 Found 196 hits with Last Name = 'gibson' and Initial = 'n'
Found 196 hits with Last Name = 'gibson' and Initial = 'n' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183224
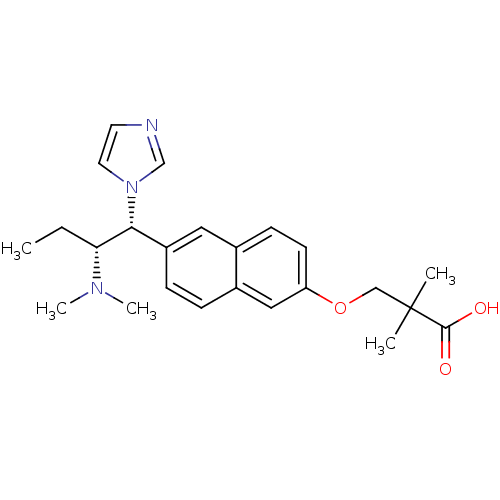
(3-[6-(2-dimethylamino-1-imidazol-1-yl-butyl)-napht...)Show SMILES CC[C@H]([C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C24H31N3O3/c1-6-21(26(4)5)22(27-12-11-25-16-27)19-8-7-18-14-20(10-9-17(18)13-19)30-15-24(2,3)23(28)29/h7-14,16,21-22H,6,15H2,1-5H3,(H,28,29)/t21-,22-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183243

(1-((6-((1R,2R)-2-(diethylamino)-1-(1H-imidazol-1-y...)Show SMILES CCN(CC)[C@H](C)[C@@H](c1ccc2cc(OCC3(CCCC3)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C27H35N3O3/c1-4-29(5-2)20(3)25(30-15-14-28-19-30)23-9-8-22-17-24(11-10-21(22)16-23)33-18-27(26(31)32)12-6-7-13-27/h8-11,14-17,19-20,25H,4-7,12-13,18H2,1-3H3,(H,31,32)/t20-,25+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183229
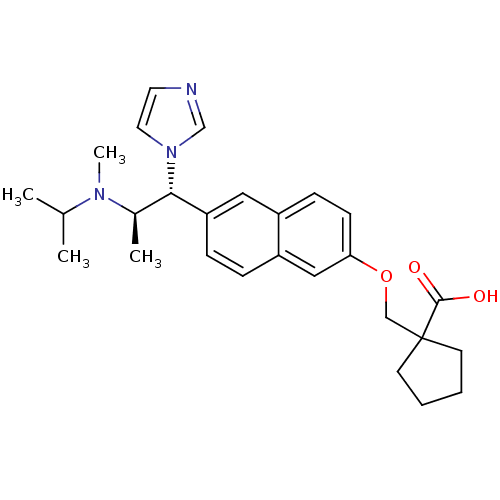
(1-((6-((1R,2R)-1-(1H-imidazol-1-yl)-2-(isopropyl(m...)Show SMILES CC(C)N(C)[C@H](C)[C@@H](c1ccc2cc(OCC3(CCCC3)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C27H35N3O3/c1-19(2)29(4)20(3)25(30-14-13-28-18-30)23-8-7-22-16-24(10-9-21(22)15-23)33-17-27(26(31)32)11-5-6-12-27/h7-10,13-16,18-20,25H,5-6,11-12,17H2,1-4H3,(H,31,32)/t20-,25+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183237
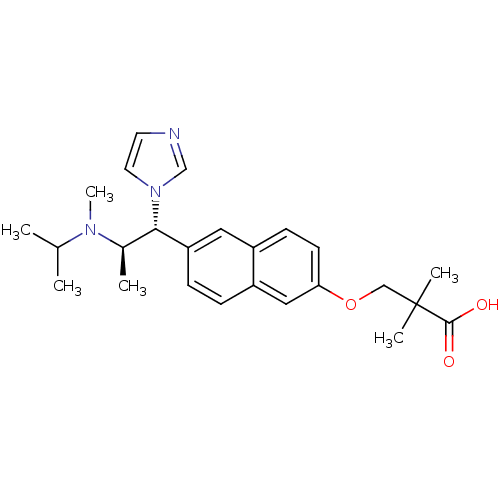
(3-(6-((1R,2R)-1-(1H-imidazol-1-yl)-2-(isopropyl(me...)Show SMILES CC(C)N(C)[C@H](C)[C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C25H33N3O3/c1-17(2)27(6)18(3)23(28-12-11-26-16-28)21-8-7-20-14-22(10-9-19(20)13-21)31-15-25(4,5)24(29)30/h7-14,16-18,23H,15H2,1-6H3,(H,29,30)/t18-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162787
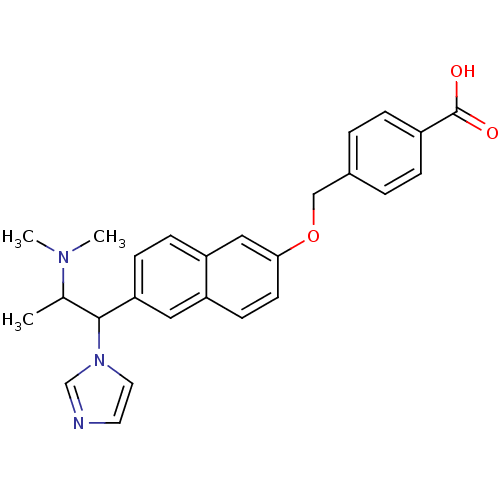
(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(cc3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H27N3O3/c1-18(28(2)3)25(29-13-12-27-17-29)23-9-8-22-15-24(11-10-21(22)14-23)32-16-19-4-6-20(7-5-19)26(30)31/h4-15,17-18,25H,16H2,1-3H3,(H,30,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162787

(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(cc3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H27N3O3/c1-18(28(2)3)25(29-13-12-27-17-29)23-9-8-22-15-24(11-10-21(22)14-23)32-16-19-4-6-20(7-5-19)26(30)31/h4-15,17-18,25H,16H2,1-3H3,(H,30,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162776
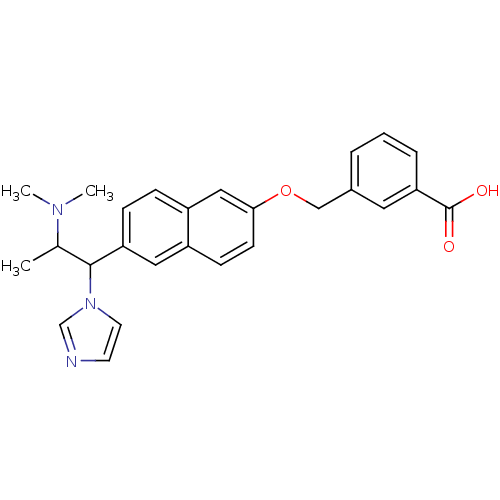
(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3cccc(c3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H27N3O3/c1-18(28(2)3)25(29-12-11-27-17-29)22-8-7-21-15-24(10-9-20(21)14-22)32-16-19-5-4-6-23(13-19)26(30)31/h4-15,17-18,25H,16H2,1-3H3,(H,30,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162776

(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3cccc(c3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H27N3O3/c1-18(28(2)3)25(29-12-11-27-17-29)22-8-7-21-15-24(10-9-20(21)14-22)32-16-19-5-4-6-23(13-19)26(30)31/h4-15,17-18,25H,16H2,1-3H3,(H,30,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50162780
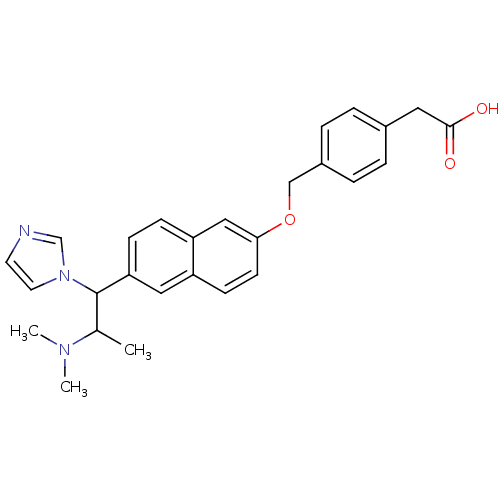
(CHEMBL180348 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(CC(O)=O)cc3)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)27(30-13-12-28-18-30)24-9-8-23-16-25(11-10-22(23)15-24)33-17-21-6-4-20(5-7-21)14-26(31)32/h4-13,15-16,18-19,27H,14,17H2,1-3H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Selectivity towards cytochrome P450 3A4 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50162783
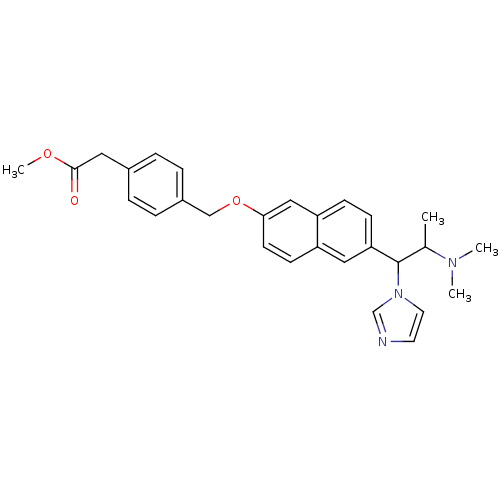
(CHEMBL179825 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES COC(=O)Cc1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C28H31N3O3/c1-20(30(2)3)28(31-14-13-29-19-31)25-10-9-24-17-26(12-11-23(24)16-25)34-18-22-7-5-21(6-8-22)15-27(32)33-4/h5-14,16-17,19-20,28H,15,18H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Selectivity towards cytochrome P450 3A4 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183231

(1-((6-((1R,2R)-2-(ethyl(methyl)amino)-1-(1H-imidaz...)Show SMILES CCN(C)[C@H](C)[C@@H](c1ccc2cc(OCC3(CCCC3)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C26H33N3O3/c1-4-28(3)19(2)24(29-14-13-27-18-29)22-8-7-21-16-23(10-9-20(21)15-22)32-17-26(25(30)31)11-5-6-12-26/h7-10,13-16,18-19,24H,4-6,11-12,17H2,1-3H3,(H,30,31)/t19-,24+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183225
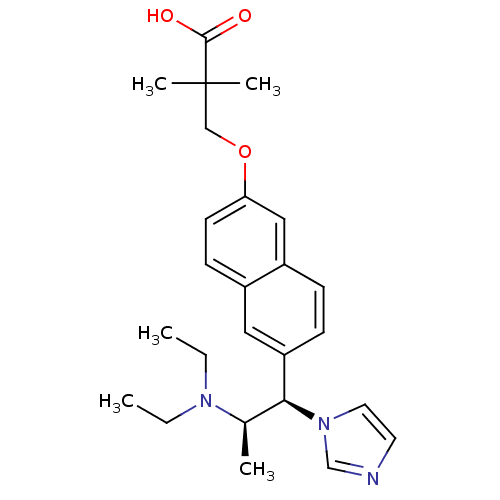
(3-(6-((1R,2R)-2-(diethylamino)-1-(1H-imidazol-1-yl...)Show SMILES CCN(CC)[C@H](C)[C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C25H33N3O3/c1-6-27(7-2)18(3)23(28-13-12-26-17-28)21-9-8-20-15-22(11-10-19(20)14-21)31-16-25(4,5)24(29)30/h8-15,17-18,23H,6-7,16H2,1-5H3,(H,29,30)/t18-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162791
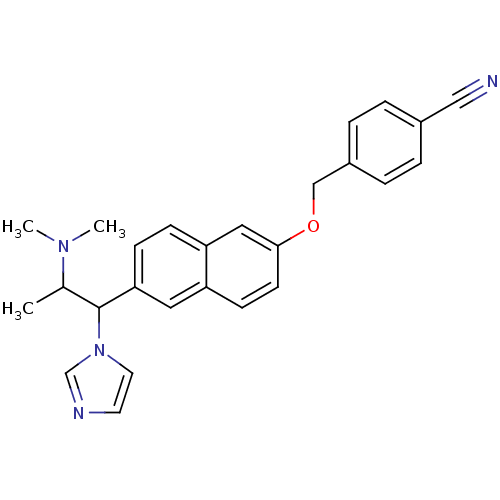
(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(cc3)C#N)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H26N4O/c1-19(29(2)3)26(30-13-12-28-18-30)24-9-8-23-15-25(11-10-22(23)14-24)31-17-21-6-4-20(16-27)5-7-21/h4-15,18-19,26H,17H2,1-3H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162791

(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(cc3)C#N)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H26N4O/c1-19(29(2)3)26(30-13-12-28-18-30)24-9-8-23-15-25(11-10-22(23)14-24)31-17-21-6-4-20(16-27)5-7-21/h4-15,18-19,26H,17H2,1-3H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162788

(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1cccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)c1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-13-12-28-18-30)23-9-8-22-16-25(11-10-21(22)15-23)33-17-20-6-5-7-24(14-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162788

(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1cccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)c1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-13-12-28-18-30)23-9-8-22-16-25(11-10-21(22)15-23)33-17-20-6-5-7-24(14-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183228
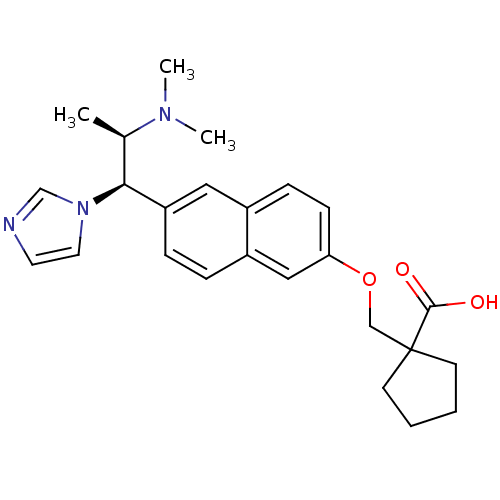
(1-((6-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC3(CCCC3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C25H31N3O3/c1-18(27(2)3)23(28-13-12-26-17-28)21-7-6-20-15-22(9-8-19(20)14-21)31-16-25(24(29)30)10-4-5-11-25/h6-9,12-15,17-18,23H,4-5,10-11,16H2,1-3H3,(H,29,30)/t18-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.30 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162782
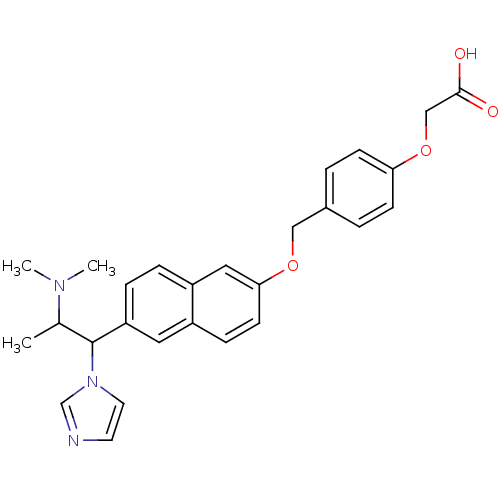
(CHEMBL179550 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(OCC(O)=O)cc3)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C27H29N3O4/c1-19(29(2)3)27(30-13-12-28-18-30)23-7-6-22-15-25(11-8-21(22)14-23)33-16-20-4-9-24(10-5-20)34-17-26(31)32/h4-15,18-19,27H,16-17H2,1-3H3,(H,31,32) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183241

(3-(6-((1R,2R)-2-(ethyl(methyl)amino)-1-(1H-imidazo...)Show SMILES CCN(C)[C@H](C)[C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C24H31N3O3/c1-6-26(5)17(2)22(27-12-11-25-16-27)20-8-7-19-14-21(10-9-18(19)13-20)30-15-24(3,4)23(28)29/h7-14,16-17,22H,6,15H2,1-5H3,(H,28,29)/t17-,22+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183230
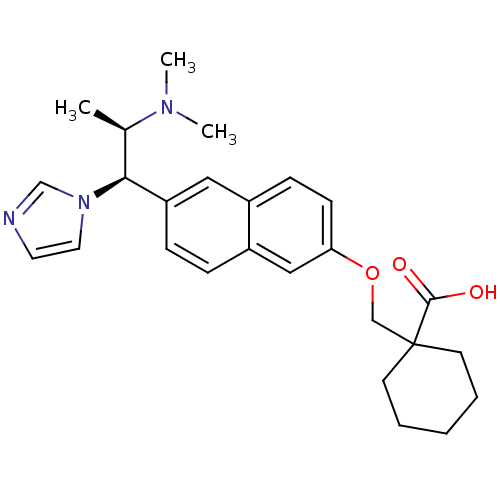
(1-((6-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC3(CCCCC3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C26H33N3O3/c1-19(28(2)3)24(29-14-13-27-18-29)22-8-7-21-16-23(10-9-20(21)15-22)32-17-26(25(30)31)11-5-4-6-12-26/h7-10,13-16,18-19,24H,4-6,11-12,17H2,1-3H3,(H,30,31)/t19-,24+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162772

(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCc3cccc(c3)C(=O)N(C)C)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C28H32N4O2/c1-20(30(2)3)27(32-14-13-29-19-32)24-10-9-23-17-26(12-11-22(23)16-24)34-18-21-7-6-8-25(15-21)28(33)31(4)5/h6-17,19-20,27H,18H2,1-5H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14.4 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162793
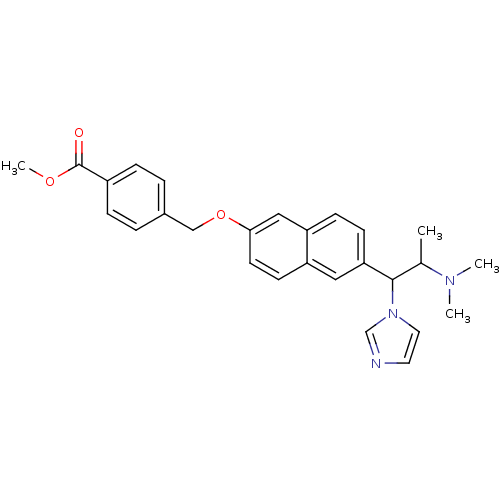
(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-14-13-28-18-30)24-10-9-23-16-25(12-11-22(23)15-24)33-17-20-5-7-21(8-6-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162793

(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-14-13-28-18-30)24-10-9-23-16-25(12-11-22(23)15-24)33-17-20-5-7-21(8-6-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375331
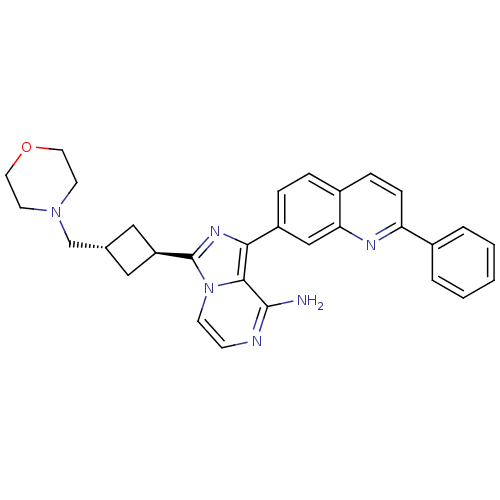
(CHEMBL261253)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@H]1C[C@H](CN2CCOCC2)C1 |wU:26.30,wD:28.33,(5.75,-42.52,;5.75,-44.06,;4.43,-44.83,;4.43,-46.37,;5.76,-47.14,;7.1,-46.37,;8.57,-46.85,;9.48,-45.59,;8.57,-44.34,;9.35,-43.01,;8.58,-41.68,;9.35,-40.35,;10.9,-40.36,;11.66,-39.02,;13.21,-39.04,;13.99,-40.37,;13.21,-41.71,;11.67,-41.7,;10.89,-43.02,;15.53,-40.38,;16.3,-41.71,;17.84,-41.71,;18.61,-40.38,;17.83,-39.04,;16.3,-39.05,;7.09,-44.82,;9.05,-48.31,;10.43,-49.01,;9.73,-50.38,;10.21,-51.85,;9.44,-53.18,;7.91,-53.17,;7.13,-54.5,;7.9,-55.83,;9.44,-55.84,;10.2,-54.51,;8.36,-49.68,)| Show InChI InChI=1S/C30H30N6O/c31-29-28-27(23-7-6-22-8-9-25(33-26(22)18-23)21-4-2-1-3-5-21)34-30(36(28)11-10-32-29)24-16-20(17-24)19-35-12-14-37-15-13-35/h1-11,18,20,24H,12-17,19H2,(H2,31,32)/t20-,24- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375334

(CHEMBL411074)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@@H]1C[C@H](CN2CCOCC2)C1 |wU:26.30,28.33,(-9.18,-42.53,;-9.17,-44.07,;-10.49,-44.84,;-10.5,-46.39,;-9.16,-47.16,;-7.83,-46.39,;-6.35,-46.87,;-5.44,-45.61,;-6.35,-44.35,;-5.57,-43.03,;-6.34,-41.7,;-5.57,-40.37,;-4.02,-40.37,;-3.26,-39.04,;-1.71,-39.05,;-.93,-40.39,;-1.71,-41.72,;-3.25,-41.72,;-4.03,-43.04,;.61,-40.39,;1.38,-41.73,;2.92,-41.73,;3.69,-40.4,;2.91,-39.06,;1.37,-39.06,;-7.83,-44.83,;-5.87,-48.33,;-6.57,-49.7,;-5.19,-50.4,;-4.72,-51.86,;-5.5,-53.2,;-7.02,-53.19,;-7.8,-54.52,;-7.03,-55.85,;-5.49,-55.86,;-4.73,-54.53,;-4.49,-49.03,)| Show InChI InChI=1S/C30H30N6O/c31-29-28-27(23-7-6-22-8-9-25(33-26(22)18-23)21-4-2-1-3-5-21)34-30(36(28)11-10-32-29)24-16-20(17-24)19-35-12-14-37-15-13-35/h1-11,18,20,24H,12-17,19H2,(H2,31,32)/t20-,24+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375339
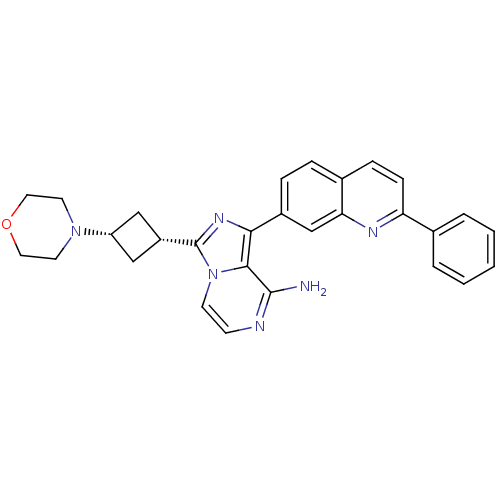
(CHEMBL258648)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@@H]1C[C@@H](C1)N1CCOCC1 |wU:26.30,28.35,(6.69,-13.61,;6.7,-15.15,;5.37,-15.92,;5.37,-17.47,;6.7,-18.24,;8.04,-17.47,;9.51,-17.95,;10.43,-16.69,;9.51,-15.44,;10.29,-14.11,;9.53,-12.78,;10.29,-11.45,;11.84,-11.45,;12.61,-10.12,;14.15,-10.13,;14.93,-11.47,;14.15,-12.8,;12.61,-12.8,;11.83,-14.12,;16.47,-11.47,;17.24,-12.81,;18.78,-12.81,;19.55,-11.48,;18.77,-10.14,;17.24,-10.14,;8.04,-15.92,;9.99,-19.41,;9.3,-20.78,;10.67,-21.48,;11.37,-20.11,;11.14,-22.94,;10.25,-24.19,;10.88,-25.58,;12.41,-25.73,;13.3,-24.48,;12.67,-23.08,)| Show InChI InChI=1S/C29H28N6O/c30-28-27-26(21-7-6-20-8-9-24(32-25(20)18-21)19-4-2-1-3-5-19)33-29(35(27)11-10-31-28)22-16-23(17-22)34-12-14-36-15-13-34/h1-11,18,22-23H,12-17H2,(H2,30,31)/t22-,23+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162778
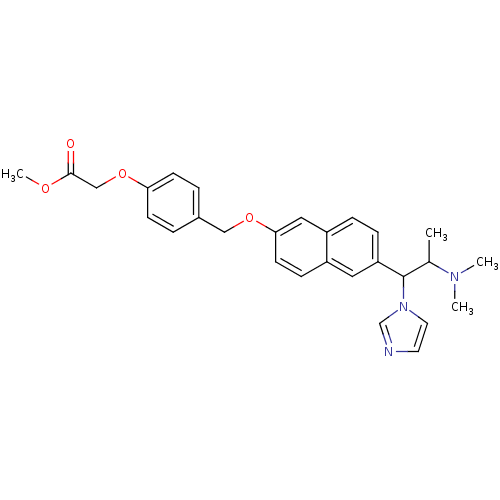
(CHEMBL362412 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES COC(=O)COc1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C28H31N3O4/c1-20(30(2)3)28(31-14-13-29-19-31)24-8-7-23-16-26(12-9-22(23)15-24)34-17-21-5-10-25(11-6-21)35-18-27(32)33-4/h5-16,19-20,28H,17-18H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375337
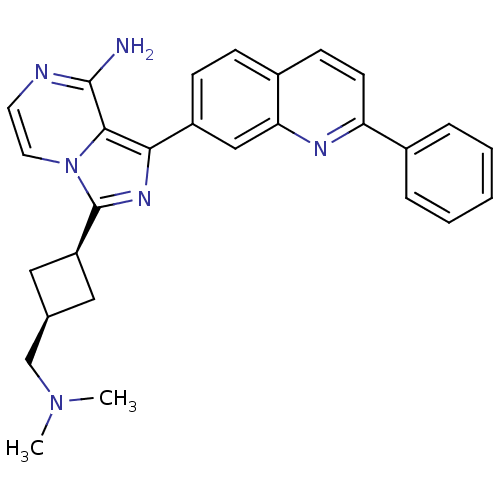
(CHEMBL413828)Show SMILES CN(C)C[C@H]1C[C@H](C1)c1nc(-c2ccc3ccc(nc3c2)-c2ccccc2)c2c(N)nccn12 |wU:6.8,4.3,(25.03,-21.69,;24.56,-20.23,;23.05,-19.91,;25.59,-19.08,;25.11,-17.62,;23.74,-16.92,;24.43,-15.55,;25.81,-16.24,;23.95,-14.09,;24.86,-12.83,;23.95,-11.57,;24.73,-10.25,;23.97,-8.92,;24.73,-7.59,;26.28,-7.59,;27.05,-6.26,;28.59,-6.27,;29.37,-7.61,;28.59,-8.94,;27.05,-8.94,;26.27,-10.26,;30.91,-7.61,;31.68,-8.95,;33.22,-8.95,;33.99,-7.62,;33.21,-6.28,;31.68,-6.29,;22.48,-12.05,;21.14,-11.29,;21.13,-9.75,;19.81,-12.06,;19.81,-13.61,;21.14,-14.38,;22.48,-13.61,)| Show InChI InChI=1S/C28H28N6/c1-33(2)17-18-14-22(15-18)28-32-25(26-27(29)30-12-13-34(26)28)21-9-8-20-10-11-23(31-24(20)16-21)19-6-4-3-5-7-19/h3-13,16,18,22H,14-15,17H2,1-2H3,(H2,29,30)/t18-,22+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162778

(CHEMBL362412 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES COC(=O)COc1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C28H31N3O4/c1-20(30(2)3)28(31-14-13-29-19-31)24-8-7-23-16-26(12-9-22(23)15-24)34-17-21-5-10-25(11-6-21)35-18-27(32)33-4/h5-16,19-20,28H,17-18H2,1-4H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162773
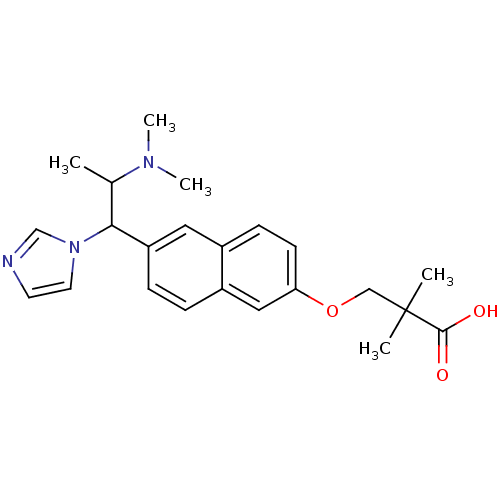
(3-(6-(2-(dimethylamino)-1-(1H-imidazol-1-yl)propyl...)Show SMILES CC(C(c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H29N3O3/c1-16(25(4)5)21(26-11-10-24-15-26)19-7-6-18-13-20(9-8-17(18)12-19)29-14-23(2,3)22(27)28/h6-13,15-16,21H,14H2,1-5H3,(H,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50336316

(CHEMBL410659 | cis-3-(3-(azetidin-1-ylmethyl)cyclo...)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@@H]1C[C@H](CN2CCC2)C1 |wU:26.30,28.33,(7.04,-25.52,;7.05,-27.06,;5.72,-27.83,;5.72,-29.37,;7.05,-30.15,;8.39,-29.37,;9.86,-29.85,;10.77,-28.6,;9.86,-27.34,;10.64,-26.02,;9.88,-24.69,;10.64,-23.36,;12.19,-23.36,;12.96,-22.02,;14.5,-22.04,;15.28,-23.37,;14.5,-24.71,;12.96,-24.7,;12.18,-26.03,;16.82,-23.38,;17.59,-24.72,;19.13,-24.72,;19.9,-23.38,;19.12,-22.05,;17.59,-22.05,;8.39,-27.82,;10.34,-31.32,;9.65,-32.69,;11.02,-33.38,;11.5,-34.85,;10.47,-35.99,;8.93,-36.07,;9.01,-37.61,;10.55,-37.53,;11.72,-32.01,)| Show InChI InChI=1S/C29H28N6/c30-28-27-26(22-8-7-21-9-10-24(32-25(21)17-22)20-5-2-1-3-6-20)33-29(35(27)14-11-31-28)23-15-19(16-23)18-34-12-4-13-34/h1-3,5-11,14,17,19,23H,4,12-13,15-16,18H2,(H2,30,31)/t19-,23+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162773

(3-(6-(2-(dimethylamino)-1-(1H-imidazol-1-yl)propyl...)Show SMILES CC(C(c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H29N3O3/c1-16(25(4)5)21(26-11-10-24-15-26)19-7-6-18-13-20(9-8-17(18)12-19)29-14-23(2,3)22(27)28/h6-13,15-16,21H,14H2,1-5H3,(H,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183226
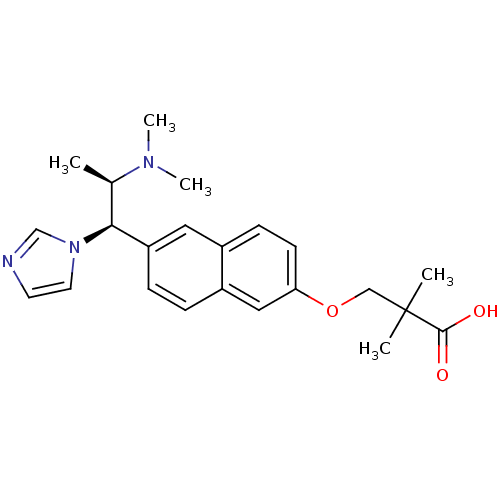
(3-[6-(2-dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H29N3O3/c1-16(25(4)5)21(26-11-10-24-15-26)19-7-6-18-13-20(9-8-17(18)12-19)29-14-23(2,3)22(27)28/h6-13,15-16,21H,14H2,1-5H3,(H,27,28)/t16-,21+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375336
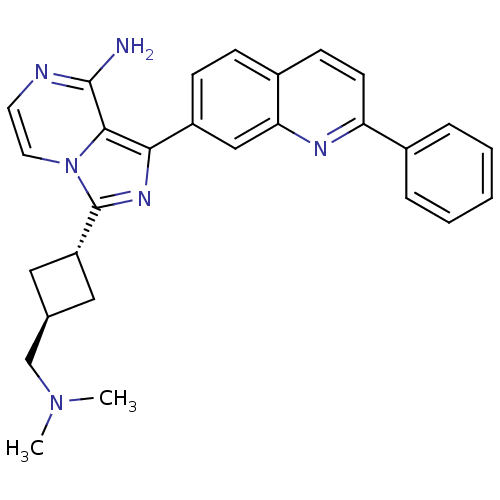
(CHEMBL411062)Show SMILES CN(C)C[C@H]1C[C@@H](C1)c1nc(-c2ccc3ccc(nc3c2)-c2ccccc2)c2c(N)nccn12 |wU:6.8,wD:4.3,(-5.34,-34.06,;-5.81,-32.6,;-7.32,-32.28,;-4.78,-31.45,;-5.26,-29.99,;-4.56,-28.62,;-5.94,-27.92,;-6.63,-29.29,;-6.42,-26.46,;-5.51,-25.2,;-6.42,-23.95,;-5.64,-22.62,;-6.41,-21.29,;-5.64,-19.96,;-4.09,-19.96,;-3.33,-18.63,;-1.78,-18.64,;-1,-19.98,;-1.78,-21.31,;-3.32,-21.31,;-4.1,-22.63,;.54,-19.98,;1.31,-21.32,;2.85,-21.32,;3.62,-19.99,;2.84,-18.65,;1.3,-18.66,;-7.89,-24.43,;-9.24,-23.66,;-9.24,-22.12,;-10.56,-24.43,;-10.56,-25.98,;-9.23,-26.75,;-7.89,-25.98,)| Show InChI InChI=1S/C28H28N6/c1-33(2)17-18-14-22(15-18)28-32-25(26-27(29)30-12-13-34(26)28)21-9-8-20-10-11-23(31-24(20)16-21)19-6-4-3-5-7-19/h3-13,16,18,22H,14-15,17H2,1-2H3,(H2,29,30)/t18-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162777
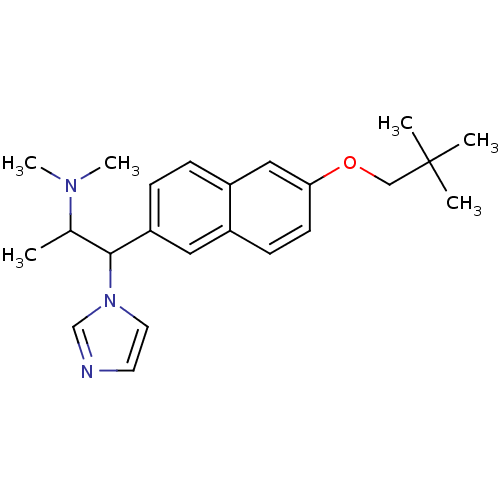
(CHEMBL180805 | {2-[6-(2,2-Dimethyl-propoxy)-naphth...)Show SMILES CC(C(c1ccc2cc(OCC(C)(C)C)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H31N3O/c1-17(25(5)6)22(26-12-11-24-16-26)20-8-7-19-14-21(10-9-18(19)13-20)27-15-23(2,3)4/h7-14,16-17,22H,15H2,1-6H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21.4 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162777

(CHEMBL180805 | {2-[6-(2,2-Dimethyl-propoxy)-naphth...)Show SMILES CC(C(c1ccc2cc(OCC(C)(C)C)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H31N3O/c1-17(25(5)6)22(26-12-11-24-16-26)20-8-7-19-14-21(10-9-18(19)13-20)27-15-23(2,3)4/h7-14,16-17,22H,15H2,1-6H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21.4 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162780

(CHEMBL180348 | {4-[6-(2-Dimethylamino-1-imidazol-1...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(CC(O)=O)cc3)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)27(30-13-12-28-18-30)24-9-8-23-16-25(11-10-22(23)15-24)33-17-21-6-4-20(5-7-21)14-26(31)32/h4-13,15-16,18-19,27H,14,17H2,1-3H3,(H,31,32) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183232
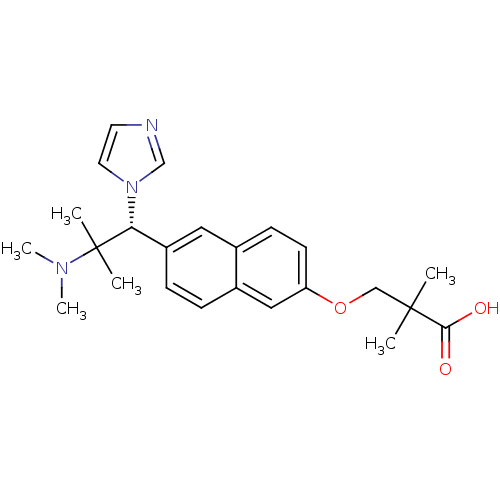
((R)-3-(6-(2-(dimethylamino)-1-(1H-imidazol-1-yl)-2...)Show SMILES CN(C)C(C)(C)[C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1 Show InChI InChI=1S/C24H31N3O3/c1-23(2,22(28)29)15-30-20-10-9-17-13-19(8-7-18(17)14-20)21(24(3,4)26(5)6)27-12-11-25-16-27/h7-14,16,21H,15H2,1-6H3,(H,28,29)/t21-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162784
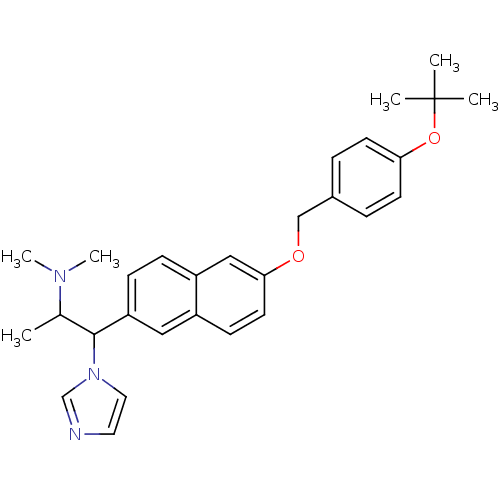
(CHEMBL181385 | {2-[6-(4-tert-Butoxy-benzyloxy)-nap...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(OC(C)(C)C)cc3)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C29H35N3O2/c1-21(31(5)6)28(32-16-15-30-20-32)25-10-9-24-18-27(14-11-23(24)17-25)33-19-22-7-12-26(13-8-22)34-29(2,3)4/h7-18,20-21,28H,19H2,1-6H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183242
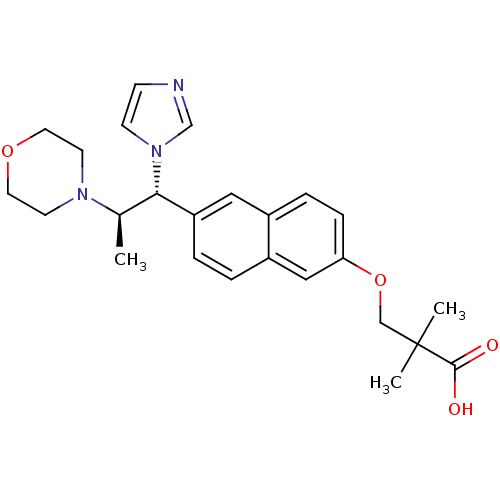
(3-(6-((1R,2R)-1-(1H-imidazol-1-yl)-2-morpholinopro...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC(C)(C)C(O)=O)ccc2c1)n1ccnc1)N1CCOCC1 Show InChI InChI=1S/C25H31N3O4/c1-18(27-10-12-31-13-11-27)23(28-9-8-26-17-28)21-5-4-20-15-22(7-6-19(20)14-21)32-16-25(2,3)24(29)30/h4-9,14-15,17-18,23H,10-13,16H2,1-3H3,(H,29,30)/t18-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162784

(CHEMBL181385 | {2-[6-(4-tert-Butoxy-benzyloxy)-nap...)Show SMILES CC(C(c1ccc2cc(OCc3ccc(OC(C)(C)C)cc3)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C29H35N3O2/c1-21(31(5)6)28(32-16-15-30-20-32)25-10-9-24-18-27(14-11-23(24)17-25)33-19-22-7-12-26(13-8-22)34-29(2,3)4/h7-18,20-21,28H,19H2,1-6H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162775
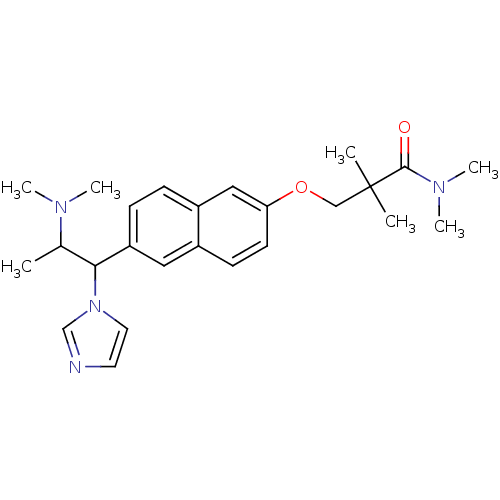
(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CC(C(c1ccc2cc(OCC(C)(C)C(=O)N(C)C)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C25H34N4O2/c1-18(27(4)5)23(29-13-12-26-17-29)21-9-8-20-15-22(11-10-19(20)14-21)31-16-25(2,3)24(30)28(6)7/h8-15,17-18,23H,16H2,1-7H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50336316

(CHEMBL410659 | cis-3-(3-(azetidin-1-ylmethyl)cyclo...)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@@H]1C[C@H](CN2CCC2)C1 |wU:26.30,28.33,(7.04,-25.52,;7.05,-27.06,;5.72,-27.83,;5.72,-29.37,;7.05,-30.15,;8.39,-29.37,;9.86,-29.85,;10.77,-28.6,;9.86,-27.34,;10.64,-26.02,;9.88,-24.69,;10.64,-23.36,;12.19,-23.36,;12.96,-22.02,;14.5,-22.04,;15.28,-23.37,;14.5,-24.71,;12.96,-24.7,;12.18,-26.03,;16.82,-23.38,;17.59,-24.72,;19.13,-24.72,;19.9,-23.38,;19.12,-22.05,;17.59,-22.05,;8.39,-27.82,;10.34,-31.32,;9.65,-32.69,;11.02,-33.38,;11.5,-34.85,;10.47,-35.99,;8.93,-36.07,;9.01,-37.61,;10.55,-37.53,;11.72,-32.01,)| Show InChI InChI=1S/C29H28N6/c30-28-27-26(22-8-7-21-9-10-24(32-25(21)17-22)20-5-2-1-3-6-20)33-29(35(27)14-11-31-28)23-15-19(16-23)18-34-12-4-13-34/h1-3,5-11,14,17,19,23H,4,12-13,15-16,18H2,(H2,30,31)/t19-,23+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183233

(1-((6-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC3(CC3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C23H27N3O3/c1-16(25(2)3)21(26-11-10-24-15-26)19-5-4-18-13-20(7-6-17(18)12-19)29-14-23(8-9-23)22(27)28/h4-7,10-13,15-16,21H,8-9,14H2,1-3H3,(H,27,28)/t16-,21+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183239
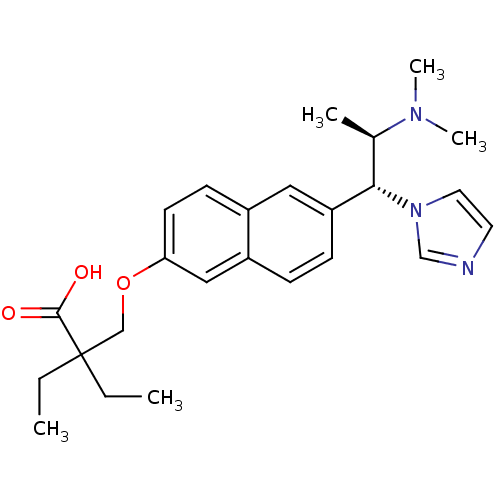
(2-((6-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-...)Show SMILES CCC(CC)(COc1ccc2cc(ccc2c1)[C@H]([C@@H](C)N(C)C)n1ccnc1)C(O)=O Show InChI InChI=1S/C25H33N3O3/c1-6-25(7-2,24(29)30)16-31-22-11-10-19-14-21(9-8-20(19)15-22)23(18(3)27(4)5)28-13-12-26-17-28/h8-15,17-18,23H,6-7,16H2,1-5H3,(H,29,30)/t18-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50162785
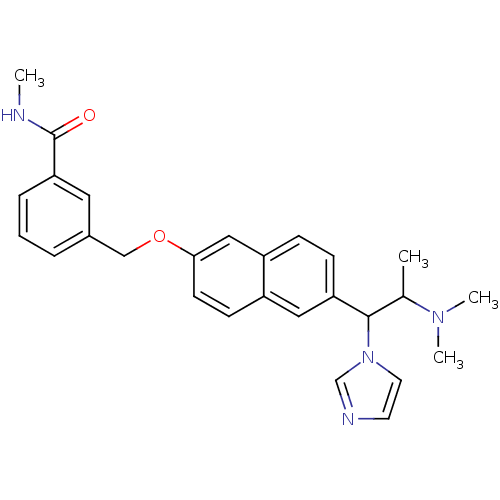
(3-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES CNC(=O)c1cccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)c1 Show InChI InChI=1S/C27H30N4O2/c1-19(30(3)4)26(31-13-12-29-18-31)23-9-8-22-16-25(11-10-21(22)15-23)33-17-20-6-5-7-24(14-20)27(32)28-2/h5-16,18-19,26H,17H2,1-4H3,(H,28,32) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Potency towards cytochrome P 450 26 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50162793

(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-14-13-28-18-30)24-10-9-23-16-25(12-11-22(23)15-24)33-17-20-5-7-21(8-6-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Selectivity towards cytochrome P450 3A4 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Cytochrome P450 26A1
(Homo sapiens (Human)) | BDBM50183227

(1-((6-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-...)Show SMILES C[C@H]([C@@H](c1ccc2cc(OCC3(CCC3)C(O)=O)ccc2c1)n1ccnc1)N(C)C Show InChI InChI=1S/C24H29N3O3/c1-17(26(2)3)22(27-12-11-25-16-27)20-6-5-19-14-21(8-7-18(19)13-20)30-15-24(23(28)29)9-4-10-24/h5-8,11-14,16-17,22H,4,9-10,15H2,1-3H3,(H,28,29)/t17-,22+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CYP26 expressed in human T47D cell line |
Bioorg Med Chem Lett 16: 2729-33 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.020
BindingDB Entry DOI: 10.7270/Q24Q7TK9 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50162793

(4-[6-(2-Dimethylamino-1-imidazol-1-yl-propyl)-naph...)Show SMILES COC(=O)c1ccc(COc2ccc3cc(ccc3c2)C(C(C)N(C)C)n2ccnc2)cc1 Show InChI InChI=1S/C27H29N3O3/c1-19(29(2)3)26(30-14-13-28-18-30)24-10-9-23-16-25(12-11-22(23)15-24)33-17-20-5-7-21(8-6-20)27(31)32-4/h5-16,18-19,26H,17H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Selectivity towards cytochrome P450 3A4 enzyme activity |
Bioorg Med Chem Lett 15: 1669-73 (2005)
Article DOI: 10.1016/j.bmcl.2005.01.044
BindingDB Entry DOI: 10.7270/Q2WW7H5R |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50375335

(CHEMBL260298)Show SMILES Nc1nccn2c(nc(-c3ccc4ccc(nc4c3)-c3ccccc3)c12)[C@H]1C[C@H](CN2CCC2)C1 |wU:26.30,wD:28.33,(20.91,-25.07,;20.92,-26.61,;19.6,-27.38,;19.59,-28.92,;20.93,-29.69,;22.26,-28.92,;23.74,-29.4,;24.65,-28.14,;23.74,-26.89,;24.51,-25.56,;23.75,-24.23,;24.52,-22.9,;26.07,-22.91,;26.83,-21.57,;28.38,-21.59,;29.16,-22.92,;28.38,-24.26,;26.83,-24.25,;26.06,-25.57,;30.7,-22.93,;31.47,-24.26,;33,-24.27,;33.78,-22.93,;33,-21.59,;31.46,-21.6,;22.26,-27.37,;24.22,-30.86,;25.59,-31.56,;24.9,-32.93,;25.37,-34.4,;24.34,-35.54,;22.81,-35.62,;22.89,-37.16,;24.43,-37.07,;23.52,-32.23,)| Show InChI InChI=1S/C29H28N6/c30-28-27-26(22-8-7-21-9-10-24(32-25(21)17-22)20-5-2-1-3-6-20)33-29(35(27)14-11-31-28)23-15-19(16-23)18-34-12-4-13-34/h1-3,5-11,14,17,19,23H,4,12-13,15-16,18H2,(H2,30,31)/t19-,23- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length IGF1R expressed in mouse 3T3 cells |
Bioorg Med Chem 16: 1359-75 (2008)
Article DOI: 10.1016/j.bmc.2007.10.061
BindingDB Entry DOI: 10.7270/Q2WH2QVH |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data