

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092393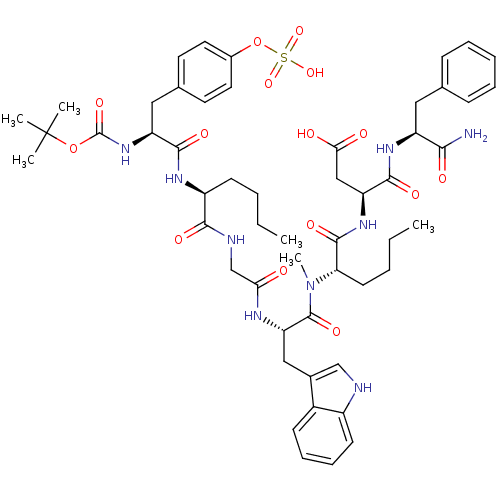 (3-(2-{[2-(2-{2-[2-tert-Butoxycarbonylamino-3-(4-su...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092405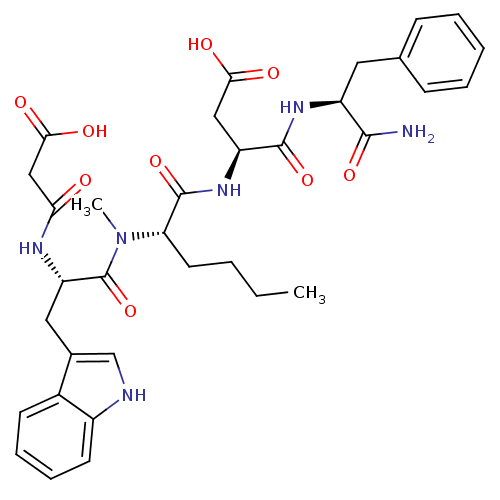 ((S)-N-((S)-1-Carbamoyl-2-phenyl-ethyl)-3-((S)-2-{[...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM21147 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (RAT) | BDBM50092393 (3-(2-{[2-(2-{2-[2-tert-Butoxycarbonylamino-3-(4-su...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for binding affinity against Cholecystokinin type B receptor expressed in CHO cells on the rat brain. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50449787 (CHEMBL2062154 | PD-134308) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 40: 3947-56 (1998) Article DOI: 10.1021/jm970439a BindingDB Entry DOI: 10.7270/Q27H1K8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50061266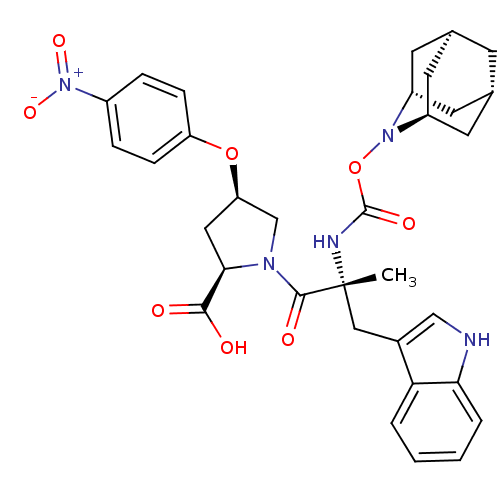 ((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells | J Med Chem 40: 3947-56 (1998) Article DOI: 10.1021/jm970439a BindingDB Entry DOI: 10.7270/Q27H1K8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092408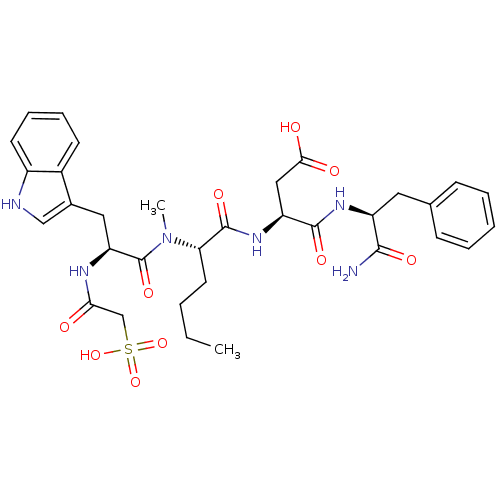 (CHEMBL120150 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (RAT) | BDBM21147 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for binding affinity against Cholecystokinin type B receptor expressed in CHO cells on the rat brain. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092398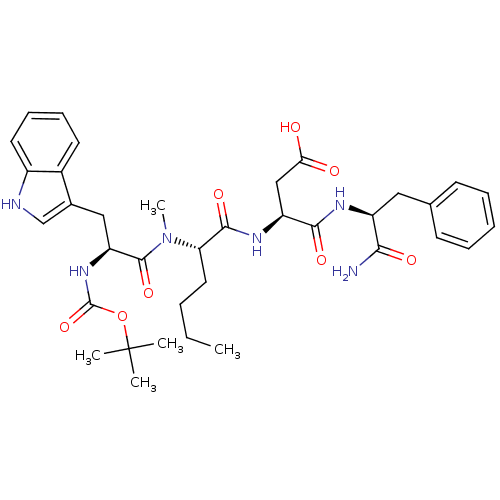 ((S)-3-((S)-2-{[(S)-2-tert-Butoxycarbonylamino-3-(1...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092405 ((S)-N-((S)-1-Carbamoyl-2-phenyl-ethyl)-3-((S)-2-{[...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | >0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM21147 ((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for binding affinity against CCK1 (cholecystokinin) receptor on guinea pig pancreatic membranes | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (RAT) | BDBM50092405 ((S)-N-((S)-1-Carbamoyl-2-phenyl-ethyl)-3-((S)-2-{[...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for binding affinity against Cholecystokinin type B receptor expressed in CHO cells on the rat brain. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092398 ((S)-3-((S)-2-{[(S)-2-tert-Butoxycarbonylamino-3-(1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamyl aminopeptidase (Homo sapiens (Human)) | BDBM50083386 (1-{[1-(2,3-Dicarboxy-pyrrolidine-1-carbonyl)-2-met...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.873 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against recombinant Aminopeptidase A | J Med Chem 42: 5197-211 (2000) BindingDB Entry DOI: 10.7270/Q2028S8F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087104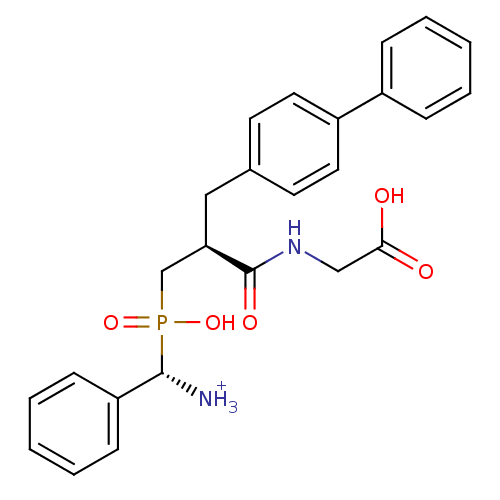 (C-{[3-Biphenyl-4-yl-2-(carboxymethyl-carbamoyl)-pr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087106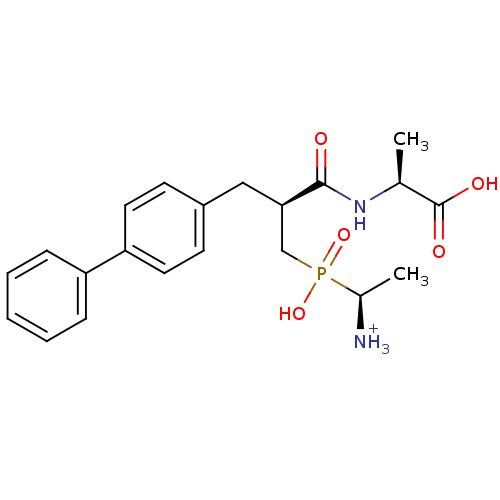 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumylethyl](hydroxy)ph...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092388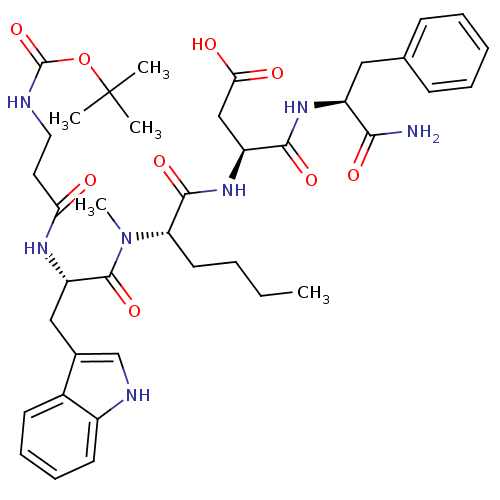 (3-(2-{[2-(3-tert-Butoxycarbonylamino-propionylamin...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092387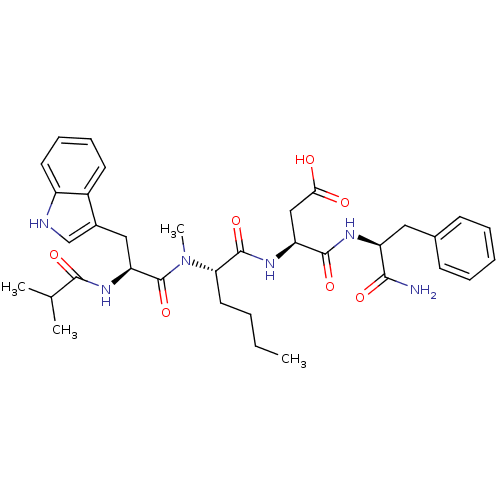 (CHEMBL118610 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087088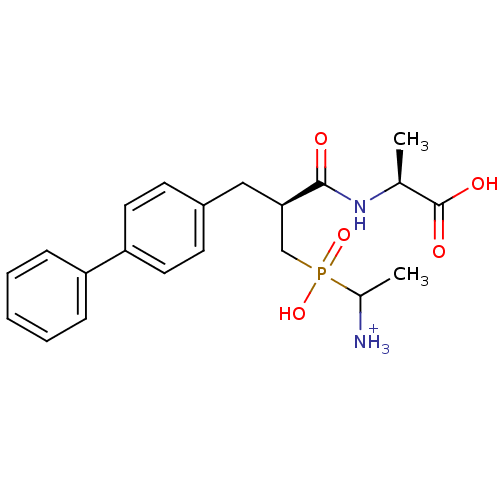 ((2S)-2-[(2S)-3-[(1-azaniumylethyl)(hydroxy)phospho...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087089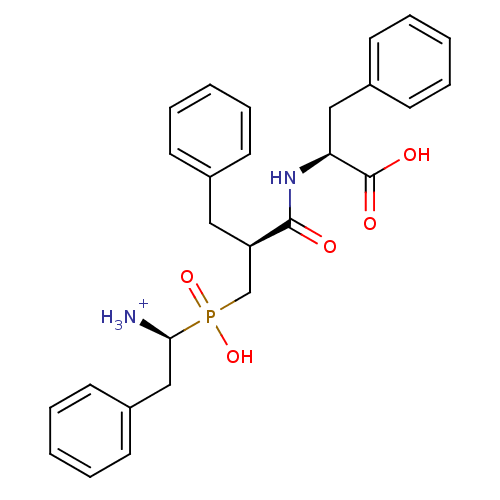 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumyl-2-phenylethyl](h...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092399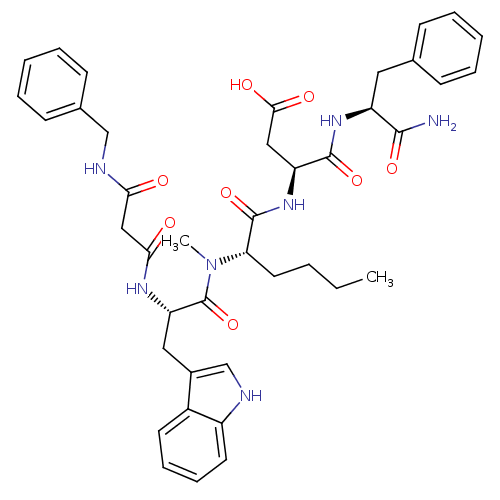 (3-(2-{[2-(2-Benzylcarbamoyl-acetylamino)-3-(1H-ind...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087095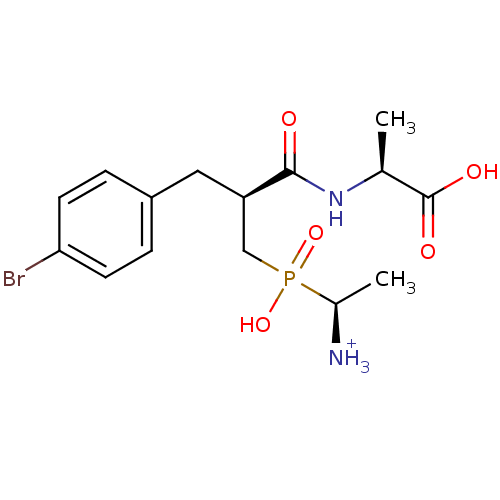 (1-{[3-(4-Bromo-phenyl)-2-(1-carboxy-ethylcarbamoyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50061270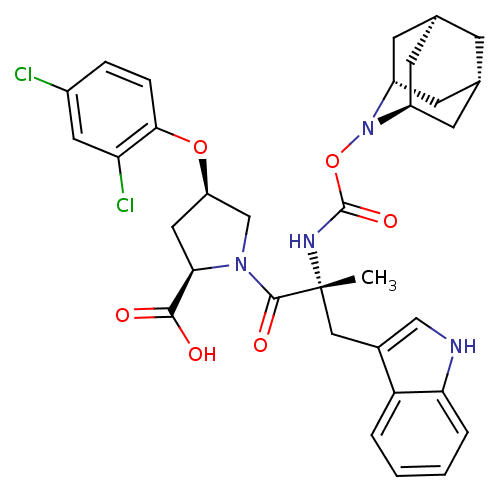 ((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells | J Med Chem 40: 3947-56 (1998) Article DOI: 10.1021/jm970439a BindingDB Entry DOI: 10.7270/Q27H1K8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087113 ((2S)-2-[(2S)-3-{[(S)-azaniumyl(phenyl)methyl](hydr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM81962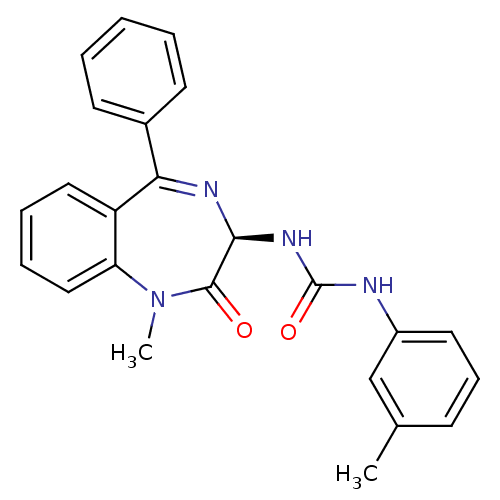 (S-L-365,260) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 40: 3947-56 (1998) Article DOI: 10.1021/jm970439a BindingDB Entry DOI: 10.7270/Q27H1K8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092391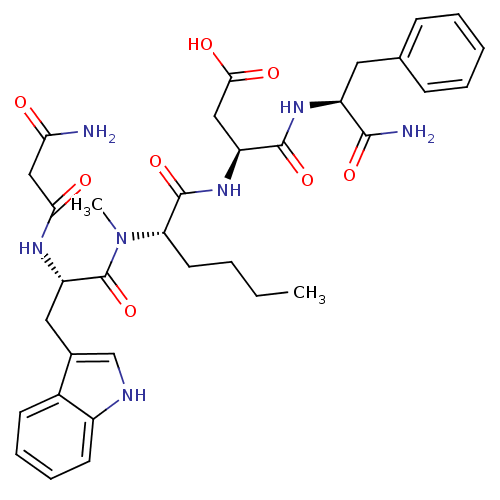 (3-(2-{[2-(2-Carbamoyl-acetylamino)-3-(1H-indol-3-y...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.05 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087085 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumyl-2-phenylethyl](h...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087097 (1-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-3-phenyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087084 (1-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-3-phenyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087085 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumyl-2-phenylethyl](h...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087094 ((R,S,S)1-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-4...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087093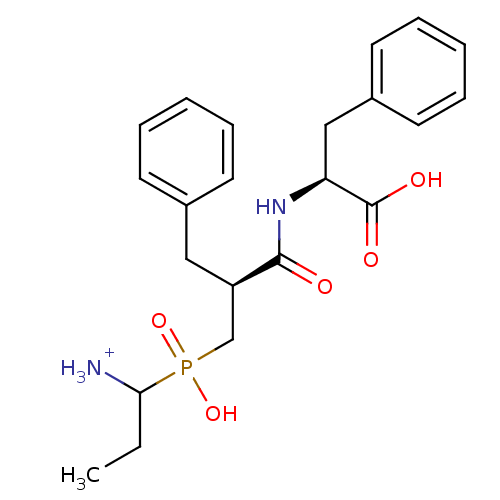 (1-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-3-phenyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092404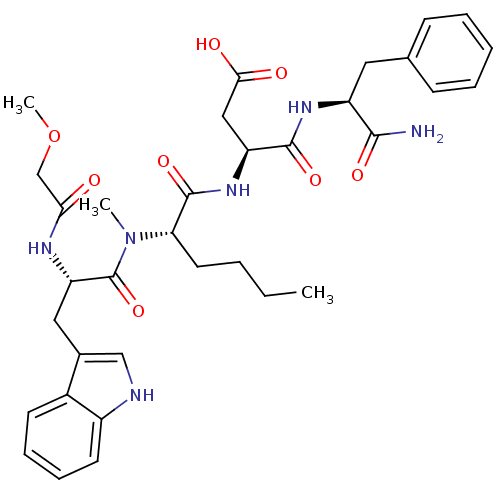 (CHEMBL261934 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092396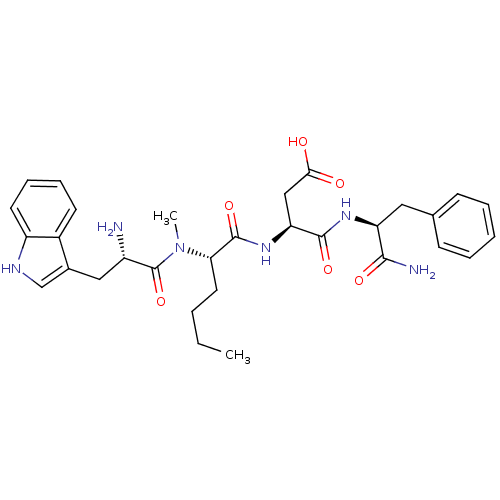 (3-(2-{[2-Amino-3-(1H-indol-3-yl)-propionyl]-methyl...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (RAT) | BDBM50092398 ((S)-3-((S)-2-{[(S)-2-tert-Butoxycarbonylamino-3-(1...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Compound was tested for binding affinity against Cholecystokinin type B receptor expressed in CHO cells on the rat brain. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092410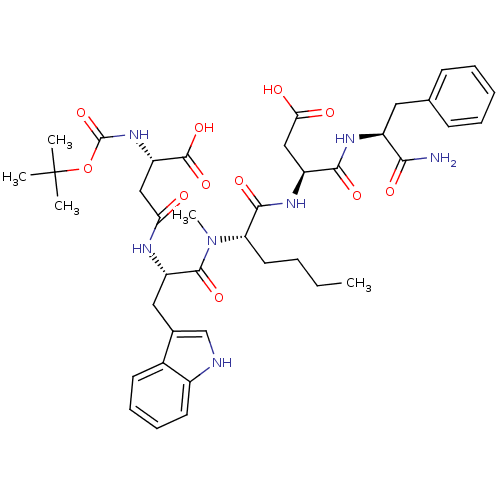 (2-tert-Butoxycarbonylamino-N-[1-({1-[1-(1-carbamoy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087106 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumylethyl](hydroxy)ph...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087089 ((2S)-2-[(2S)-3-{[(1S)-1-azaniumyl-2-phenylethyl](h...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087100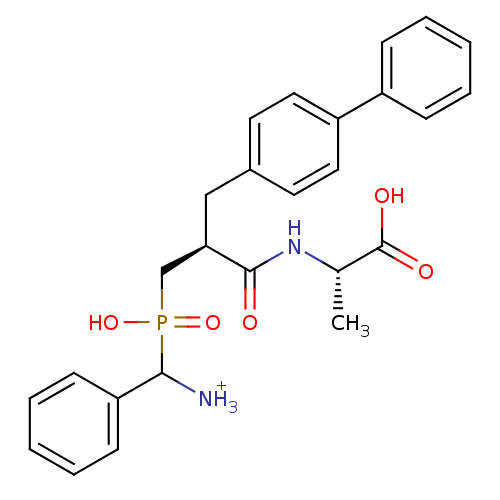 ((2S)-2-[(2S)-3-{[azaniumyl(phenyl)methyl](hydroxy)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholecystokinin receptor type A (Cavia porcellus) | BDBM50092384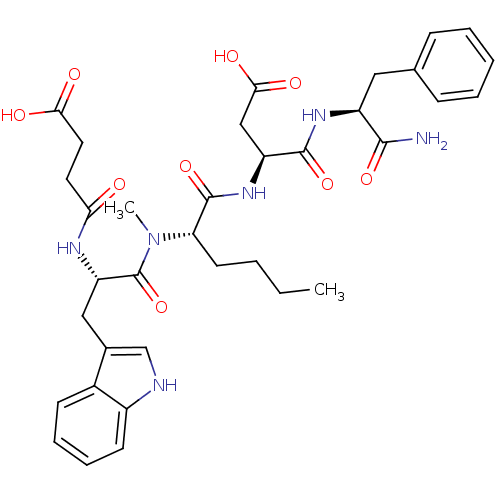 (CHEMBL100538 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB MMDB Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.02 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Affinity against Cholecystokinin type A receptor on guinea pig pancreatic membranes. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087114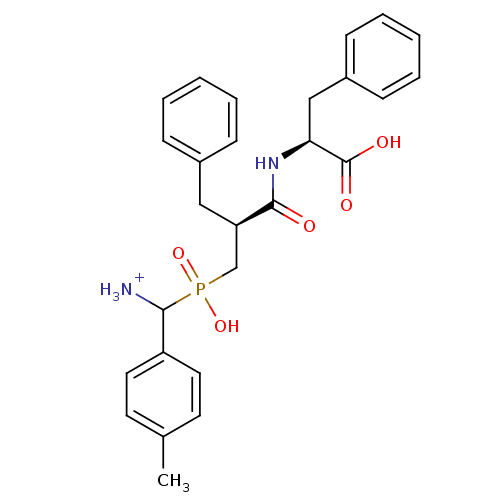 (C-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-3-phenyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamyl aminopeptidase (Homo sapiens (Human)) | BDBM50083394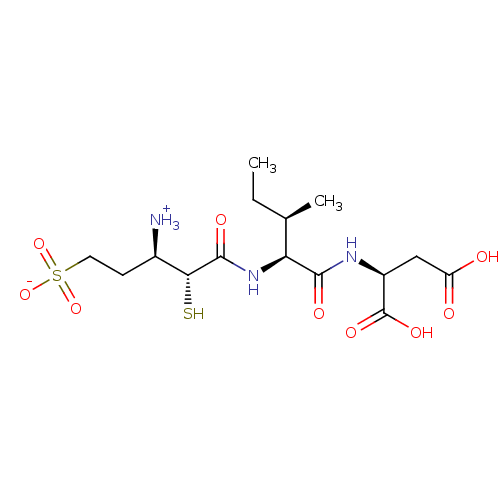 (1-{[1-(1,2-Dicarboxy-ethylcarbamoyl)-2-methyl-buty...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against recombinant Aminopeptidase A | J Med Chem 42: 5197-211 (2000) BindingDB Entry DOI: 10.7270/Q2028S8F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087096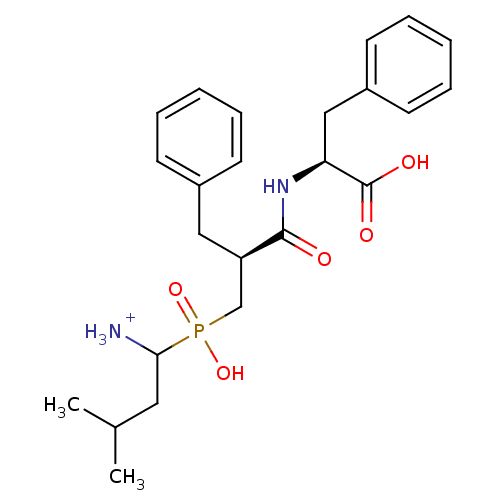 (1-{[2-(1-Carboxy-2-phenyl-ethylcarbamoyl)-3-phenyl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087101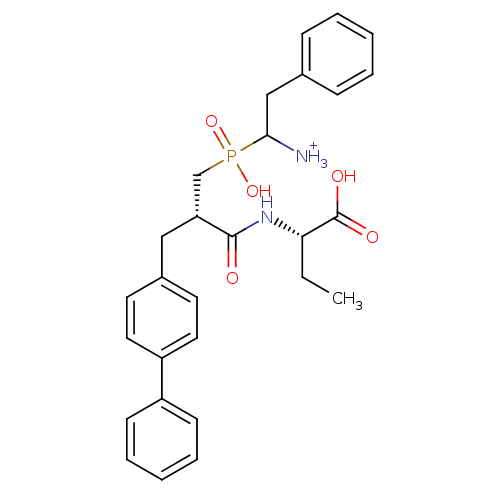 (1-{[3-Biphenyl-4-yl-2-(1-carboxy-propylcarbamoyl)-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamyl aminopeptidase (Homo sapiens (Human)) | BDBM50083378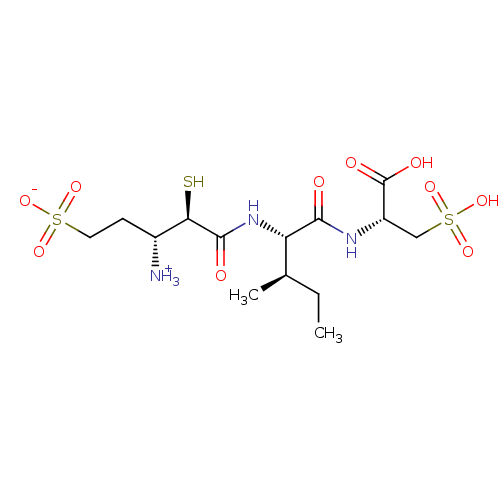 (1-{[1-(1-Carboxy-2-sulfo-ethylcarbamoyl)-2-methyl-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against recombinant Aminopeptidase A | J Med Chem 42: 5197-211 (2000) BindingDB Entry DOI: 10.7270/Q2028S8F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamyl aminopeptidase (Homo sapiens (Human)) | BDBM50083376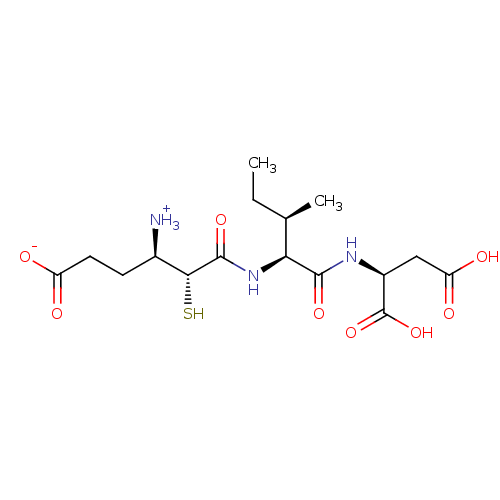 (3-Carboxy-1-{[1-(1,2-dicarboxy-ethylcarbamoyl)-2-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against recombinant Aminopeptidase A | J Med Chem 42: 5197-211 (2000) BindingDB Entry DOI: 10.7270/Q2028S8F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092389 (CHEMBL321456 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.61 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087105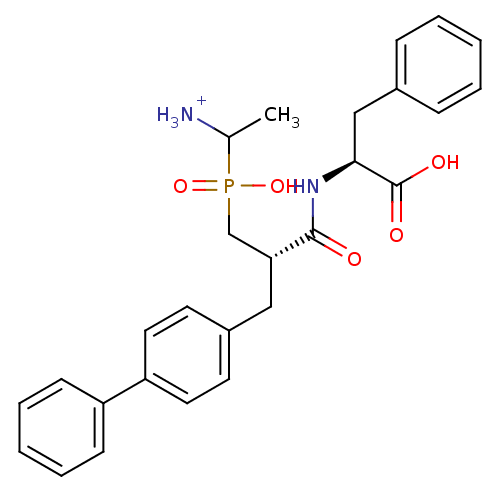 (1-{[3-Biphenyl-4-yl-2-(1-carboxy-2-phenyl-ethylcar...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamyl aminopeptidase (Homo sapiens (Human)) | BDBM50083385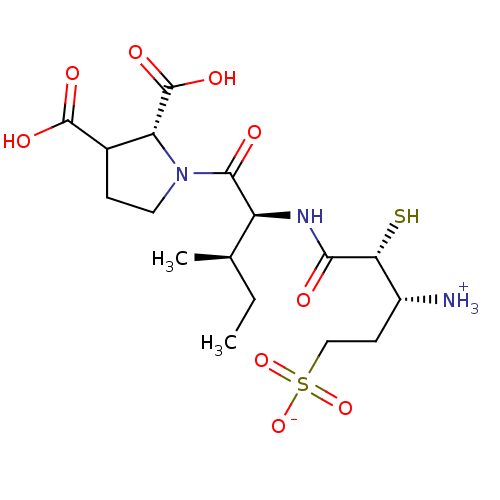 (1-{[1-(2,3-Dicarboxy-pyrrolidine-1-carbonyl)-2-met...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against recombinant Aminopeptidase A | J Med Chem 42: 5197-211 (2000) BindingDB Entry DOI: 10.7270/Q2028S8F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin/cholecystokinin type B receptor (Homo sapiens (Human)) | BDBM50092414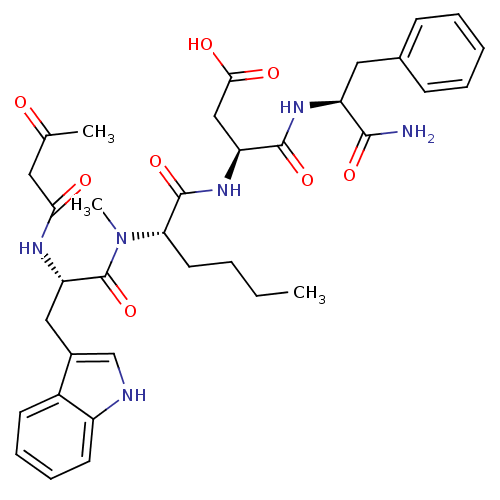 (CHEMBL120335 | N-(1-Carbamoyl-2-phenyl-ethyl)-3-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.84 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Binding affinity against cholecystokinin type B receptor on guinea pig cortex. | J Med Chem 43: 3614-23 (2000) BindingDB Entry DOI: 10.7270/Q2RN373C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 289 total ) | Next | Last >> |