 Found 34 hits with Last Name = 'griffon' and Initial = 'y'
Found 34 hits with Last Name = 'griffon' and Initial = 'y' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50336729

(4-(3,3-Dimethyl-butyrylamino)-N-thiazol-2-yl-benza...)Show InChI InChI=1S/C16H19N3O2S/c1-16(2,3)10-13(20)18-12-6-4-11(5-7-12)14(21)19-15-17-8-9-22-15/h4-9H,10H2,1-3H3,(H,18,20)(H,17,19,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50336728
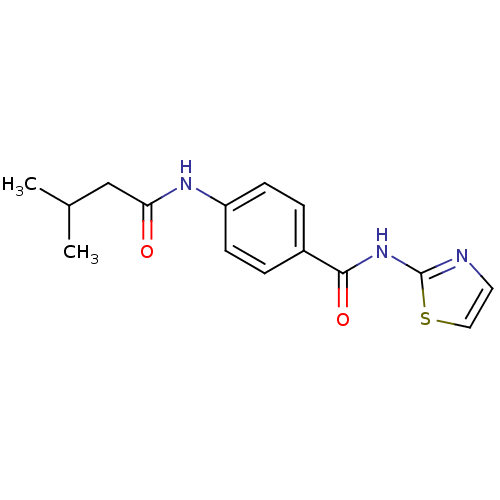
(4-(3-Methyl-butyrylamino)-N-thiazol-2-yl-benzamide...)Show InChI InChI=1S/C15H17N3O2S/c1-10(2)9-13(19)17-12-5-3-11(4-6-12)14(20)18-15-16-7-8-21-15/h3-8,10H,9H2,1-2H3,(H,17,19)(H,16,18,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 59 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50336714
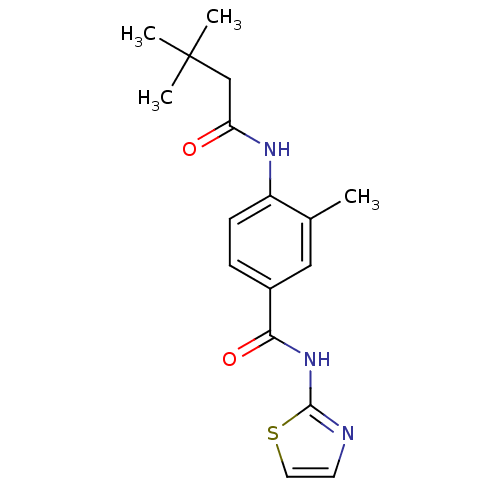
(4-(3,3-Dimethyl-butyrylamino)-3-methyl-N-thiazol-2...)Show InChI InChI=1S/C17H21N3O2S/c1-11-9-12(15(22)20-16-18-7-8-23-16)5-6-13(11)19-14(21)10-17(2,3)4/h5-9H,10H2,1-4H3,(H,19,21)(H,18,20,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065010
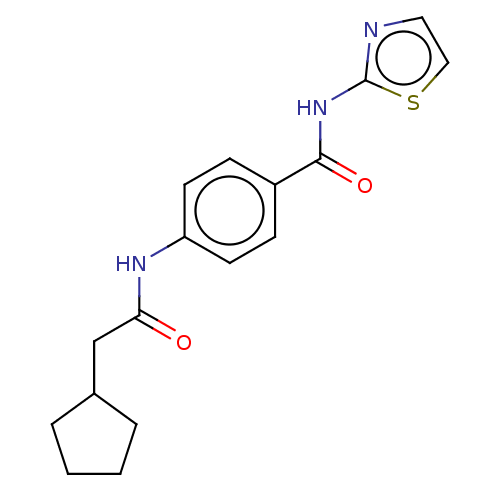
(CHEMBL3401300)Show InChI InChI=1S/C17H19N3O2S/c21-15(11-12-3-1-2-4-12)19-14-7-5-13(6-8-14)16(22)20-17-18-9-10-23-17/h5-10,12H,1-4,11H2,(H,19,21)(H,18,20,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50176050
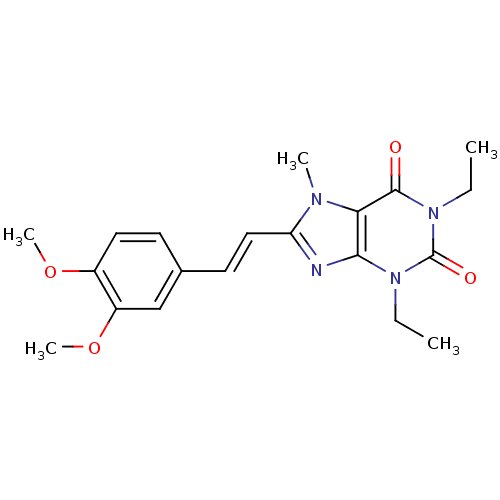
(8-(3,4-dimethoxystyryl)-1,3-diethyl-7-methyl-1H-pu...)Show SMILES CCn1c2nc(\C=C\c3ccc(OC)c(OC)c3)n(C)c2c(=O)n(CC)c1=O Show InChI InChI=1S/C20H24N4O4/c1-6-23-18-17(19(25)24(7-2)20(23)26)22(3)16(21-18)11-9-13-8-10-14(27-4)15(12-13)28-5/h8-12H,6-7H2,1-5H3/b11-9+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 72 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065004
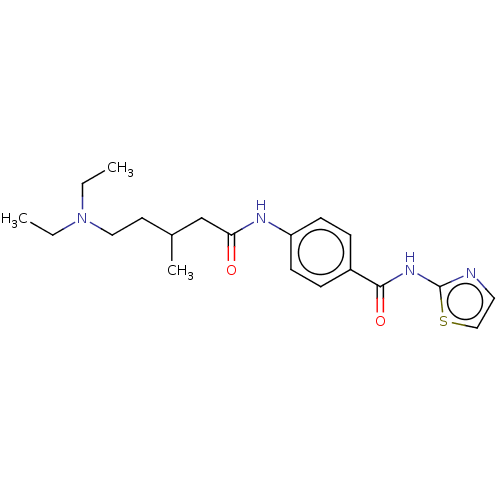
(CHEMBL3402640)Show SMILES CCN(CC)CCC(C)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H28N4O2S/c1-4-24(5-2)12-10-15(3)14-18(25)22-17-8-6-16(7-9-17)19(26)23-20-21-11-13-27-20/h6-9,11,13,15H,4-5,10,12,14H2,1-3H3,(H,22,25)(H,21,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065005

(CHEMBL3401305)Show SMILES CCN(CC)CCCCCCCC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C22H32N4O2S/c1-3-26(4-2)16-9-7-5-6-8-10-20(27)24-19-13-11-18(12-14-19)21(28)25-22-23-15-17-29-22/h11-15,17H,3-10,16H2,1-2H3,(H,24,27)(H,23,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50336725
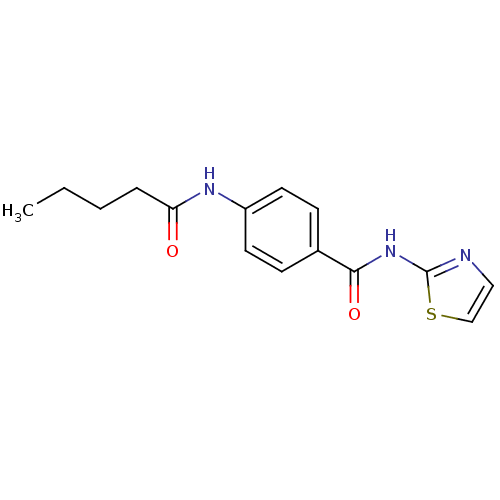
(4-pentanamido-N-(thiazol-2-yl)benzamide | CHEMBL16...)Show InChI InChI=1S/C15H17N3O2S/c1-2-3-4-13(19)17-12-7-5-11(6-8-12)14(20)18-15-16-9-10-21-15/h5-10H,2-4H2,1H3,(H,17,19)(H,16,18,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065006

(CHEMBL3401304)Show InChI InChI=1S/C20H28N4O2S/c1-3-24(4-2)14-7-5-6-8-18(25)22-17-11-9-16(10-12-17)19(26)23-20-21-13-15-27-20/h9-13,15H,3-8,14H2,1-2H3,(H,22,25)(H,21,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50064903
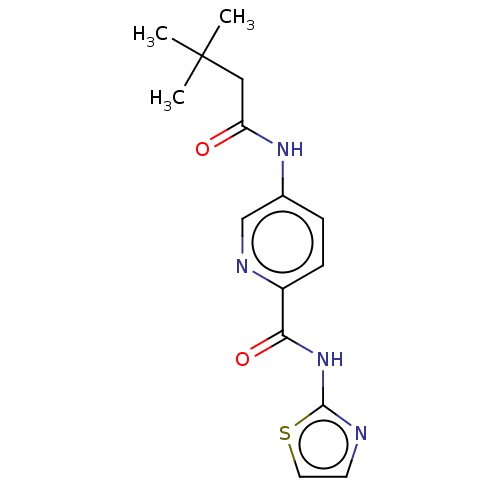
(CHEMBL3401296)Show InChI InChI=1S/C15H18N4O2S/c1-15(2,3)8-12(20)18-10-4-5-11(17-9-10)13(21)19-14-16-6-7-22-14/h4-7,9H,8H2,1-3H3,(H,18,20)(H,16,19,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50064904
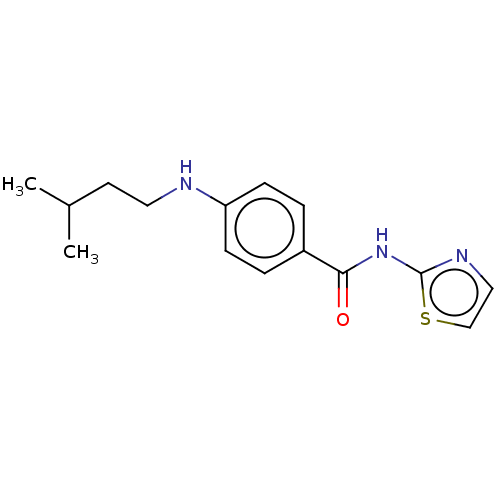
(CHEMBL3401295)Show InChI InChI=1S/C15H19N3OS/c1-11(2)7-8-16-13-5-3-12(4-6-13)14(19)18-15-17-9-10-20-15/h3-6,9-11,16H,7-8H2,1-2H3,(H,17,18,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065007

(CHEMBL3401303)Show InChI InChI=1S/C19H26N4O2S/c1-3-23(4-2)13-6-5-7-17(24)21-16-10-8-15(9-11-16)18(25)22-19-20-12-14-26-19/h8-12,14H,3-7,13H2,1-2H3,(H,21,24)(H,20,22,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50064902
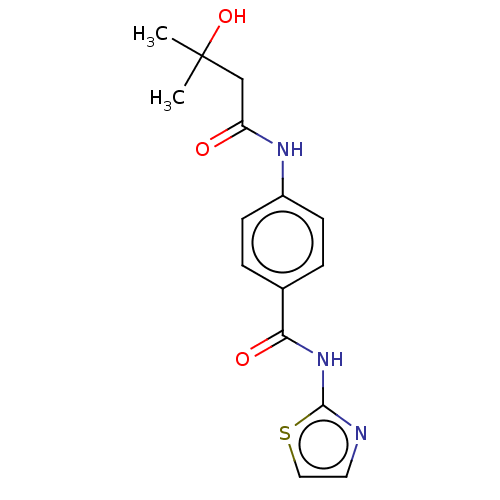
(CHEMBL3401298)Show InChI InChI=1S/C15H17N3O3S/c1-15(2,21)9-12(19)17-11-5-3-10(4-6-11)13(20)18-14-16-7-8-22-14/h3-8,21H,9H2,1-2H3,(H,17,19)(H,16,18,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065008
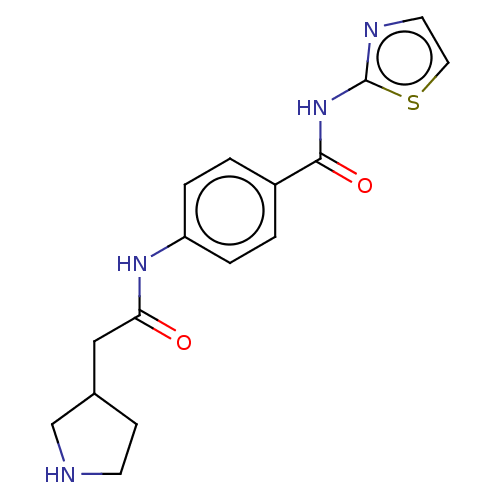
(CHEMBL3401301)Show InChI InChI=1S/C16H18N4O2S/c21-14(9-11-5-6-17-10-11)19-13-3-1-12(2-4-13)15(22)20-16-18-7-8-23-16/h1-4,7-8,11,17H,5-6,9-10H2,(H,19,21)(H,18,20,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50176050

(8-(3,4-dimethoxystyryl)-1,3-diethyl-7-methyl-1H-pu...)Show SMILES CCn1c2nc(\C=C\c3ccc(OC)c(OC)c3)n(C)c2c(=O)n(CC)c1=O Show InChI InChI=1S/C20H24N4O4/c1-6-23-18-17(19(25)24(7-2)20(23)26)22(3)16(21-18)11-9-13-8-10-14(27-4)15(12-13)28-5/h8-12H,6-7H2,1-5H3/b11-9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A1 receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50064913
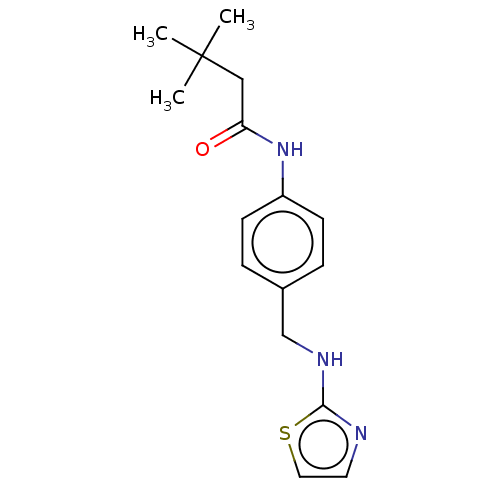
(CHEMBL3401294)Show InChI InChI=1S/C16H21N3OS/c1-16(2,3)10-14(20)19-13-6-4-12(5-7-13)11-18-15-17-8-9-21-15/h4-9H,10-11H2,1-3H3,(H,17,18)(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50065009
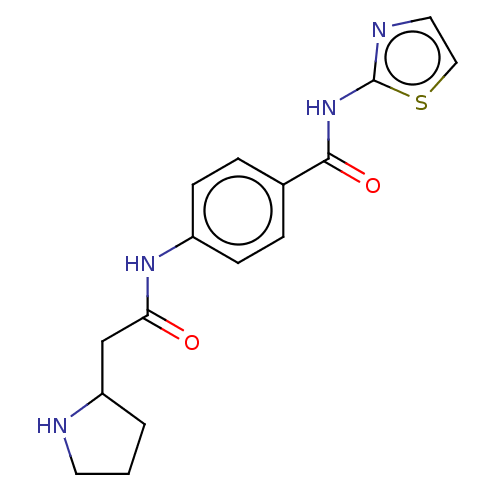
(CHEMBL3400161)Show InChI InChI=1S/C16H18N4O2S/c21-14(10-13-2-1-7-17-13)19-12-5-3-11(4-6-12)15(22)20-16-18-8-9-23-16/h3-6,8-9,13,17H,1-2,7,10H2,(H,19,21)(H,18,20,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Antagonist activity at human adenosine A2A receptor |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50142916
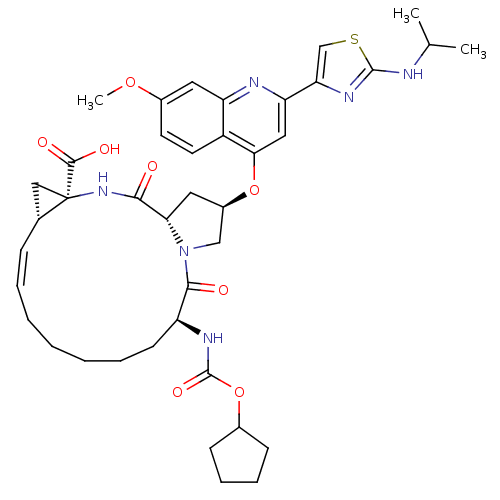
((1S,4R,6S,14S,18R)-14-Cyclopentyloxycarbonylamino-...)Show SMILES COc1ccc2c(O[C@@H]3C[C@@H]4N(C3)C(=O)[C@H](CCCCC\C=C/[C@@H]3C[C@]3(NC4=O)C(O)=O)NC(=O)OC3CCCC3)cc(nc2c1)-c1csc(NC(C)C)n1 |r,c:22| Show InChI InChI=1S/C40H50N6O8S/c1-23(2)41-38-43-32(22-55-38)31-19-34(28-16-15-26(52-3)17-30(28)42-31)53-27-18-33-35(47)45-40(37(49)50)20-24(40)11-7-5-4-6-8-14-29(36(48)46(33)21-27)44-39(51)54-25-12-9-10-13-25/h7,11,15-17,19,22-25,27,29,33H,4-6,8-10,12-14,18,20-21H2,1-3H3,(H,41,43)(H,44,51)(H,45,47)(H,49,50)/b11-7-/t24-,27-,29+,33+,40-/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023510
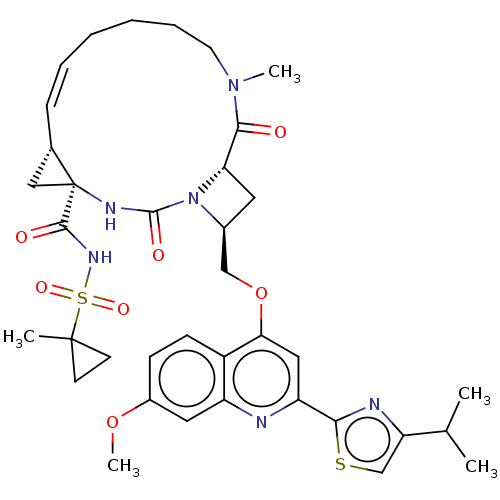
(CHEMBL3326826)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4cc(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:47,@:7| Show InChI InChI=1S/C37H46N6O7S2/c1-22(2)29-21-51-32(39-29)28-18-31(26-12-11-25(49-5)17-27(26)38-28)50-20-24-16-30-33(44)42(4)15-9-7-6-8-10-23-19-37(23,40-35(46)43(24)30)34(45)41-52(47,48)36(3)13-14-36/h8,10-12,17-18,21-24,30H,6-7,9,13-16,19-20H2,1-5H3,(H,40,46)(H,41,45)/b10-8-/t23-,24-,30-,37-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023579

(CHEMBL3326830)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4c(Cl)c(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:48,@:7| Show InChI InChI=1S/C37H45ClN6O7S2/c1-21(2)26-20-52-32(40-26)25-17-29(24-11-12-28(50-5)30(38)31(24)39-25)51-19-23-16-27-33(45)43(4)15-9-7-6-8-10-22-18-37(22,41-35(47)44(23)27)34(46)42-53(48,49)36(3)13-14-36/h8,10-12,17,20-23,27H,6-7,9,13-16,18-19H2,1-5H3,(H,41,47)(H,42,46)/b10-8-/t22-,23-,27-,37-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023508
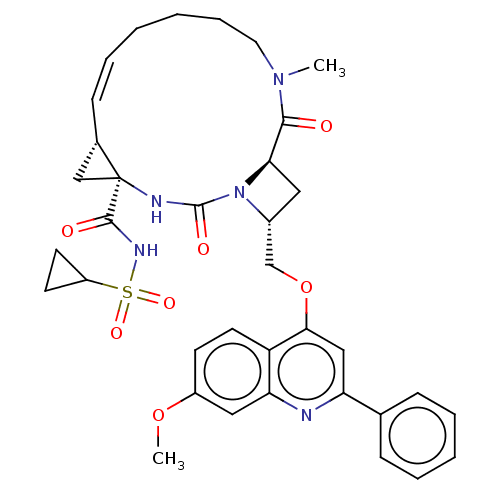
(CHEMBL3326539)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@@H](COc3cc(nc4cc(OC)ccc34)-c3ccccc3)C[C@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:45,@:7| Show InChI InChI=1S/C36H41N5O7S/c1-40-17-9-4-3-8-12-24-21-36(24,34(43)39-49(45,46)27-14-15-27)38-35(44)41-25(18-31(41)33(40)42)22-48-32-20-29(23-10-6-5-7-11-23)37-30-19-26(47-2)13-16-28(30)32/h5-8,10-13,16,19-20,24-25,27,31H,3-4,9,14-15,17-18,21-22H2,1-2H3,(H,38,44)(H,39,43)/b12-8-/t24-,25+,31+,36-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023578
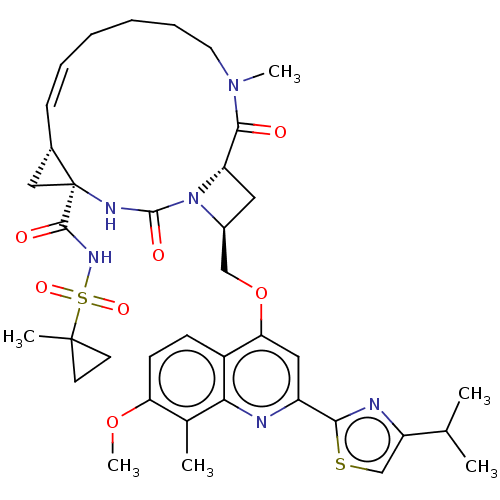
(CHEMBL3326829)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4c(C)c(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:48,@:7| Show InChI InChI=1S/C38H48N6O7S2/c1-22(2)28-21-52-33(40-28)27-18-31(26-12-13-30(50-6)23(3)32(26)39-27)51-20-25-17-29-34(45)43(5)16-10-8-7-9-11-24-19-38(24,41-36(47)44(25)29)35(46)42-53(48,49)37(4)14-15-37/h9,11-13,18,21-22,24-25,29H,7-8,10,14-17,19-20H2,1-6H3,(H,41,47)(H,42,46)/b11-9-/t24-,25-,29-,38-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023509
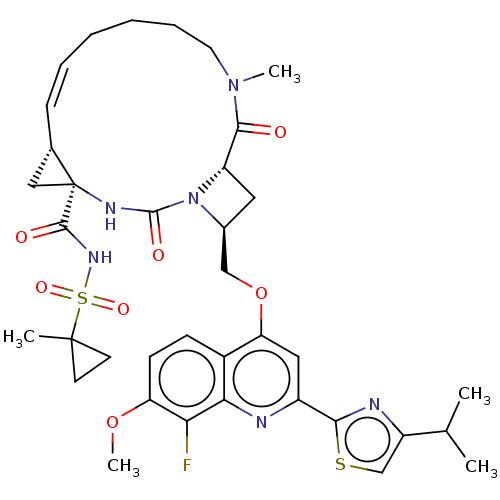
(CHEMBL3326540)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4c(F)c(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:48,@:7| Show InChI InChI=1S/C37H45FN6O7S2/c1-21(2)26-20-52-32(40-26)25-17-29(24-11-12-28(50-5)30(38)31(24)39-25)51-19-23-16-27-33(45)43(4)15-9-7-6-8-10-22-18-37(22,41-35(47)44(23)27)34(46)42-53(48,49)36(3)13-14-36/h8,10-12,17,20-23,27H,6-7,9,13-16,18-19H2,1-5H3,(H,41,47)(H,42,46)/b10-8-/t22-,23-,27-,37-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023507
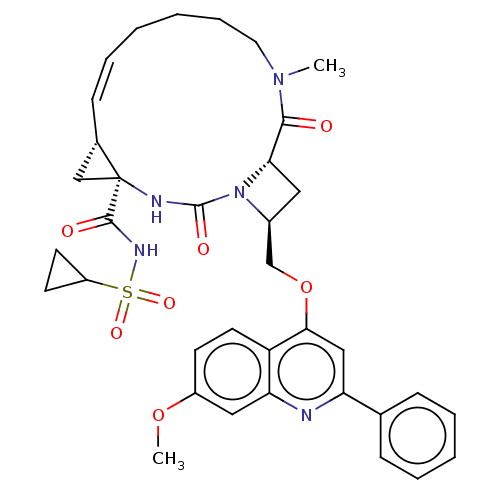
(CHEMBL3326538)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4cc(OC)ccc34)-c3ccccc3)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:45,@:7| Show InChI InChI=1S/C36H41N5O7S/c1-40-17-9-4-3-8-12-24-21-36(24,34(43)39-49(45,46)27-14-15-27)38-35(44)41-25(18-31(41)33(40)42)22-48-32-20-29(23-10-6-5-7-11-23)37-30-19-26(47-2)13-16-28(30)32/h5-8,10-13,16,19-20,24-25,27,31H,3-4,9,14-15,17-18,21-22H2,1-2H3,(H,38,44)(H,39,43)/b12-8-/t24-,25-,31-,36-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023581

(CHEMBL3326537)Show SMILES [H][C@]12C[C@@]1(NC(=O)[C@@]1([H])C[C@H](COc3cc(nc4cc(OC)ccc34)-c3ccccc3)N1C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:45| Show InChI InChI=1S/C36H41N5O7S/c1-40-17-9-4-3-8-12-24-21-36(24,34(43)39-49(45,46)27-14-15-27)38-33(42)31-18-25(41(31)35(40)44)22-48-32-20-29(23-10-6-5-7-11-23)37-30-19-26(47-2)13-16-28(30)32/h5-8,10-13,16,19-20,24-25,27,31H,3-4,9,14-15,17-18,21-22H2,1-2H3,(H,38,42)(H,39,43)/b12-8-/t24-,25+,31+,36-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 278 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023576

(CHEMBL3326827)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4c(C)c(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:48,@:7| Show InChI InChI=1S/C37H46N6O7S2/c1-21(2)28-20-51-33(39-28)27-17-31(26-13-14-30(49-5)22(3)32(26)38-27)50-19-24-16-29-34(44)42(4)15-9-7-6-8-10-23-18-37(23,40-36(46)43(24)29)35(45)41-52(47,48)25-11-12-25/h8,10,13-14,17,20-21,23-25,29H,6-7,9,11-12,15-16,18-19H2,1-5H3,(H,40,46)(H,41,45)/b10-8-/t23-,24-,29-,37-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023577

(CHEMBL3326828)Show SMILES [H][C@]12C[C@@]1(NC(=O)N1[C@H](COc3cc(nc4c(Cl)c(OC)ccc34)-c3nc(cs3)C(C)C)C[C@@]1([H])C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:48,@:7| Show InChI InChI=1S/C36H43ClN6O7S2/c1-20(2)26-19-51-32(39-26)25-16-29(24-12-13-28(49-4)30(37)31(24)38-25)50-18-22-15-27-33(44)42(3)14-8-6-5-7-9-21-17-36(21,40-35(46)43(22)27)34(45)41-52(47,48)23-10-11-23/h7,9,12-13,16,19-23,27H,5-6,8,10-11,14-15,17-18H2,1-4H3,(H,40,46)(H,41,45)/b9-7-/t21-,22-,27-,36-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50023580
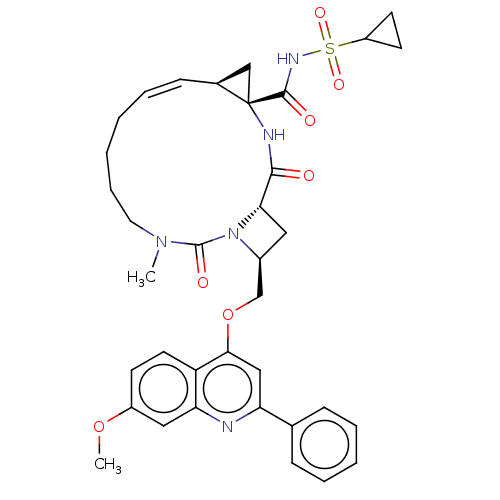
(CHEMBL3326536)Show SMILES [H][C@]12C[C@@]1(NC(=O)[C@]1([H])C[C@@H](COc3cc(nc4cc(OC)ccc34)-c3ccccc3)N1C(=O)N(C)CCCC\C=C/2)C(=O)NS(=O)(=O)C1CC1 |r,c:45| Show InChI InChI=1S/C36H41N5O7S/c1-40-17-9-4-3-8-12-24-21-36(24,34(43)39-49(45,46)27-14-15-27)38-33(42)31-18-25(41(31)35(40)44)22-48-32-20-29(23-10-6-5-7-11-23)37-30-19-26(47-2)13-16-28(30)32/h5-8,10-13,16,19-20,24-25,27,31H,3-4,9,14-15,17-18,21-22H2,1-2H3,(H,38,42)(H,39,43)/b12-8-/t24-,25-,31-,36-/m0/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
IDENIX Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of full length Hepatitis C virus genotype 1b Con1 NS3/4A by FRET assay |
Bioorg Med Chem Lett 24: 4444-9 (2014)
Article DOI: 10.1016/j.bmcl.2014.08.002
BindingDB Entry DOI: 10.7270/Q20C4XBK |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50065003

(CHEMBL3402641)Show SMILES CC(CCN1CCOCC1)CC(=O)Nc1ccc(cc1)C(=O)Nc1nccs1 Show InChI InChI=1S/C20H26N4O3S/c1-15(6-8-24-9-11-27-12-10-24)14-18(25)22-17-4-2-16(3-5-17)19(26)23-20-21-7-13-28-20/h2-5,7,13,15H,6,8-12,14H2,1H3,(H,22,25)(H,21,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
H. Lundbeck A/S
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 25: 1212-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.01.062
BindingDB Entry DOI: 10.7270/Q22Z1779 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data