

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Apelin (Homo sapiens (Human)) | BDBM50009575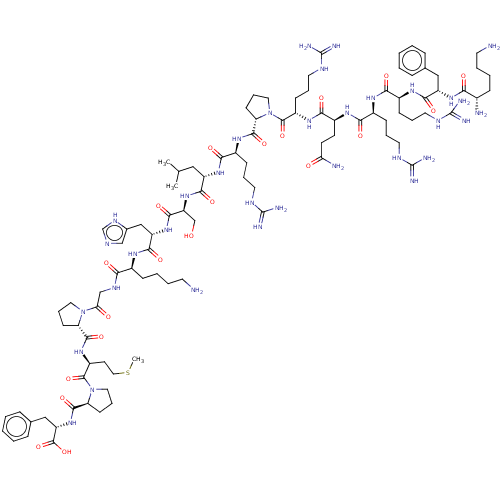 (CHEMBL3234446) | KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [125I]-pE13F from human apelin receptor expressed in CHO cell membranes after 1 hr by gamma counting analysis | J Med Chem 57: 2908-19 (2014) Article DOI: 10.1021/jm401789v BindingDB Entry DOI: 10.7270/Q21J9C9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50241132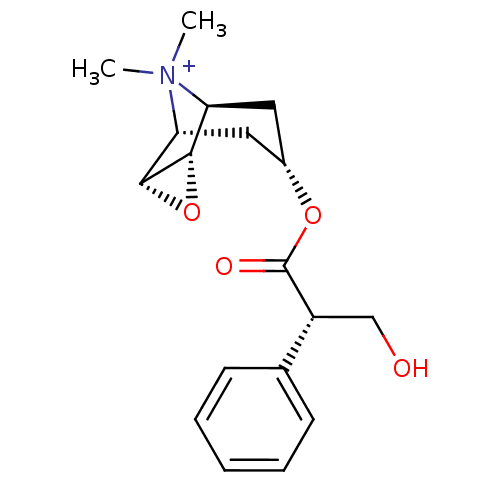 (3-Hydroxy-2-phenyl-propionic acid 9-methyl-3-oxa-9...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.0955 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]NMS from EGFP-fused human M1 receptor N-terminal truncated at 17 residues expressed in HEK293 cells after 22 hrs by liquid scinti... | J Med Chem 55: 2125-43 (2012) Article DOI: 10.1021/jm201348t BindingDB Entry DOI: 10.7270/Q2XW4KZ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50241132 (3-Hydroxy-2-phenyl-propionic acid 9-methyl-3-oxa-9...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive inhibition of EGFP-fused human M1 receptor N-terminal truncated at 17 residues expressed in HEK293 cells after 4 hrs by FRET assay in pre... | J Med Chem 55: 2125-43 (2012) Article DOI: 10.1021/jm201348t BindingDB Entry DOI: 10.7270/Q2XW4KZ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50077141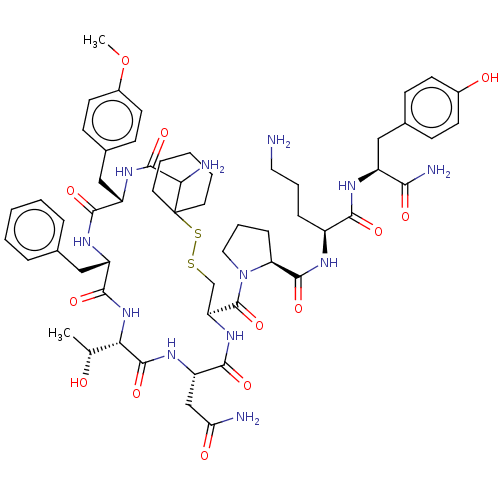 (CHEMBL3416758) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to human oxytocin receptor by radioligand binding assay | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50459408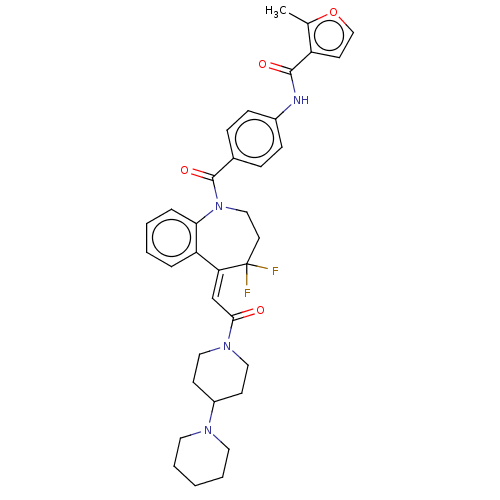 (CHEMBL3307200) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human V1A receptor expressed in CHO cell membranes | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50077141 (CHEMBL3416758) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (RAT) | BDBM85095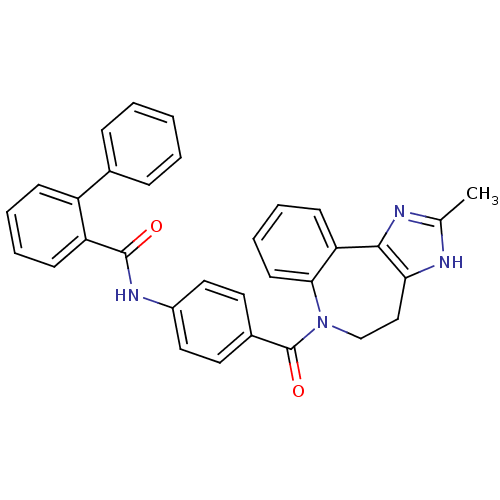 (CAS_151171 | CONIVAPTAN | NSC_151171 | YM087) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]AVP from V1A receptor in Wistar rat liver membranes incubated for 60 mins by microplate scintillation counting method | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1b receptor (Homo sapiens (Human)) | BDBM35667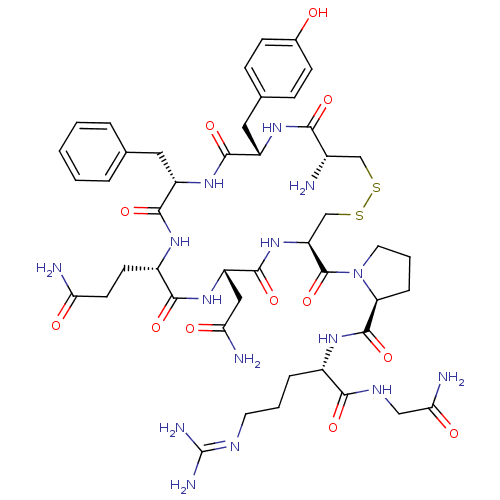 (AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V1b receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50307117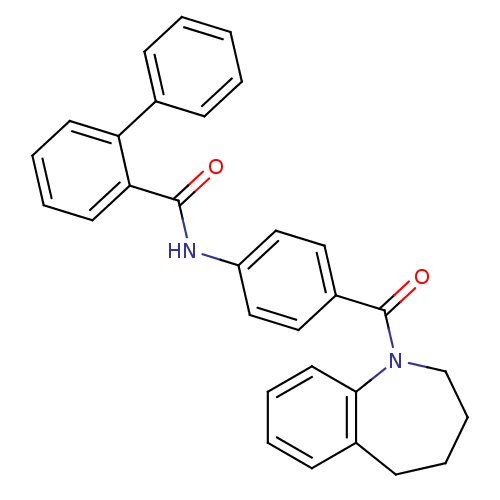 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR 7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]AVP from human vasopressin V2 receptor expressed in CHO cells | J Med Chem 53: 1546-62 (2010) Article DOI: 10.1021/jm901084f BindingDB Entry DOI: 10.7270/Q2FX7BD4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human V2 receptor expressed in CHO cell membranes by radioligand binding assay | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50000483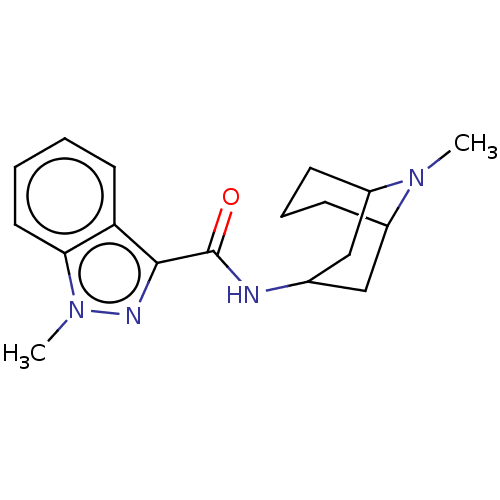 ((BRL 43694)1-Methyl-1H-indazole-3-carboxylic acid ...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 0.589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Potency at neuronal 5-hydroxytryptamine 3 receptor in the rabbit heart | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50108392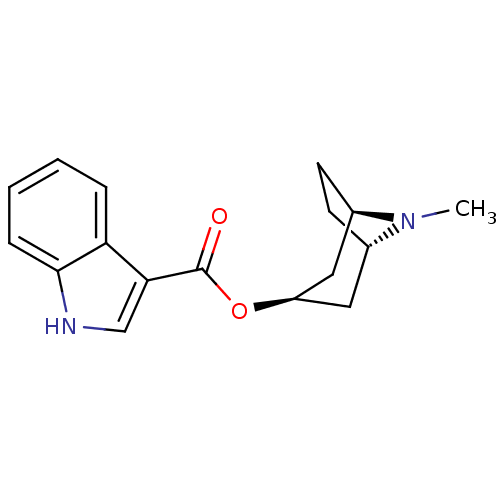 ((3-ENDO)-8-METHYL-8-AZABICYCLO[3.2.1]OCT-3-YL 1H-I...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 0.603 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity to 5-hydroxytryptamine 3 receptor entirely in guinea pig ileum | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50077141 (CHEMBL3416758) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM35667 (AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V1a receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50205990 (CHEMBL395429 | OXYTOCIN) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 0.790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to oxytocin receptor (unknown origin) by radioligand binding assay | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50205990 (CHEMBL395429 | OXYTOCIN) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 0.790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to OTR (unknown origin) | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397209 (CHEMBL2172291) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from SNAP-tagged vasopressin V2 receptor expressed in HEK293 cells by FRET assay | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50014407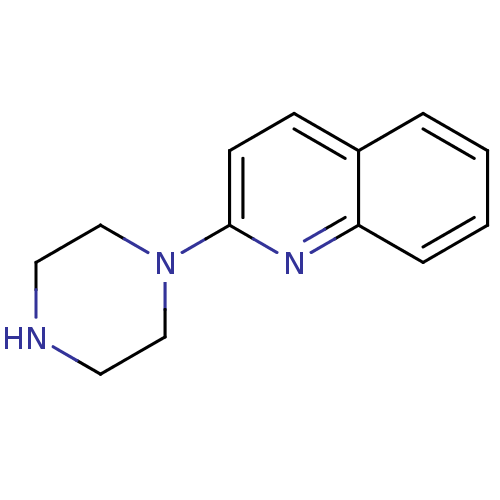 (2-(piperazin-1-yl)quinoline | 2-Piperazin-1-yl-qui...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Potency at neuronal 5-hydroxytryptamine 3 receptors in the rabbit heart | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM35667 (AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 1.36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins by saturation binding assay | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50014407 (2-(piperazin-1-yl)quinoline | 2-Piperazin-1-yl-qui...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Potency at neuronal 5-hydroxytryptamine 3 receptor in the rabbit heart | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM35667 (AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 1.48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM35667 (AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 1.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human oxytocin receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397210 (CHEMBL2172290) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from SNAP-tagged vasopressin V2 receptor expressed in HEK293 cells by FRET assay | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50459401 (CHEMBL4210854) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human V1A receptor expressed in CHO cell membranes by radioligand binding assay | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50052954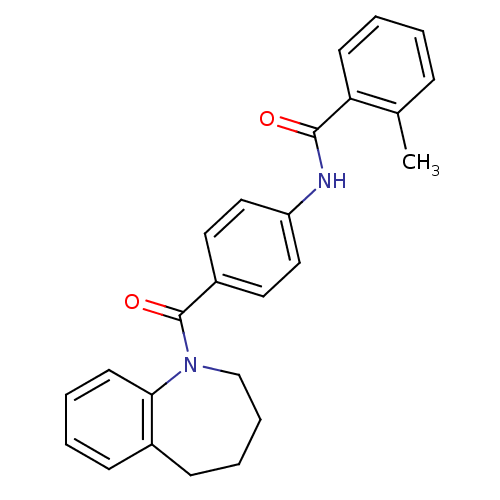 (2-Methyl-N-[4-(2,3,4,5-tetrahydro-benzo[b]azepine-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human V1A receptor expressed in CHO cell membranes by radioligand binding assay | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.74 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V1a receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.74 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR 7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]AVP from human vasopressin V1a receptor expressed in CHO cells | J Med Chem 53: 1546-62 (2010) Article DOI: 10.1021/jm901084f BindingDB Entry DOI: 10.7270/Q2FX7BD4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Rattus norvegicus (Rat)) | BDBM85095 (CAS_151171 | CONIVAPTAN | NSC_151171 | YM087) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Patents Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]AVP from V2 receptor in Wistar rat kidney membranes incubated for 60 mins by microplate scintillation counting method | J Med Chem 61: 8670-8692 (2018) Article DOI: 10.1021/acs.jmedchem.8b00697 BindingDB Entry DOI: 10.7270/Q24J0HRF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50108392 ((3-ENDO)-8-METHYL-8-AZABICYCLO[3.2.1]OCT-3-YL 1H-I...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity to 5-hydroxytryptamine 3 receptor of neuronal in the afferent rabbit vagus | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM85330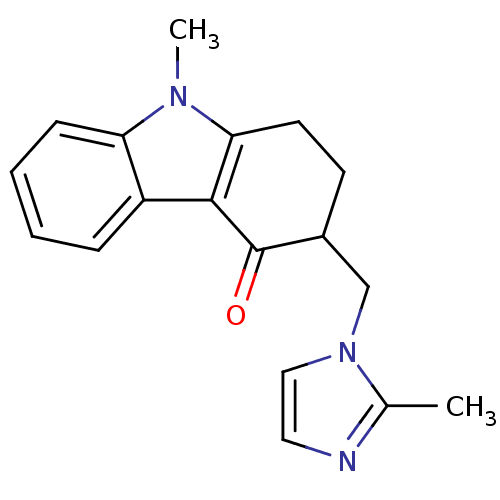 (CAS_68647 | NSC_68647 | ONDANSETRON | Ondansetron ...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid PDB UniChem Similars | PDB PubMed | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity to 5-hydroxytryptamine 3 receptor of neuronal in the afferent rabbit vagus | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM85330 (CAS_68647 | NSC_68647 | ONDANSETRON | Ondansetron ...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid PDB UniChem Similars | PDB PubMed | 3.31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Potency at neuronal 5-hydroxytryptamine 3 receptors in the rabbit heart | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397215 (CHEMBL2172295) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397206 (CHEMBL2172289) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50077140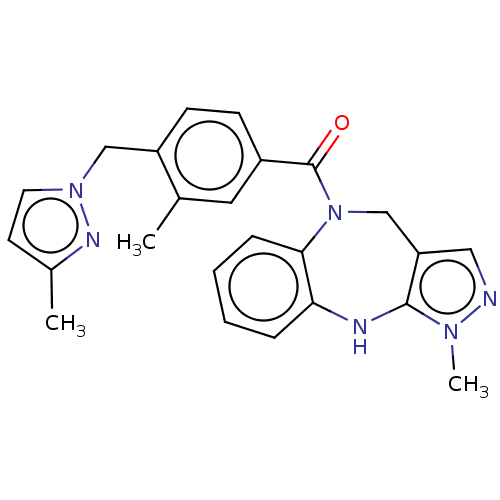 (CHEMBL3416757) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50307120 (5-[4-[(1,1'-Biphenyl-2-ylcarbonyl)amino]-3-methylb...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR 7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]AVP from human vasopressin V2 receptor expressed in CHO cells | J Med Chem 53: 1546-62 (2010) Article DOI: 10.1021/jm901084f BindingDB Entry DOI: 10.7270/Q2FX7BD4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50077140 (CHEMBL3416757) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50205990 (CHEMBL395429 | OXYTOCIN) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50052954 (2-Methyl-N-[4-(2,3,4,5-tetrahydro-benzo[b]azepine-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to vasopressin V2 receptor | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50307117 (CHEMBL603708 | N-(4-(2,3,4,5-tetrahydro-1H-benzo[b...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to vasopressin V2 receptor | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397213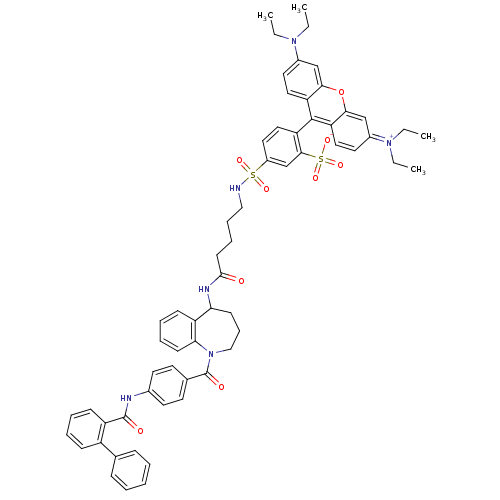 (CHEMBL2172391) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50052954 (2-Methyl-N-[4-(2,3,4,5-tetrahydro-benzo[b]azepine-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 8.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to vasopressin V1a receptor | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50397219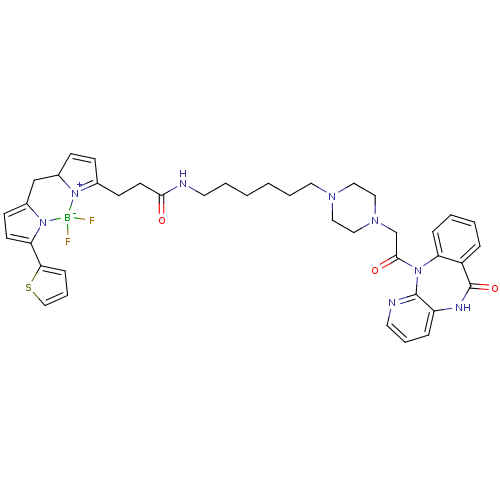 (CHEMBL2172285) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to human muscarinic M1 receptor | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50151212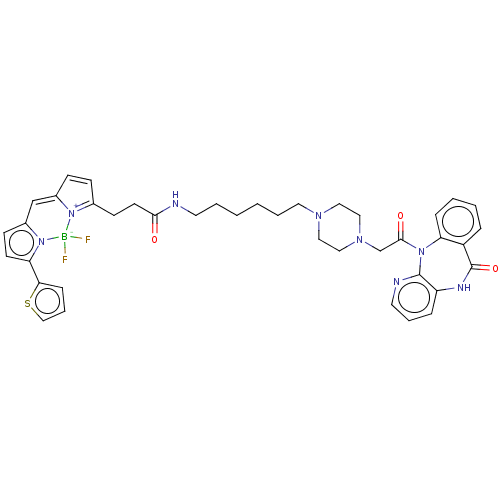 (CHEMBL2368393 | difluoro(2-{[(2Z)-1-methyl-5-[2-({...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR CNRS/ULP 7081 Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding to human muscarinic receptor M1 | J Med Chem 47: 4300-15 (2004) Article DOI: 10.1021/jm040800a BindingDB Entry DOI: 10.7270/Q2T43TVQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50151218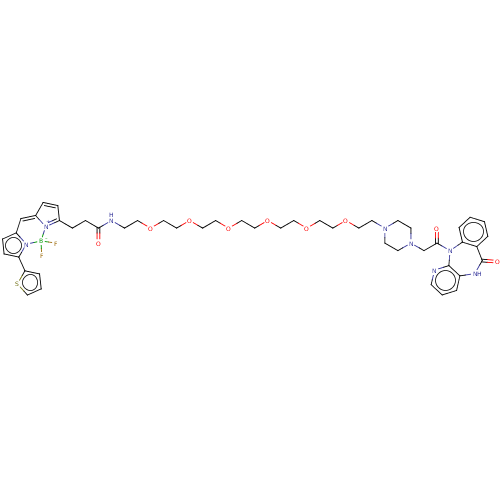 (CHEMBL2368392 | difluoro(2-{[(2Z)-1-methyl-5-(2-{[...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR CNRS/ULP 7081 Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding to human muscarinic receptor M1 | J Med Chem 47: 4300-15 (2004) Article DOI: 10.1021/jm040800a BindingDB Entry DOI: 10.7270/Q2T43TVQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50397210 (CHEMBL2172290) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.02 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Displacement of [3H]-AVP from human vasopressin V2 receptor expressed in CHO cells after 30 mins | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A/3B (Rattus norvegicus-RAT) | BDBM50000483 ((BRL 43694)1-Methyl-1H-indazole-3-carboxylic acid ...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 9.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Binding affinity to 5-hydroxytryptamine 3 receptor of neuronal in the afferent rabbit vagus | J Med Chem 33: 1594-600 (1990) BindingDB Entry DOI: 10.7270/Q25Q4X94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50205990 (CHEMBL395429 | OXYTOCIN) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 9.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Competitive binding to SNAP-tagged oxytocin receptor (unknown origin) expressed in HEK293 cells incubated for 1 hr at RT followed by 4 hrs at 4 degC ... | J Med Chem 58: 2547-52 (2015) Article DOI: 10.1021/jm501395b BindingDB Entry DOI: 10.7270/Q2TB18KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM35714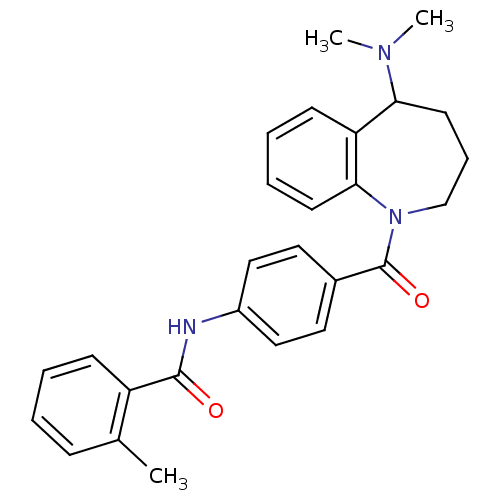 (CHEMBL420762 | Mozavaptan | N-[4-(5-Dimethylamino-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UMR7200 CNRS/Universit£ de Strasbourg Curated by ChEMBL | Assay Description Binding affinity to vasopressin V2 receptor | J Med Chem 55: 8588-602 (2012) Article DOI: 10.1021/jm3006146 BindingDB Entry DOI: 10.7270/Q28G8MT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 536 total ) | Next | Last >> |