 Found 53642 hits with Last Name = 'ge' and Initial = 'l'
Found 53642 hits with Last Name = 'ge' and Initial = 'l' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50293089
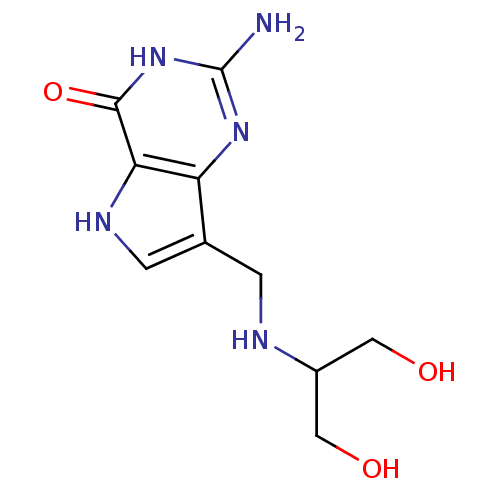
(2-Amino-7-{[(1,3-dihydroxypropan-2-yl)amino]methyl...)Show InChI InChI=1S/C10H15N5O3/c11-10-14-7-5(1-12-6(3-16)4-17)2-13-8(7)9(18)15-10/h2,6,12-13,16-17H,1,3-4H2,(H3,11,14,15,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 3 [306-416]
(Homo sapiens (Human)) | BDBM205431
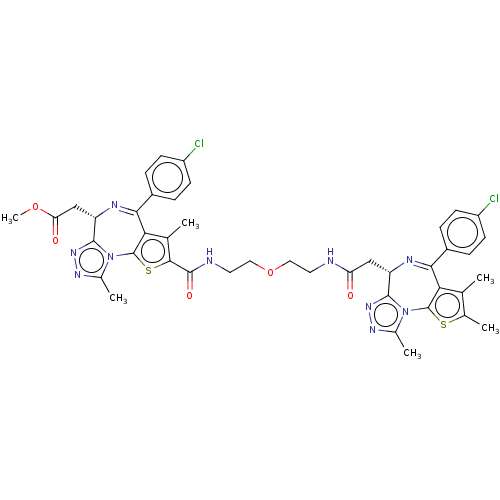
((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Neurotensin receptor type 2
(Homo sapiens (Human)) | BDBM50553771
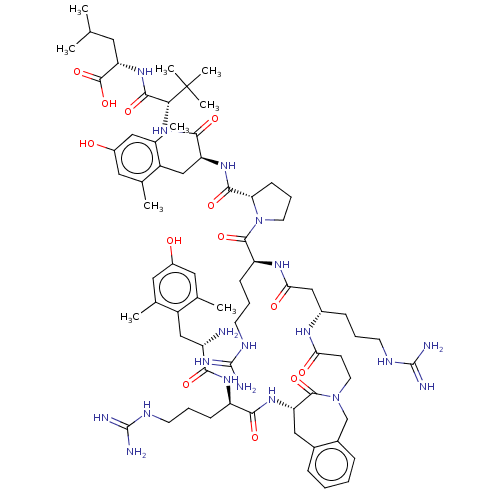
(CHEMBL4776986)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1c(C)cc(O)cc1C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)C[C@H](CCCNC(N)=N)NC(=O)CCN1Cc2ccccc2C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc2c(C)cc(O)cc2C)C1=O)C(C)(C)C)C(O)=O |r| | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [125I]-neurotensin from human recombinant NTS2 stably expressed in human 1321N1 cell membranes incubated for 60 mins by gamma counter... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01376
BindingDB Entry DOI: 10.7270/Q2H41W2P |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50293090
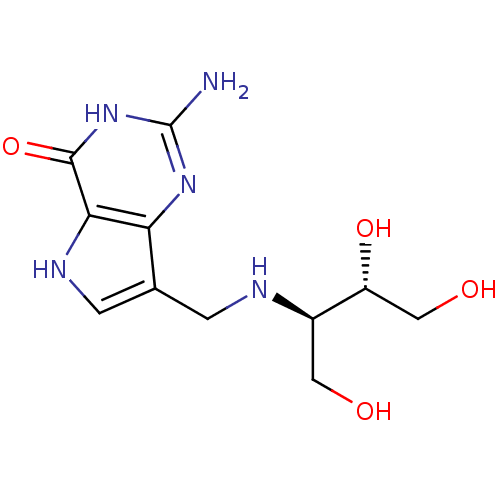
(2-Amino-7-({[(2R,3S)-1,3,4-trihydroxybutan-2-yl]am...)Show SMILES Nc1nc2c(CN[C@H](CO)[C@H](O)CO)c[nH]c2c(=O)[nH]1 |r| Show InChI InChI=1S/C11H17N5O4/c12-11-15-8-5(2-14-9(8)10(20)16-11)1-13-6(3-17)7(19)4-18/h2,6-7,13-14,17-19H,1,3-4H2,(H3,12,15,16,20)/t6-,7-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 4 [333-460]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50246593
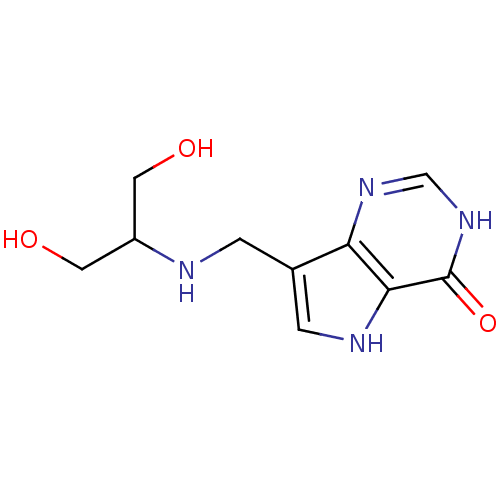
(7-((1,3-dihydroxypropan-2-ylamino)methyl)-3H-pyrro...)Show InChI InChI=1S/C10H14N4O3/c15-3-7(4-16)11-1-6-2-12-9-8(6)13-5-14-10(9)17/h2,5,7,11-12,15-16H,1,3-4H2,(H,13,14,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bromodomain-containing protein 2 [348-455]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50293087
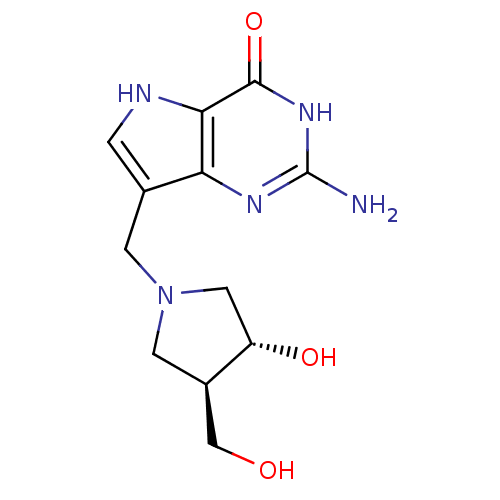
(2-amino-7-(((3R,4R)-3-hydroxy-4-(hydroxymethyl)pyr...)Show SMILES Nc1nc2c(CN3C[C@H](O)[C@@H](CO)C3)c[nH]c2c(=O)[nH]1 |r| Show InChI InChI=1S/C12H17N5O3/c13-12-15-9-6(1-14-10(9)11(20)16-12)2-17-3-7(5-18)8(19)4-17/h1,7-8,14,18-19H,2-5H2,(H3,13,15,16,20)/t7-,8+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Isoform C of Bromodomain-containing protein 4 (Short)
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50293091

(7-({[(1R,2S)-2,3-DIHYDROXY-1-(HYDROXYMETHYL)PROPYL...)Show SMILES OC[C@@H](O)[C@@H](CO)NCc1c[nH]c2c1nc[nH]c2=O |r| Show InChI InChI=1S/C11H16N4O4/c16-3-7(8(18)4-17)12-1-6-2-13-10-9(6)14-5-15-11(10)19/h2,5,7-8,12-13,16-18H,1,3-4H2,(H,14,15,19)/t7-,8-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM22109
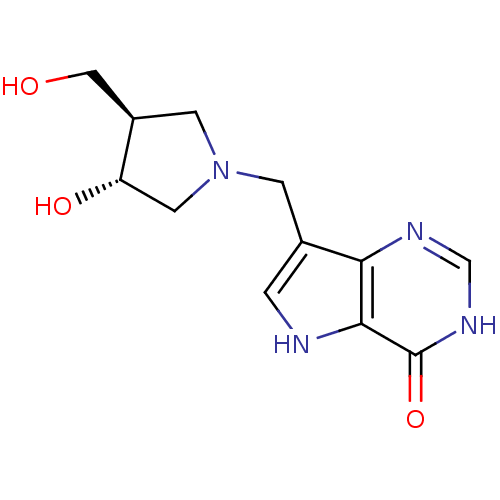
(7-{[(3R,4R)-3-hydroxy-4-(hydroxymethyl)pyrrolidin-...)Show SMILES OC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C12H16N4O3/c17-5-8-3-16(4-9(8)18)2-7-1-13-11-10(7)14-6-15-12(11)19/h1,6,8-9,13,17-18H,2-5H2,(H,14,15,19)/t8-,9+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bromodomain-containing protein 2 [71-194,348-455]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 3 [24-144,306-416]
(Homo sapiens (Human)) | BDBM205453
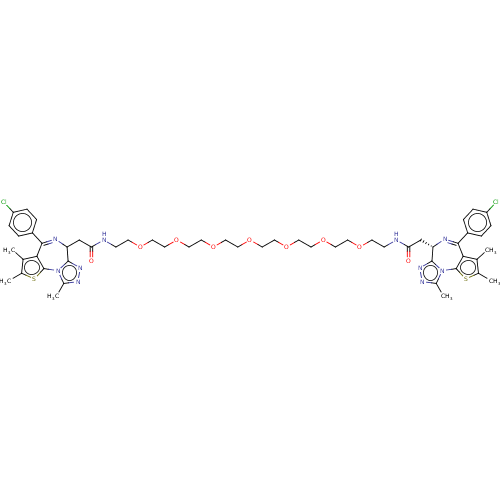
(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 4 [44-168,333-460]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 2 [71-194]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50327601
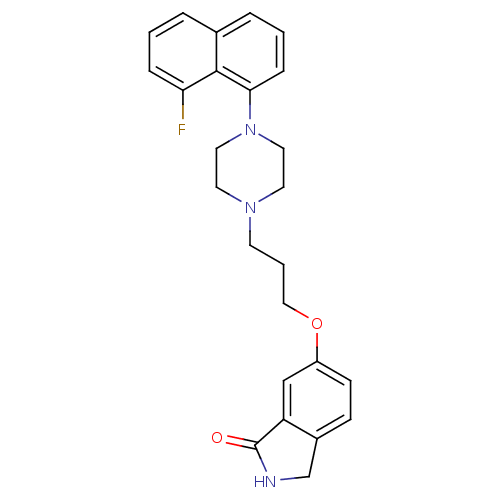
(6-(3-(4-(8-fluoronaphthalen-1-yl)piperazin-1-yl)pr...)Show SMILES Fc1cccc2cccc(N3CCN(CCCOc4ccc5CNC(=O)c5c4)CC3)c12 Show InChI InChI=1S/C25H26FN3O2/c26-22-6-1-4-18-5-2-7-23(24(18)22)29-13-11-28(12-14-29)10-3-15-31-20-9-8-19-17-27-25(30)21(19)16-20/h1-2,4-9,16H,3,10-15,17H2,(H,27,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0141 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from human 5HT1A receptor expressed in HeLa cells |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50073120
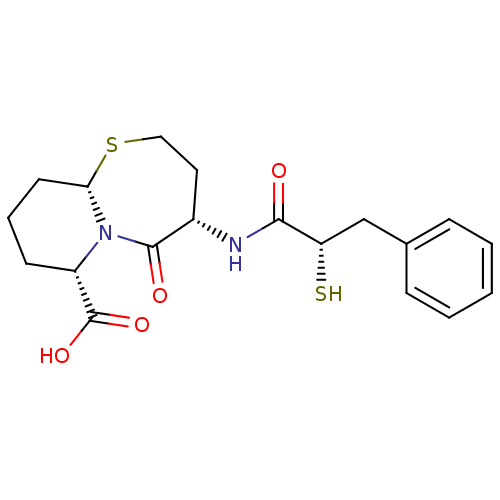
((4S,6S,9aS)-6-((S)-2-Mercapto-3-phenyl-propionylam...)Show SMILES OC(=O)[C@@H]1CCC[C@@H]2SCC[C@H](NC(=O)[C@@H](S)Cc3ccccc3)C(=O)N12 |r| Show InChI InChI=1S/C19H24N2O4S2/c22-17(15(26)11-12-5-2-1-3-6-12)20-13-9-10-27-16-8-4-7-14(19(24)25)21(16)18(13)23/h1-3,5-6,13-16,26H,4,7-11H2,(H,20,22)(H,24,25)/t13-,14-,15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bath
Curated by ChEMBL
| Assay Description
Inhibition of human fully glycosylated ACE N-terminal domain expressed in CHO cells using Cbz-Phe-His-Leu as substrate preincubated for 15 mins follo... |
J Med Chem 61: 10141-10154 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01309
BindingDB Entry DOI: 10.7270/Q2862K4R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50327602
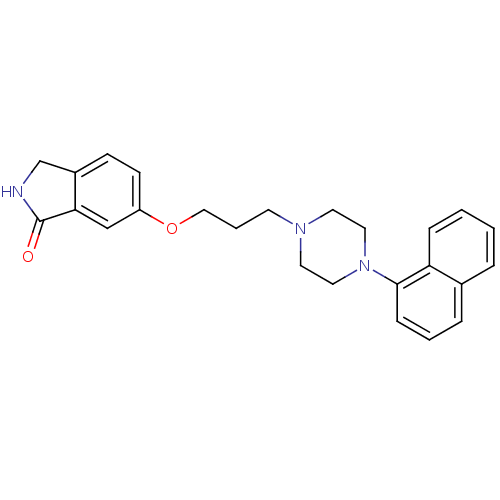
(6-(3-(4-(naphthalen-1-yl)piperazin-1-yl)propoxy)is...)Show SMILES O=C1NCc2ccc(OCCCN3CCN(CC3)c3cccc4ccccc34)cc12 Show InChI InChI=1S/C25H27N3O2/c29-25-23-17-21(10-9-20(23)18-26-25)30-16-4-11-27-12-14-28(15-13-27)24-8-3-6-19-5-1-2-7-22(19)24/h1-3,5-10,17H,4,11-16,18H2,(H,26,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0152 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from human 5HT1A receptor expressed in HeLa cells |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50073120

((4S,6S,9aS)-6-((S)-2-Mercapto-3-phenyl-propionylam...)Show SMILES OC(=O)[C@@H]1CCC[C@@H]2SCC[C@H](NC(=O)[C@@H](S)Cc3ccccc3)C(=O)N12 |r| Show InChI InChI=1S/C19H24N2O4S2/c22-17(15(26)11-12-5-2-1-3-6-12)20-13-9-10-27-16-8-4-7-14(19(24)25)21(16)18(13)23/h1-3,5-6,13-16,26H,4,7-11H2,(H,20,22)(H,24,25)/t13-,14-,15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bath
Curated by ChEMBL
| Assay Description
Inhibition of human fully glycosylated ACE C-terminal domain expressed in CHO cells using Cbz-Phe-His-Leu as substrate preincubated for 15 mins follo... |
J Med Chem 61: 10141-10154 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01309
BindingDB Entry DOI: 10.7270/Q2862K4R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50063292
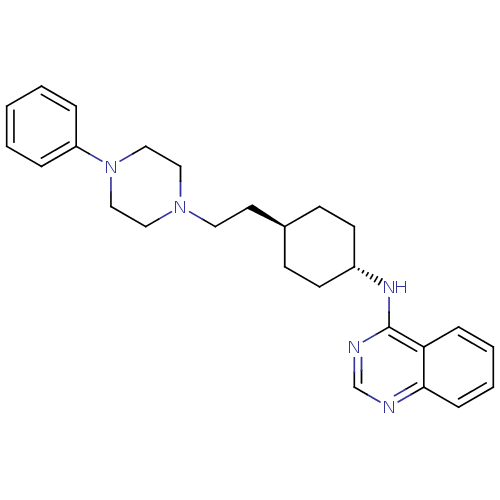
(CHEMBL349426 | {4-[2-(4-Phenyl-piperazin-1-yl)-eth...)Show SMILES C(CN1CCN(CC1)c1ccccc1)[C@H]1CC[C@@H](CC1)Nc1ncnc2ccccc12 |wU:17.22,wD:14.15,(10.31,-6.02,;11.15,-7.32,;12.69,-7.24,;13.39,-5.86,;14.93,-5.79,;15.76,-7.07,;15.07,-8.45,;13.53,-8.53,;17.3,-7,;17.99,-5.63,;19.5,-5.53,;20.36,-6.83,;19.66,-8.19,;18.13,-8.28,;8.79,-6.11,;7.95,-4.81,;6.42,-4.88,;5.72,-6.26,;6.55,-7.54,;8.09,-7.47,;4.18,-6.33,;3.34,-5.04,;4.04,-3.66,;3.23,-2.36,;1.69,-2.43,;.99,-3.81,;-.55,-3.87,;-1.26,-5.23,;-.44,-6.54,;1.1,-6.47,;1.8,-5.11,)| Show InChI InChI=1S/C26H33N5/c1-2-6-23(7-3-1)31-18-16-30(17-19-31)15-14-21-10-12-22(13-11-21)29-26-24-8-4-5-9-25(24)27-20-28-26/h1-9,20-22H,10-19H2,(H,27,28,29)/t21-,22- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Binding affinity determined by measuring displacement of [3H]-spiperone from cloned Human Dopamine receptor D3 in CHO-K1 cells |
J Med Chem 41: 760-71 (1998)
Article DOI: 10.1021/jm9707378
BindingDB Entry DOI: 10.7270/Q20G3J97 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50290221
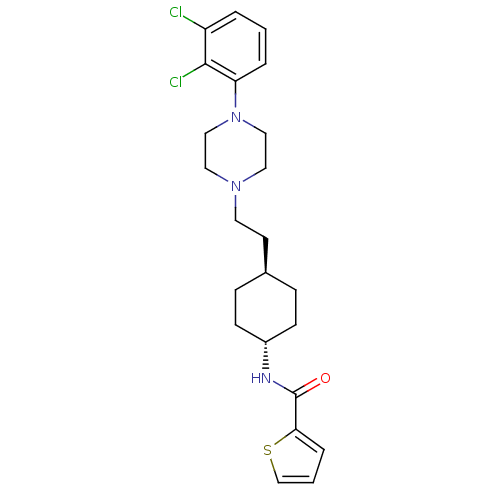
(CHEMBL80919 | Thiophene-2-carboxylic acid (4-{2-[4...)Show SMILES Clc1cccc(N2CCN(CC[C@H]3CC[C@@H](CC3)NC(=O)c3cccs3)CC2)c1Cl |wU:12.11,wD:15.18,(20.53,-4.53,;19.79,-5.88,;20.6,-7.19,;19.85,-8.55,;18.31,-8.59,;17.52,-7.26,;15.98,-7.29,;15.24,-8.63,;13.7,-8.66,;12.91,-7.36,;11.37,-7.38,;10.6,-8.73,;9.06,-8.73,;8.29,-10.06,;6.75,-10.06,;5.98,-8.73,;6.74,-7.4,;8.28,-7.4,;4.44,-8.75,;3.67,-10.08,;2.13,-10.09,;4.44,-11.41,;5.98,-11.49,;5.83,-14.15,;4.3,-14.05,;3.59,-12.7,;13.66,-6,;15.2,-5.98,;18.25,-5.91,;17.46,-4.6,)| Show InChI InChI=1S/C23H29Cl2N3OS/c24-19-3-1-4-20(22(19)25)28-14-12-27(13-15-28)11-10-17-6-8-18(9-7-17)26-23(29)21-5-2-16-30-21/h1-5,16-18H,6-15H2,(H,26,29)/t17-,18- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Ability to displace radioligand [3H]N-0437 from human dopamine D2 receptor transfected chinese hamster ovary cell membranes. |
Bioorg Med Chem Lett 7: 2403-2408 (1997)
Article DOI: 10.1016/S0960-894X(97)00443-5
BindingDB Entry DOI: 10.7270/Q27W6CQ0 |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 2 [71-194,348-455]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12693
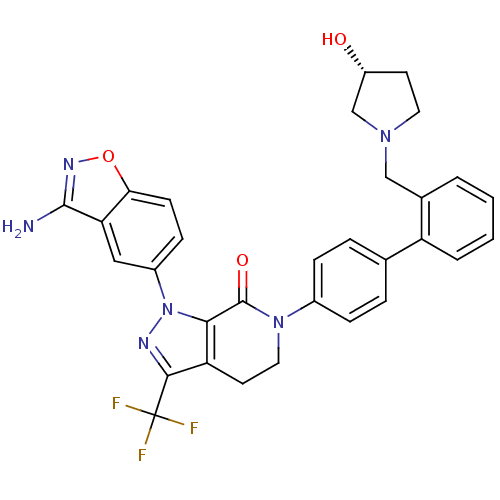
(1-(3-amino-1,2-benzoxazol-5-yl)-6-[4-(2-{[(3R)-3-h...)Show SMILES Nc1noc2ccc(cc12)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)-c1ccccc1CN1CC[C@@H](O)C1)C(F)(F)F |r| Show InChI InChI=1S/C31H27F3N6O3/c32-31(33,34)28-24-12-14-39(30(42)27(24)40(36-28)21-9-10-26-25(15-21)29(35)37-43-26)20-7-5-18(6-8-20)23-4-2-1-3-19(23)16-38-13-11-22(41)17-38/h1-10,15,22,41H,11-14,16-17H2,(H2,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0300 | -59.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 4141-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.069
BindingDB Entry DOI: 10.7270/Q26T0JW1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345133
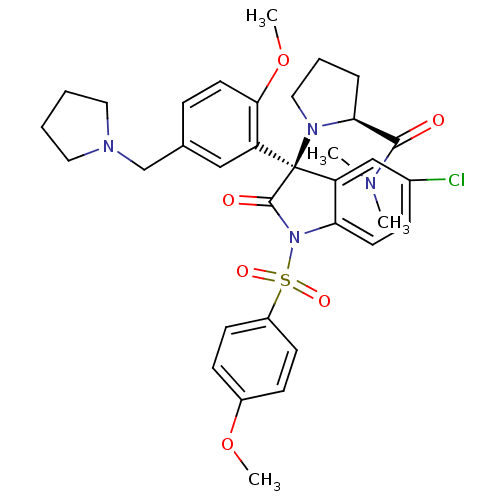
((S)-1-((R)-5-chloro-3-(2-methoxy-5-(pyrrolidin-1-y...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1cc(CN2CCCC2)ccc1OC |r| Show InChI InChI=1S/C34H39ClN4O6S/c1-36(2)32(40)30-8-7-19-38(30)34(28-20-23(9-16-31(28)45-4)22-37-17-5-6-18-37)27-21-24(35)10-15-29(27)39(33(34)41)46(42,43)26-13-11-25(44-3)12-14-26/h9-16,20-21,30H,5-8,17-19,22H2,1-4H3/t30-,34+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Neurotensin receptor type 2
(Homo sapiens (Human)) | BDBM50553769
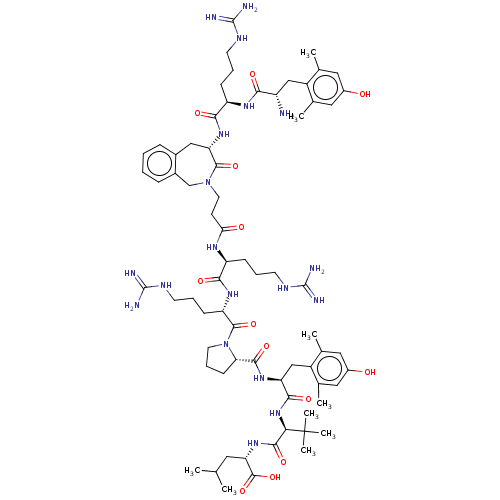
(CHEMBL4798887)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1c(C)cc(O)cc1C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)CCN1Cc2ccccc2C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc2c(C)cc(O)cc2C)C1=O)C(C)(C)C)C(O)=O |r| | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [125I]-neurotensin from human recombinant NTS2 stably expressed in human 1321N1 cell membranes incubated for 60 mins by gamma counter... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01376
BindingDB Entry DOI: 10.7270/Q2H41W2P |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345134
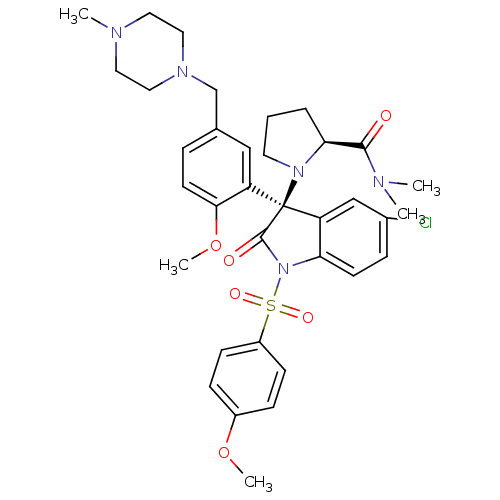
((S)-1-((R)-5-chloro-3-(2-methoxy-5-((4-methylpiper...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1cc(CN2CCN(C)CC2)ccc1OC |r| Show InChI InChI=1S/C35H42ClN5O6S/c1-37(2)33(42)31-7-6-16-40(31)35(29-21-24(8-15-32(29)47-5)23-39-19-17-38(3)18-20-39)28-22-25(36)9-14-30(28)41(34(35)43)48(44,45)27-12-10-26(46-4)11-13-27/h8-15,21-22,31H,6-7,16-20,23H2,1-5H3/t31-,35+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 2 [348-455]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Cysteine protease
(Trypanosoma brucei rhodesiense) | BDBM50258507

(CHEMBL4078345)Show SMILES CC(=O)\C=C\[C@H](CCc1ccccc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCc1ccccc1 |r| Show InChI InChI=1S/C30H32N2O4/c1-23(33)17-19-27(20-18-24-11-5-2-6-12-24)31-29(34)28(21-25-13-7-3-8-14-25)32-30(35)36-22-26-15-9-4-10-16-26/h2-17,19,27-28H,18,20-22H2,1H3,(H,31,34)(H,32,35)/b19-17+/t27-,28+/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemical, Biological, Pharmaceutical and Environmental Sciences, University of Messina , Viale Annunziata, 98168 Messina, Italy.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Trypanosoma brucei rhodesiense rhodesain expressed in Pichia pastoris using Cbz-Phe-Arg-AMC as substrate after 30 min by fl... |
J Med Chem 60: 6911-6923 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00405
BindingDB Entry DOI: 10.7270/Q2FJ2K73 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12681
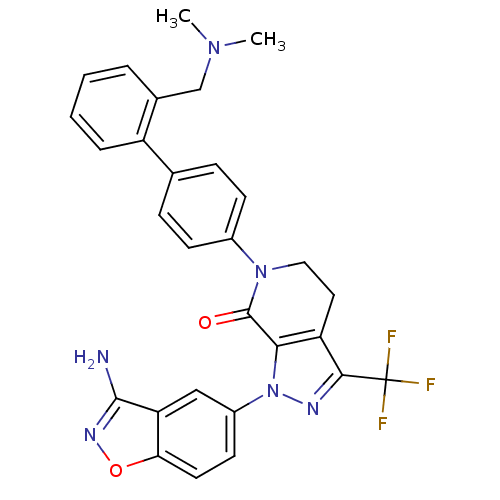
(1-(3-amino-1,2-benzoxazol-5-yl)-6-(4-{2-[(dimethyl...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)N1CCc2c(nn(c2C1=O)-c1ccc2onc(N)c2c1)C(F)(F)F Show InChI InChI=1S/C29H25F3N6O2/c1-36(2)16-18-5-3-4-6-21(18)17-7-9-19(10-8-17)37-14-13-22-25(28(37)39)38(34-26(22)29(30,31)32)20-11-12-24-23(15-20)27(33)35-40-24/h3-12,15H,13-14,16H2,1-2H3,(H2,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | -58.8 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 4141-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.069
BindingDB Entry DOI: 10.7270/Q26T0JW1 |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345131
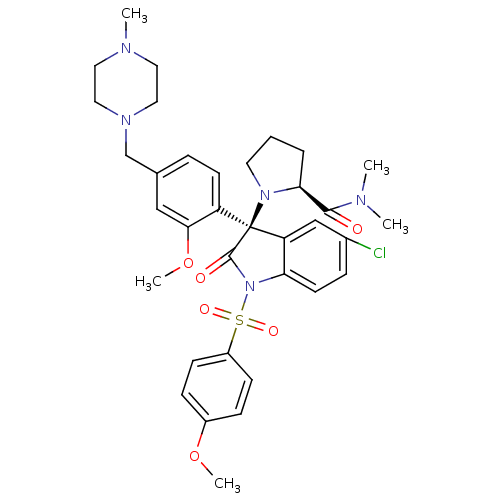
((S)-1-((R)-5-chloro-3-(2-methoxy-4-((4-methylpiper...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1ccc(CN2CCN(C)CC2)cc1OC |r| Show InChI InChI=1S/C35H42ClN5O6S/c1-37(2)33(42)31-7-6-16-40(31)35(28-14-8-24(21-32(28)47-5)23-39-19-17-38(3)18-20-39)29-22-25(36)9-15-30(29)41(34(35)43)48(44,45)27-12-10-26(46-4)11-13-27/h8-15,21-22,31H,6-7,16-20,23H2,1-5H3/t31-,35-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50433530
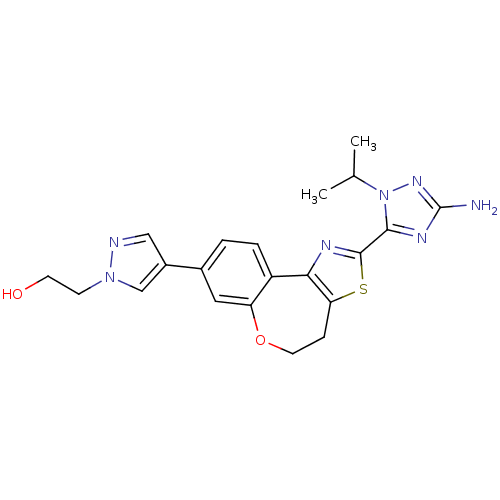
(CHEMBL2381382)Show SMILES CC(C)n1nc(N)nc1-c1nc-2c(CCOc3cc(ccc-23)-c2cnn(CCO)c2)s1 Show InChI InChI=1S/C21H23N7O2S/c1-12(2)28-19(25-21(22)26-28)20-24-18-15-4-3-13(14-10-23-27(11-14)6-7-29)9-16(15)30-8-5-17(18)31-20/h3-4,9-12,29H,5-8H2,1-2H3,(H2,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of GST-fused human recombinant PI3Kalpha expressed in baculovirus infected SF9 cells after 1 hr by scintillation proximity assay in presen... |
Bioorg Med Chem Lett 23: 2606-13 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.102
BindingDB Entry DOI: 10.7270/Q2W95BKF |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50293086
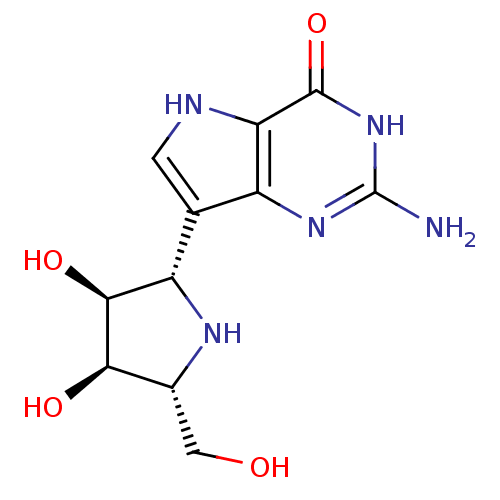
((1S)-1,4-dideoxy-1,4-imino-1-(9-deazaguanin-9-yl)-...)Show SMILES Nc1nc2c(c[nH]c2c(=O)[nH]1)[C@@H]1N[C@H](CO)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C11H15N5O4/c12-11-15-5-3(1-13-7(5)10(20)16-11)6-9(19)8(18)4(2-17)14-6/h1,4,6,8-9,13-14,17-19H,2H2,(H3,12,15,16,20)/t4-,6+,8-,9+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
Article
PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50327603
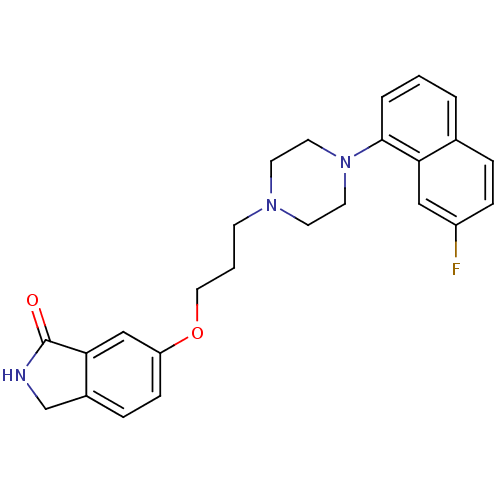
(6-(3-(4-(7-fluoronaphthalen-1-yl)piperazin-1-yl)pr...)Show SMILES Fc1ccc2cccc(N3CCN(CCCOc4ccc5CNC(=O)c5c4)CC3)c2c1 Show InChI InChI=1S/C25H26FN3O2/c26-20-7-5-18-3-1-4-24(22(18)15-20)29-12-10-28(11-13-29)9-2-14-31-21-8-6-19-17-27-25(30)23(19)16-21/h1,3-8,15-16H,2,9-14,17H2,(H,27,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0447 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from human 5HT1A receptor expressed in HeLa cells |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50098242

(3-[4-(Amino-hydrazino-methyl)-phenyl]-N-cyclopenty...)Show SMILES CN(C1CCCC1)C(=O)[C@H](Cc1ccc(cc1)C(N)NN)NS(=O)(=O)c1ccc2CCCCCc2c1 Show InChI InChI=1S/C27H39N5O3S/c1-32(23-9-5-6-10-23)27(33)25(17-19-11-13-21(14-12-19)26(28)30-29)31-36(34,35)24-16-15-20-7-3-2-4-8-22(20)18-24/h11-16,18,23,25-26,30-31H,2-10,17,28-29H2,1H3/t25-,26?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Binding affinity was determined against human thrombin |
J Med Chem 44: 1043-50 (2001)
BindingDB Entry DOI: 10.7270/Q2KS6S86 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50327604
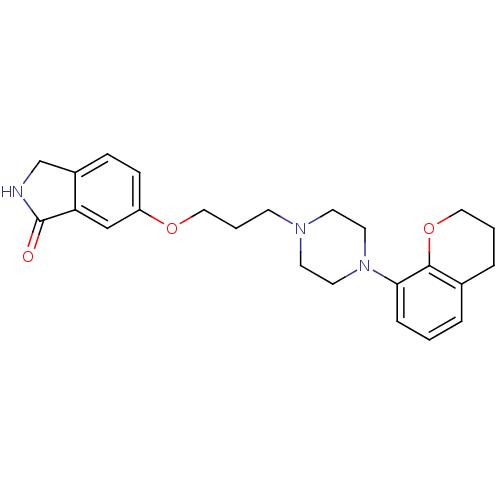
(6-(3-(4-(chroman-8-yl)piperazin-1-yl)propoxy)isoin...)Show SMILES O=C1NCc2ccc(OCCCN3CCN(CC3)c3cccc4CCCOc34)cc12 Show InChI InChI=1S/C24H29N3O3/c28-24-21-16-20(8-7-19(21)17-25-24)29-15-3-9-26-10-12-27(13-11-26)22-6-1-4-18-5-2-14-30-23(18)22/h1,4,6-8,16H,2-3,5,9-15,17H2,(H,25,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1A receptor expressed in HeLa cells by scintillation proximity assay |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50327605
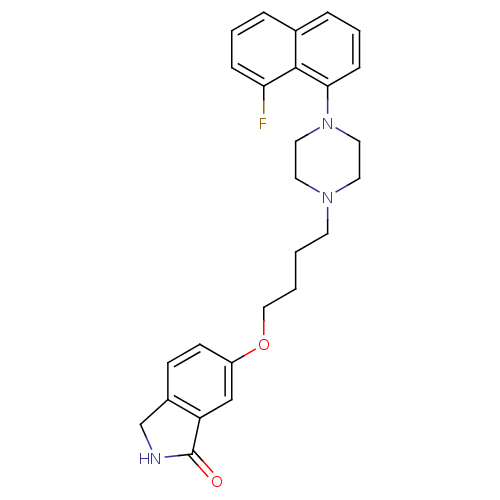
(6-(4-(4-(8-fluoronaphthalen-1-yl)piperazin-1-yl)bu...)Show SMILES Fc1cccc2cccc(N3CCN(CCCCOc4ccc5CNC(=O)c5c4)CC3)c12 Show InChI InChI=1S/C26H28FN3O2/c27-23-7-3-5-19-6-4-8-24(25(19)23)30-14-12-29(13-15-30)11-1-2-16-32-21-10-9-20-18-28-26(31)22(20)17-21/h3-10,17H,1-2,11-16,18H2,(H,28,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0504 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from human 5HT1A receptor expressed in HeLa cells |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 4 [333-460]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM50195587
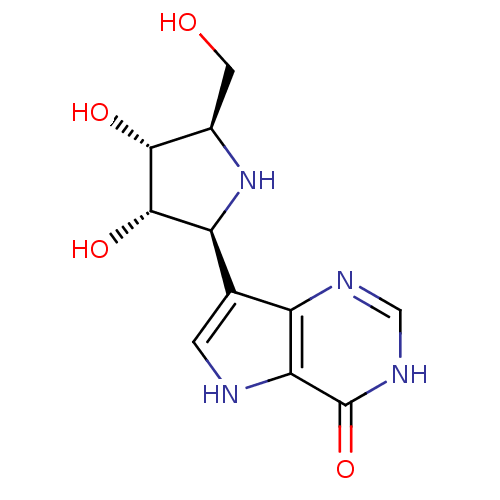
(1,4-DIDEOXY-4-AZA-1-(S)-(9-DEAZAHYPOXANTHIN-9-YL)-...)Show SMILES OC[C@H]1N[C@H]([C@H](O)[C@@H]1O)c1c[nH]c2c1nc[nH]c2=O |r| Show InChI InChI=1S/C11H14N4O4/c16-2-5-9(17)10(18)7(15-5)4-1-12-8-6(4)13-3-14-11(8)19/h1,3,5,7,9-10,12,15-18H,2H2,(H,13,14,19)/t5-,7+,9-,10+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human PNP by xanthine-oxidase coupled assay |
J Med Chem 52: 1126-43 (2009)
Article DOI: 10.1021/jm801421q
BindingDB Entry DOI: 10.7270/Q2QR4Z18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50327606
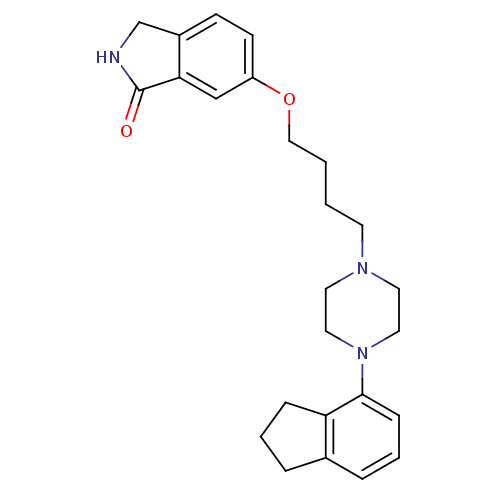
(6-(4-(4-(2,3-dihydro-1H-inden-4-yl)piperazin-1-yl)...)Show SMILES O=C1NCc2ccc(OCCCCN3CCN(CC3)c3cccc4CCCc34)cc12 Show InChI InChI=1S/C25H31N3O2/c29-25-23-17-21(10-9-20(23)18-26-25)30-16-2-1-11-27-12-14-28(15-13-27)24-8-4-6-19-5-3-7-22(19)24/h4,6,8-10,17H,1-3,5,7,11-16,18H2,(H,26,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0575 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-Spiperone from human dopamine D2L receptor expressed in CHO cells |
Bioorg Med Chem Lett 20: 5666-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.023
BindingDB Entry DOI: 10.7270/Q2HH6K9S |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 3 [24-144]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50007692
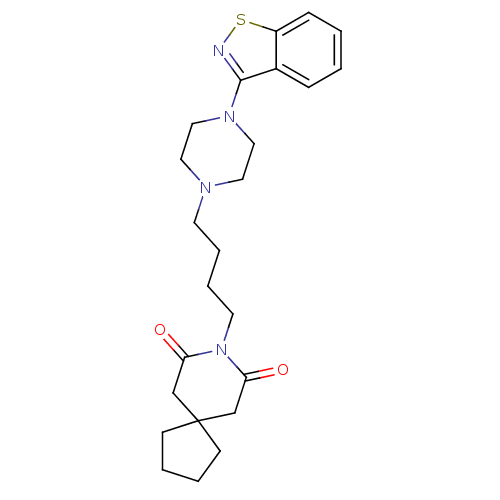
(8-[4-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-bu...)Show SMILES O=C1CC2(CCCC2)CC(=O)N1CCCCN1CCN(CC1)c1nsc2ccccc12 Show InChI InChI=1S/C24H32N4O2S/c29-21-17-24(9-3-4-10-24)18-22(30)28(21)12-6-5-11-26-13-15-27(16-14-26)23-19-7-1-2-8-20(19)31-25-23/h1-2,7-8H,3-6,9-18H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Case Western Reserve University
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 120: 365-8 (1995)
Article DOI: 10.1007/bf02311185
BindingDB Entry DOI: 10.7270/Q2VT1QKB |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM21397
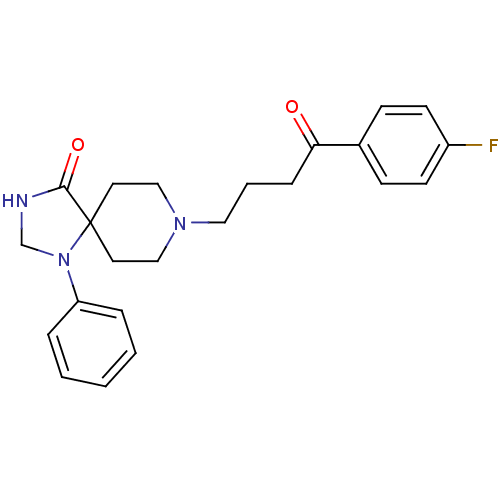
(8-[4-(4-fluorophenyl)-4-keto-butyl]-1-phenyl-1,3,8...)Show SMILES Fc1ccc(cc1)C(=O)CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1 Show InChI InChI=1S/C23H26FN3O2/c24-19-10-8-18(9-11-19)21(28)7-4-14-26-15-12-23(13-16-26)22(29)25-17-27(23)20-5-2-1-3-6-20/h1-3,5-6,8-11H,4,7,12-17H2,(H,25,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Case Western Reserve University
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 120: 365-8 (1995)
Article DOI: 10.1007/bf02311185
BindingDB Entry DOI: 10.7270/Q2VT1QKB |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
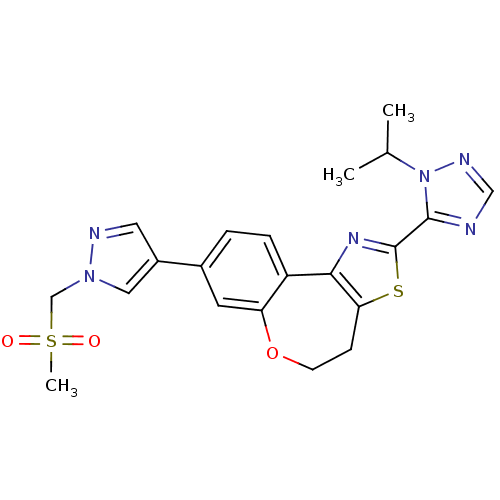
(Homo sapiens (Human)) | BDBM50433534
(CHEMBL2381375)Show SMILES CC(C)n1ncnc1-c1nc-2c(CCOc3cc(ccc-23)-c2cnn(CS(C)(=O)=O)c2)s1 Show InChI InChI=1S/C21H22N6O3S2/c1-13(2)27-20(22-11-24-27)21-25-19-16-5-4-14(8-17(16)30-7-6-18(19)31-21)15-9-23-26(10-15)12-32(3,28)29/h4-5,8-11,13H,6-7,12H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of GST-fused human recombinant PI3Kalpha expressed in baculovirus infected SF9 cells after 1 hr by scintillation proximity assay in presen... |
Bioorg Med Chem Lett 23: 2606-13 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.102
BindingDB Entry DOI: 10.7270/Q2W95BKF |
More data for this
Ligand-Target Pair | |
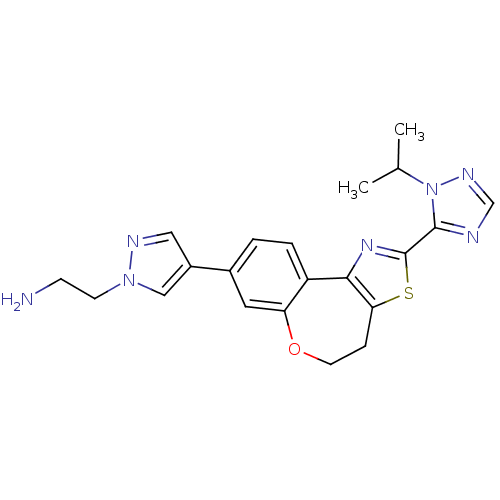
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50433533
(CHEMBL2381376)Show SMILES CC(C)n1ncnc1-c1nc-2c(CCOc3cc(ccc-23)-c2cnn(CCN)c2)s1 Show InChI InChI=1S/C21H23N7OS/c1-13(2)28-20(23-12-25-28)21-26-19-16-4-3-14(15-10-24-27(11-15)7-6-22)9-17(16)29-8-5-18(19)30-21/h3-4,9-13H,5-8,22H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of GST-fused human recombinant PI3Kalpha expressed in baculovirus infected SF9 cells after 1 hr by scintillation proximity assay in presen... |
Bioorg Med Chem Lett 23: 2606-13 (2013)
Article DOI: 10.1016/j.bmcl.2013.02.102
BindingDB Entry DOI: 10.7270/Q2W95BKF |
More data for this
Ligand-Target Pair | |
Isoform C of Bromodomain-containing protein 4 (Short)
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Bromodomain testis-specific protein [251-382]
(Homo sapiens (Human)) | BDBM205431

((6S+2S)-PEG1 (7))Show SMILES COC(=O)C[C@@H]1N=C(c2c(C)c(sc2-n2c(C)nnc12)C(=O)NCCOCCNC(=O)C[C@@H]1N=C(c2c(C)c(C)sc2-n2c(C)nnc12)c1ccc(Cl)cc1)c1ccc(Cl)cc1 |r,c:6,36| Show InChI InChI=1S/C43H42Cl2N10O5S2/c1-21-23(3)61-42-34(21)36(26-7-11-28(44)12-8-26)48-30(39-52-50-24(4)54(39)42)19-32(56)46-15-17-60-18-16-47-41(58)38-22(2)35-37(27-9-13-29(45)14-10-27)49-31(20-33(57)59-6)40-53-51-25(5)55(40)43(35)62-38/h7-14,30-31H,15-20H2,1-6H3,(H,46,56)(H,47,58)/t30-,31-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 3 [306-416]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |
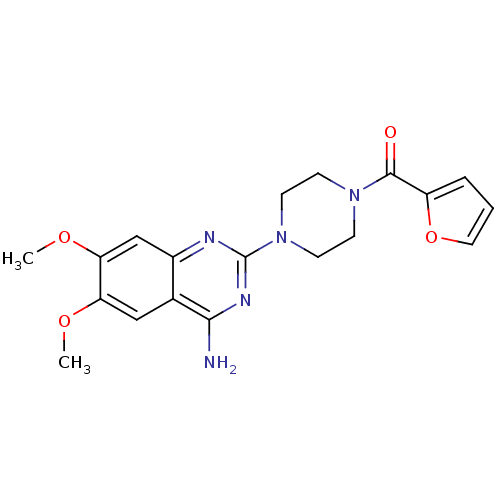
Alpha-1A adrenergic receptor
(Rattus norvegicus (Rat)) | BDBM29568
(CHEMBL2 | PRAZOSIN | PRAZOSIN HYDROCHLORIDE | [3H]...)Show SMILES COc1cc2nc(nc(N)c2cc1OC)N1CCN(CC1)C(=O)c1ccco1 Show InChI InChI=1S/C19H21N5O4/c1-26-15-10-12-13(11-16(15)27-2)21-19(22-17(12)20)24-7-5-23(6-8-24)18(25)14-4-3-9-28-14/h3-4,9-11H,5-8H2,1-2H3,(H2,20,21,22) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]- prazosin binding against Alpha-1A adrenergic receptor from rat submaxillary gland |
J Med Chem 42: 5181-7 (2000)
BindingDB Entry DOI: 10.7270/Q23T9HZ0 |
More data for this
Ligand-Target Pair | |
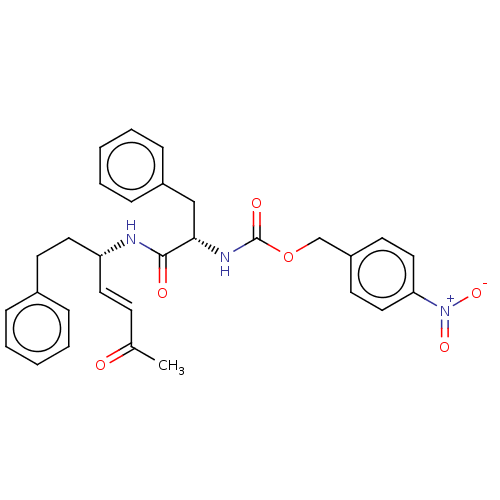
Cysteine protease
(Trypanosoma brucei rhodesiense) | BDBM50258514
(CHEMBL4062015)Show SMILES CC(=O)\C=C\[C@H](CCc1ccccc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCc1ccc(cc1)[N+]([O-])=O |r| Show InChI InChI=1S/C30H31N3O6/c1-22(34)12-16-26(17-13-23-8-4-2-5-9-23)31-29(35)28(20-24-10-6-3-7-11-24)32-30(36)39-21-25-14-18-27(19-15-25)33(37)38/h2-12,14-16,18-19,26,28H,13,17,20-21H2,1H3,(H,31,35)(H,32,36)/b16-12+/t26-,28+/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemical, Biological, Pharmaceutical and Environmental Sciences, University of Messina , Viale Annunziata, 98168 Messina, Italy.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Trypanosoma brucei rhodesiense rhodesain expressed in Pichia pastoris using Cbz-Phe-Arg-AMC as substrate after 30 min by fl... |
J Med Chem 60: 6911-6923 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00405
BindingDB Entry DOI: 10.7270/Q2FJ2K73 |
More data for this
Ligand-Target Pair | |
Bromodomain testis-specific protein [21-137,251-382]
(Homo sapiens (Human)) | BDBM205453

(MTI (35))Show SMILES Cc1sc-2c(c1C)C(=NC(CC(=O)NCCOCCOCCOCCOCCOCCOCCOCCNC(=O)C[C@@H]1N=C(c3c(C)c(C)sc3-n3c(C)nnc13)c1ccc(Cl)cc1)c1nnc(C)n-21)c1ccc(Cl)cc1 |r,c:8,43| Show InChI InChI=1S/C54H66Cl2N10O9S2/c1-33-35(3)76-53-47(33)49(39-7-11-41(55)12-8-39)59-43(51-63-61-37(5)65(51)53)31-45(67)57-15-17-69-19-21-71-23-25-73-27-29-75-30-28-74-26-24-72-22-20-70-18-16-58-46(68)32-44-52-64-62-38(6)66(52)54-48(34(2)36(4)77-54)50(60-44)40-9-13-42(56)14-10-40/h7-14,43-44H,15-32H2,1-6H3,(H,57,67)(H,58,68)/t43-,44?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dana-Farber Cancer Institute
| Assay Description
Refer to DiscoverX |
Nat Chem Biol 12: 1089-1096 (2016)
Article DOI: 10.1038/nchembio.2209
BindingDB Entry DOI: 10.7270/Q2VQ31HW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data