 Found 149 hits with Last Name = 'encinas' and Initial = 'a'
Found 149 hits with Last Name = 'encinas' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50095181
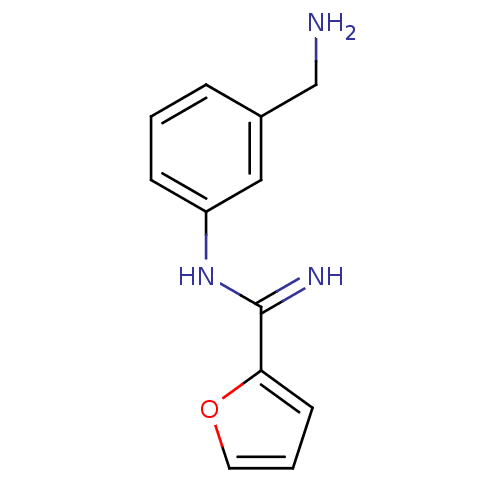
(CHEMBL96680 | N-(3-Aminomethyl-phenyl)-furan-2-car...)Show InChI InChI=1S/C12H13N3O/c13-8-9-3-1-4-10(7-9)15-12(14)11-5-2-6-16-11/h1-7H,8,13H2,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50095172

(CHEMBL262040 | N-(3-Aminomethyl-phenyl)-thiophene-...)Show InChI InChI=1S/C12H13N3S/c13-8-9-3-1-4-10(7-9)15-12(14)11-5-2-6-16-11/h1-7H,8,13H2,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271464
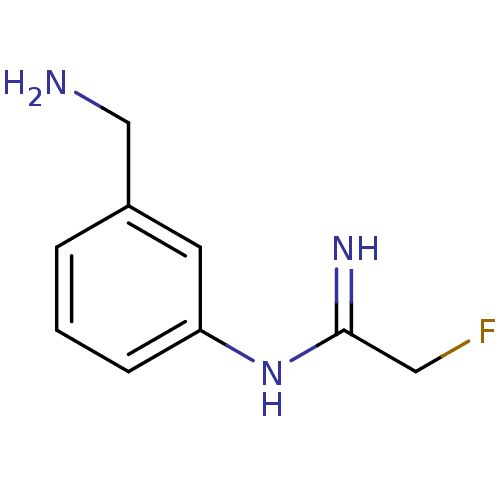
(CHEMBL455310 | N-(3-Aminomethyl-phenyl)-2-fluoro-a...)Show InChI InChI=1S/C9H12FN3/c10-5-9(12)13-8-3-1-2-7(4-8)6-11/h1-4H,5-6,11H2,(H2,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271363
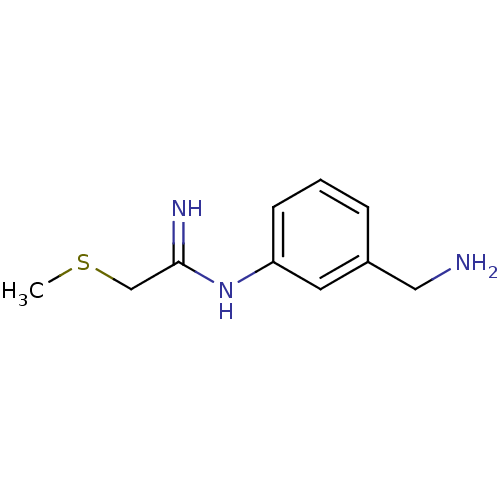
(CHEMBL476988 | N-(3-Aminomethyl-phenyl)-2-methylsu...)Show InChI InChI=1S/C10H15N3S/c1-14-7-10(12)13-9-4-2-3-8(5-9)6-11/h2-5H,6-7,11H2,1H3,(H2,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271882
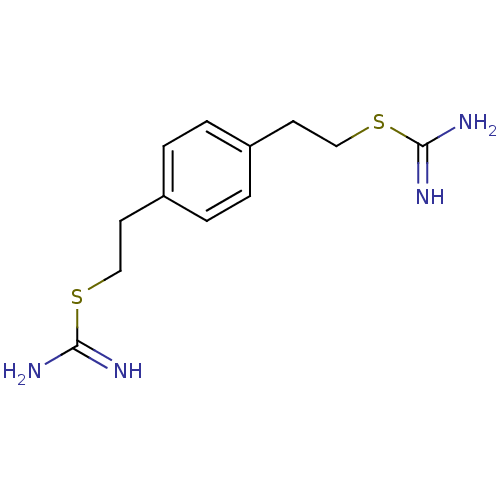
(2-{2-[4-(2-CARBAMIMIDOYLSULFANYL-ETHYL)-PHENYL]-ET...)Show InChI InChI=1S/C12H18N4S2/c13-11(14)17-7-5-9-1-2-10(4-3-9)6-8-18-12(15)16/h1-4H,5-8H2,(H3,13,14)(H3,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50055281
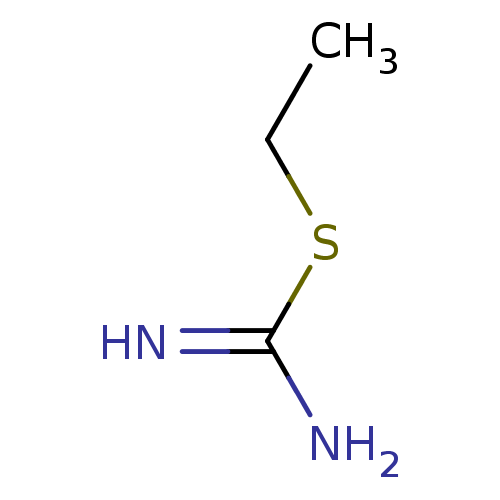
(2-Ethyl-isothiourea | CHEMBL321691 | ETHYLISOTHIOU...)Show InChI InChI=1S/C3H8N2S/c1-2-6-3(4)5/h2H2,1H3,(H3,4,5) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271827
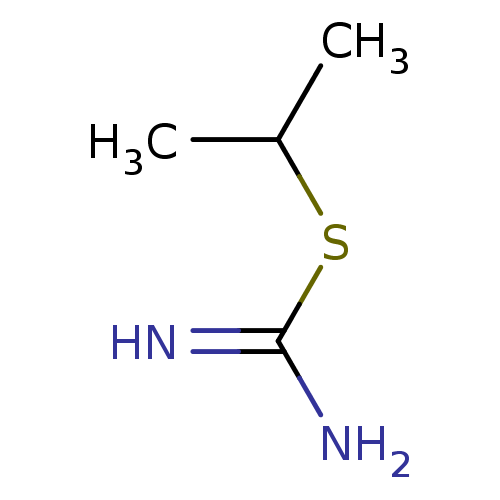
(2-Isopropyl-isothiourea | CHEMBL483091 | S-ISOPROP...)Show InChI InChI=1S/C4H10N2S/c1-3(2)7-4(5)6/h3H,1-2H3,(H3,5,6) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50138977

(CHEMBL419740 | N-(3-Aminomethyl-phenyl)-acetamidin...)Show InChI InChI=1S/C9H13N3/c1-7(11)12-9-4-2-3-8(5-9)6-10/h2-5H,6,10H2,1H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271316

(CHEMBL482699 | N-(3-{[3-(1-Imino-ethyl)-benzylamin...)Show InChI InChI=1S/C18H22N4/c1-13(19)17-7-3-5-15(9-17)11-21-12-16-6-4-8-18(10-16)22-14(2)20/h3-10,19,21H,11-12H2,1-2H3,(H2,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50272045
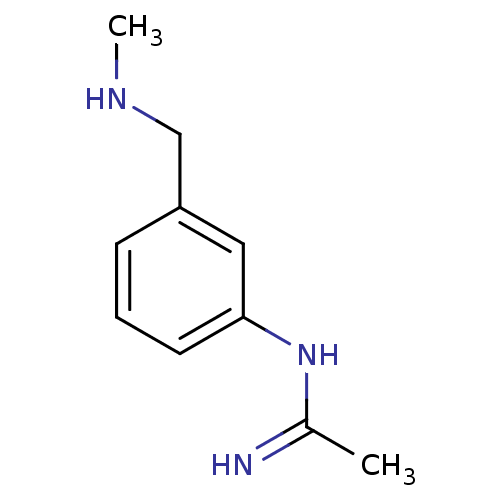
(CHEMBL496134 | N-(3-Methylaminomethyl-phenyl)-acet...)Show InChI InChI=1S/C10H15N3/c1-8(11)13-10-5-3-4-9(6-10)7-12-2/h3-6,12H,7H2,1-2H3,(H2,11,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271883
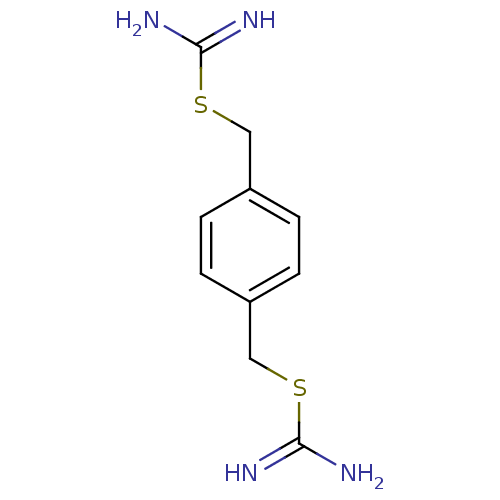
(2-(4-Carbamimidoylsulfanylmethyl-benzyl)-isothiour...)Show InChI InChI=1S/C10H14N4S2/c11-9(12)15-5-7-1-2-8(4-3-7)6-16-10(13)14/h1-4H,5-6H2,(H3,11,12)(H3,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50240715
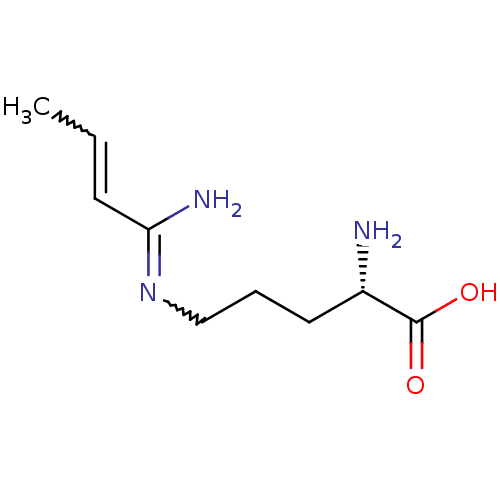
((S)-2-Amino-5-but-3-enimidoylamino-pentanoic acid ...)Show InChI InChI=1S/C9H17N3O2/c1-2-4-8(11)12-6-3-5-7(10)9(13)14/h2,4,7H,3,5-6,10H2,1H3,(H2,11,12)(H,13,14)/t7-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| MMDB
Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM21961
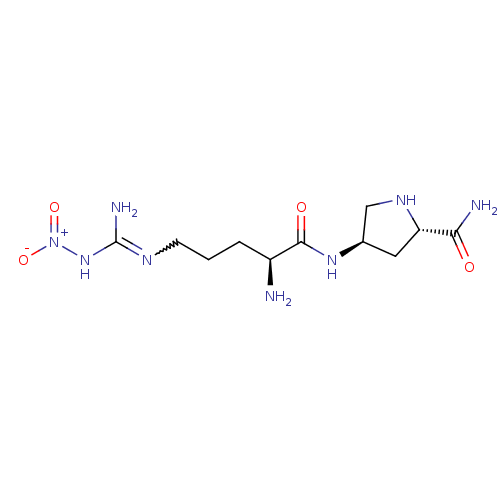
((2S,4R)-4-[(2S)-2-amino-5-(1-nitrocarbamimidamido)...)Show SMILES N[C@@H](CCCN=C(N)N[N+]([O-])=O)C(=O)N[C@H]1CN[C@@H](C1)C(N)=O |w:5.4| Show InChI InChI=1S/C11H22N8O4/c12-7(2-1-3-15-11(14)18-19(22)23)10(21)17-6-4-8(9(13)20)16-5-6/h6-8,16H,1-5,12H2,(H2,13,20)(H,17,21)(H3,14,15,18)/t6-,7+,8+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058459
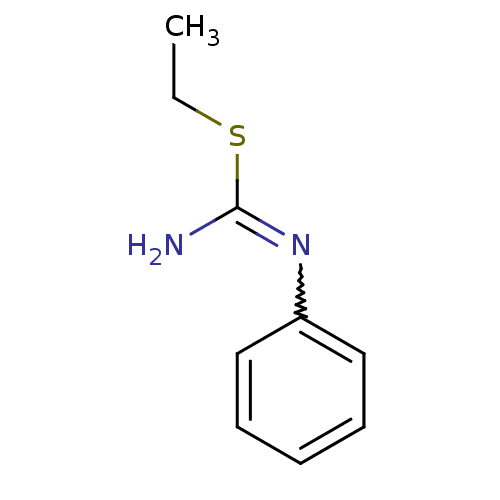
(2-Ethyl-1-phenyl-isothiourea; hydriodide | CHEMBL4...)Show InChI InChI=1S/C9H12N2S/c1-2-12-9(10)11-8-6-4-3-5-7-8/h3-7H,2H2,1H3,(H2,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM22030
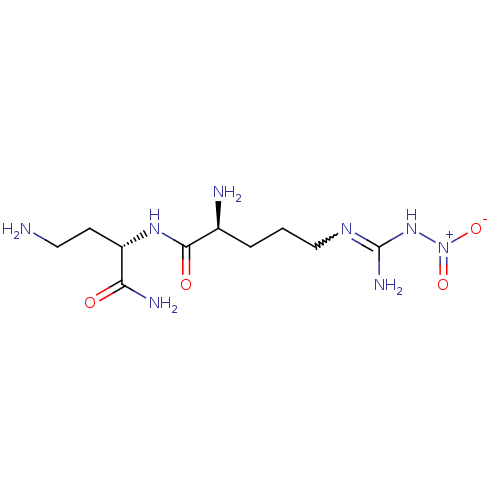
((2S)-2-amino-N-[(1S)-3-amino-1-(aminocarbonyl)prop...)Show SMILES NCC[C@H](NC(=O)[C@@H](N)CCCN=C(N)N[N+]([O-])=O)C(N)=O |r,w:12.11| Show InChI InChI=1S/C10H22N8O4/c11-4-3-7(8(13)19)16-9(20)6(12)2-1-5-15-10(14)17-18(21)22/h6-7H,1-5,11-12H2,(H2,13,19)(H,16,20)(H3,14,15,17)/t6-,7-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058469
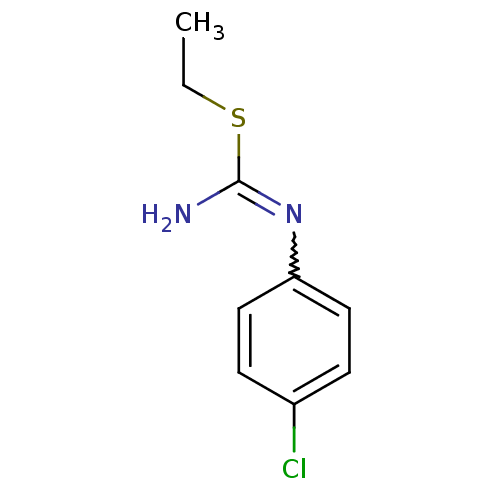
(1-(4-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...)Show InChI InChI=1S/C9H11ClN2S/c1-2-13-9(11)12-8-5-3-7(10)4-6-8/h3-6H,2H2,1H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50055282

(2-Methyl-isothiourea | CHEMBL356703)Show InChI InChI=1S/C2H6N2S/c1-5-2(3)4/h1H3,(H3,3,4) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50272046

(CHEMBL502800 | N-(3-Dimethylaminomethyl-phenyl)-ac...)Show InChI InChI=1S/C11H17N3/c1-9(12)13-11-6-4-5-10(7-11)8-14(2)3/h4-7H,8H2,1-3H3,(H2,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058441
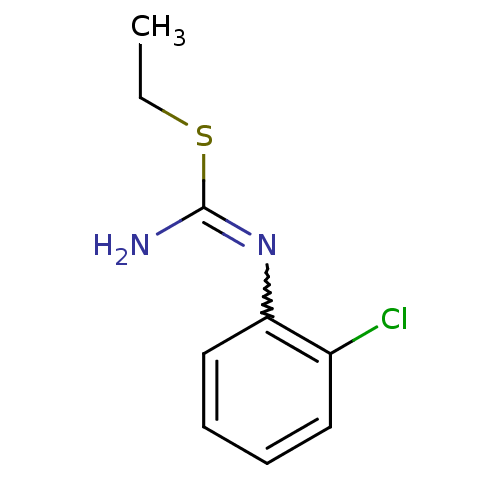
(1-(2-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...)Show InChI InChI=1S/C9H11ClN2S/c1-2-13-9(11)12-8-6-4-3-5-7(8)10/h3-6H,2H2,1H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058448
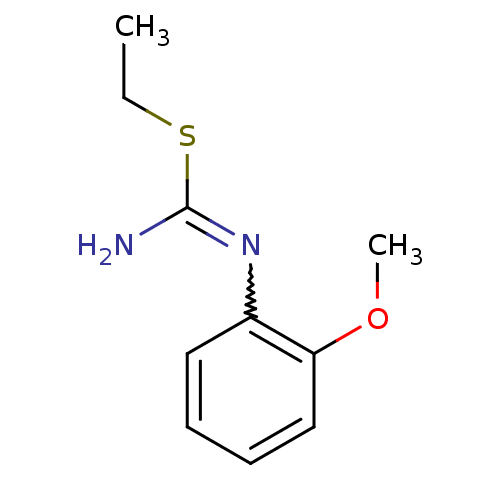
(2-Ethyl-1-(2-methoxy-phenyl)-isothiourea; hydrochl...)Show InChI InChI=1S/C10H14N2OS/c1-3-14-10(11)12-8-6-4-5-7-9(8)13-2/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058443
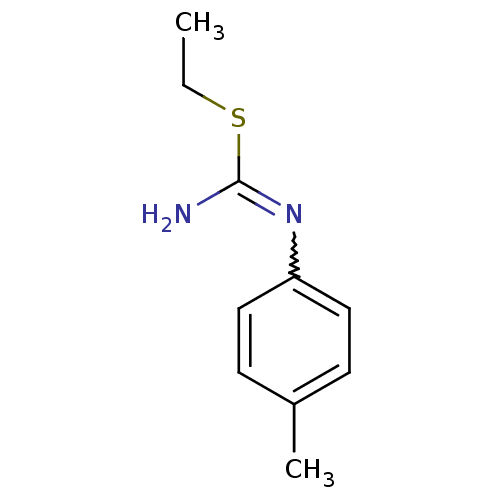
(2-Ethyl-1-p-tolyl-isothiourea; hydrochloride | CHE...)Show InChI InChI=1S/C10H14N2S/c1-3-13-10(11)12-9-6-4-8(2)5-7-9/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271322

(2-Amino-N-(3-aminomethyl-phenyl)-acetamidine | CHE...)Show InChI InChI=1S/C9H14N4/c10-5-7-2-1-3-8(4-7)13-9(12)6-11/h1-4H,5-6,10-11H2,(H2,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058445
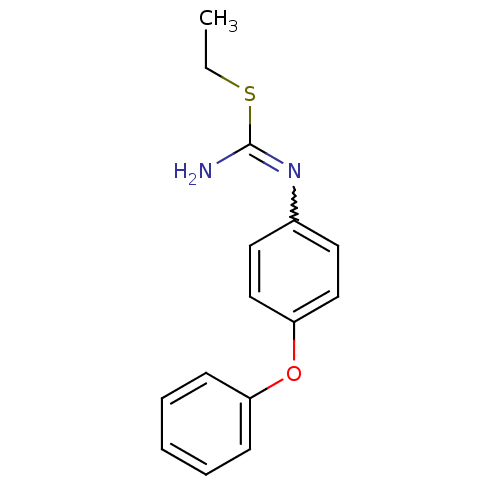
(2-Ethyl-1-(4-phenoxy-phenyl)-isothiourea; hydriodi...)Show InChI InChI=1S/C15H16N2OS/c1-2-19-15(16)17-12-8-10-14(11-9-12)18-13-6-4-3-5-7-13/h3-11H,2H2,1H3,(H2,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058468
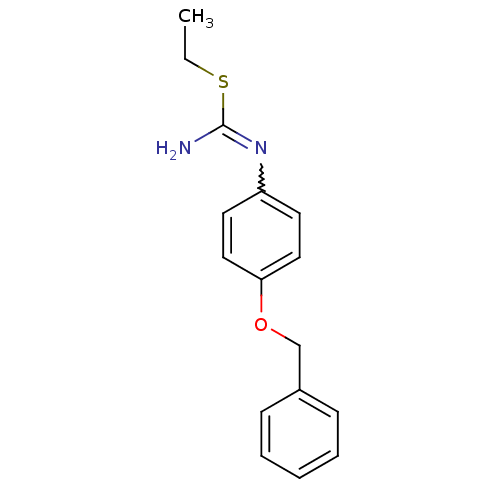
(1-(4-Benzyloxy-phenyl)-2-ethyl-isothiourea; hydrio...)Show InChI InChI=1S/C16H18N2OS/c1-2-20-16(17)18-14-8-10-15(11-9-14)19-12-13-6-4-3-5-7-13/h3-11H,2,12H2,1H3,(H2,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50240717
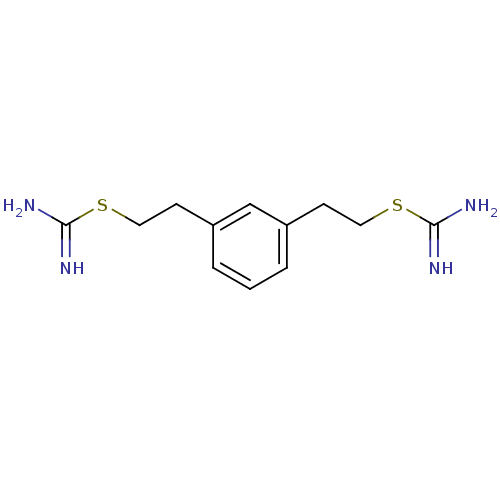
(2-{2-[3-(2-Carbamimidoylsulfanyl-ethyl)-phenyl]-et...)Show InChI InChI=1S/C12H18N4S2/c13-11(14)17-6-4-9-2-1-3-10(8-9)5-7-18-12(15)16/h1-3,8H,4-7H2,(H3,13,14)(H3,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058440
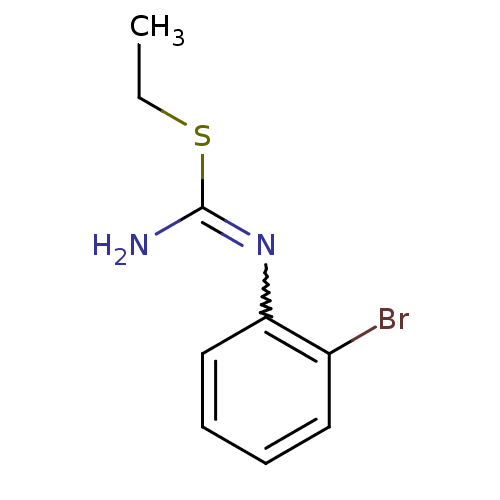
(1-(2-Bromo-phenyl)-2-ethyl-isothiourea; hydriodide...)Show InChI InChI=1S/C9H11BrN2S/c1-2-13-9(11)12-8-6-4-3-5-7(8)10/h3-6H,2H2,1H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058447
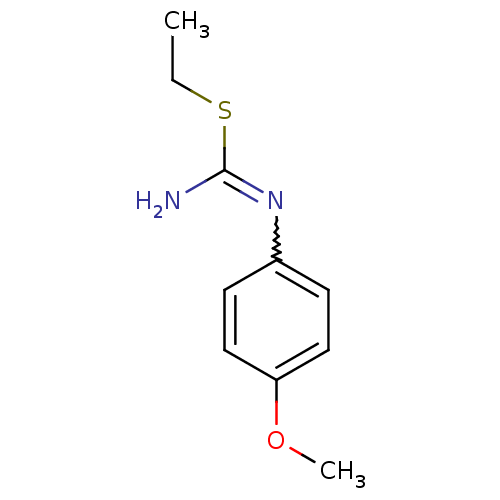
(2-Ethyl-1-(4-methoxy-phenyl)-isothiourea; hydriodi...)Show InChI InChI=1S/C10H14N2OS/c1-3-14-10(11)12-8-4-6-9(13-2)7-5-8/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271465

(2-Fluoro-N-(3-hydroxymethyl-phenyl)-acetamidine | ...)Show InChI InChI=1S/C9H11FN2O/c10-5-9(11)12-8-3-1-2-7(4-8)6-13/h1-4,13H,5-6H2,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058464

(2-Ethyl-1-(4-trifluoromethyl-phenyl)-isothiourea; ...)Show InChI InChI=1S/C10H11F3N2S/c1-2-16-9(14)15-8-5-3-7(4-6-8)10(11,12)13/h3-6H,2H2,1H3,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058451
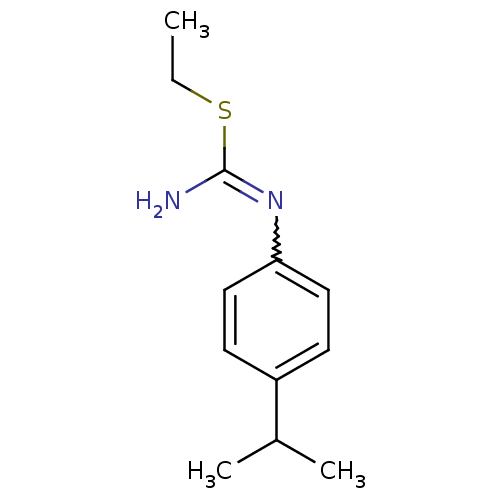
(2-Ethyl-1-(4-isopropyl-phenyl)-isothiourea; hydroc...)Show InChI InChI=1S/C12H18N2S/c1-4-15-12(13)14-11-7-5-10(6-8-11)9(2)3/h5-9H,4H2,1-3H3,(H2,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271466

(CHEMBL444240 | N-(3-Aminomethyl-phenyl)-pyridine-2...)Show InChI InChI=1S/C13H14N4/c14-9-10-4-3-5-11(8-10)17-13(15)12-6-1-2-7-16-12/h1-8H,9,14H2,(H2,15,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058444

(2-Ethyl-1-(4-hydroxy-phenyl)-isothiourea; hydriodi...)Show InChI InChI=1S/C9H12N2OS/c1-2-13-9(10)11-7-3-5-8(12)6-4-7/h3-6,12H,2H2,1H3,(H2,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271317

(CHEMBL482700 | N-(3-Carbamimidoylsulfanylmethyl-ph...)Show InChI InChI=1S/C10H14N4S/c1-7(11)14-9-4-2-3-8(5-9)6-15-10(12)13/h2-5H,6H2,1H3,(H2,11,14)(H3,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271315

(CHEMBL466888 | N-[3-(Acetimidoylamino-methyl)-phen...)Show InChI InChI=1S/C11H16N4/c1-8(12)14-7-10-4-3-5-11(6-10)15-9(2)13/h3-6H,7H2,1-2H3,(H2,12,14)(H2,13,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271510
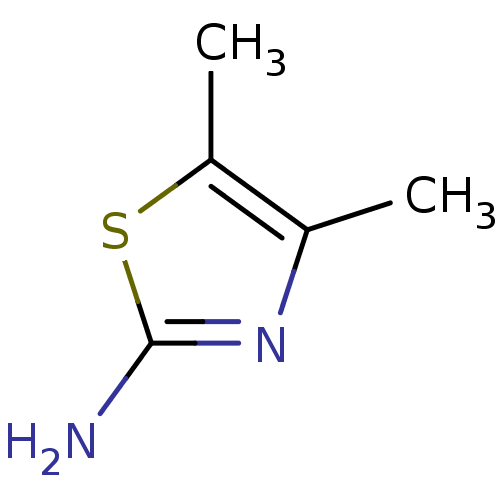
(4,5-dimethylthiazol-2-amine | CHEMBL450172)Show InChI InChI=1S/C5H8N2S/c1-3-4(2)8-5(6)7-3/h1-2H3,(H2,6,7) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50095177
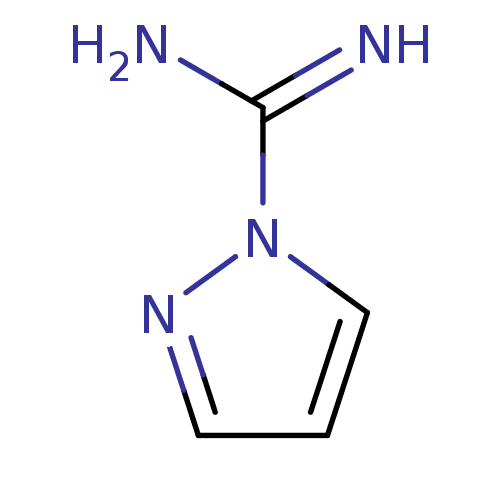
(CHEMBL503356 | CHEMBL542185 | Pyrazole-1-carboxami...)Show InChI InChI=1S/C4H6N4/c5-4(6)8-3-1-2-7-8/h1-3H,(H3,5,6) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50150920
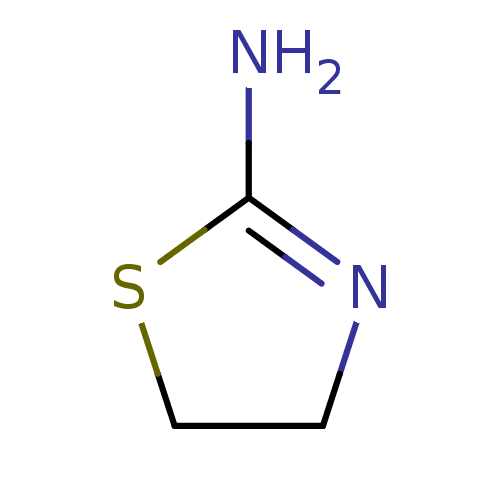
(2-AMINOTHIAZOLINE | 4,5-dihydrothiazol-2-amine | C...)Show InChI InChI=1S/C3H6N2S/c4-3-5-1-2-6-3/h1-2H2,(H2,4,5) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058455
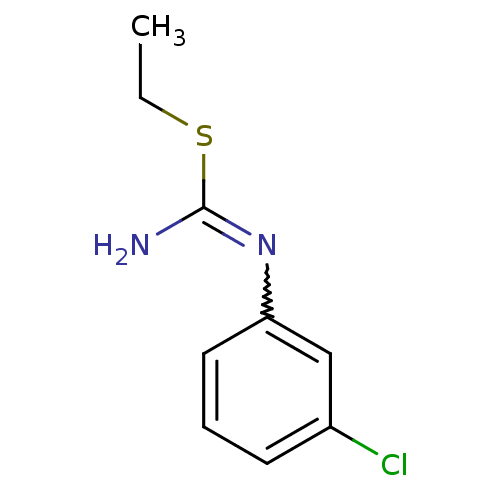
(1-(3-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...)Show InChI InChI=1S/C9H11ClN2S/c1-2-13-9(11)12-8-5-3-4-7(10)6-8/h3-6H,2H2,1H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50225106
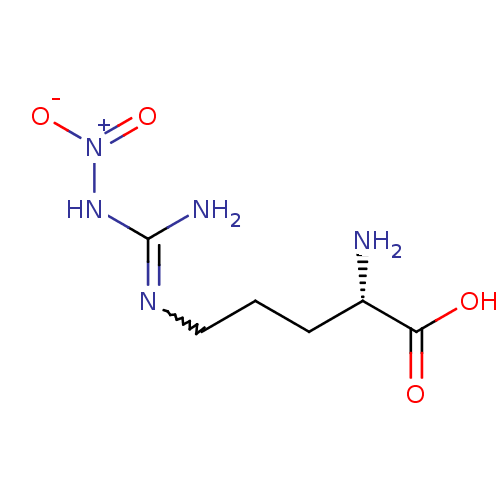
((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...)Show SMILES N[C@@H](CCCNC(N)=N[N+]([O-])=O)C(O)=O |r,w:8.8| Show InChI InChI=1S/C6H13N5O4/c7-4(5(12)13)2-1-3-9-6(8)10-11(14)15/h4H,1-3,7H2,(H,12,13)(H3,8,9,10)/t4-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058442

(2-Ethyl-1-(3-methoxy-phenyl)-isothiourea; hydriodi...)Show InChI InChI=1S/C10H14N2OS/c1-3-14-10(11)12-8-5-4-6-9(7-8)13-2/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271320

(3-Acetimidoylamino-benzamidine | CHEMBL463507)Show InChI InChI=1S/C9H12N4/c1-6(10)13-8-4-2-3-7(5-8)9(11)12/h2-5H,1H3,(H2,10,13)(H3,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271828
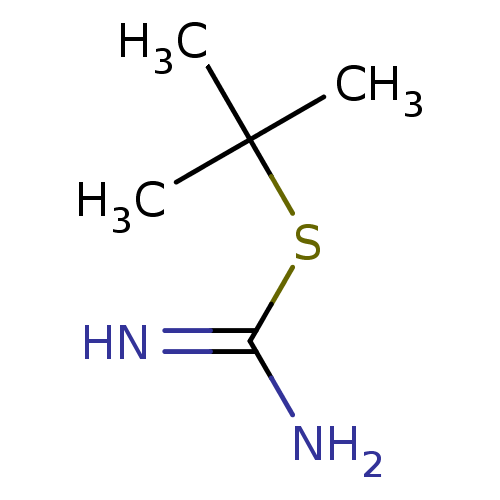
(2-tert-Butyl-isothiourea | CHEMBL451408)Show InChI InChI=1S/C5H12N2S/c1-5(2,3)8-4(6)7/h1-3H3,(H3,6,7) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271829

(2-Propyl-isothiourea | CHEMBL483092)Show InChI InChI=1S/C4H10N2S/c1-2-3-7-4(5)6/h2-3H2,1H3,(H3,5,6) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271972

(2-Ethyl-1-(4-nitro-phenyl)-isothiourea | CHEMBL500...)Show InChI InChI=1S/C9H11N3O2S/c1-2-15-9(10)11-7-3-5-8(6-4-7)12(13)14/h3-6H,2H2,1H3,(H2,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50240621

(1-amino-3-hydroxyguanidine tosylate | CHEMBL80352 ...)Show InChI InChI=1S/CH6N4O/c2-1(4-3)5-6/h6H,3H2,(H3,2,4,5) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058453

(2-Ethyl-1-(4-trifluoromethoxy-phenyl)-isothiourea;...)Show InChI InChI=1S/C10H11F3N2OS/c1-2-17-9(14)15-7-3-5-8(6-4-7)16-10(11,12)13/h3-6H,2H2,1H3,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271831

(2-Phenethyl-isothiourea | CHEMBL483093)Show InChI InChI=1S/C9H12N2S/c10-9(11)12-7-6-8-4-2-1-3-5-8/h1-5H,6-7H2,(H3,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50058456

(2-Ethyl-1-pyridin-4-yl-isothiourea; hydrofluoride ...)Show InChI InChI=1S/C8H11N3S/c1-2-12-8(9)11-7-3-5-10-6-4-7/h3-6H,2H2,1H3,(H2,9,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50271511
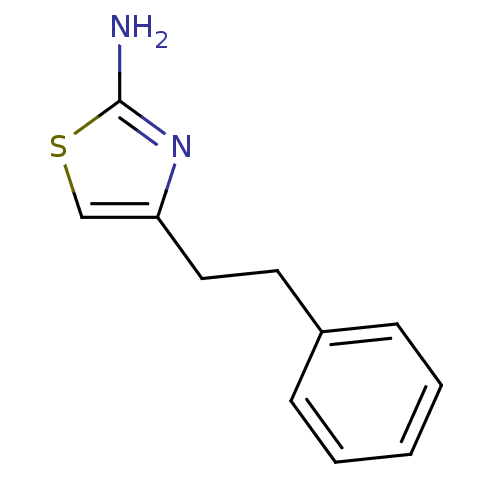
(4-phenethylthiazol-2-amine | CHEMBL484101)Show InChI InChI=1S/C11H12N2S/c12-11-13-10(8-14-11)7-6-9-4-2-1-3-5-9/h1-5,8H,6-7H2,(H2,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 890 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50272004
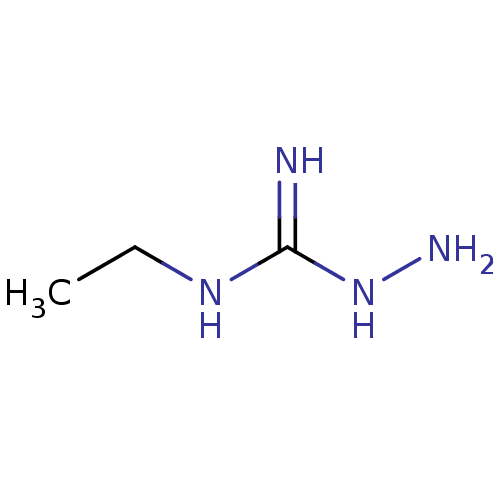
(CHEMBL507240 | N-ethylhydrazinecarboximidamide)Show InChI InChI=1S/C3H10N4/c1-2-6-3(4)7-5/h2,5H2,1H3,(H3,4,6,7) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto de Química Médica
Curated by ChEMBL
| Assay Description
Inhibition of nNOS (unknown origin) assessed as conversion of L-[3H]arginine to L-[3H]citrulline |
Bioorg Med Chem 16: 6193-206 (2008)
Article DOI: 10.1016/j.bmc.2008.04.036
BindingDB Entry DOI: 10.7270/Q2611040 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data