 Found 2518 hits with Last Name = 'hawkins' and Initial = 'j'
Found 2518 hits with Last Name = 'hawkins' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Chymase
(Homo sapiens (Human)) | BDBM50208224
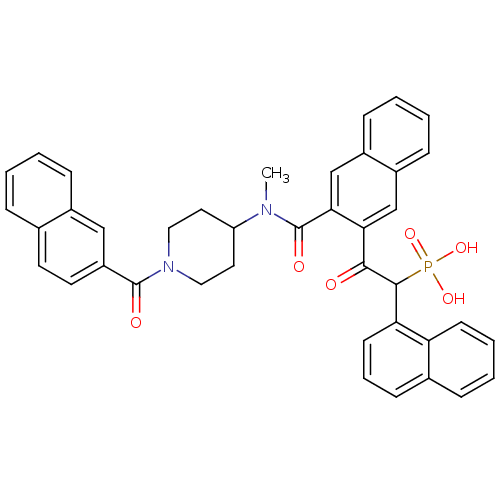
(2-(3-((1-(2-naphthoyl)piperidin-4-yl)(methyl)carba...)Show SMILES CN(C1CCN(CC1)C(=O)c1ccc2ccccc2c1)C(=O)c1cc2ccccc2cc1C(=O)C(c1cccc2ccccc12)P(O)(O)=O Show InChI InChI=1S/C40H35N2O6P/c1-41(32-19-21-42(22-20-32)39(44)31-18-17-26-9-2-3-11-28(26)23-31)40(45)36-25-30-13-5-4-12-29(30)24-35(36)37(43)38(49(46,47)48)34-16-8-14-27-10-6-7-15-33(27)34/h2-18,23-25,32,38H,19-22H2,1H3,(H2,46,47,48) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human skin chymase |
J Med Chem 50: 1727-30 (2007)
Article DOI: 10.1021/jm0700619
BindingDB Entry DOI: 10.7270/Q26W99RF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Chymase
(Homo sapiens (Human)) | BDBM50208222

((E)-2-(3-chloro-5-fluorostyrylamino)-1-(5-chlorobe...)Show SMILES CP(O)(=O)C(C(=O)NC=Cc1cc(F)cc(Cl)c1)c1csc2ccc(Cl)cc12 |w:9.9| Show InChI InChI=1S/C19H15Cl2FNO3PS/c1-27(25,26)18(16-10-28-17-3-2-12(20)9-15(16)17)19(24)23-5-4-11-6-13(21)8-14(22)7-11/h2-10,18H,1H3,(H,23,24)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human skin chymase |
J Med Chem 50: 1727-30 (2007)
Article DOI: 10.1021/jm0700619
BindingDB Entry DOI: 10.7270/Q26W99RF |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-1
(Rattus norvegicus (Rat)) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 17.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-3
(RAT) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-5
(RAT) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Chymase
(Homo sapiens (Human)) | BDBM50208228
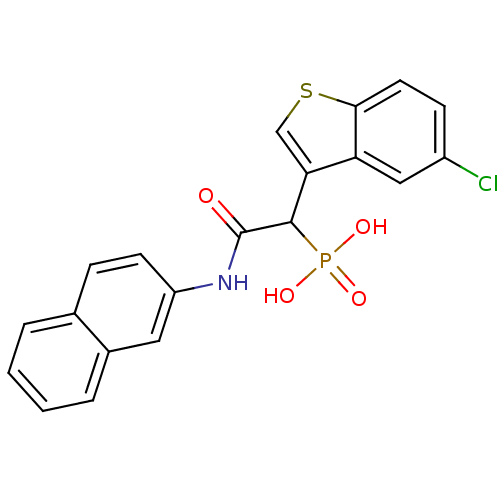
(1-(5-chlorobenzo[b]thiophen-3-yl)-2-(naphthalen-2-...)Show SMILES OP(O)(=O)C(C(=O)Nc1ccc2ccccc2c1)c1csc2ccc(Cl)cc12 Show InChI InChI=1S/C20H15ClNO4PS/c21-14-6-8-18-16(10-14)17(11-28-18)19(27(24,25)26)20(23)22-15-7-5-12-3-1-2-4-13(12)9-15/h1-11,19H,(H,22,23)(H2,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human skin chymase |
J Med Chem 50: 1727-30 (2007)
Article DOI: 10.1021/jm0700619
BindingDB Entry DOI: 10.7270/Q26W99RF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin G
(Homo sapiens (Human)) | BDBM50208224

(2-(3-((1-(2-naphthoyl)piperidin-4-yl)(methyl)carba...)Show SMILES CN(C1CCN(CC1)C(=O)c1ccc2ccccc2c1)C(=O)c1cc2ccccc2cc1C(=O)C(c1cccc2ccccc12)P(O)(O)=O Show InChI InChI=1S/C40H35N2O6P/c1-41(32-19-21-42(22-20-32)39(44)31-18-17-26-9-2-3-11-28(26)23-31)40(45)36-25-30-13-5-4-12-29(30)24-35(36)37(43)38(49(46,47)48)34-16-8-14-27-10-6-7-15-33(27)34/h2-18,23-25,32,38H,19-22H2,1H3,(H2,46,47,48) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil Cat G |
J Med Chem 50: 1727-30 (2007)
Article DOI: 10.1021/jm0700619
BindingDB Entry DOI: 10.7270/Q26W99RF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gamma-aminobutyric acid receptor subunit alpha-2
(Rattus norvegicus (Rat)) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-6
(RAT) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50222653
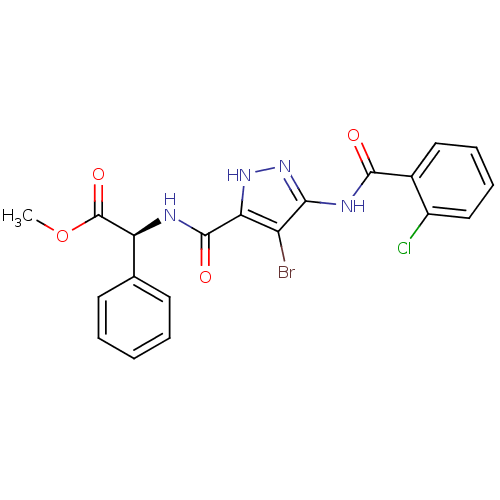
((S)-alpha-[[[4-bromo-5-[(2-chlorobenzoyl)amino]-1H...)Show SMILES COC(=O)[C@@H](NC(=O)c1[nH]nc(NC(=O)c2ccccc2Cl)c1Br)c1ccccc1 Show InChI InChI=1S/C20H16BrClN4O4/c1-30-20(29)15(11-7-3-2-4-8-11)23-19(28)16-14(21)17(26-25-16)24-18(27)12-9-5-6-10-13(12)22/h2-10,15H,1H3,(H,23,28)(H2,24,25,26,27)/t15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAKA from human bradykinin B1 receptor in IL1beta stimulated IMR90 cells |
J Med Chem 50: 5161-7 (2007)
Article DOI: 10.1021/jm051292n
BindingDB Entry DOI: 10.7270/Q2G73DF9 |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-3
(RAT) | BDBM85683
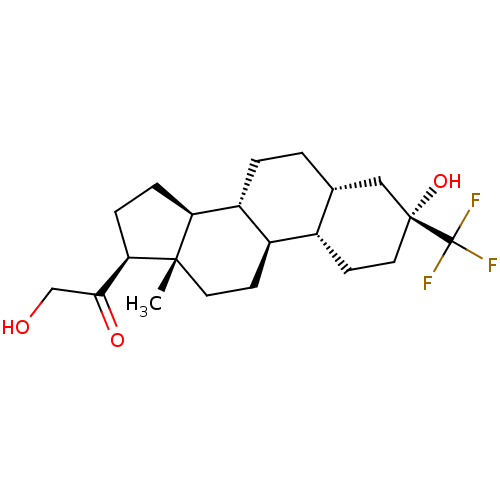
(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 96 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50222669
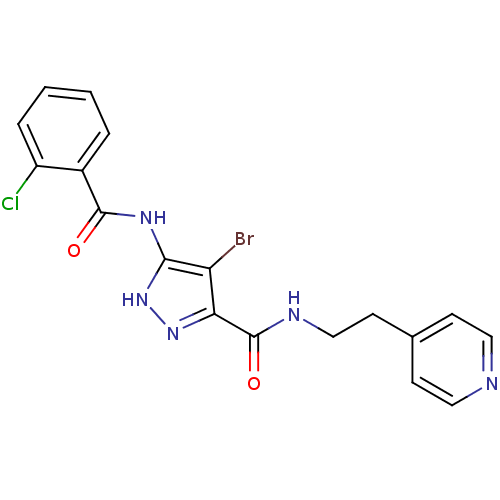
(4-bromo-5-[(2-chlorobenzoyl)amino]-N-[2-(4-pyridin...)Show SMILES Clc1ccccc1C(=O)Nc1[nH]nc(C(=O)NCCc2ccncc2)c1Br Show InChI InChI=1S/C18H15BrClN5O2/c19-14-15(18(27)22-10-7-11-5-8-21-9-6-11)24-25-16(14)23-17(26)12-3-1-2-4-13(12)20/h1-6,8-9H,7,10H2,(H,22,27)(H2,23,24,25,26) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAKA from human bradykinin B1 receptor in IL1beta stimulated IMR90 cells |
J Med Chem 50: 5161-7 (2007)
Article DOI: 10.1021/jm051292n
BindingDB Entry DOI: 10.7270/Q2G73DF9 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50222657
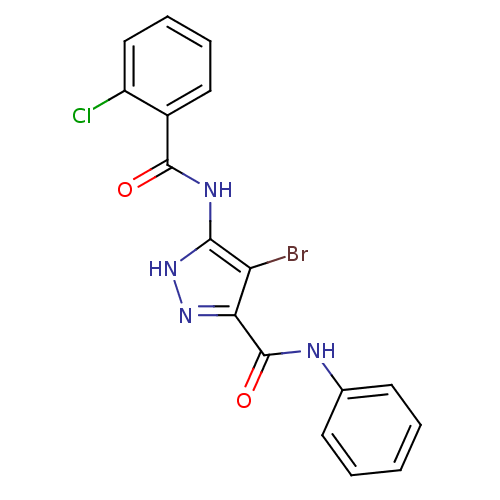
(4-bromo-5-[(2-chlorobenzoyl)amino]-N-phenyl-1H-pyr...)Show SMILES Clc1ccccc1C(=O)Nc1[nH]nc(C(=O)Nc2ccccc2)c1Br Show InChI InChI=1S/C17H12BrClN4O2/c18-13-14(17(25)20-10-6-2-1-3-7-10)22-23-15(13)21-16(24)11-8-4-5-9-12(11)19/h1-9H,(H,20,25)(H2,21,22,23,24) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAKA from human bradykinin B1 receptor in IL1beta stimulated IMR90 cells |
J Med Chem 50: 5161-7 (2007)
Article DOI: 10.1021/jm051292n
BindingDB Entry DOI: 10.7270/Q2G73DF9 |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-1
(Rattus norvegicus (Rat)) | BDBM85683

(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-2
(Rattus norvegicus (Rat)) | BDBM85683

(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-5
(RAT) | BDBM85683

(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-6
(RAT) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50045050

(1-(3-Hydroxy-10,13-dimethyl-hexadecahydro-cyclopen...)Show SMILES CC(=O)C1CCC2C3CC[C@H]4C[C@H](O)CC[C@]4(C)C3CC[C@]12C Show InChI InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16?,17?,18?,19?,20-,21+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-6
(RAT) | BDBM85683

(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM85683

(Co 2-6749 | Co-2-6749 | GMA-839 | WAY-141839)Show SMILES C[C@]12CC[C@H]3[C@@H](CC[C@@H]4C[C@](O)(CC[C@H]34)C(F)(F)F)[C@@H]1CC[C@@H]2C(=O)CO Show InChI InChI=1S/C21H31F3O3/c1-19-8-6-14-13-7-9-20(27,21(22,23)24)10-12(13)2-3-15(14)16(19)4-5-17(19)18(26)11-25/h12-17,25,27H,2-11H2,1H3/t12-,13+,14-,15-,16+,17-,19+,20-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CoCensys, Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 295: 337-45 (2000)
BindingDB Entry DOI: 10.7270/Q2251GQJ |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50208228

(1-(5-chlorobenzo[b]thiophen-3-yl)-2-(naphthalen-2-...)Show SMILES OP(O)(=O)C(C(=O)Nc1ccc2ccccc2c1)c1csc2ccc(Cl)cc12 Show InChI InChI=1S/C20H15ClNO4PS/c21-14-6-8-18-16(10-14)17(11-28-18)19(27(24,25)26)20(23)22-15-7-5-12-3-1-2-4-13(12)9-15/h1-11,19H,(H,22,23)(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil Cat G |
J Med Chem 50: 1727-30 (2007)
Article DOI: 10.1021/jm0700619
BindingDB Entry DOI: 10.7270/Q26W99RF |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-1
(Rattus norvegicus) | BDBM50255109
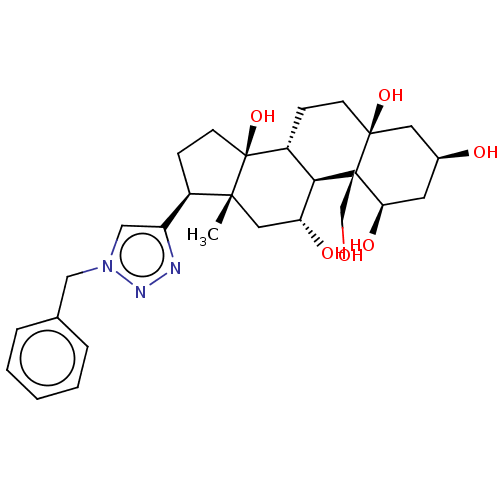
(CHEMBL4081196)Show SMILES [H][C@@]12CC[C@]3(O)C[C@@H](O)C[C@@H](O)[C@]3(CO)[C@@]1([H])[C@H](O)C[C@]1(C)[C@H](CC[C@]21O)c1cn(Cc2ccccc2)nn1 |r| Show InChI InChI=1S/C28H39N3O6/c1-25-13-22(34)24-20(7-9-26(36)12-18(33)11-23(35)27(24,26)16-32)28(25,37)10-8-19(25)21-15-31(30-29-21)14-17-5-3-2-4-6-17/h2-6,15,18-20,22-24,32-37H,7-14,16H2,1H3/t18-,19+,20+,22+,23+,24+,25+,26-,27+,28-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta1 expressed in baculovirus infected insect Sf9 cell membranes using [gamma-32P]ATP as substrat... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-1
(Rattus norvegicus) | BDBM50255111
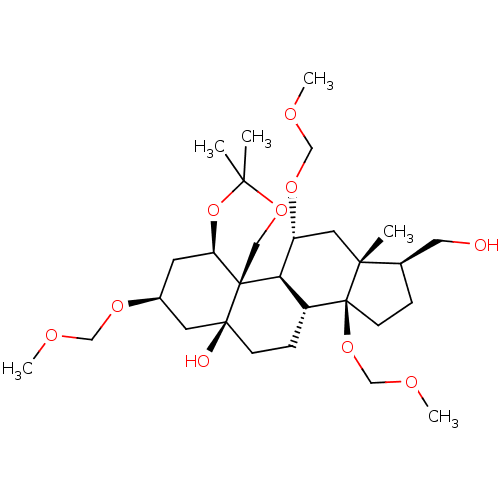
(CHEMBL4092961)Show SMILES [H][C@@]12C[C@@H](C[C@@]3(O)CC[C@]4([H])[C@]([H])([C@@H](C[C@]5(C)[C@@H](CO)CC[C@]45OCOC)OCOC)[C@@]13COC(C)(C)O2)OCOC |r| Show InChI InChI=1S/C29H50O10/c1-25(2)37-15-28-23(39-25)11-20(35-16-32-4)12-27(28,31)9-8-21-24(28)22(36-17-33-5)13-26(3)19(14-30)7-10-29(21,26)38-18-34-6/h19-24,30-31H,7-18H2,1-6H3/t19-,20+,21-,22-,23-,24-,26-,27+,28-,29+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta1 expressed in baculovirus infected insect Sf9 cell membranes using [gamma-32P]ATP as substrat... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50222656
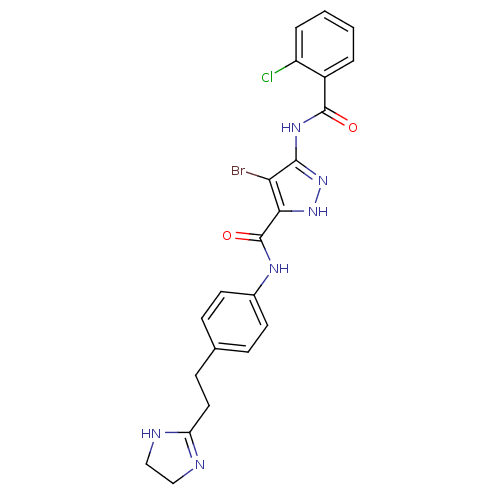
(4-bromo-5-[(2-chlorobenzoyl)amino]-N-[4-[2-(4,5-di...)Show SMILES Clc1ccccc1C(=O)Nc1n[nH]c(C(=O)Nc2ccc(CCC3=NCCN3)cc2)c1Br |t:24| Show InChI InChI=1S/C22H20BrClN6O2/c23-18-19(29-30-20(18)28-21(31)15-3-1-2-4-16(15)24)22(32)27-14-8-5-13(6-9-14)7-10-17-25-11-12-26-17/h1-6,8-9H,7,10-12H2,(H,25,26)(H,27,32)(H2,28,29,30,31) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.0750 | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human bradykinin B1 receptor in IL1beta stimulated IMR90 cells by FLIPR assay |
J Med Chem 50: 5161-7 (2007)
Article DOI: 10.1021/jm051292n
BindingDB Entry DOI: 10.7270/Q2G73DF9 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50167611

((2R,5R)-1-[4-(2-Chloro-4-fluoro-benzyloxy)-benzene...)Show SMILES CC1(C)C[C@@H](O)CN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(F)cc2Cl)cc1 Show InChI InChI=1S/C21H24ClFN2O6S/c1-21(2)10-15(26)11-25(19(21)20(27)24-28)32(29,30)17-7-5-16(6-8-17)31-12-13-3-4-14(23)9-18(13)22/h3-9,15,19,26,28H,10-12H2,1-2H3,(H,24,27)/t15-,19+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Groton Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Matrix metalloproteinase-13 (MMP-13) |
Bioorg Med Chem Lett 15: 2808-11 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.105
BindingDB Entry DOI: 10.7270/Q2BC3Z24 |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-1
(Rattus norvegicus) | BDBM50255122
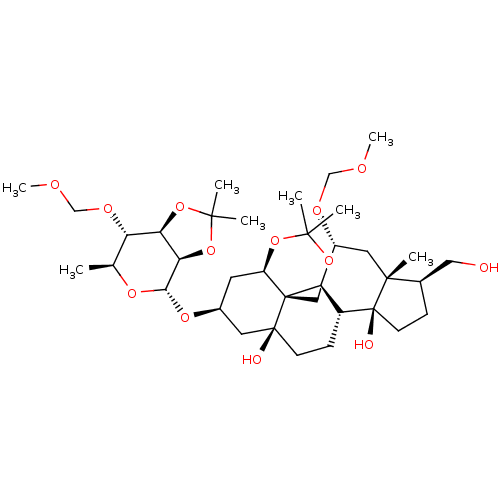
(CHEMBL4059538)Show SMILES [H][C@]12OC(C)(C)O[C@@]1([H])[C@]([H])(O[C@H]1C[C@@]3([H])OC(C)(C)OC[C@]33[C@@]4([H])[C@@H](C[C@]5(C)[C@@H](CO)CC[C@]5(O)[C@]4([H])CC[C@]3(O)C1)OCOC)O[C@@H](C)[C@@H]2OCOC |r| Show InChI InChI=1S/C36H60O13/c1-20-27(43-19-41-8)28-29(49-32(4,5)48-28)30(45-20)46-22-13-25-35(17-44-31(2,3)47-25)26-23(10-11-34(35,38)14-22)36(39)12-9-21(16-37)33(36,6)15-24(26)42-18-40-7/h20-30,37-39H,9-19H2,1-8H3/t20-,21+,22-,23+,24+,25+,26+,27-,28+,29+,30-,33+,34-,35+,36-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta1 expressed in baculovirus infected insect Sf9 cell membranes using [gamma-32P]ATP as substrat... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50167619
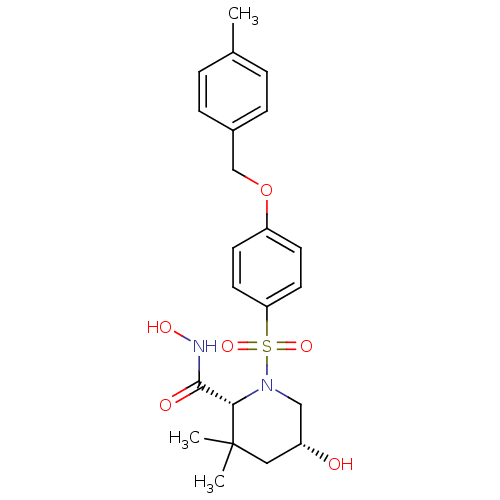
((2R,5R)-5-Hydroxy-3,3-dimethyl-1-[4-(4-methyl-benz...)Show SMILES Cc1ccc(COc2ccc(cc2)S(=O)(=O)N2C[C@H](O)CC(C)(C)[C@@H]2C(=O)NO)cc1 Show InChI InChI=1S/C22H28N2O6S/c1-15-4-6-16(7-5-15)14-30-18-8-10-19(11-9-18)31(28,29)24-13-17(25)12-22(2,3)20(24)21(26)23-27/h4-11,17,20,25,27H,12-14H2,1-3H3,(H,23,26)/t17-,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Groton Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Matrix metalloproteinase-13 (MMP-13) |
Bioorg Med Chem Lett 15: 2808-11 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.105
BindingDB Entry DOI: 10.7270/Q2BC3Z24 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50167618

((2R,5R)-1-[4-(4-Bromo-benzyloxy)-benzenesulfonyl]-...)Show SMILES CC1(C)C[C@@H](O)CN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(Br)cc2)cc1 Show InChI InChI=1S/C21H25BrN2O6S/c1-21(2)11-16(25)12-24(19(21)20(26)23-27)31(28,29)18-9-7-17(8-10-18)30-13-14-3-5-15(22)6-4-14/h3-10,16,19,25,27H,11-13H2,1-2H3,(H,23,26)/t16-,19+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Groton Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Matrix metalloproteinase-13 (MMP-13) |
Bioorg Med Chem Lett 15: 2808-11 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.105
BindingDB Entry DOI: 10.7270/Q2BC3Z24 |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-1
(Rattus norvegicus) | BDBM50255138

(CHEMBL4066361)Show SMILES [H][C@@]12C[C@@H](C[C@@]3(O)CC[C@]4([H])[C@]([H])([C@@H](C[C@]5(C)[C@H](CC[C@]45OCOC)\C=N\O)OCOC)[C@@]13COC(C)(C)O2)OCOC |r| Show InChI InChI=1S/C29H49NO10/c1-25(2)38-15-28-23(40-25)11-20(36-16-33-4)12-27(28,31)9-8-21-24(28)22(37-17-34-5)13-26(3)19(14-30-32)7-10-29(21,26)39-18-35-6/h14,19-24,31-32H,7-13,15-18H2,1-6H3/b30-14+/t19-,20+,21-,22-,23-,24-,26-,27+,28-,29+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta1 expressed in baculovirus infected insect Sf9 cell membranes using [gamma-32P]ATP as substrat... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-3
(Rattus norvegicus) | BDBM50286739
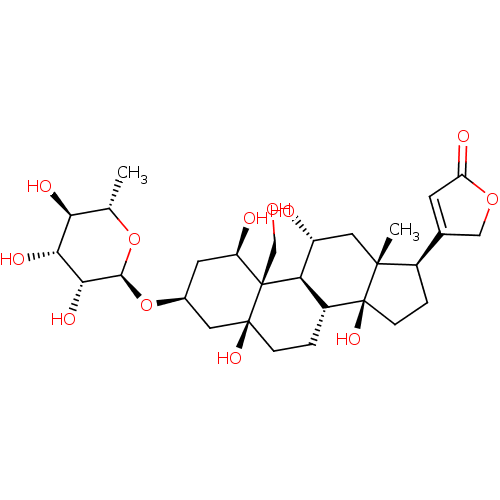
(4-((1R,3S,5S,8R,10R,11R,13R,14S,17R)-1,5,11,14-tet...)Show SMILES C[C@@H]1O[C@@H](O[C@H]2C[C@@H](O)[C@]3(CO)[C@H]4[C@H](O)C[C@]5(C)[C@H](CC[C@]5(O)[C@@H]4CC[C@]3(O)C2)C2=CC(=O)OC2)[C@H](O)[C@H](O)[C@H]1O |r,t:33| Show InChI InChI=1S/C29H44O12/c1-13-22(34)23(35)24(36)25(40-13)41-15-8-19(32)28(12-30)21-17(3-5-27(28,37)9-15)29(38)6-4-16(14-7-20(33)39-11-14)26(29,2)10-18(21)31/h7,13,15-19,21-25,30-32,34-38H,3-6,8-12H2,1-2H3/t13-,15-,16+,17+,18+,19+,21+,22-,23+,24+,25-,26+,27-,28+,29-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta3 expressed in baculovirus infected Sf9 cell membranes using [gamma-32P]ATP as substrate prein... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50222678
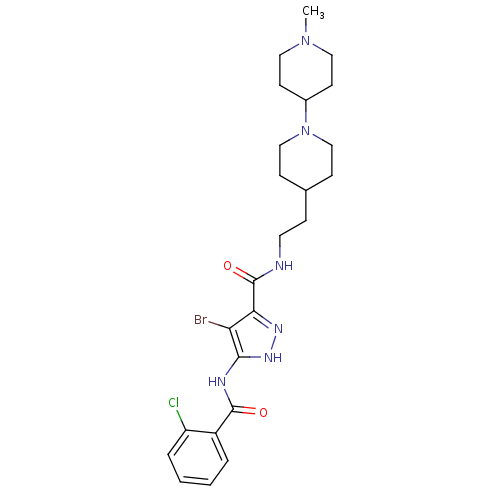
(4-bromo-5-(2-chlorobenzamido)-N-(2-(1'-methyl-1,4'...)Show SMILES CN1CCC(CC1)N1CCC(CCNC(=O)c2n[nH]c(NC(=O)c3ccccc3Cl)c2Br)CC1 Show InChI InChI=1S/C24H32BrClN6O2/c1-31-12-9-17(10-13-31)32-14-7-16(8-15-32)6-11-27-24(34)21-20(25)22(30-29-21)28-23(33)18-4-2-3-5-19(18)26/h2-5,16-17H,6-15H2,1H3,(H,27,34)(H2,28,29,30,33) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human bradykinin B1 receptor in IL1beta stimulated IMR90 cells by FLIPR assay |
J Med Chem 50: 5161-7 (2007)
Article DOI: 10.1021/jm051292n
BindingDB Entry DOI: 10.7270/Q2G73DF9 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM26788
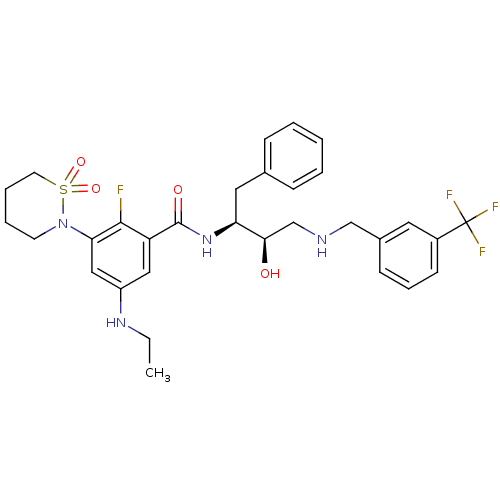
(3-(1,1-dioxo-1,2-thiazinan-2-yl)-5-(ethylamino)-2-...)Show SMILES CCNc1cc(N2CCCCS2(=O)=O)c(F)c(c1)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C31H36F4N4O4S/c1-2-37-24-17-25(29(32)27(18-24)39-13-6-7-14-44(39,42)43)30(41)38-26(16-21-9-4-3-5-10-21)28(40)20-36-19-22-11-8-12-23(15-22)31(33,34)35/h3-5,8-12,15,17-18,26,28,36-37,40H,2,6-7,13-14,16,19-20H2,1H3,(H,38,41)/t26-,28+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK
| Assay Description
Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... |
Bioorg Med Chem Lett 19: 3664-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.165
BindingDB Entry DOI: 10.7270/Q2F18X23 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM26503
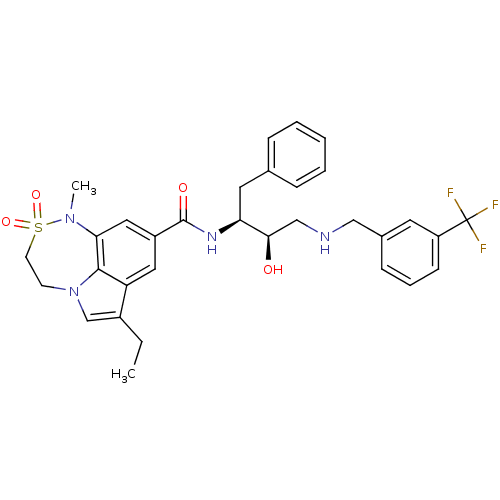
(3-ethyl-N-[(2S,3R)-3-hydroxy-1-phenyl-4-({[3-(trif...)Show SMILES CCc1cn2CCS(=O)(=O)N(C)c3cc(cc1c23)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C32H35F3N4O4S/c1-3-23-20-39-12-13-44(42,43)38(2)28-17-24(16-26(23)30(28)39)31(41)37-27(15-21-8-5-4-6-9-21)29(40)19-36-18-22-10-7-11-25(14-22)32(33,34)35/h4-11,14,16-17,20,27,29,36,40H,3,12-13,15,18-19H2,1-2H3,(H,37,41)/t27-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK
| Assay Description
Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... |
J Med Chem 51: 3313-7 (2008)
Article DOI: 10.1021/jm800138h
BindingDB Entry DOI: 10.7270/Q2XS5SQR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM26503

(3-ethyl-N-[(2S,3R)-3-hydroxy-1-phenyl-4-({[3-(trif...)Show SMILES CCc1cn2CCS(=O)(=O)N(C)c3cc(cc1c23)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C32H35F3N4O4S/c1-3-23-20-39-12-13-44(42,43)38(2)28-17-24(16-26(23)30(28)39)31(41)37-27(15-21-8-5-4-6-9-21)29(40)19-36-18-22-10-7-11-25(14-22)32(33,34)35/h4-11,14,16-17,20,27,29,36,40H,3,12-13,15,18-19H2,1-2H3,(H,37,41)/t27-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK
| Assay Description
Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... |
Bioorg Med Chem Lett 19: 3669-73 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.150
BindingDB Entry DOI: 10.7270/Q29885BR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM29782
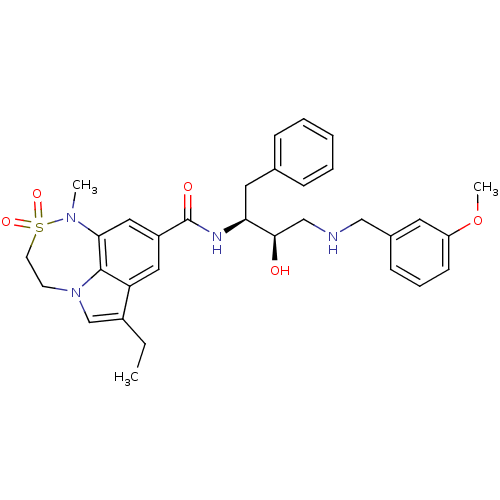
(7,6,5 tricyclic sulfonamide, 22)Show SMILES CCc1cn2CCS(=O)(=O)N(C)c3cc(cc1c23)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(OC)c1 |r| Show InChI InChI=1S/C32H38N4O5S/c1-4-24-21-36-13-14-42(39,40)35(2)29-18-25(17-27(24)31(29)36)32(38)34-28(16-22-9-6-5-7-10-22)30(37)20-33-19-23-11-8-12-26(15-23)41-3/h5-12,15,17-18,21,28,30,33,37H,4,13-14,16,19-20H2,1-3H3,(H,34,38)/t28-,30+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK
| Assay Description
Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... |
Bioorg Med Chem Lett 19: 3669-73 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.150
BindingDB Entry DOI: 10.7270/Q29885BR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM26503

(3-ethyl-N-[(2S,3R)-3-hydroxy-1-phenyl-4-({[3-(trif...)Show SMILES CCc1cn2CCS(=O)(=O)N(C)c3cc(cc1c23)C(=O)N[C@@H](Cc1ccccc1)[C@H](O)CNCc1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C32H35F3N4O4S/c1-3-23-20-39-12-13-44(42,43)38(2)28-17-24(16-26(23)30(28)39)31(41)37-27(15-21-8-5-4-6-9-21)29(40)19-36-18-22-10-7-11-25(14-22)32(33,34)35/h4-11,14,16-17,20,27,29,36,40H,3,12-13,15,18-19H2,1-2H3,(H,37,41)/t27-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | 4.5 | 22 |
GSK
| Assay Description
Enzyme activity and inhibition were assayed using a fluorescent FAM-[SEVNLDAEFK]-TAMRA substrate. Control reactions with no enzyme were included in e... |
Bioorg Med Chem Lett 19: 3674-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.149
BindingDB Entry DOI: 10.7270/Q25H7DKK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Collagenase 3
(Homo sapiens (Human)) | BDBM50167620
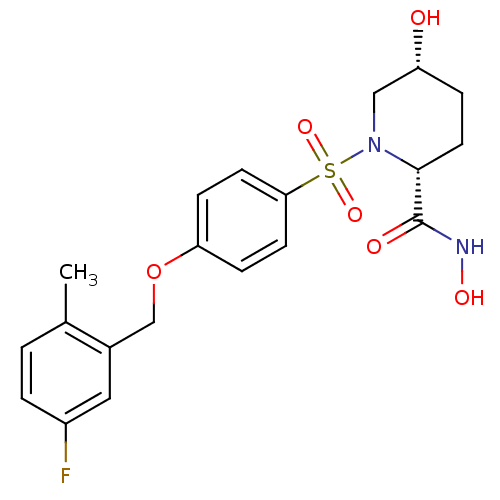
((2R,5R)-1-[4-(5-Fluoro-2-methyl-benzyloxy)-benzene...)Show SMILES Cc1ccc(F)cc1COc1ccc(cc1)S(=O)(=O)N1C[C@H](O)CC[C@@H]1C(=O)NO Show InChI InChI=1S/C20H23FN2O6S/c1-13-2-3-15(21)10-14(13)12-29-17-5-7-18(8-6-17)30(27,28)23-11-16(24)4-9-19(23)20(25)22-26/h2-3,5-8,10,16,19,24,26H,4,9,11-12H2,1H3,(H,22,25)/t16-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Groton Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Matrix metalloproteinase-13 (MMP-13) |
Bioorg Med Chem Lett 15: 2808-11 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.105
BindingDB Entry DOI: 10.7270/Q2BC3Z24 |
More data for this
Ligand-Target Pair | |
Sodium/potassium-transporting ATPase subunit alpha-4/beta-1
(Rattus norvegicus) | BDBM50255120
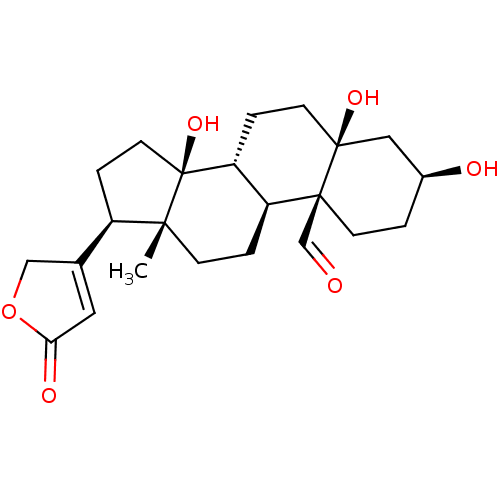
(CHEBI:38178 | STROPHANTHIDIN)Show SMILES [H][C@@]12CC[C@]3(O)C[C@@H](O)CC[C@]3(C=O)[C@@]1([H])CC[C@]1(C)[C@H](CC[C@]21O)C1=CC(=O)OC1 |r,t:29| Show InChI InChI=1S/C23H32O6/c1-20-6-3-17-18(4-8-22(27)11-15(25)2-7-21(17,22)13-24)23(20,28)9-5-16(20)14-10-19(26)29-12-14/h10,13,15-18,25,27-28H,2-9,11-12H2,1H3/t15-,16+,17-,18+,20+,21-,22-,23-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, College of Pharmacy , University of Minnesota , Minneapolis , Minnesota 55414 , United States.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant rat Na+/K+-ATPase alpha4/beta1 expressed in baculovirus infected insect Sf9 cell membranes using [gamma-32P]ATP as substrat... |
J Med Chem 61: 1800-1820 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00925
BindingDB Entry DOI: 10.7270/Q2T43WJH |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1/M2/M3/M4/M5
(RAT) | BDBM50229676

(CHEMBL78697)Show InChI InChI=1S/C7H12N6/c8-7-9-11-13(10-7)6-4-12-2-1-5(6)3-12/h5-6H,1-4H2,(H2,8,10)/t5-,6-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro ability to displace [3H]oxotremorine-M (OXO-M) from rat cerebral cortex muscarinic receptor. |
J Med Chem 35: 2392-406 (1992)
BindingDB Entry DOI: 10.7270/Q2TF00K5 |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50167621

((2R,5R)-5-Hydroxy-3,3-dimethyl-1-[4-(3-methyl-benz...)Show SMILES Cc1cccc(COc2ccc(cc2)S(=O)(=O)N2C[C@H](O)CC(C)(C)[C@@H]2C(=O)NO)c1 Show InChI InChI=1S/C22H28N2O6S/c1-15-5-4-6-16(11-15)14-30-18-7-9-19(10-8-18)31(28,29)24-13-17(25)12-22(2,3)20(24)21(26)23-27/h4-11,17,20,25,27H,12-14H2,1-3H3,(H,23,26)/t17-,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Groton Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Matrix metalloproteinase-13 (MMP-13) |
Bioorg Med Chem Lett 15: 2808-11 (2005)
Article DOI: 10.1016/j.bmcl.2005.03.105
BindingDB Entry DOI: 10.7270/Q2BC3Z24 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1/M2/M3/M4/M5
(RAT) | BDBM50229669
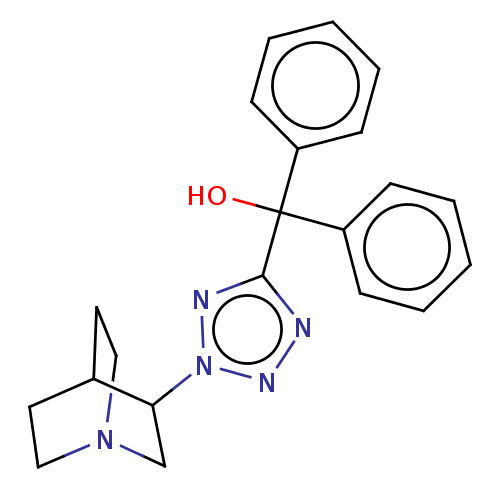
(CHEMBL309432)Show SMILES OC(c1nnn(n1)C1CN2CCC1CC2)(c1ccccc1)c1ccccc1 |(9.08,-4.44,;7.54,-4.44,;6.01,-4.28,;5.24,-2.95,;3.73,-3.27,;3.57,-4.81,;4.97,-5.43,;2.22,-5.58,;2.22,-7.12,;.9,-7.89,;-.44,-7.12,;-.44,-5.58,;.9,-4.81,;1.58,-6.16,;.11,-6.56,;8.45,-3.21,;7.81,-1.81,;8.71,-.56,;10.25,-.72,;10.88,-2.13,;9.98,-3.37,;8.16,-5.85,;7.26,-7.1,;7.87,-8.5,;9.41,-8.66,;10.31,-7.41,;9.69,-6.01,)| Show InChI InChI=1S/C21H23N5O/c27-21(17-7-3-1-4-8-17,18-9-5-2-6-10-18)20-22-24-26(23-20)19-15-25-13-11-16(19)12-14-25/h1-10,16,19,27H,11-15H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro displacement of [3H]quinuclidinyl benzilate (QNB) from rat cerebral cortex muscarinic receptor. |
J Med Chem 35: 2392-406 (1992)
BindingDB Entry DOI: 10.7270/Q2TF00K5 |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454570
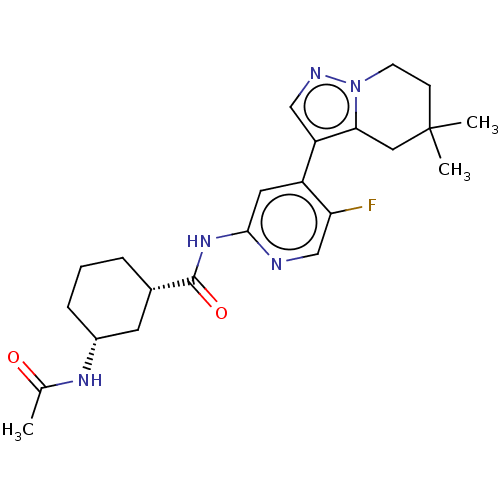
(US10717746, Example 51)Show SMILES CC(=O)N[C@@H]1CCC[C@@H](C1)C(=O)Nc1cc(-c2cnn3CCC(C)(C)Cc23)c(F)cn1 |r| Show InChI InChI=1S/C23H30FN5O2/c1-14(30)27-16-6-4-5-15(9-16)22(31)28-21-10-17(19(24)13-25-21)18-12-26-29-8-7-23(2,3)11-20(18)29/h10,12-13,15-16H,4-9,11H2,1-3H3,(H,27,30)(H,25,28,31)/t15-,16+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTRAZENECA AB
US Patent
| Assay Description
Activity of CDK9 was determined in-vitro using a mobility shift assay on a Caliper LC3000 reader (Caliper/PerkinElmer), which measures fluorescence o... |
US Patent US10717746 (2020)
BindingDB Entry DOI: 10.7270/Q2NV9N9F |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454571

(US10717746, Example 51a)Show SMILES CC1(C)CCn2ncc(c2C1)-c1cc(NC(=O)[C@H]2CCC[C@@H](N)C2)ncc1F |r| Show InChI InChI=1S/C21H28FN5O/c1-21(2)6-7-27-18(10-21)16(11-25-27)15-9-19(24-12-17(15)22)26-20(28)13-4-3-5-14(23)8-13/h9,11-14H,3-8,10,23H2,1-2H3,(H,24,26,28)/t13-,14+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTRAZENECA AB
US Patent
| Assay Description
Activity of CDK9 was determined in-vitro using a mobility shift assay on a Caliper LC3000 reader (Caliper/PerkinElmer), which measures fluorescence o... |
US Patent US10717746 (2020)
BindingDB Entry DOI: 10.7270/Q2NV9N9F |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454572
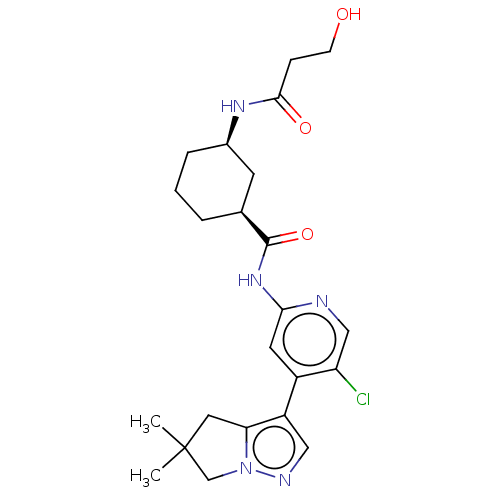
(US10717746, Example 52)Show SMILES CC1(C)Cc2c(cnn2C1)-c1cc(NC(=O)[C@H]2CCC[C@H](C2)NC(=O)CCO)ncc1Cl |r| Show InChI InChI=1S/C23H30ClN5O3/c1-23(2)10-19-17(11-26-29(19)13-23)16-9-20(25-12-18(16)24)28-22(32)14-4-3-5-15(8-14)27-21(31)6-7-30/h9,11-12,14-15,30H,3-8,10,13H2,1-2H3,(H,27,31)(H,25,28,32)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTRAZENECA AB
US Patent
| Assay Description
Activity of CDK9 was determined in-vitro using a mobility shift assay on a Caliper LC3000 reader (Caliper/PerkinElmer), which measures fluorescence o... |
US Patent US10717746 (2020)
BindingDB Entry DOI: 10.7270/Q2NV9N9F |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM50588263
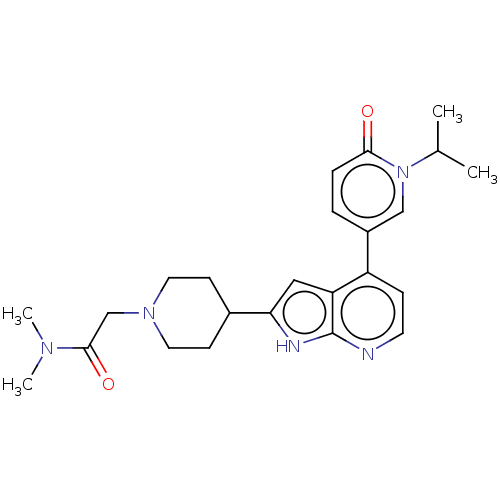
(CHEMBL5198038)Show SMILES CC(C)n1cc(ccc1=O)-c1ccnc2[nH]c(cc12)C1CCN(CC(=O)N(C)C)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01249
BindingDB Entry DOI: 10.7270/Q2F193PM |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM50588264

(CHEMBL5207871)Show SMILES CC(C)n1cc(ccc1=O)-c1ccnc2[nH]c(cc12)C1CCN(CC(=O)N2CCCC2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01249
BindingDB Entry DOI: 10.7270/Q2F193PM |
More data for this
Ligand-Target Pair | |
Cyclin-T1/Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454567

(US10717746, Example 48)Show SMILES CC(=O)N[C@@H]1CC[C@@H](C1)C(=O)Nc1cc(-c2cnn3CC(C)(C)Cc23)c(Cl)cn1 |r| Show InChI InChI=1S/C21H26ClN5O2/c1-12(28)25-14-5-4-13(6-14)20(29)26-19-7-15(17(22)10-23-19)16-9-24-27-11-21(2,3)8-18(16)27/h7,9-10,13-14H,4-6,8,11H2,1-3H3,(H,25,28)(H,23,26,29)/t13-,14+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length human N-terminal GST-fused CDK9 (1 to 372 residues)/His-tagged CyclinT1 (1 to 726 residues) expressed in baculo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01754
BindingDB Entry DOI: 10.7270/Q2M90D8V |
More data for this
Ligand-Target Pair | |
Cyclin-T1/Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454570

(US10717746, Example 51)Show SMILES CC(=O)N[C@@H]1CCC[C@@H](C1)C(=O)Nc1cc(-c2cnn3CCC(C)(C)Cc23)c(F)cn1 |r| Show InChI InChI=1S/C23H30FN5O2/c1-14(30)27-16-6-4-5-15(9-16)22(31)28-21-10-17(19(24)13-25-21)18-12-26-29-8-7-23(2,3)11-20(18)29/h10,12-13,15-16H,4-9,11H2,1-3H3,(H,27,30)(H,25,28,31)/t15-,16+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length human N-terminal GST-fused CDK9 (1 to 372 residues)/His-tagged CyclinT1 (1 to 726 residues) expressed in baculo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01754
BindingDB Entry DOI: 10.7270/Q2M90D8V |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454577

(US10717746, Example 57)Show SMILES CC1(C)Cc2ncc(-c3cc(NC(=O)[C@H]4CCC[C@H](C4)NC(=O)C4COC4)ncc3Cl)n2C1 |r| Show InChI InChI=1S/C24H30ClN5O3/c1-24(2)8-21-27-10-19(30(21)13-24)17-7-20(26-9-18(17)25)29-22(31)14-4-3-5-16(6-14)28-23(32)15-11-33-12-15/h7,9-10,14-16H,3-6,8,11-13H2,1-2H3,(H,28,32)(H,26,29,31)/t14-,16+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTRAZENECA AB
US Patent
| Assay Description
Activity of CDK9 was determined in-vitro using a mobility shift assay on a Caliper LC3000 reader (Caliper/PerkinElmer), which measures fluorescence o... |
US Patent US10717746 (2020)
BindingDB Entry DOI: 10.7270/Q2NV9N9F |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM454578
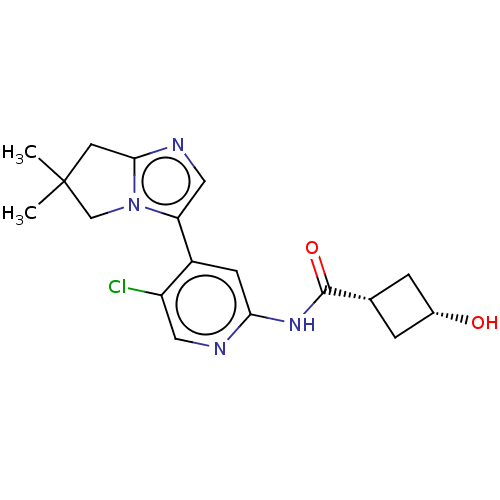
(US10717746, Example 58)Show SMILES CC1(C)Cc2ncc(-c3cc(NC(=O)[C@@H]4C[C@H](O)C4)ncc3Cl)n2C1 |r,wD:14.13,16.16,(1.09,5.09,;.32,3.76,;1.81,3.36,;-.93,4.66,;-2.17,3.76,;-3.71,3.76,;-4.19,2.29,;-2.94,1.39,;-2.94,-.15,;-1.61,-.92,;-1.61,-2.46,;-.28,-3.23,;1.06,-2.46,;1.06,-.92,;2.39,-3.23,;3.88,-2.83,;4.28,-4.32,;5.61,-5.09,;2.79,-4.72,;-2.94,-3.23,;-4.28,-2.46,;-4.28,-.92,;-5.61,-.15,;-1.7,2.29,;-.16,2.29,)| Show InChI InChI=1S/C18H21ClN4O2/c1-18(2)6-16-21-8-14(23(16)9-18)12-5-15(20-7-13(12)19)22-17(25)10-3-11(24)4-10/h5,7-8,10-11,24H,3-4,6,9H2,1-2H3,(H,20,22,25)/t10-,11+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <3 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTRAZENECA AB
US Patent
| Assay Description
Activity of CDK9 was determined in-vitro using a mobility shift assay on a Caliper LC3000 reader (Caliper/PerkinElmer), which measures fluorescence o... |
US Patent US10717746 (2020)
BindingDB Entry DOI: 10.7270/Q2NV9N9F |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data