

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM160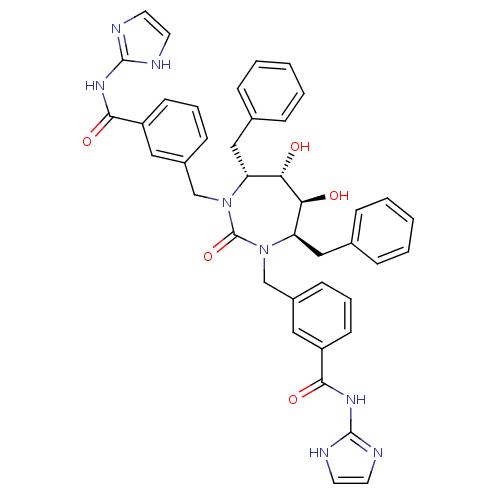 (3-{[(4R,5S,6S,7R)-4,7-dibenzyl-5,6-dihydroxy-3-{[3...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055590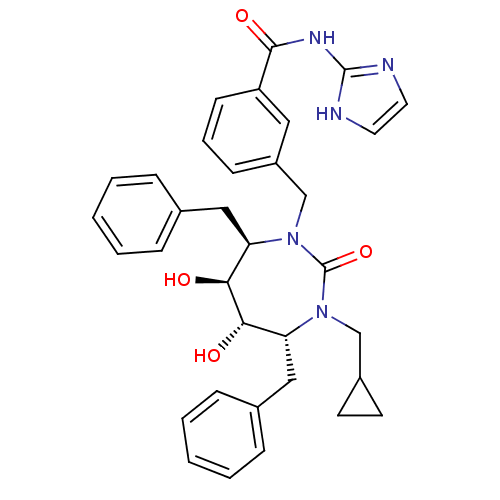 (3-((4R,5S,6S,7R)-4,7-Dibenzyl-3-cyclopropylmethyl-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM155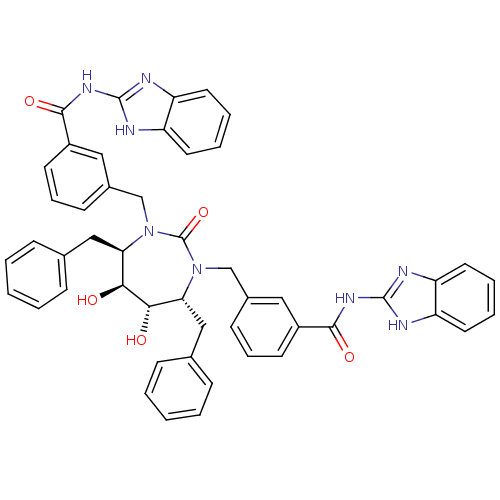 (CHEMBL11266 | N-(1H-1,3-benzodiazol-2-yl)-3-{[(4R,...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.0240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055587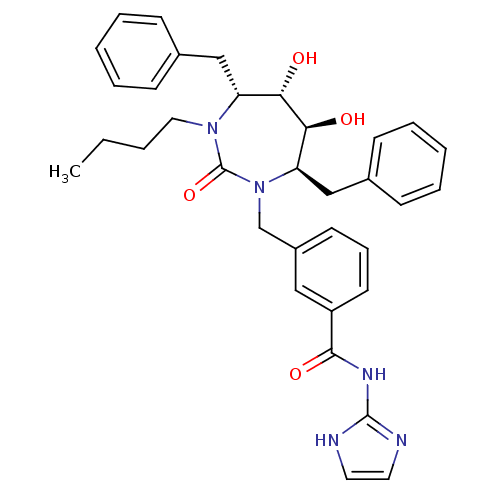 (3-((4R,5S,6S,7R)-4,7-Dibenzyl-3-butyl-5,6-dihydrox...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM154 (3-{[(4R,5S,6S,7R)-4,7-dibenzyl-5,6-dihydroxy-2-oxo...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055593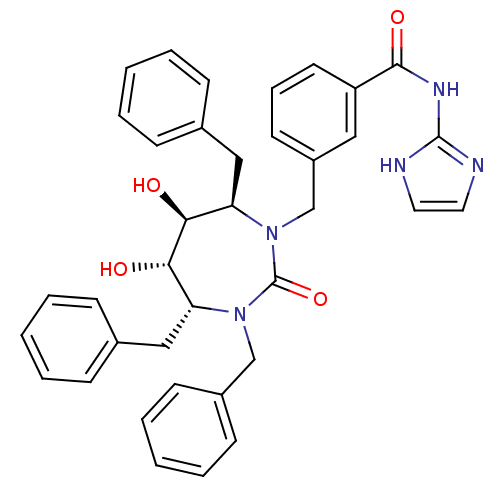 (CHEMBL292905 | N-(1H-Imidazol-2-yl)-3-((4R,5S,6S,7...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055588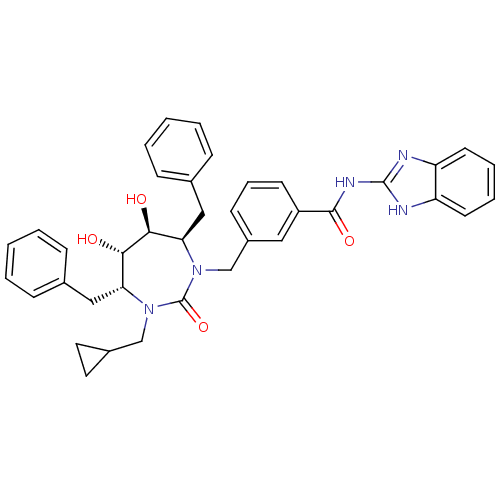 (CHEMBL301219 | N-(1H-Benzoimidazol-2-yl)-3-((4R,5S...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055589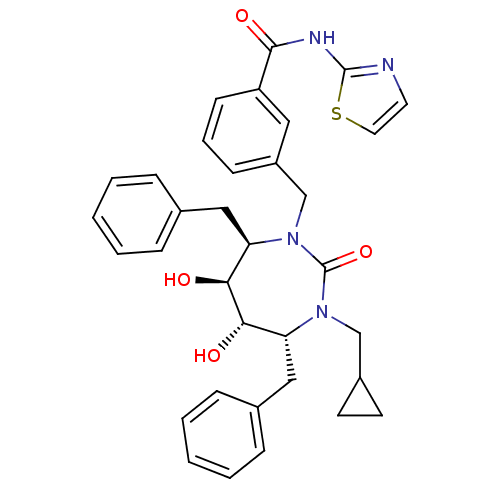 (3-((4R,5S,6S,7R)-4,7-Dibenzyl-3-cyclopropylmethyl-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055592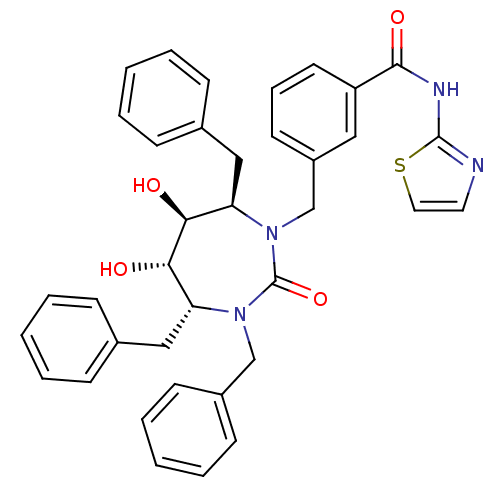 (CHEMBL56117 | N-Thiazol-2-yl-3-((4R,5S,6S,7R)-3,4,...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM577 ((3S)-oxolan-3-yl N-[(2S,3R)-4-[(4-aminobenzene)(2-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM519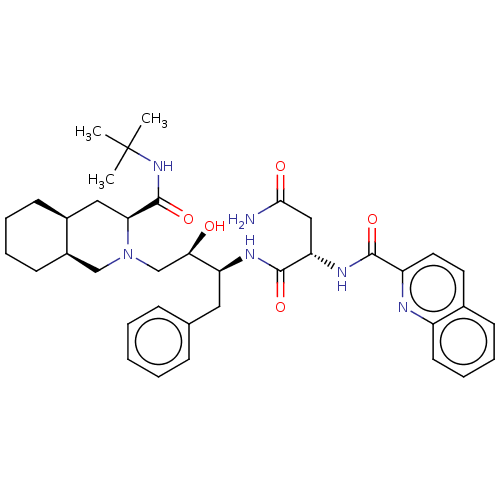 ((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50055591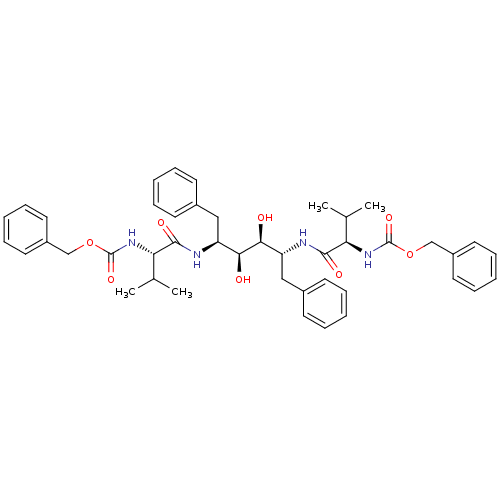 (CHEMBL56387 | {(R)-1-[(1R,2S,3S,4S)-1-Benzyl-4-((S...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM150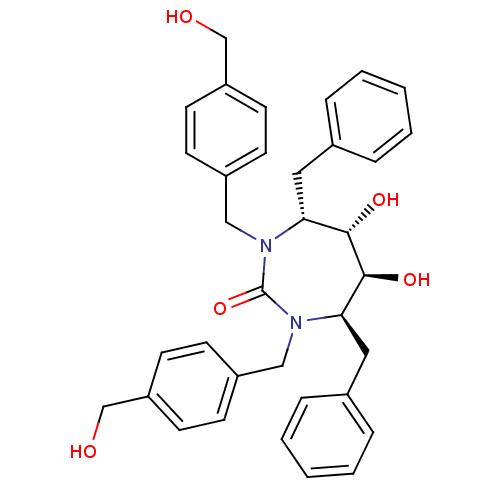 ((4R,5S,6S,7R)-4,7-dibenzyl-5,6-dihydroxy-1,3-bis({...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM151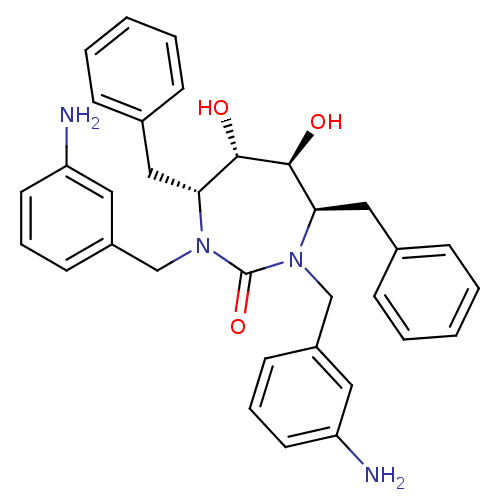 ((4R,5S,6S,7R)-1,3-bis[(3-aminophenyl)methyl]-4,7-d...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM520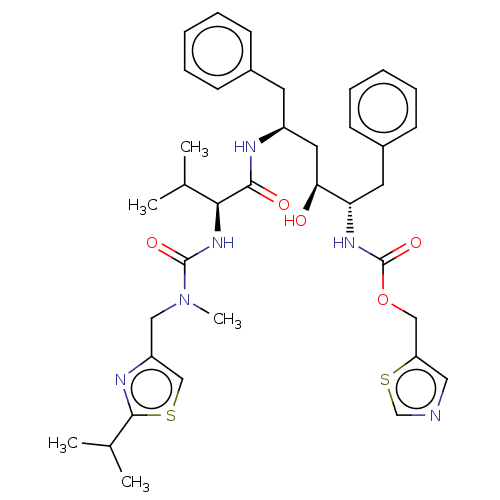 (1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| HIV-1 protease (Human immunodeficiency virus) | BDBM517 ((2S)-1-[(2S,4R)-4-benzyl-2-hydroxy-4-{[(1S,2R)-2-h...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York | Assay Description Enzyme inhibition assay for silanediols, carbinols, and indinavir using assay system C. | Chem Biol 8: 1161-6 (2001) Article DOI: 10.1016/S1074-5521(01)00079-5 BindingDB Entry DOI: 10.7270/Q20V8B63 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM517 ((2S)-1-[(2S,4R)-4-benzyl-2-hydroxy-4-{[(1S,2R)-2-h...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM32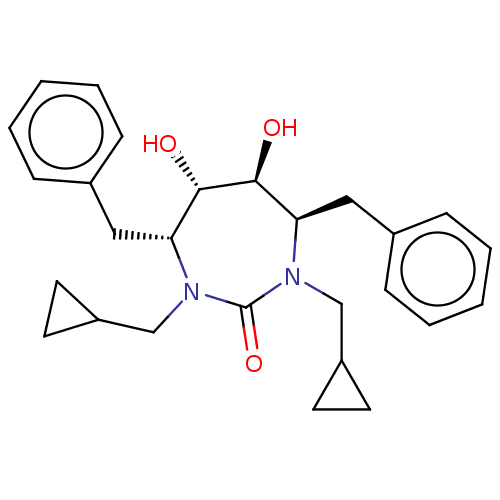 ((4R,5S,6S,7R)-4,7-dibenzyl-1,3-bis(cyclopropylmeth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to inhibit the purified wild-type HIV-1 Protease | J Med Chem 40: 181-91 (1997) Article DOI: 10.1021/jm960586t BindingDB Entry DOI: 10.7270/Q2ST7NZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease (Human immunodeficiency virus) | BDBM40773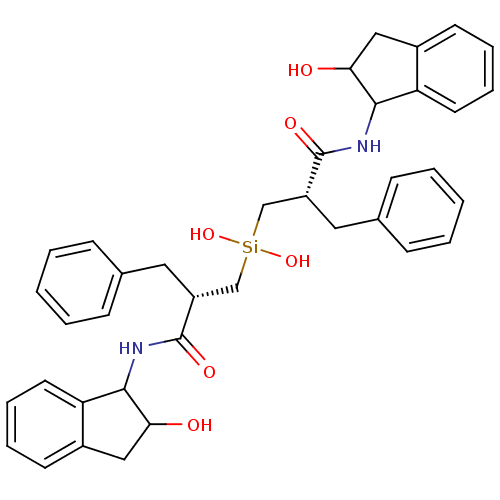 (Silanediol, 17b) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York | Assay Description Enzyme inhibition assay for silanediols, carbinols, and indinavir using assay system C. | Chem Biol 8: 1161-6 (2001) Article DOI: 10.1016/S1074-5521(01)00079-5 BindingDB Entry DOI: 10.7270/Q20V8B63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease (Human immunodeficiency virus) | BDBM40774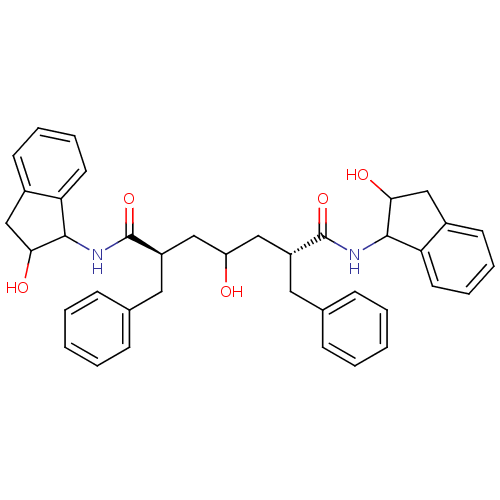 (Carbinol, 18b) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York | Assay Description Enzyme inhibition assay for silanediols, carbinols, and indinavir using assay system C. | Chem Biol 8: 1161-6 (2001) Article DOI: 10.1016/S1074-5521(01)00079-5 BindingDB Entry DOI: 10.7270/Q20V8B63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease (Human immunodeficiency virus) | BDBM40775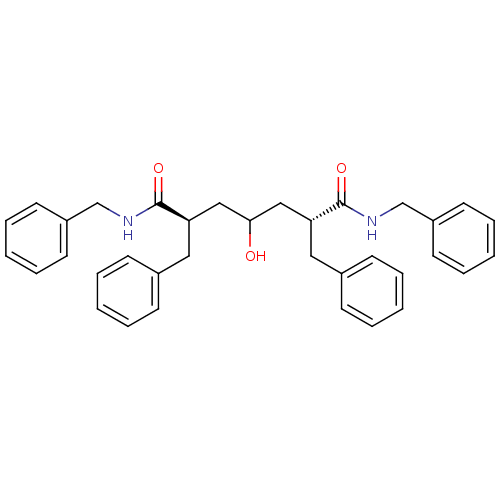 (Carbinol, 18a) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York | Assay Description Enzyme inhibition assay for silanediols, carbinols, and indinavir using assay system C. | Chem Biol 8: 1161-6 (2001) Article DOI: 10.1016/S1074-5521(01)00079-5 BindingDB Entry DOI: 10.7270/Q20V8B63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 protease (Human immunodeficiency virus) | BDBM40772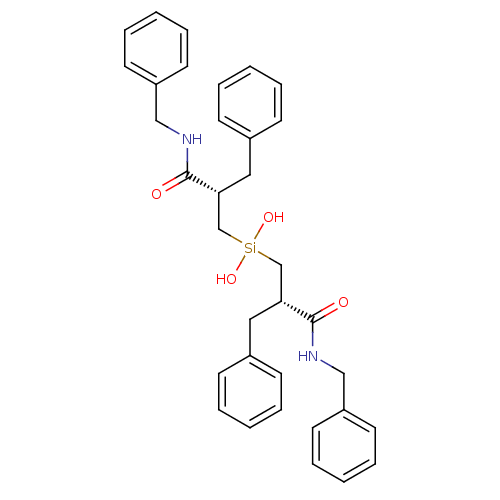 (Silanediol, 17a) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York | Assay Description Enzyme inhibition assay for silanediols, carbinols, and indinavir using assay system C. | Chem Biol 8: 1161-6 (2001) Article DOI: 10.1016/S1074-5521(01)00079-5 BindingDB Entry DOI: 10.7270/Q20V8B63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||