 Found 486 hits with Last Name = 'coumar' and Initial = 'ms'
Found 486 hits with Last Name = 'coumar' and Initial = 'ms' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aurora kinase A
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase B
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora B kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase C
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora C kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM12985
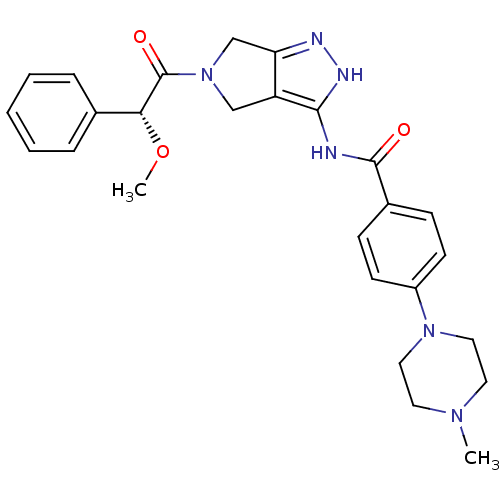
(5-Amido-pyrrolopyrazole 9d | CHEMBL402548 | N-{5-[...)Show SMILES CO[C@@H](C(=O)N1Cc2n[nH]c(NC(=O)c3ccc(cc3)N3CCN(C)CC3)c2C1)c1ccccc1 |r| Show InChI InChI=1S/C26H30N6O3/c1-30-12-14-31(15-13-30)20-10-8-19(9-11-20)25(33)27-24-21-16-32(17-22(21)28-29-24)26(34)23(35-2)18-6-4-3-5-7-18/h3-11,23H,12-17H2,1-2H3,(H2,27,28,29,33)/t23-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | |
Aurora kinase C
(Homo sapiens (Human)) | BDBM12985

(5-Amido-pyrrolopyrazole 9d | CHEMBL402548 | N-{5-[...)Show SMILES CO[C@@H](C(=O)N1Cc2n[nH]c(NC(=O)c3ccc(cc3)N3CCN(C)CC3)c2C1)c1ccccc1 |r| Show InChI InChI=1S/C26H30N6O3/c1-30-12-14-31(15-13-30)20-10-8-19(9-11-20)25(33)27-24-21-16-32(17-22(21)28-29-24)26(34)23(35-2)18-6-4-3-5-7-18/h3-11,23H,12-17H2,1-2H3,(H2,27,28,29,33)/t23-/m1/s1 | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 61 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora C kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM12985

(5-Amido-pyrrolopyrazole 9d | CHEMBL402548 | N-{5-[...)Show SMILES CO[C@@H](C(=O)N1Cc2n[nH]c(NC(=O)c3ccc(cc3)N3CCN(C)CC3)c2C1)c1ccccc1 |r| Show InChI InChI=1S/C26H30N6O3/c1-30-12-14-31(15-13-30)20-10-8-19(9-11-20)25(33)27-24-21-16-32(17-22(21)28-29-24)26(34)23(35-2)18-6-4-3-5-7-18/h3-11,23H,12-17H2,1-2H3,(H2,27,28,29,33)/t23-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora B kinase |
Bioorg Med Chem Lett 18: 1623-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.068
BindingDB Entry DOI: 10.7270/Q261115V |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM12178
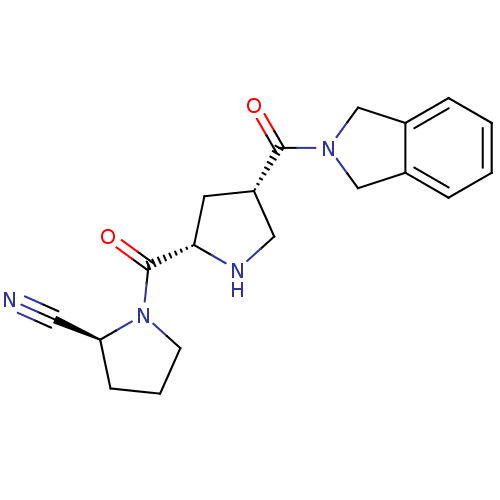
((2S)-1-{[(2S,4S)-4-(2,3-dihydro-1H-isoindol-2-ylca...)Show SMILES O=C([C@@H]1CN[C@@H](C1)C(=O)N1CCC[C@H]1C#N)N1Cc2ccccc2C1 |r| Show InChI InChI=1S/C19H22N4O2/c20-9-16-6-3-7-23(16)19(25)17-8-15(10-21-17)18(24)22-11-13-4-1-2-5-14(13)12-22/h1-2,4-5,15-17,21H,3,6-8,10-12H2/t15-,16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
Bioorg Med Chem Lett 16: 3268-72 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.037
BindingDB Entry DOI: 10.7270/Q22805V8 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50322822
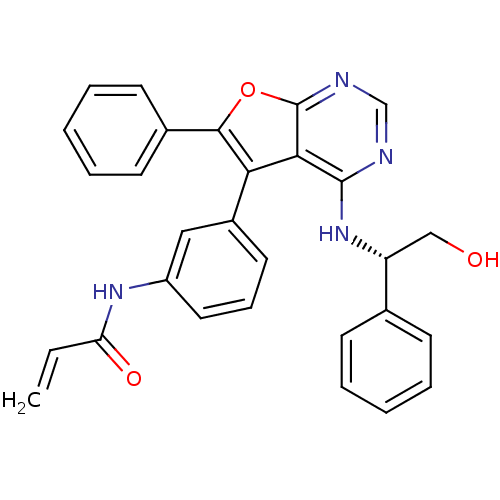
((S)-N-(3-(4-(2-hydroxy-1-phenylethylamino)-6-pheny...)Show SMILES OC[C@@H](Nc1ncnc2oc(c(-c3cccc(NC(=O)C=C)c3)c12)-c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C29H24N4O3/c1-2-24(35)32-22-15-9-14-21(16-22)25-26-28(33-23(17-34)19-10-5-3-6-11-19)30-18-31-29(26)36-27(25)20-12-7-4-8-13-20/h2-16,18,23,34H,1,17H2,(H,32,35)(H,30,31,33)/t23-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged EGFR expressed in Escherichia coli |
J Med Chem 53: 4980-8 (2010)
Checked by Author
Article DOI: 10.1021/jm1000198
BindingDB Entry DOI: 10.7270/Q2Z60P87 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330230
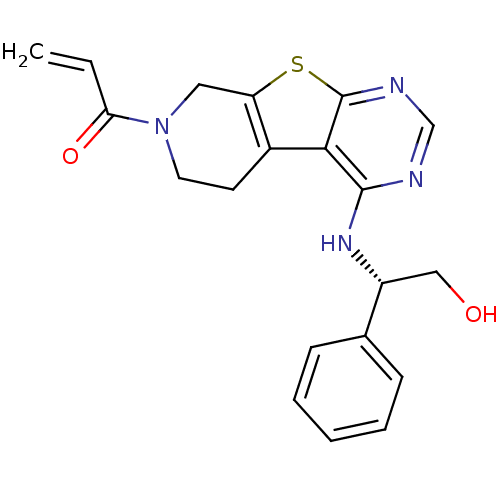
(1-(4-(((1S)-2-Hydroxy-1-phenylethyl)amino)-5,6,7,8...)Show SMILES OC[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)C=C)c1ccccc1 |r| Show InChI InChI=1S/C20H20N4O2S/c1-2-17(26)24-9-8-14-16(10-24)27-20-18(14)19(21-12-22-20)23-15(11-25)13-6-4-3-5-7-13/h2-7,12,15,25H,1,8-11H2,(H,21,22,23)/t15-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50432686
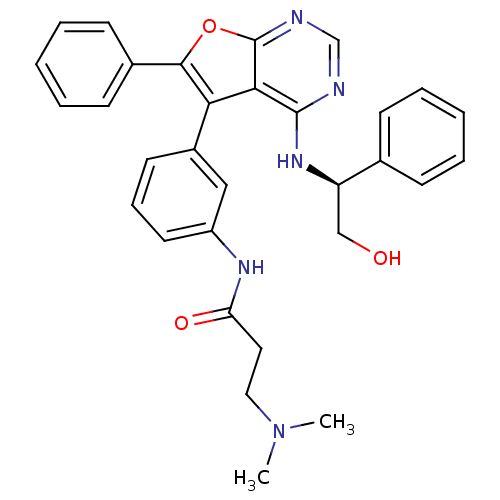
(CHEMBL2347963)Show SMILES CN(C)CCC(=O)Nc1cccc(c1)-c1c(oc2ncnc(N[C@H](CO)c3ccccc3)c12)-c1ccccc1 |r| Show InChI InChI=1S/C31H31N5O3/c1-36(2)17-16-26(38)34-24-15-9-14-23(18-24)27-28-30(35-25(19-37)21-10-5-3-6-11-21)32-20-33-31(28)39-29(27)22-12-7-4-8-13-22/h3-15,18,20,25,37H,16-17,19H2,1-2H3,(H,34,38)(H,32,33,35)/t25-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330238

((E)-4-(Diethylamino)-1-(4-(((1R)-2-hydroxy-1-pheny...)Show SMILES CCN(CC)C\C=C\C(=O)N1CCc2c(C1)sc1ncnc(N[C@H](CO)c3ccccc3)c21 |r| Show InChI InChI=1S/C25H31N5O2S/c1-3-29(4-2)13-8-11-22(32)30-14-12-19-21(15-30)33-25-23(19)24(26-17-27-25)28-20(16-31)18-9-6-5-7-10-18/h5-11,17,20,31H,3-4,12-16H2,1-2H3,(H,26,27,28)/b11-8+/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50322823

((S)-N-(4-(3-chloro-4-fluorophenylamino)-7-(tetrahy...)Show SMILES CN(C)C\C=C\C(=O)Nc1cc2c(Nc3ccc(F)c(Cl)c3)ncnc2cc1O[C@H]1CCOC1 |r| Show InChI InChI=1S/C24H25ClFN5O3/c1-31(2)8-3-4-23(32)30-21-11-17-20(12-22(21)34-16-7-9-33-13-16)27-14-28-24(17)29-15-5-6-19(26)18(25)10-15/h3-6,10-12,14,16H,7-9,13H2,1-2H3,(H,30,32)(H,27,28,29)/b4-3+/t16-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged EGFR expressed in Escherichia coli |
J Med Chem 53: 4980-8 (2010)
Checked by Author
Article DOI: 10.1021/jm1000198
BindingDB Entry DOI: 10.7270/Q2Z60P87 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330242
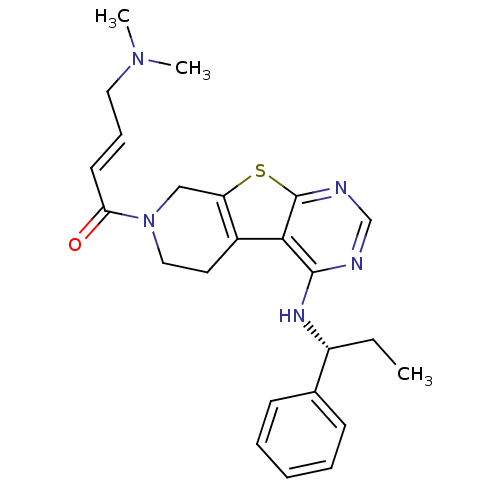
((E)-4-(Dimethylamino)-1-(4-[(1S)-1-phenylpropyl]am...)Show SMILES CC[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)\C=C\CN(C)C)c1ccccc1 |r| Show InChI InChI=1S/C24H29N5OS/c1-4-19(17-9-6-5-7-10-17)27-23-22-18-12-14-29(21(30)11-8-13-28(2)3)15-20(18)31-24(22)26-16-25-23/h5-11,16,19H,4,12-15H2,1-3H3,(H,25,26,27)/b11-8+/t19-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330241
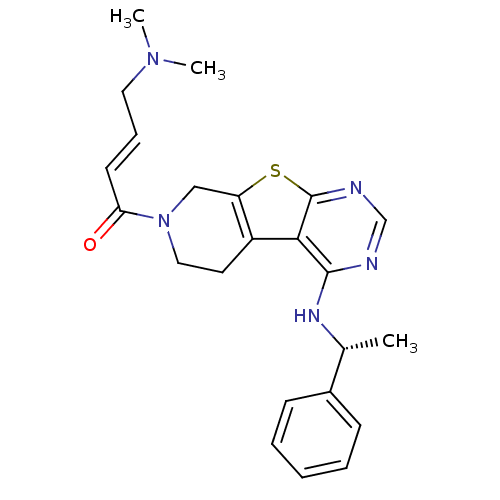
((E)-4-(Dimethylamino)-1-(4-[(1R)-1-phenylethyl]ami...)Show SMILES C[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)\C=C\CN(C)C)c1ccccc1 |r| Show InChI InChI=1S/C23H27N5OS/c1-16(17-8-5-4-6-9-17)26-22-21-18-11-13-28(20(29)10-7-12-27(2)3)14-19(18)30-23(21)25-15-24-22/h4-10,15-16H,11-14H2,1-3H3,(H,24,25,26)/b10-7+/t16-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11694

((2S)-1-[(2S)-2-amino-2-cyclohexylacetyl]pyrrolidin...)Show InChI InChI=1S/C13H21N3O/c14-9-11-7-4-8-16(11)13(17)12(15)10-5-2-1-3-6-10/h10-12H,1-8,15H2/t11-,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11719
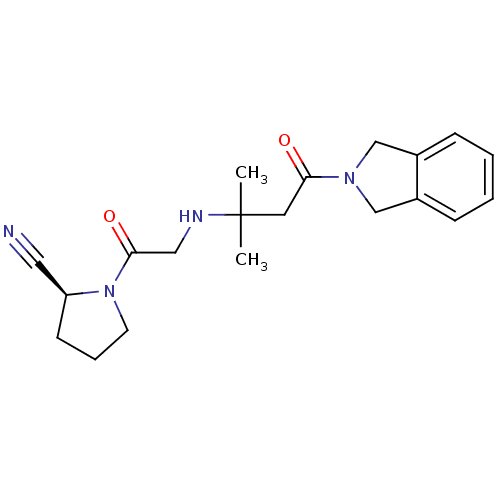
((2S)-1-(2-{[4-(2,3-dihydro-1H-isoindol-2-yl)-2-met...)Show SMILES CC(C)(CC(=O)N1Cc2ccccc2C1)NCC(=O)N1CCC[C@H]1C#N |r| Show InChI InChI=1S/C20H26N4O2/c1-20(2,22-12-19(26)24-9-5-8-17(24)11-21)10-18(25)23-13-15-6-3-4-7-16(15)14-23/h3-4,6-7,17,22H,5,8-10,12-14H2,1-2H3/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11719

((2S)-1-(2-{[4-(2,3-dihydro-1H-isoindol-2-yl)-2-met...)Show SMILES CC(C)(CC(=O)N1Cc2ccccc2C1)NCC(=O)N1CCC[C@H]1C#N |r| Show InChI InChI=1S/C20H26N4O2/c1-20(2,22-12-19(26)24-9-5-8-17(24)11-21)10-18(25)23-13-15-6-3-4-7-16(15)14-23/h3-4,6-7,17,22H,5,8-10,12-14H2,1-2H3/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
Bioorg Med Chem Lett 16: 3268-72 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.037
BindingDB Entry DOI: 10.7270/Q22805V8 |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50436695
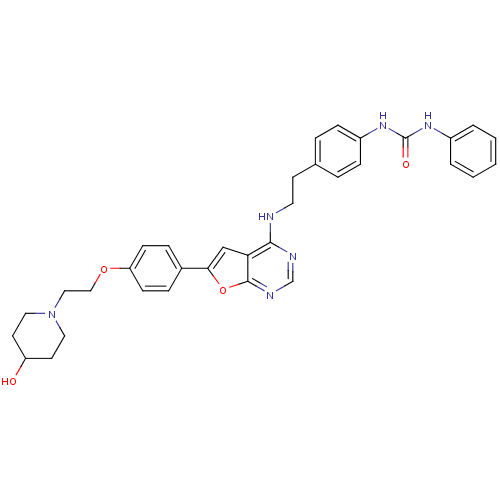
(CHEMBL2398642)Show SMILES OC1CCN(CCOc2ccc(cc2)-c2cc3c(NCCc4ccc(NC(=O)Nc5ccccc5)cc4)ncnc3o2)CC1 Show InChI InChI=1S/C34H36N6O4/c41-28-15-18-40(19-16-28)20-21-43-29-12-8-25(9-13-29)31-22-30-32(36-23-37-33(30)44-31)35-17-14-24-6-10-27(11-7-24)39-34(42)38-26-4-2-1-3-5-26/h1-13,22-23,28,41H,14-21H2,(H,35,36,37)(H2,38,39,42) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase B (unknown origin) |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436696
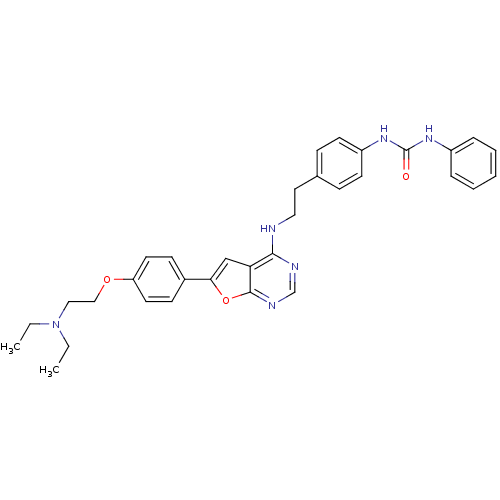
(CHEMBL2398658)Show SMILES CCN(CC)CCOc1ccc(cc1)-c1cc2c(NCCc3ccc(NC(=O)Nc4ccccc4)cc3)ncnc2o1 Show InChI InChI=1S/C33H36N6O3/c1-3-39(4-2)20-21-41-28-16-12-25(13-17-28)30-22-29-31(35-23-36-32(29)42-30)34-19-18-24-10-14-27(15-11-24)38-33(40)37-26-8-6-5-7-9-26/h5-17,22-23H,3-4,18-21H2,1-2H3,(H,34,35,36)(H2,37,38,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged Aurora kinase A catalytic domain (123 to 401 amino acids) (unknown origin) expressed in sf9 cells using tetra(LRRWSLG) as su... |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5447
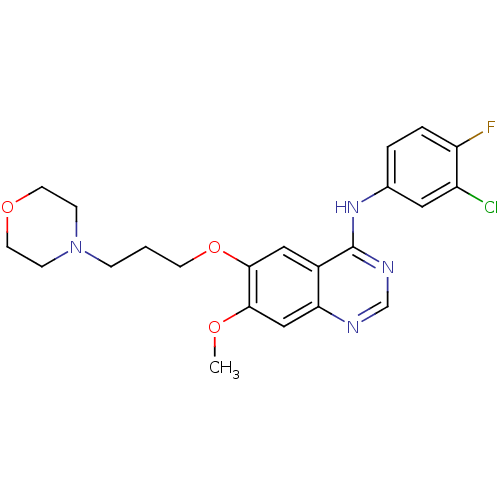
(CHEMBL939 | GEFITINIB | Iressa | N-(3-Chloro-4-flu...)Show SMILES COc1cc2ncnc(Nc3ccc(F)c(Cl)c3)c2cc1OCCCN1CCOCC1 Show InChI InChI=1S/C22H24ClFN4O3/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged EGFR expressed in Escherichia coli |
J Med Chem 53: 4980-8 (2010)
Checked by Author
Article DOI: 10.1021/jm1000198
BindingDB Entry DOI: 10.7270/Q2Z60P87 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A |
J Med Chem 53: 4980-8 (2010)
Checked by Author
Article DOI: 10.1021/jm1000198
BindingDB Entry DOI: 10.7270/Q2Z60P87 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged Aurora kinase A catalytic domain (123 to 401 amino acids) (unknown origin) expressed in sf9 cells using tetra(LRRWSLG) as su... |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A (unknown origin) |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330234

(1-(4-[(1R)-1-Phenylethyl]amino-5,6,7,8-tetrahydrop...)Show SMILES C[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)C=C)c1ccccc1 |r| Show InChI InChI=1S/C20H20N4OS/c1-3-17(25)24-10-9-15-16(11-24)26-20-18(15)19(21-12-22-20)23-13(2)14-7-5-4-6-8-14/h3-8,12-13H,1,9-11H2,2H3,(H,21,22,23)/t13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5447

(CHEMBL939 | GEFITINIB | Iressa | N-(3-Chloro-4-flu...)Show SMILES COc1cc2ncnc(Nc3ccc(F)c(Cl)c3)c2cc1OCCCN1CCOCC1 Show InChI InChI=1S/C22H24ClFN4O3/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330239
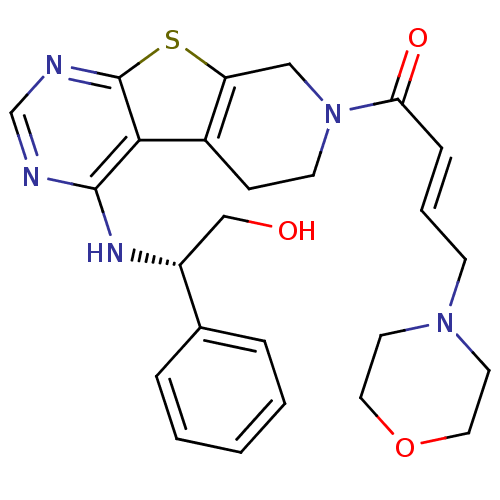
((E)-1-(4-[(1R)-2-Hydroxy-1-phenylethyl]amino-5,6,7...)Show SMILES OC[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)\C=C\CN1CCOCC1)c1ccccc1 |r| Show InChI InChI=1S/C25H29N5O3S/c31-16-20(18-5-2-1-3-6-18)28-24-23-19-8-10-30(15-21(19)34-25(23)27-17-26-24)22(32)7-4-9-29-11-13-33-14-12-29/h1-7,17,20,31H,8-16H2,(H,26,27,28)/b7-4+/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11718
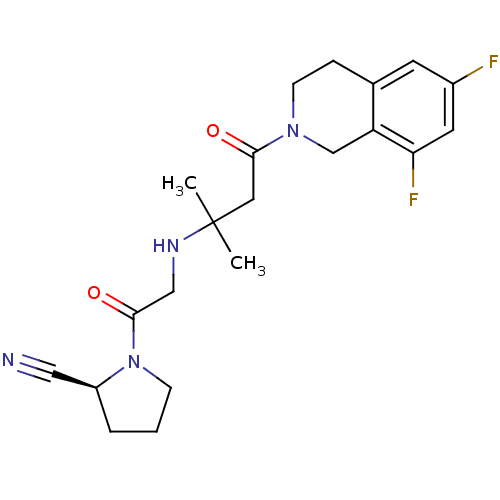
((2S)-1-(2-{[4-(6,8-difluoro-1,2,3,4-tetrahydroisoq...)Show SMILES CC(C)(CC(=O)N1CCc2cc(F)cc(F)c2C1)NCC(=O)N1CCC[C@H]1C#N |r| Show InChI InChI=1S/C21H26F2N4O2/c1-21(2,25-12-20(29)27-6-3-4-16(27)11-24)10-19(28)26-7-5-14-8-15(22)9-18(23)17(14)13-26/h8-9,16,25H,3-7,10,12-13H2,1-2H3/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436691

(CHEMBL2398657)Show SMILES CN(C)CCOc1ccc(cc1)-c1cc2c(NCCc3ccc(NC(=O)Nc4ccccc4)cc3)ncnc2o1 Show InChI InChI=1S/C31H32N6O3/c1-37(2)18-19-39-26-14-10-23(11-15-26)28-20-27-29(33-21-34-30(27)40-28)32-17-16-22-8-12-25(13-9-22)36-31(38)35-24-6-4-3-5-7-24/h3-15,20-21H,16-19H2,1-2H3,(H,32,33,34)(H2,35,36,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A (unknown origin) |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 8
(Homo sapiens (Human)) | BDBM11694

((2S)-1-[(2S)-2-amino-2-cyclohexylacetyl]pyrrolidin...)Show InChI InChI=1S/C13H21N3O/c14-9-11-7-4-8-16(11)13(17)12(15)10-5-2-1-3-6-10/h10-12H,1-8,15H2/t11-,12-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50432685
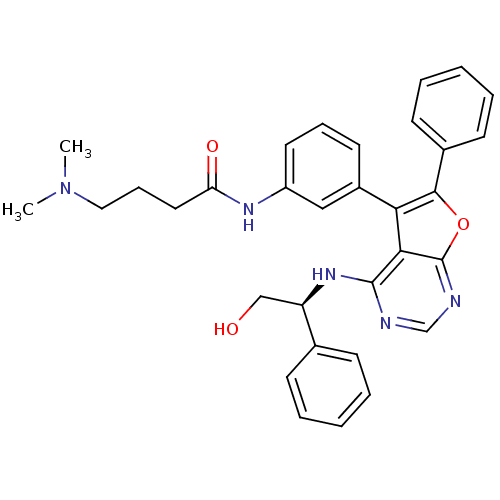
(CHEMBL2347960)Show SMILES CN(C)CCCC(=O)Nc1cccc(c1)-c1c(oc2ncnc(N[C@H](CO)c3ccccc3)c12)-c1ccccc1 |r| Show InChI InChI=1S/C32H33N5O3/c1-37(2)18-10-17-27(39)35-25-16-9-15-24(19-25)28-29-31(36-26(20-38)22-11-5-3-6-12-22)33-21-34-32(29)40-30(28)23-13-7-4-8-14-23/h3-9,11-16,19,21,26,38H,10,17-18,20H2,1-2H3,(H,35,39)(H,33,34,36)/t26-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase B
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase B (unknown origin) |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11162
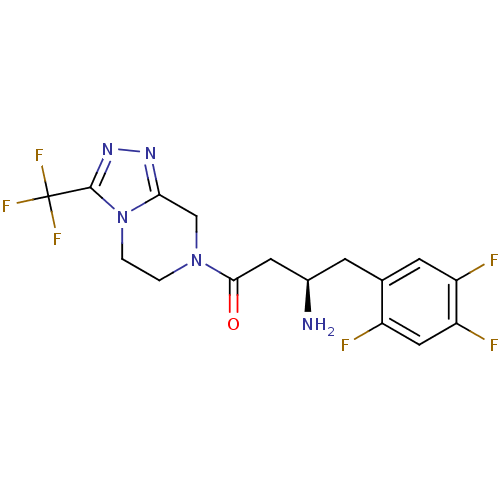
((1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4...)Show SMILES N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
Bioorg Med Chem Lett 16: 3268-72 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.037
BindingDB Entry DOI: 10.7270/Q22805V8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11717
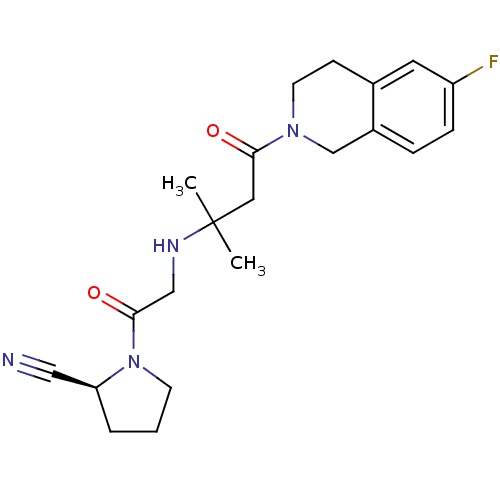
((2S)-1-(2-{[4-(6-fluoro-1,2,3,4-tetrahydroisoquino...)Show SMILES CC(C)(CC(=O)N1CCc2cc(F)ccc2C1)NCC(=O)N1CCC[C@H]1C#N |r| Show InChI InChI=1S/C21H27FN4O2/c1-21(2,24-13-20(28)26-8-3-4-18(26)12-23)11-19(27)25-9-7-15-10-17(22)6-5-16(15)14-25/h5-6,10,18,24H,3-4,7-9,11,13-14H2,1-2H3/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5447

(CHEMBL939 | GEFITINIB | Iressa | N-(3-Chloro-4-flu...)Show SMILES COc1cc2ncnc(Nc3ccc(F)c(Cl)c3)c2cc1OCCCN1CCOCC1 Show InChI InChI=1S/C22H24ClFN4O3/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50479526
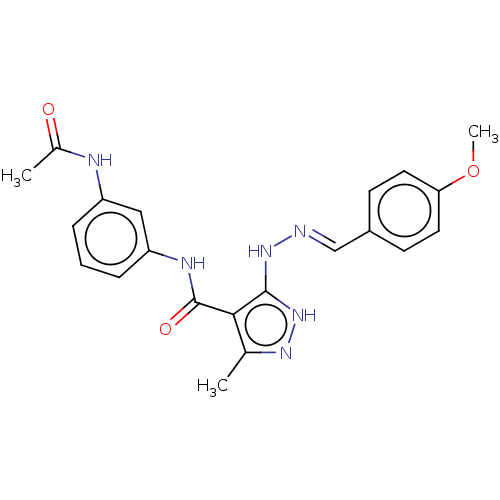
(CHEMBL515560)Show SMILES COc1ccc(\C=N\Nc2[nH]nc(C)c2C(=O)Nc2cccc(NC(C)=O)c2)cc1 Show InChI InChI=1S/C21H22N6O3/c1-13-19(21(29)24-17-6-4-5-16(11-17)23-14(2)28)20(27-25-13)26-22-12-15-7-9-18(30-3)10-8-15/h4-12H,1-3H3,(H,23,28)(H,24,29)(H2,25,26,27)/b22-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A T288D mutant expressed in Escherichia coli BL21 DE3 |
J Med Chem 52: 1050-62 (2009)
Article DOI: 10.1021/jm801270e
BindingDB Entry DOI: 10.7270/Q27M0BQJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50432684
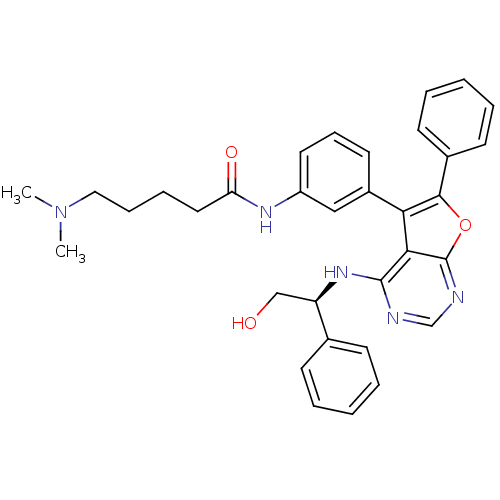
(CHEMBL2347961)Show SMILES CN(C)CCCCC(=O)Nc1cccc(c1)-c1c(oc2ncnc(N[C@H](CO)c3ccccc3)c12)-c1ccccc1 |r| Show InChI InChI=1S/C33H35N5O3/c1-38(2)19-10-9-18-28(40)36-26-17-11-16-25(20-26)29-30-32(37-27(21-39)23-12-5-3-6-13-23)34-22-35-33(30)41-31(29)24-14-7-4-8-15-24/h3-8,11-17,20,22,27,39H,9-10,18-19,21H2,1-2H3,(H,36,40)(H,34,35,37)/t27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50432681
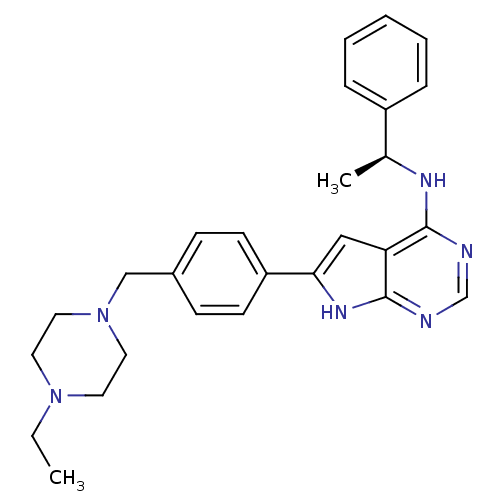
(CHEMBL484270)Show SMILES CCN1CCN(Cc2ccc(cc2)-c2cc3c(N[C@@H](C)c4ccccc4)ncnc3[nH]2)CC1 |r| Show InChI InChI=1S/C27H32N6/c1-3-32-13-15-33(16-14-32)18-21-9-11-23(12-10-21)25-17-24-26(28-19-29-27(24)31-25)30-20(2)22-7-5-4-6-8-22/h4-12,17,19-20H,3,13-16,18H2,1-2H3,(H2,28,29,30,31)/t20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436695

(CHEMBL2398642)Show SMILES OC1CCN(CCOc2ccc(cc2)-c2cc3c(NCCc4ccc(NC(=O)Nc5ccccc5)cc4)ncnc3o2)CC1 Show InChI InChI=1S/C34H36N6O4/c41-28-15-18-40(19-16-28)20-21-43-29-12-8-25(9-13-29)31-22-30-32(36-23-37-33(30)44-31)35-17-14-24-6-10-27(11-7-24)39-34(42)38-26-4-2-1-3-5-26/h1-13,22-23,28,41H,14-21H2,(H,35,36,37)(H2,38,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora kinase A (unknown origin) |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50201594
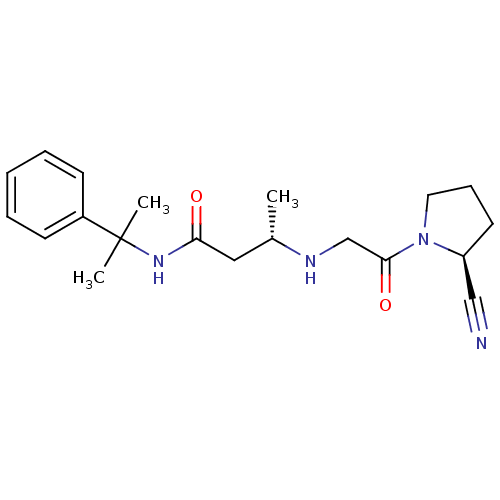
((S)-3-(2-((S)-2-cyanopyrrolidin-1-yl)-2-oxoethylam...)Show SMILES C[C@@H](CC(=O)NC(C)(C)c1ccccc1)NCC(=O)N1CCC[C@H]1C#N Show InChI InChI=1S/C20H28N4O2/c1-15(22-14-19(26)24-11-7-10-17(24)13-21)12-18(25)23-20(2,3)16-8-5-4-6-9-16/h4-6,8-9,15,17,22H,7,10-12,14H2,1-3H3,(H,23,25)/t15-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
In vitro inhibition of DPP-4 |
Bioorg Med Chem Lett 17: 1274-9 (2007)
Article DOI: 10.1016/j.bmcl.2006.12.019
BindingDB Entry DOI: 10.7270/Q23T9GV7 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50322816
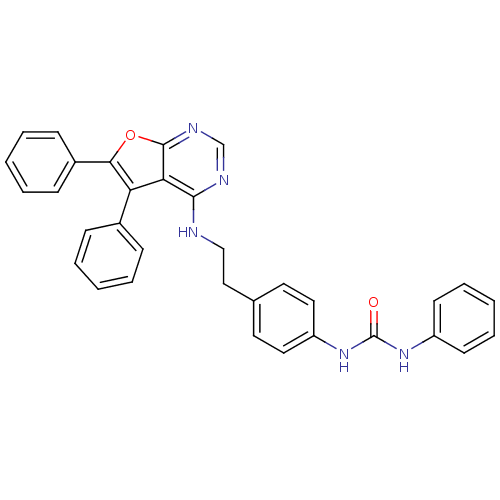
(1-{4-[2-(5,6-Diphenylfuro[2,3-d]pyrimidin-4-ylamin...)Show SMILES O=C(Nc1ccccc1)Nc1ccc(CCNc2ncnc3oc(c(-c4ccccc4)c23)-c2ccccc2)cc1 Show InChI InChI=1S/C33H27N5O2/c39-33(37-26-14-8-3-9-15-26)38-27-18-16-23(17-19-27)20-21-34-31-29-28(24-10-4-1-5-11-24)30(25-12-6-2-7-13-25)40-32(29)36-22-35-31/h1-19,22H,20-21H2,(H,34,35,36)(H2,37,38,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A |
J Med Chem 53: 4980-8 (2010)
Checked by Author
Article DOI: 10.1021/jm1000198
BindingDB Entry DOI: 10.7270/Q2Z60P87 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50330235
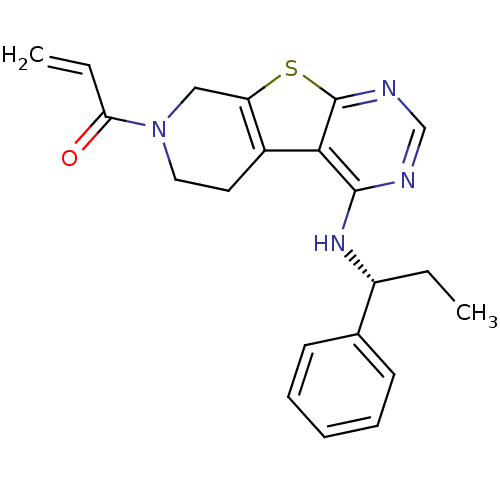
(1-(4-[(1R)-1-Phenylpropyl]amino-5,6,7,8-tetrahydro...)Show SMILES CC[C@@H](Nc1ncnc2sc3CN(CCc3c12)C(=O)C=C)c1ccccc1 |r| Show InChI InChI=1S/C21H22N4OS/c1-3-16(14-8-6-5-7-9-14)24-20-19-15-10-11-25(18(26)4-2)12-17(15)27-21(19)23-13-22-20/h4-9,13,16H,2-3,10-12H2,1H3,(H,22,23,24)/t16-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
J Med Chem 53: 7316-26 (2010)
Article DOI: 10.1021/jm100607r
BindingDB Entry DOI: 10.7270/Q26D5T64 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50432687
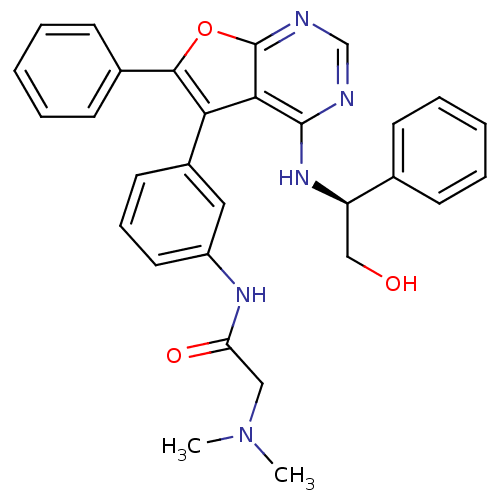
(CHEMBL2347966)Show SMILES CN(C)CC(=O)Nc1cccc(c1)-c1c(oc2ncnc(N[C@H](CO)c3ccccc3)c12)-c1ccccc1 |r| Show InChI InChI=1S/C30H29N5O3/c1-35(2)17-25(37)33-23-15-9-14-22(16-23)26-27-29(34-24(18-36)20-10-5-3-6-11-20)31-19-32-30(27)38-28(26)21-12-7-4-8-13-21/h3-16,19,24,36H,17-18H2,1-2H3,(H,33,37)(H,31,32,34)/t24-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of human wild type GST-tagged EGFR kinase domain expressed in Sf9 cells by luminescence assay |
J Med Chem 56: 3889-903 (2013)
Article DOI: 10.1021/jm400072p
BindingDB Entry DOI: 10.7270/Q27W6DJ5 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436694
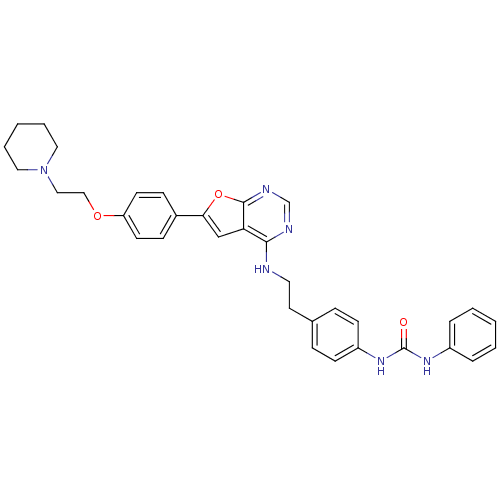
(CHEMBL2398638)Show SMILES O=C(Nc1ccccc1)Nc1ccc(CCNc2ncnc3oc(cc23)-c2ccc(OCCN3CCCCC3)cc2)cc1 Show InChI InChI=1S/C34H36N6O3/c41-34(38-27-7-3-1-4-8-27)39-28-13-9-25(10-14-28)17-18-35-32-30-23-31(43-33(30)37-24-36-32)26-11-15-29(16-12-26)42-22-21-40-19-5-2-6-20-40/h1,3-4,7-16,23-24H,2,5-6,17-22H2,(H,35,36,37)(H2,38,39,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged Aurora kinase A catalytic domain (123 to 401 amino acids) (unknown origin) expressed in sf9 cells using tetra(LRRWSLG) as su... |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436693
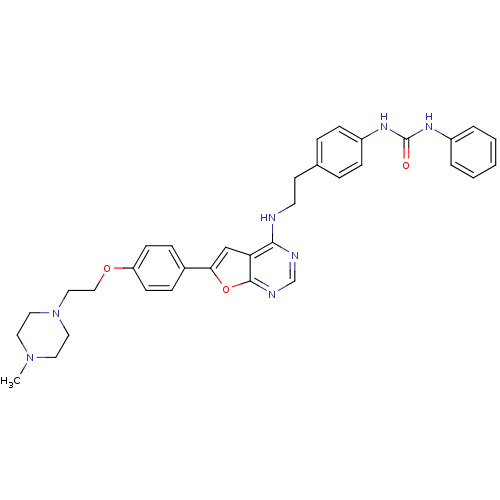
(CHEMBL2398639)Show SMILES CN1CCN(CCOc2ccc(cc2)-c2cc3c(NCCc4ccc(NC(=O)Nc5ccccc5)cc4)ncnc3o2)CC1 Show InChI InChI=1S/C34H37N7O3/c1-40-17-19-41(20-18-40)21-22-43-29-13-9-26(10-14-29)31-23-30-32(36-24-37-33(30)44-31)35-16-15-25-7-11-28(12-8-25)39-34(42)38-27-5-3-2-4-6-27/h2-14,23-24H,15-22H2,1H3,(H,35,36,37)(H2,38,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged Aurora kinase A catalytic domain (123 to 401 amino acids) (unknown origin) expressed in sf9 cells using tetra(LRRWSLG) as su... |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11716

((2S)-1-(2-{[2-methyl-4-oxo-4-(1,2,3,4-tetrahydrois...)Show SMILES CC(C)(CC(=O)N1CCc2ccccc2C1)NCC(=O)N1CCC[C@H]1C#N |r| Show InChI InChI=1S/C21H28N4O2/c1-21(2,23-14-20(27)25-10-5-8-18(25)13-22)12-19(26)24-11-9-16-6-3-4-7-17(16)15-24/h3-4,6-7,18,23H,5,8-12,14-15H2,1-2H3/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM12167

(5-chloro-2-{[(3S,5S)-5-(pyrrolidin-1-ylcarbonyl)py...)Show SMILES Clc1ccc2CN(Cc2c1)C(=O)[C@@H]1CN[C@@H](C1)C(=O)N1CCCC1 |r| Show InChI InChI=1S/C18H22ClN3O2/c19-15-4-3-12-10-22(11-14(12)7-15)17(23)13-8-16(20-9-13)18(24)21-5-1-2-6-21/h3-4,7,13,16,20H,1-2,5-6,8-11H2/t13-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
Bioorg Med Chem Lett 16: 3268-72 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.037
BindingDB Entry DOI: 10.7270/Q22805V8 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50436692
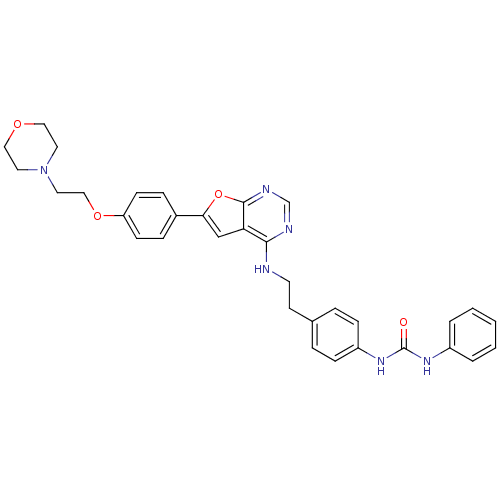
(CHEMBL2398640)Show SMILES O=C(Nc1ccccc1)Nc1ccc(CCNc2ncnc3oc(cc23)-c2ccc(OCCN3CCOCC3)cc2)cc1 Show InChI InChI=1S/C33H34N6O4/c40-33(37-26-4-2-1-3-5-26)38-27-10-6-24(7-11-27)14-15-34-31-29-22-30(43-32(29)36-23-35-31)25-8-12-28(13-9-25)42-21-18-39-16-19-41-20-17-39/h1-13,22-23H,14-21H2,(H,34,35,36)(H2,37,38,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
National Health Research Institutes
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged Aurora kinase A catalytic domain (123 to 401 amino acids) (unknown origin) expressed in sf9 cells using tetra(LRRWSLG) as su... |
J Med Chem 56: 5247-60 (2014)
Article DOI: 10.1021/jm4006059
BindingDB Entry DOI: 10.7270/Q2J104K6 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11695
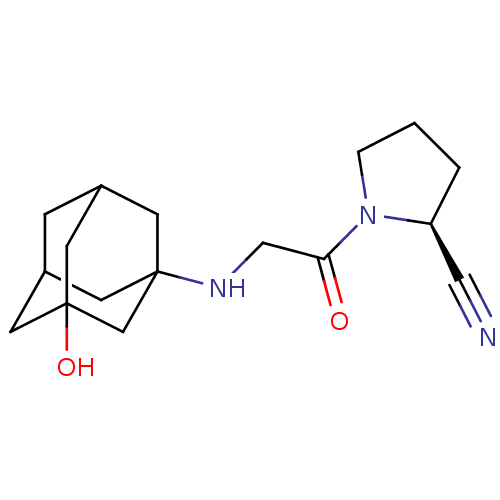
((2S)-1-{2-[(3-hydroxyadamantan-1-yl)amino]acetyl}p...)Show SMILES OC12CC3CC(C1)CC(C3)(C2)NCC(=O)N1CCC[C@H]1C#N |r,TLB:9:8:6:3.2.4,4:3:10:7.6.5,4:5:10:3.2.9,THB:9:3:6:10.7.8,11:8:6:3.2.4| Show InChI InChI=1S/C17H25N3O2/c18-9-14-2-1-3-20(14)15(21)10-19-16-5-12-4-13(6-16)8-17(22,7-12)11-16/h12-14,19,22H,1-8,10-11H2/t12?,13?,14-,16?,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
Bioorg Med Chem Lett 16: 3268-72 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.037
BindingDB Entry DOI: 10.7270/Q22805V8 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11695

((2S)-1-{2-[(3-hydroxyadamantan-1-yl)amino]acetyl}p...)Show SMILES OC12CC3CC(C1)CC(C3)(C2)NCC(=O)N1CCC[C@H]1C#N |r,TLB:9:8:6:3.2.4,4:3:10:7.6.5,4:5:10:3.2.9,THB:9:3:6:10.7.8,11:8:6:3.2.4| Show InChI InChI=1S/C17H25N3O2/c18-9-14-2-1-3-20(14)15(21)10-19-16-5-12-4-13(6-16)8-17(22,7-12)11-16/h12-14,19,22H,1-8,10-11H2/t12?,13?,14-,16?,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11113
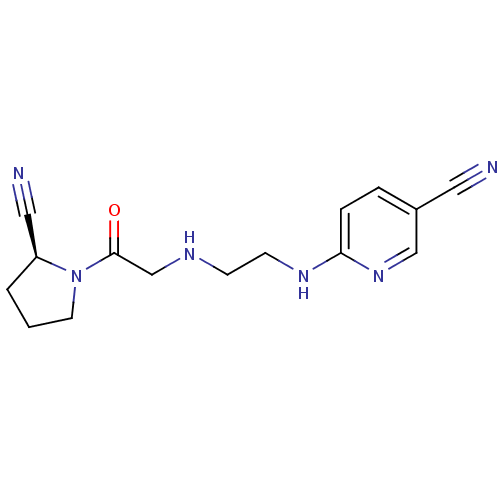
(6-{[2-({2-[(2S)-2-cyanopyrrolidin-1-yl]-2-oxoethyl...)Show InChI InChI=1S/C15H18N6O/c16-8-12-3-4-14(20-10-12)19-6-5-18-11-15(22)21-7-1-2-13(21)9-17/h3-4,10,13,18H,1-2,5-7,11H2,(H,19,20)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 53 | n/a | n/a | n/a | n/a | 8.0 | 37 |
National Health Research Institutes
| Assay Description
The enzyme activity resulted in the liberation of free pNA at 405 nm. Reaction progress was monitored using a Molecular Devices SpectraMax Plus micro... |
J Med Chem 49: 373-80 (2006)
Article DOI: 10.1021/jm0507781
BindingDB Entry DOI: 10.7270/Q28W3BJH |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data