26(20-3-7-22(32)8-4-20)19(2)27-25(35-29)12-11-24(33)28(27)30/h3-12,18-19,26,29,32-33H,13-17H2,1-2H3/t18-,19-,26+,29-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146267

((2R,3S)-5-Fluoro-3-(4-hydroxy-phenyl)-2-[4-(2-pipe...)Show SMILES Oc1ccc(cc1)[C@@H]1Sc2c(F)c(O)ccc2O[C@@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H28FNO4S/c28-24-22(31)12-13-23-27(24)34-26(19-4-8-20(30)9-5-19)25(33-23)18-6-10-21(11-7-18)32-17-16-29-14-2-1-3-15-29/h4-13,25-26,30-31H,1-3,14-17H2/t25-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor beta |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Arachidonate 5-lipoxygenase-activating protein
(Homo sapiens (Human)) | BDBM50383916

(CHEMBL2031657)Show SMILES O=c1[nH]nc(o1)-c1ccc(OCc2ccc3ccccc3n2)cc1[C@]1(C[C@H]2CC[C@H]1C2)c1ccccc1 |r,TLB:31:24:27.28:30,THB:23:24:27.28:30| Show InChI InChI=1S/C31H27N3O3/c35-30-34-33-29(37-30)26-15-14-25(36-19-24-13-11-21-6-4-5-9-28(21)32-24)17-27(26)31(22-7-2-1-3-8-22)18-20-10-12-23(31)16-20/h1-9,11,13-15,17,20,23H,10,12,16,18-19H2,(H,34,35)/t20-,23-,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [125I]L-691,831 from 5-lipoxygenase-activating protein in human polymorphonuclear cells |
Bioorg Med Chem Lett 22: 4133-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.064
BindingDB Entry DOI: 10.7270/Q2348MCB |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50187941
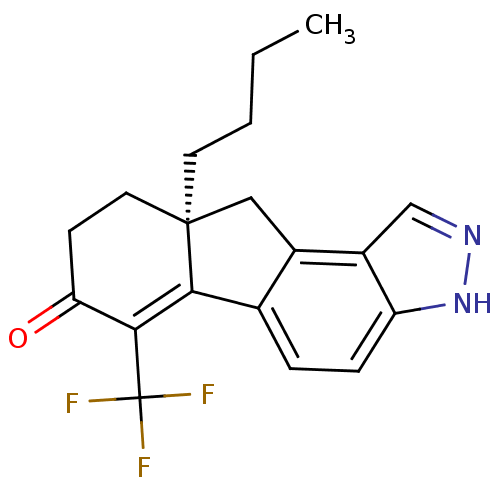
((S)-9a-butyl-6-(trifluoromethyl)-8,9,9a,10-tetrahy...)Show SMILES CCCC[C@]12Cc3c(ccc4[nH]ncc34)C1=C(C(=O)CC2)C(F)(F)F |c:18| Show InChI InChI=1S/C19H19F3N2O/c1-2-3-7-18-8-6-15(25)17(19(20,21)22)16(18)11-4-5-14-13(10-23-24-14)12(11)9-18/h4-5,10H,2-3,6-9H2,1H3,(H,23,24)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERbeta |
Bioorg Med Chem Lett 16: 3896-901 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.036
BindingDB Entry DOI: 10.7270/Q24T6J01 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50144849

((2S,3R)-2-(4-(2-(PIPERIDIN-1-YL)ETHOXY)PHENYL)-2,3...)Show SMILES Oc1ccc(cc1)[C@H]1Sc2cc(O)ccc2O[C@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H29NO4S/c29-21-8-4-20(5-9-21)27-26(32-24-13-10-22(30)18-25(24)33-27)19-6-11-23(12-7-19)31-17-16-28-14-2-1-3-15-28/h4-13,18,26-27,29-30H,1-3,14-17H2/t26-,27+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor alpha |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50153395
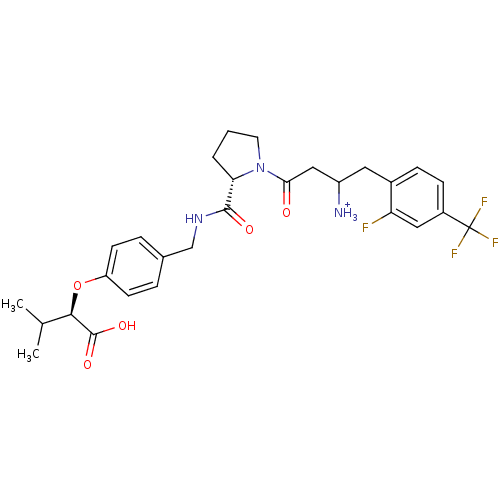
(CHEMBL364140 | Trifluoro-acetate3-{(S)-2-[4-((R)-1...)Show SMILES CC(C)[C@@H](Oc1ccc(CNC(=O)[C@@H]2CCCN2C(=O)CC([NH3+])Cc2ccc(cc2F)C(F)(F)F)cc1)C(O)=O Show InChI InChI=1S/C28H33F4N3O5/c1-16(2)25(27(38)39)40-21-9-5-17(6-10-21)15-34-26(37)23-4-3-11-35(23)24(36)14-20(33)12-18-7-8-19(13-22(18)29)28(30,31)32/h5-10,13,16,20,23,25H,3-4,11-12,14-15,33H2,1-2H3,(H,34,37)(H,38,39)/p+1/t20?,23-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.830 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant Dipeptidylpeptidase IV |
Bioorg Med Chem Lett 14: 5151-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.056
BindingDB Entry DOI: 10.7270/Q2QR4WM2 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146272
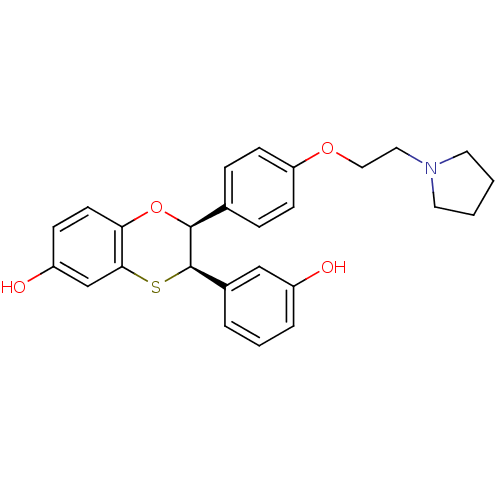
((2S,3R)-3-(3-Hydroxy-phenyl)-2-[4-(2-pyrrolidin-1-...)Show SMILES Oc1cccc(c1)[C@H]1Sc2cc(O)ccc2O[C@H]1c1ccc(OCCN2CCCC2)cc1 Show InChI InChI=1S/C26H27NO4S/c28-20-5-3-4-19(16-20)26-25(31-23-11-8-21(29)17-24(23)32-26)18-6-9-22(10-7-18)30-15-14-27-12-1-2-13-27/h3-11,16-17,25-26,28-29H,1-2,12-15H2/t25-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor alpha |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162805
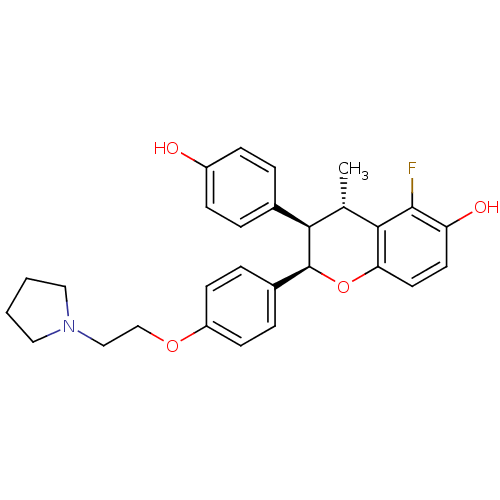
((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@H]1[C@@H]([C@@H](Oc2ccc(O)c(F)c12)c1ccc(OCCN2CCCC2)cc1)c1ccc(O)cc1 Show InChI InChI=1S/C28H30FNO4/c1-18-25(19-4-8-21(31)9-5-19)28(34-24-13-12-23(32)27(29)26(18)24)20-6-10-22(11-7-20)33-17-16-30-14-2-3-15-30/h4-13,18,25,28,31-32H,2-3,14-17H2,1H3/t18-,25+,28-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50162796

((2R,3R,4S)-5-Fluoro-3-(4-hydroxy-phenyl)-4-methyl-...)Show SMILES C[C@@H](COc1ccc(cc1)[C@@H]1Oc2ccc(O)c(F)c2[C@@H](C)[C@@H]1c1ccc(O)cc1)N1CC[C@@H](C)C1 Show InChI InChI=1S/C30H34FNO4/c1-18-14-15-32(16-18)19(2)17-35-24-10-6-22(7-11-24)30-27(21-4-8-23(33)9-5-21)20(3)28-26(36-30)13-12-25(34)29(28)31/h4-13,18-20,27,30,33-34H,14-17H2,1-3H3/t18-,19+,20+,27-,30+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of estrogen receptor alpha |
Bioorg Med Chem Lett 15: 1675-81 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.046
BindingDB Entry DOI: 10.7270/Q2S46RFB |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50187956
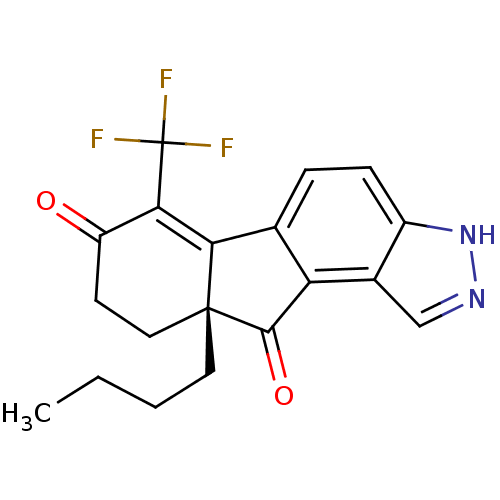
((R)-9a-butyl-6-(trifluoromethyl)-9,9a-dihydroinden...)Show SMILES CCCC[C@@]12CCC(=O)C(=C1c1ccc3[nH]ncc3c1C2=O)C(F)(F)F |c:9| Show InChI InChI=1S/C19H17F3N2O2/c1-2-3-7-18-8-6-13(25)16(19(20,21)22)15(18)10-4-5-12-11(9-23-24-12)14(10)17(18)26/h4-5,9H,2-3,6-8H2,1H3,(H,23,24)/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERbeta |
Bioorg Med Chem Lett 16: 3896-901 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.036
BindingDB Entry DOI: 10.7270/Q24T6J01 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50157933
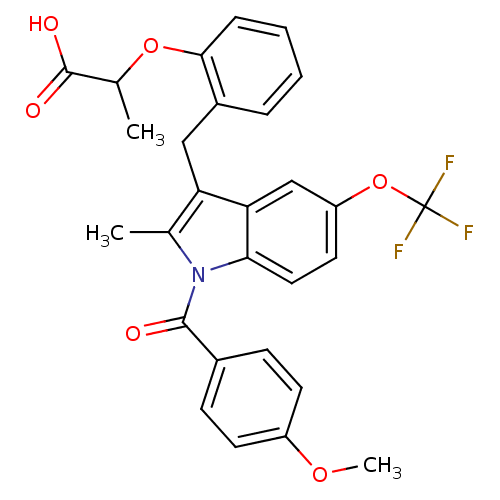
(2-{2-[1-(4-Methoxy-benzoyl)-2-methyl-5-trifluorome...)Show SMILES COc1ccc(cc1)C(=O)n1c(C)c(Cc2ccccc2OC(C)C(O)=O)c2cc(OC(F)(F)F)ccc12 Show InChI InChI=1S/C28H24F3NO6/c1-16-22(14-19-6-4-5-7-25(19)37-17(2)27(34)35)23-15-21(38-28(29,30)31)12-13-24(23)32(16)26(33)18-8-10-20(36-3)11-9-18/h4-13,15,17H,14H2,1-3H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Peroxisome proliferator activated receptor gamma |
Bioorg Med Chem Lett 15: 357-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.068
BindingDB Entry DOI: 10.7270/Q27W6BNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50267990
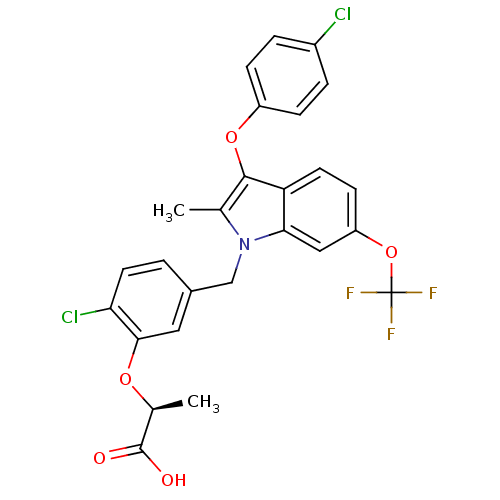
((S)-2-(2-chloro-5-((3-(4-chlorophenoxy)-2-methyl-6...)Show SMILES C[C@H](Oc1cc(Cn2c(C)c(Oc3ccc(Cl)cc3)c3ccc(OC(F)(F)F)cc23)ccc1Cl)C(O)=O |r| Show InChI InChI=1S/C26H20Cl2F3NO5/c1-14-24(36-18-6-4-17(27)5-7-18)20-9-8-19(37-26(29,30)31)12-22(20)32(14)13-16-3-10-21(28)23(11-16)35-15(2)25(33)34/h3-12,15H,13H2,1-2H3,(H,33,34)/t15-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H2]nTZD3 from human recombinant GST-fused PPARgamma expressed in Escherichia coli by scintillation proximity assay |
J Med Chem 52: 3846-54 (2009)
Article DOI: 10.1021/jm900097m
BindingDB Entry DOI: 10.7270/Q2GF0TDR |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50157917

((2S)-2-(3-{[1-(4-METHOXYBENZOYL)-2-METHYL-5-(TRIFL...)Show SMILES COc1ccc(cc1)C(=O)n1c(C)c(Cc2cccc(O[C@@H](C)C(O)=O)c2)c2cc(OC(F)(F)F)ccc12 |r| Show InChI InChI=1S/C28H24F3NO6/c1-16-23(14-18-5-4-6-21(13-18)37-17(2)27(34)35)24-15-22(38-28(29,30)31)11-12-25(24)32(16)26(33)19-7-9-20(36-3)10-8-19/h4-13,15,17H,14H2,1-3H3,(H,34,35)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H2]nTZD3 from human recombinant GST-fused PPARgamma expressed in Escherichia coli by scintillation proximity assay |
J Med Chem 52: 3846-54 (2009)
Article DOI: 10.1021/jm900097m
BindingDB Entry DOI: 10.7270/Q2GF0TDR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50268271
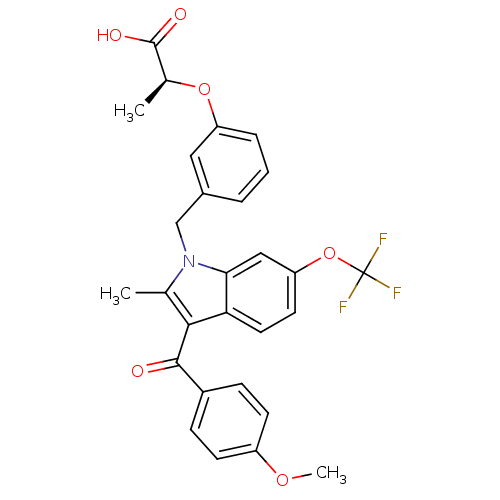
((S)-2-(3-((3-(4-methoxybenzoyl)-2-methyl-6-(triflu...)Show SMILES COc1ccc(cc1)C(=O)c1c(C)n(Cc2cccc(O[C@@H](C)C(O)=O)c2)c2cc(OC(F)(F)F)ccc12 |r| Show InChI InChI=1S/C28H24F3NO6/c1-16-25(26(33)19-7-9-20(36-3)10-8-19)23-12-11-22(38-28(29,30)31)14-24(23)32(16)15-18-5-4-6-21(13-18)37-17(2)27(34)35/h4-14,17H,15H2,1-3H3,(H,34,35)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H2]nTZD3 from human recombinant GST-fused PPARgamma expressed in Escherichia coli by scintillation proximity assay |
J Med Chem 52: 3846-54 (2009)
Article DOI: 10.1021/jm900097m
BindingDB Entry DOI: 10.7270/Q2GF0TDR |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50166300
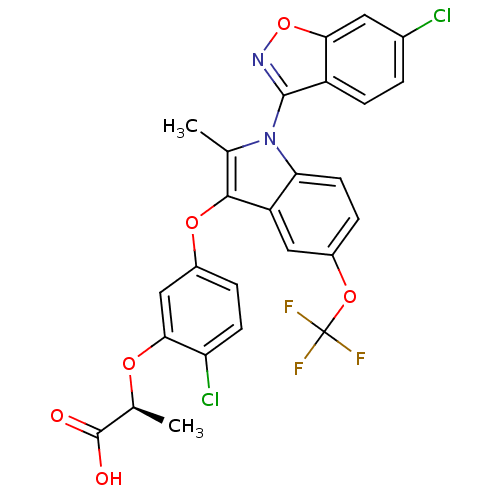
((S)-2-{2-Chloro-5-[1-(6-chloro-benzo[d]isoxazol-3-...)Show SMILES C[C@H](Oc1cc(Oc2c(C)n(-c3noc4cc(Cl)ccc34)c3ccc(OC(F)(F)F)cc23)ccc1Cl)C(O)=O |wU:1.0,(11.52,1.27,;11.04,-.19,;9.55,-.52,;8.51,.64,;7.01,.32,;5.98,1.43,;4.48,1.11,;4,-.36,;4.91,-1.61,;6.44,-1.61,;4,-2.84,;4.41,-4.33,;3.45,-5.51,;4.3,-6.81,;5.77,-6.42,;7.06,-7.25,;8.43,-6.56,;9.72,-7.37,;8.5,-5,;7.22,-4.17,;5.86,-4.89,;2.54,-2.38,;1.22,-3.14,;-.13,-2.38,;-.13,-.84,;-1.48,-.05,;-2.8,-.84,;-4.15,-1.63,;-3.59,.5,;-2.01,-2.17,;1.22,-.05,;2.54,-.82,;6.46,2.92,;7.95,3.24,;9,2.11,;10.5,2.41,;12.09,-1.33,;11.6,-2.79,;13.6,-1.01,)| Show InChI InChI=1S/C26H17Cl2F3N2O6/c1-12-23(37-15-4-7-19(28)22(11-15)36-13(2)25(34)35)18-10-16(38-26(29,30)31)5-8-20(18)33(12)24-17-6-3-14(27)9-21(17)39-32-24/h3-11,13H,1-2H3,(H,34,35)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human peroxisome proliferator activated receptor gamma binding |
Bioorg Med Chem Lett 15: 2437-40 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.092
BindingDB Entry DOI: 10.7270/Q2CF9PM8 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50166292
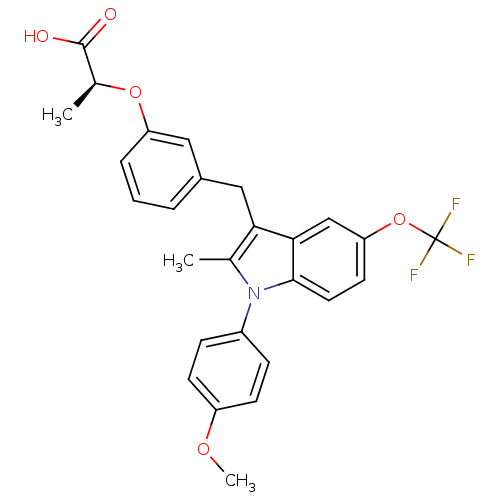
((S)-2-{3-[1-(4-Methoxy-phenyl)-2-methyl-5-trifluor...)Show SMILES COc1ccc(cc1)-n1c(C)c(Cc2cccc(O[C@@H](C)C(O)=O)c2)c2cc(OC(F)(F)F)ccc12 Show InChI InChI=1S/C27H24F3NO5/c1-16-23(14-18-5-4-6-21(13-18)35-17(2)26(32)33)24-15-22(36-27(28,29)30)11-12-25(24)31(16)19-7-9-20(34-3)10-8-19/h4-13,15,17H,14H2,1-3H3,(H,32,33)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human peroxisome proliferator activated receptor gamma binding |
Bioorg Med Chem Lett 15: 2437-40 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.092
BindingDB Entry DOI: 10.7270/Q2CF9PM8 |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50187968
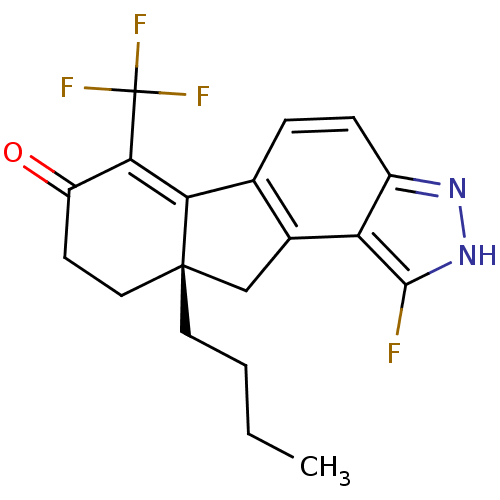
((S)-9a-butyl-1-fluoro-6-(trifluoromethyl)-8,9,9a,1...)Show SMILES CCCC[C@]12Cc3c(ccc4n[nH]c(F)c34)C1=C(C(=O)CC2)C(F)(F)F |c:19| Show InChI InChI=1S/C19H18F4N2O/c1-2-3-7-18-8-6-13(26)16(19(21,22)23)15(18)10-4-5-12-14(11(10)9-18)17(20)25-24-12/h4-5H,2-3,6-9H2,1H3,(H,24,25)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human ERbeta |
Bioorg Med Chem Lett 16: 3896-901 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.036
BindingDB Entry DOI: 10.7270/Q24T6J01 |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146264
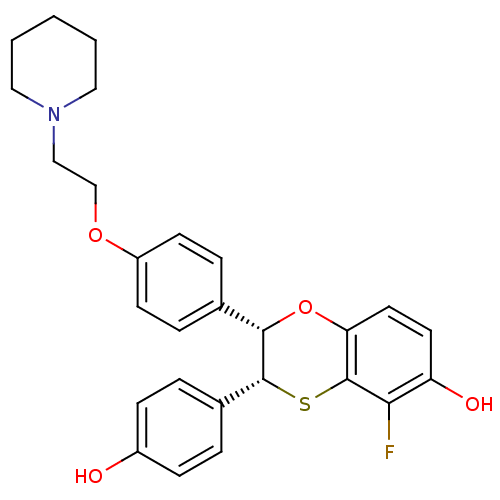
((2S,3R)-5-Fluoro-3-(4-hydroxy-phenyl)-2-[4-(2-pipe...)Show SMILES Oc1ccc(cc1)[C@H]1Sc2c(F)c(O)ccc2O[C@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H28FNO4S/c28-24-22(31)12-13-23-27(24)34-26(19-4-8-20(30)9-5-19)25(33-23)18-6-10-21(11-7-18)32-17-16-29-14-2-1-3-15-29/h4-13,25-26,30-31H,1-3,14-17H2/t25-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor alpha |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50167697

((4aR,9S)-6-Hydroxy-1,4a-dimethyl-1,2,3,4,4a,9,10,1...)Show SMILES CC1(CCC[C@@]2(C)[C@H]1CCc1ccc(O)cc21)C(=O)NC(=O)[C@@]1(C)CCC[C@@]2(C)[C@H]1CCc1ccc(O)cc21 Show InChI InChI=1S/C34H43NO4/c1-31-15-5-17-33(3,27(31)13-9-21-7-11-23(36)19-25(21)31)29(38)35-30(39)34(4)18-6-16-32(2)26-20-24(37)12-8-22(26)10-14-28(32)34/h7-8,11-12,19-20,27-28,36-37H,5-6,9-10,13-18H2,1-4H3,(H,35,38,39)/t27-,28-,31-,32-,33+,34?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration in LXRSPA alpha binding assay |
Bioorg Med Chem Lett 15: 4574-8 (2005)
Article DOI: 10.1016/j.bmcl.2005.06.100
BindingDB Entry DOI: 10.7270/Q2154GKK |
More data for this
Ligand-Target Pair | |
Estrogen receptor
(Homo sapiens (Human)) | BDBM50146261
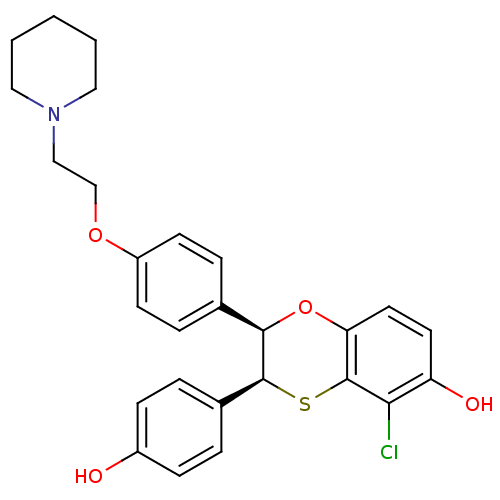
((2R,3S)-5-Chloro-3-(4-hydroxy-phenyl)-2-[4-(2-pipe...)Show SMILES Oc1ccc(cc1)[C@@H]1Sc2c(Cl)c(O)ccc2O[C@@H]1c1ccc(OCCN2CCCCC2)cc1 Show InChI InChI=1S/C27H28ClNO4S/c28-24-22(31)12-13-23-27(24)34-26(19-4-8-20(30)9-5-19)25(33-23)18-6-10-21(11-7-18)32-17-16-29-14-2-1-3-15-29/h4-13,25-26,30-31H,1-3,14-17H2/t25-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-17-beta-estradiol from full length human estrogen receptor beta |
Bioorg Med Chem Lett 14: 2741-5 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.074
BindingDB Entry DOI: 10.7270/Q28P5ZZ1 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50157931
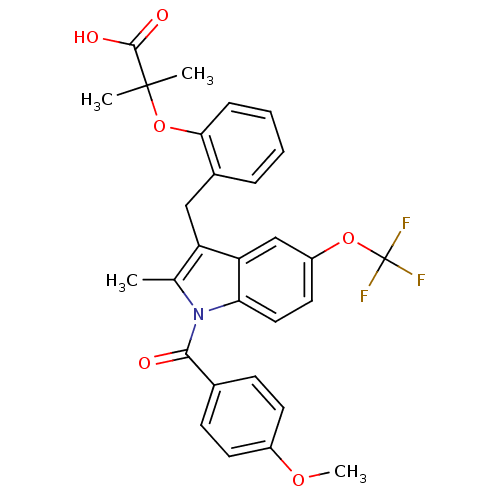
(2-{2-[1-(4-Methoxy-benzoyl)-2-methyl-5-trifluorome...)Show SMILES COc1ccc(cc1)C(=O)n1c(C)c(Cc2ccccc2OC(C)(C)C(O)=O)c2cc(OC(F)(F)F)ccc12 Show InChI InChI=1S/C29H26F3NO6/c1-17-22(15-19-7-5-6-8-25(19)39-28(2,3)27(35)36)23-16-21(38-29(30,31)32)13-14-24(23)33(17)26(34)18-9-11-20(37-4)12-10-18/h5-14,16H,15H2,1-4H3,(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Peroxisome proliferator activated receptor gamma |
Bioorg Med Chem Lett 15: 357-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.068
BindingDB Entry DOI: 10.7270/Q27W6BNP |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50157917

((2S)-2-(3-{[1-(4-METHOXYBENZOYL)-2-METHYL-5-(TRIFL...)Show SMILES COc1ccc(cc1)C(=O)n1c(C)c(Cc2cccc(O[C@@H](C)C(O)=O)c2)c2cc(OC(F)(F)F)ccc12 |r| Show InChI InChI=1S/C28H24F3NO6/c1-16-23(14-18-5-4-6-21(13-18)37-17(2)27(34)35)24-15-22(38-28(29,30)31)11-12-25(24)32(16)26(33)19-7-9-20(36-3)10-8-19/h4-13,15,17H,14H2,1-3H3,(H,34,35)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Peroxisome proliferator activated receptor gamma |
Bioorg Med Chem Lett 15: 357-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.068
BindingDB Entry DOI: 10.7270/Q27W6BNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50157926

(2-{3-[1-(4-Methoxy-benzoyl)-2-methyl-5-trifluorome...)Show SMILES COc1ccc(cc1)C(=O)n1c(C)c(Cc2cccc(OC(C)C(O)=O)c2)c2cc(OC(F)(F)F)ccc12 Show InChI InChI=1S/C28H24F3NO6/c1-16-23(14-18-5-4-6-21(13-18)37-17(2)27(34)35)24-15-22(38-28(29,30)31)11-12-25(24)32(16)26(33)19-7-9-20(36-3)10-8-19/h4-13,15,17H,14H2,1-3H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Peroxisome proliferator activated receptor gamma |
Bioorg Med Chem Lett 15: 357-62 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.068
BindingDB Entry DOI: 10.7270/Q27W6BNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50244702
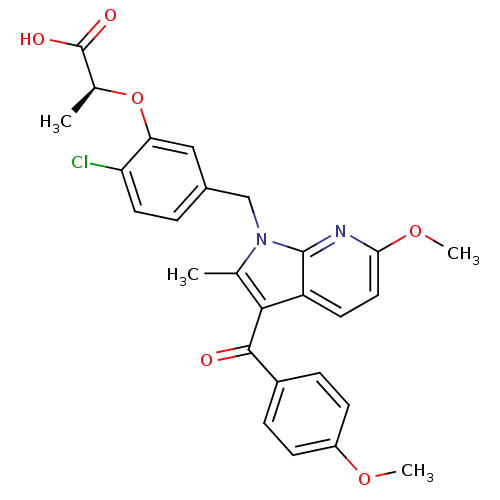
((S)-2-(2-chloro-5-((6-methoxy-3-(4-methoxybenzoyl)...)Show SMILES COc1ccc(cc1)C(=O)c1c(C)n(Cc2ccc(Cl)c(O[C@@H](C)C(O)=O)c2)c2nc(OC)ccc12 |r| Show InChI InChI=1S/C27H25ClN2O6/c1-15-24(25(31)18-6-8-19(34-3)9-7-18)20-10-12-23(35-4)29-26(20)30(15)14-17-5-11-21(28)22(13-17)36-16(2)27(32)33/h5-13,16H,14H2,1-4H3,(H,32,33)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]2AD-5075 from GST-tagged human PPARgamma receptor expressed in Escherichia coli BL21 |
Bioorg Med Chem Lett 18: 4798-801 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.103
BindingDB Entry DOI: 10.7270/Q2BC3ZC5 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50244700
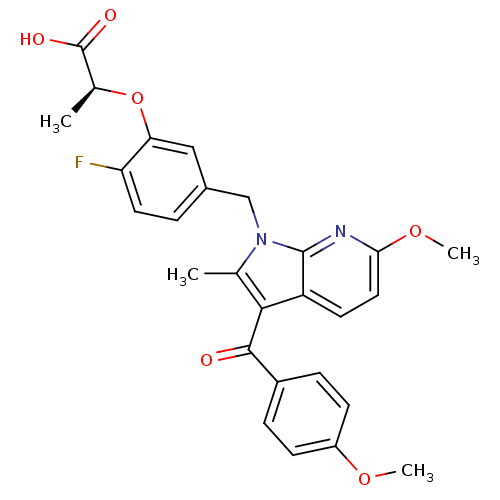
((S)-2-(2-fluoro-5-((6-methoxy-3-(4-methoxybenzoyl)...)Show SMILES COc1ccc(cc1)C(=O)c1c(C)n(Cc2ccc(F)c(O[C@@H](C)C(O)=O)c2)c2nc(OC)ccc12 |r| Show InChI InChI=1S/C27H25FN2O6/c1-15-24(25(31)18-6-8-19(34-3)9-7-18)20-10-12-23(35-4)29-26(20)30(15)14-17-5-11-21(28)22(13-17)36-16(2)27(32)33/h5-13,16H,14H2,1-4H3,(H,32,33)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]2AD-5075 from GST-tagged human PPARgamma receptor expressed in Escherichia coli BL21 |
Bioorg Med Chem Lett 18: 4798-801 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.103
BindingDB Entry DOI: 10.7270/Q2BC3ZC5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data