 Found 1678 hits with Last Name = 'hoo' and Initial = 'k'
Found 1678 hits with Last Name = 'hoo' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50329836
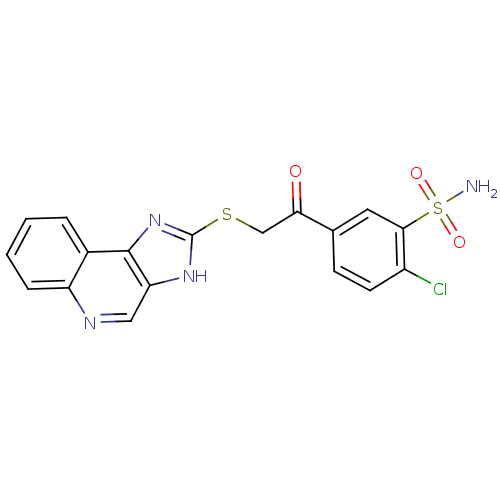
(2-Chloro-5-[(1H-imidazo[4,5-c]quinolin-2-ylsulfany...)Show SMILES NS(=O)(=O)c1cc(ccc1Cl)C(=O)CSc1nc2c(cnc3ccccc23)[nH]1 Show InChI InChI=1S/C18H13ClN4O3S2/c19-12-6-5-10(7-16(12)28(20,25)26)15(24)9-27-18-22-14-8-21-13-4-2-1-3-11(13)17(14)23-18/h1-8H,9H2,(H,22,23)(H2,20,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| MMDB
Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50329836

(2-Chloro-5-[(1H-imidazo[4,5-c]quinolin-2-ylsulfany...)Show SMILES NS(=O)(=O)c1cc(ccc1Cl)C(=O)CSc1nc2c(cnc3ccccc23)[nH]1 Show InChI InChI=1S/C18H13ClN4O3S2/c19-12-6-5-10(7-16(12)28(20,25)26)15(24)9-27-18-22-14-8-21-13-4-2-1-3-11(13)17(14)23-18/h1-8H,9H2,(H,22,23)(H2,20,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM50060627
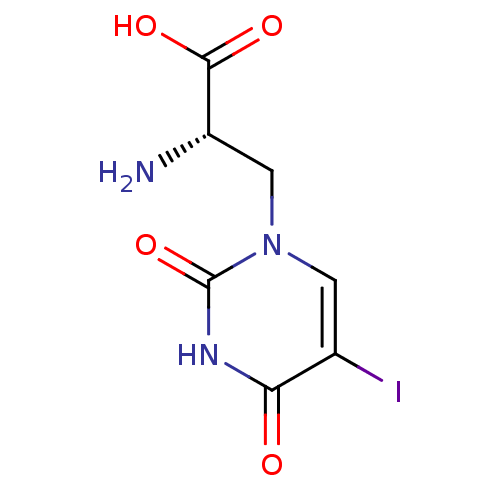
((S)-2-Amino-3-(5-iodo-2,4-dioxo-3,4-dihydro-2H-pyr...)Show InChI InChI=1S/C7H8IN3O4/c8-3-1-11(2-4(9)6(13)14)7(15)10-5(3)12/h1,4H,2,9H2,(H,13,14)(H,10,12,15)/t4-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Displacement of [3H]kainate from human Ionotropic glutamate receptor ionotropic kainate 1 expressed in HEK293 cells |
J Med Chem 40: 3645-50 (1997)
Article DOI: 10.1021/jm9702387
BindingDB Entry DOI: 10.7270/Q28G8MCB |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50187673
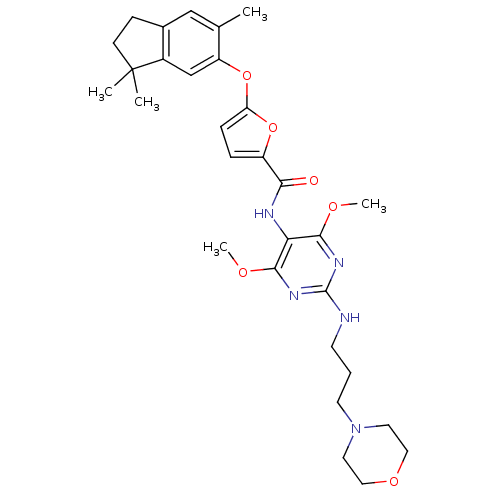
(5-(3,3,6-trimethyl-indan-5-yloxy)-furan-2-carboxyl...)Show SMILES COc1nc(NCCCN2CCOCC2)nc(OC)c1NC(=O)c1ccc(Oc2cc3c(CCC3(C)C)cc2C)o1 Show InChI InChI=1S/C30H39N5O6/c1-19-17-20-9-10-30(2,3)21(20)18-23(19)41-24-8-7-22(40-24)26(36)32-25-27(37-4)33-29(34-28(25)38-5)31-11-6-12-35-13-15-39-16-14-35/h7-8,17-18H,6,9-16H2,1-5H3,(H,32,36)(H,31,33,34) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity to rat pituitary GnRH receptor |
J Med Chem 49: 3362-7 (2006)
Article DOI: 10.1021/jm060012g
BindingDB Entry DOI: 10.7270/Q2F76C54 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50187673

(5-(3,3,6-trimethyl-indan-5-yloxy)-furan-2-carboxyl...)Show SMILES COc1nc(NCCCN2CCOCC2)nc(OC)c1NC(=O)c1ccc(Oc2cc3c(CCC3(C)C)cc2C)o1 Show InChI InChI=1S/C30H39N5O6/c1-19-17-20-9-10-30(2,3)21(20)18-23(19)41-24-8-7-22(40-24)26(36)32-25-27(37-4)33-29(34-28(25)38-5)31-11-6-12-35-13-15-39-16-14-35/h7-8,17-18H,6,9-16H2,1-5H3,(H,32,36)(H,31,33,34) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity to human recombinant GnRH receptor |
J Med Chem 49: 3362-7 (2006)
Article DOI: 10.1021/jm060012g
BindingDB Entry DOI: 10.7270/Q2F76C54 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50329836

(2-Chloro-5-[(1H-imidazo[4,5-c]quinolin-2-ylsulfany...)Show SMILES NS(=O)(=O)c1cc(ccc1Cl)C(=O)CSc1nc2c(cnc3ccccc23)[nH]1 Show InChI InChI=1S/C18H13ClN4O3S2/c19-12-6-5-10(7-16(12)28(20,25)26)15(24)9-27-18-22-14-8-21-13-4-2-1-3-11(13)17(14)23-18/h1-8H,9H2,(H,22,23)(H2,20,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121472
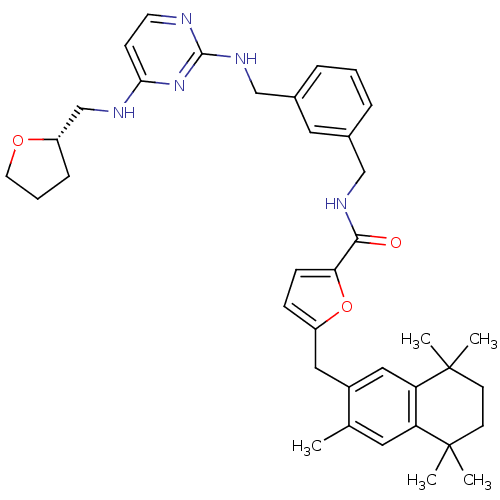
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3nccc(NC[C@@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-22-26-8-6-9-27(19-26)23-42-36-39-16-13-34(43-36)40-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053067
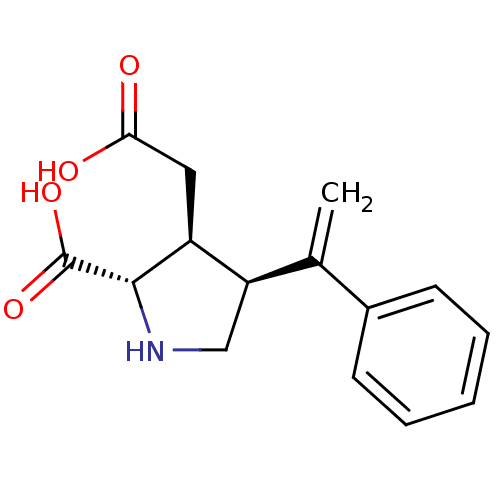
((2S,3S,4S)-3-Carboxymethyl-4-(1-phenyl-vinyl)-pyrr...)Show SMILES OC(=O)C[C@H]1[C@H](CN[C@@H]1C(O)=O)C(=C)c1ccccc1 Show InChI InChI=1S/C15H17NO4/c1-9(10-5-3-2-4-6-10)12-8-16-14(15(19)20)11(12)7-13(17)18/h2-6,11-12,14,16H,1,7-8H2,(H,17,18)(H,19,20)/t11-,12+,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053071
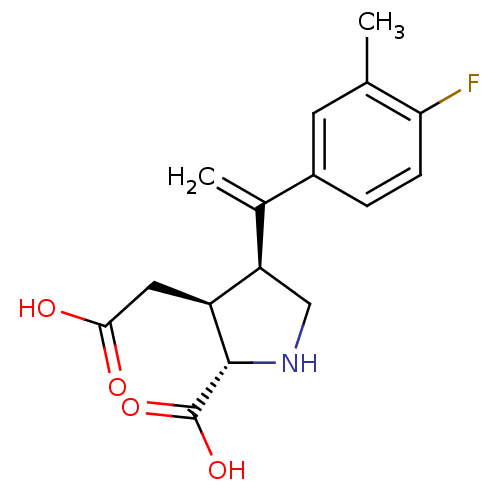
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-fluoro-3-methyl...)Show SMILES Cc1cc(ccc1F)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C16H18FNO4/c1-8-5-10(3-4-13(8)17)9(2)12-7-18-15(16(21)22)11(12)6-14(19)20/h3-5,11-12,15,18H,2,6-7H2,1H3,(H,19,20)(H,21,22)/t11-,12+,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053075
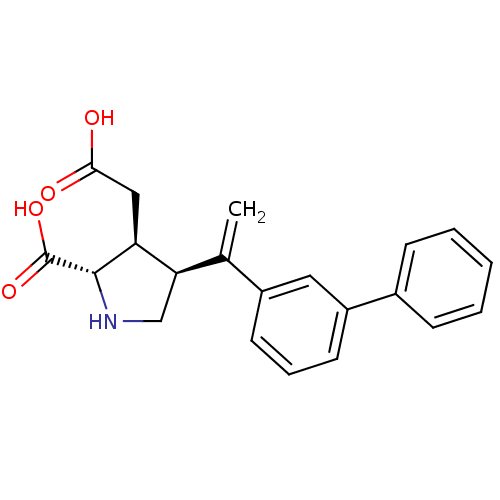
((2S,3S,4S)-4-(1-Biphenyl-3-yl-vinyl)-3-carboxymeth...)Show SMILES OC(=O)C[C@H]1[C@H](CN[C@@H]1C(O)=O)C(=C)c1cccc(c1)-c1ccccc1 Show InChI InChI=1S/C21H21NO4/c1-13(18-12-22-20(21(25)26)17(18)11-19(23)24)15-8-5-9-16(10-15)14-6-3-2-4-7-14/h2-10,17-18,20,22H,1,11-12H2,(H,23,24)(H,25,26)/t17-,18+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053083
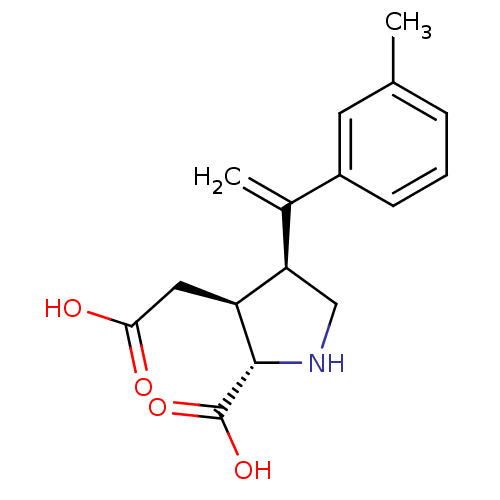
((2S,3S,4S)-3-Carboxymethyl-4-(1-m-tolyl-vinyl)-pyr...)Show SMILES Cc1cccc(c1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C16H19NO4/c1-9-4-3-5-11(6-9)10(2)13-8-17-15(16(20)21)12(13)7-14(18)19/h3-6,12-13,15,17H,2,7-8H2,1H3,(H,18,19)(H,20,21)/t12-,13+,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053080
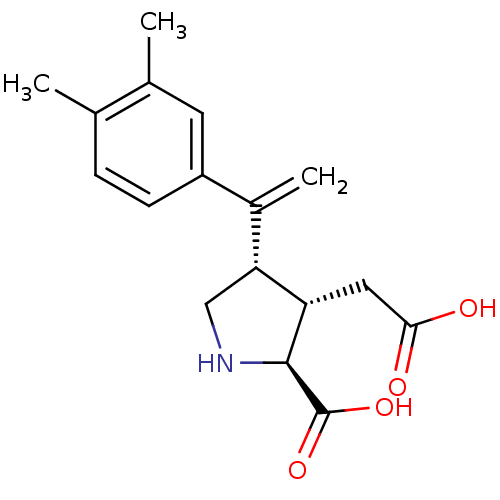
((2S,3S,4S)-3-Carboxymethyl-4-[1-(3,4-dimethyl-phen...)Show SMILES Cc1ccc(cc1C)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C17H21NO4/c1-9-4-5-12(6-10(9)2)11(3)14-8-18-16(17(21)22)13(14)7-15(19)20/h4-6,13-14,16,18H,3,7-8H2,1-2H3,(H,19,20)(H,21,22)/t13-,14+,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50596811

(CHEMBL5181231)Show SMILES NS(=O)(=O)c1ccc(NC(=O)Cn2cc(\C=C\C(=O)c3cccc(c3)C(F)(F)F)c3ccccc23)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121475
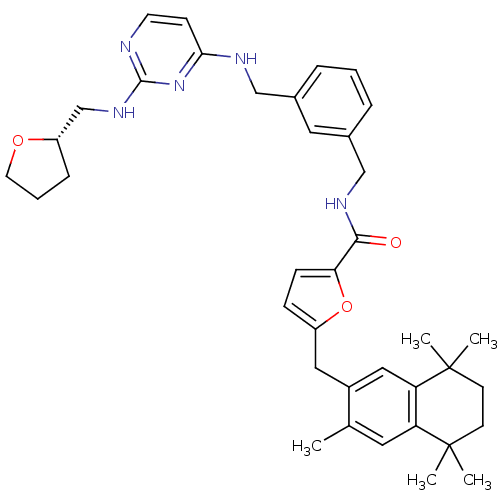
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3ccnc(NC[C@@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-23-27-9-6-8-26(19-27)22-40-34-13-16-39-36(43-34)42-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053088
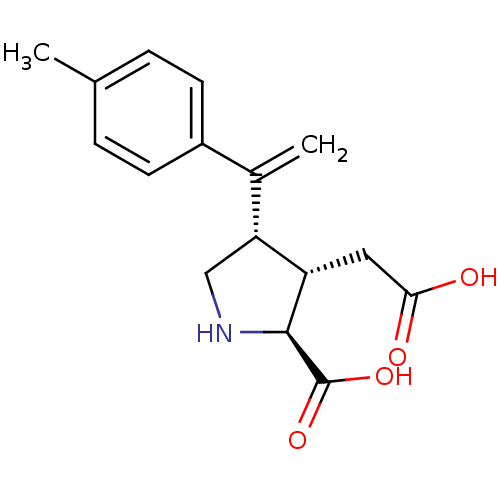
((2S,3S,4S)-3-Carboxymethyl-4-(1-p-tolyl-vinyl)-pyr...)Show SMILES Cc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C16H19NO4/c1-9-3-5-11(6-4-9)10(2)13-8-17-15(16(20)21)12(13)7-14(18)19/h3-6,12-13,15,17H,2,7-8H2,1H3,(H,18,19)(H,20,21)/t12-,13+,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053087
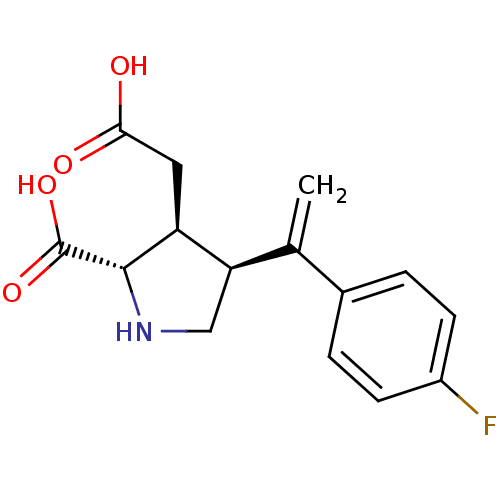
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-fluoro-phenyl)-...)Show SMILES OC(=O)C[C@H]1[C@H](CN[C@@H]1C(O)=O)C(=C)c1ccc(F)cc1 Show InChI InChI=1S/C15H16FNO4/c1-8(9-2-4-10(16)5-3-9)12-7-17-14(15(20)21)11(12)6-13(18)19/h2-5,11-12,14,17H,1,6-7H2,(H,18,19)(H,20,21)/t11-,12+,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053064
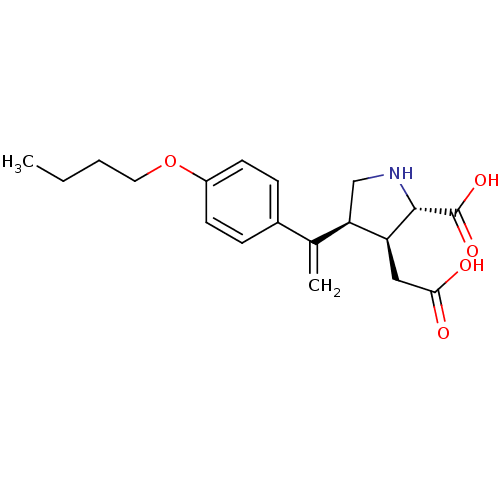
((2S,3S,4S)-4-[1-(4-Butoxy-phenyl)-vinyl]-3-carboxy...)Show SMILES CCCCOc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C19H25NO5/c1-3-4-9-25-14-7-5-13(6-8-14)12(2)16-11-20-18(19(23)24)15(16)10-17(21)22/h5-8,15-16,18,20H,2-4,9-11H2,1H3,(H,21,22)(H,23,24)/t15-,16+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM50060630
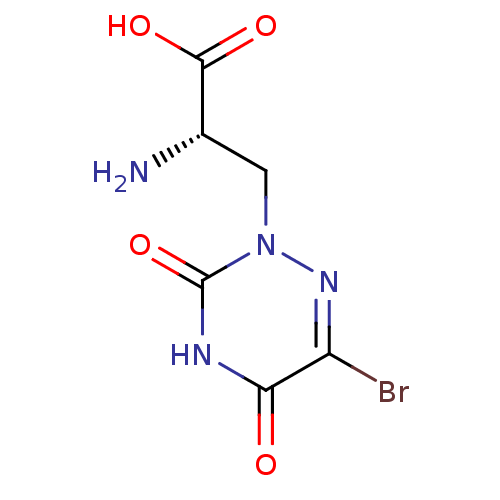
((S)-2-Amino-3-(6-bromo-3,5-dioxo-4,5-dihydro-3H-[1...)Show InChI InChI=1S/C6H7BrN4O4/c7-3-4(12)9-6(15)11(10-3)1-2(8)5(13)14/h2H,1,8H2,(H,13,14)(H,9,12,15)/t2-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Displacement of [3H]kainate from human Ionotropic glutamate receptor ionotropic kainate 1 expressed in HEK293 cells |
J Med Chem 40: 3645-50 (1997)
Article DOI: 10.1021/jm9702387
BindingDB Entry DOI: 10.7270/Q28G8MCB |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121461
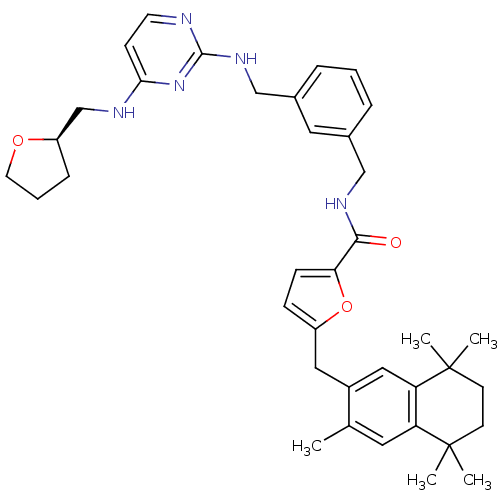
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3nccc(NC[C@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-22-26-8-6-9-27(19-26)23-42-36-39-16-13-34(43-36)40-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50031704
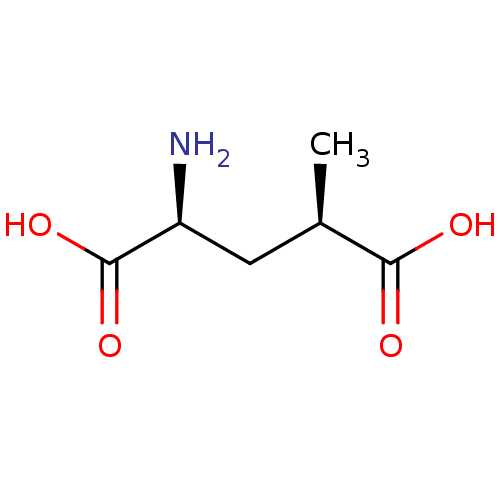
((2S,4R)-2-Amino-4-methyl-pentanedioic acid | (2S,4...)Show InChI InChI=1S/C6H11NO4/c1-3(5(8)9)2-4(7)6(10)11/h3-4H,2,7H2,1H3,(H,8,9)(H,10,11)/t3-,4+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly, S.A.
Curated by ChEMBL
| Assay Description
Binding affinity against human ionotropic glutamate receptor kainate 1 in HK293 cells using [3H]-kainate as radioligand |
J Med Chem 43: 1958-68 (2000)
BindingDB Entry DOI: 10.7270/Q2FX78QB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053063

((2S,3S,4S)-3-Carboxymethyl-4-[1-(3-methoxy-phenyl)...)Show SMILES COc1cccc(c1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C16H19NO5/c1-9(10-4-3-5-11(6-10)22-2)13-8-17-15(16(20)21)12(13)7-14(18)19/h3-6,12-13,15,17H,1,7-8H2,2H3,(H,18,19)(H,20,21)/t12-,13+,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053068
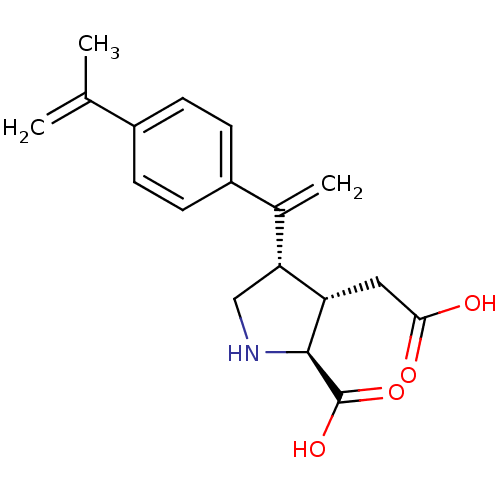
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-isopropenyl-phe...)Show SMILES CC(=C)c1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C18H21NO4/c1-10(2)12-4-6-13(7-5-12)11(3)15-9-19-17(18(22)23)14(15)8-16(20)21/h4-7,14-15,17,19H,1,3,8-9H2,2H3,(H,20,21)(H,22,23)/t14-,15+,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053059

((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-propyl-phenyl)-...)Show SMILES CCCc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C18H23NO4/c1-3-4-12-5-7-13(8-6-12)11(2)15-10-19-17(18(22)23)14(15)9-16(20)21/h5-8,14-15,17,19H,2-4,9-10H2,1H3,(H,20,21)(H,22,23)/t14-,15+,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50060628

((S)-2-Amino-3-(6-chloro-3,5-dioxo-4,5-dihydro-3H-[...)Show InChI InChI=1S/C6H7ClN4O4/c7-3-4(12)9-6(15)11(10-3)1-2(8)5(13)14/h2H,1,8H2,(H,13,14)(H,9,12,15)/t2-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Displacement of [3H]AMPA from human Ionotropic glutamate receptor AMPA 4 expressed in HEK293 cells |
J Med Chem 40: 3645-50 (1997)
Article DOI: 10.1021/jm9702387
BindingDB Entry DOI: 10.7270/Q28G8MCB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50002369
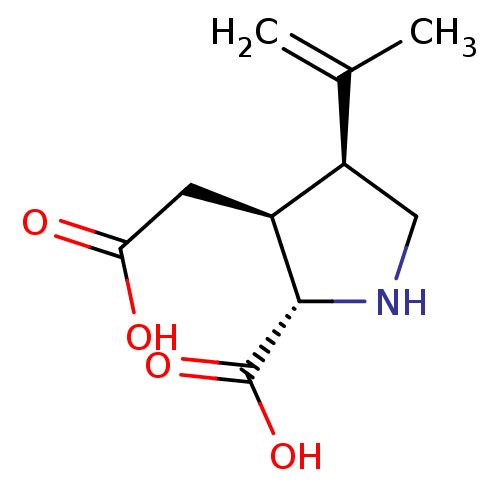
((2S-(2alpha,3beta,4beta))-2-carboxy-4-(1-methyleth...)Show SMILES CC(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O |r| Show InChI InChI=1S/C10H15NO4/c1-5(2)7-4-11-9(10(14)15)6(7)3-8(12)13/h6-7,9,11H,1,3-4H2,2H3,(H,12,13)(H,14,15)/t6-,7+,9-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053061
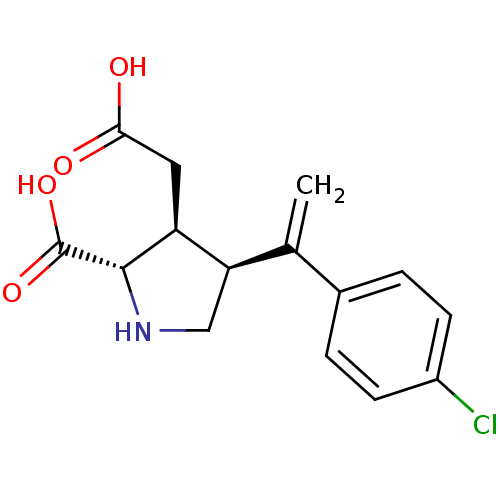
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-chloro-phenyl)-...)Show SMILES OC(=O)C[C@H]1[C@H](CN[C@@H]1C(O)=O)C(=C)c1ccc(Cl)cc1 Show InChI InChI=1S/C15H16ClNO4/c1-8(9-2-4-10(16)5-3-9)12-7-17-14(15(20)21)11(12)6-13(18)19/h2-5,11-12,14,17H,1,6-7H2,(H,18,19)(H,20,21)/t11-,12+,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053073

((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-methoxy-phenyl)...)Show SMILES COc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C16H19NO5/c1-9(10-3-5-11(22-2)6-4-10)13-8-17-15(16(20)21)12(13)7-14(18)19/h3-6,12-13,15,17H,1,7-8H2,2H3,(H,18,19)(H,20,21)/t12-,13+,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121484
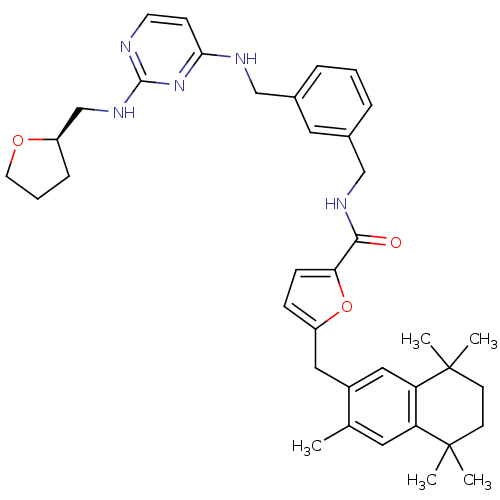
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3ccnc(NC[C@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-23-27-9-6-8-26(19-27)22-40-34-13-16-39-36(43-34)42-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
Competitive inhibition of GnRH-stimulated extracellular acidification in cells expressing recombinant rat GnRH receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121484

(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3ccnc(NC[C@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-23-27-9-6-8-26(19-27)22-40-34-13-16-39-36(43-34)42-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053082
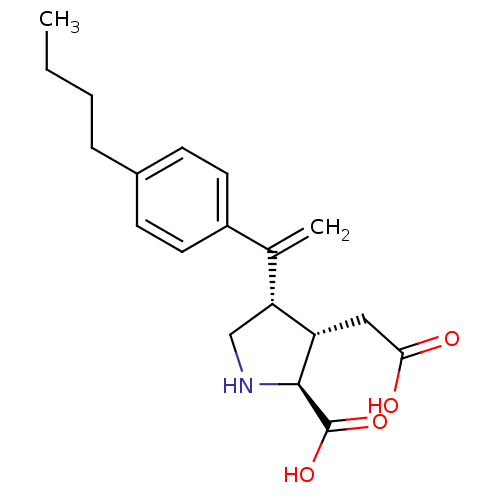
((2S,3S,4S)-4-[1-(4-Butyl-phenyl)-vinyl]-3-carboxym...)Show SMILES CCCCc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C19H25NO4/c1-3-4-5-13-6-8-14(9-7-13)12(2)16-11-20-18(19(23)24)15(16)10-17(21)22/h6-9,15-16,18,20H,2-5,10-11H2,1H3,(H,21,22)(H,23,24)/t15-,16+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50596809

(CHEMBL5201409)Show SMILES CCN(C1CC(C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O)S(=O)(=O)c1cccc2cccnc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 5.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121464
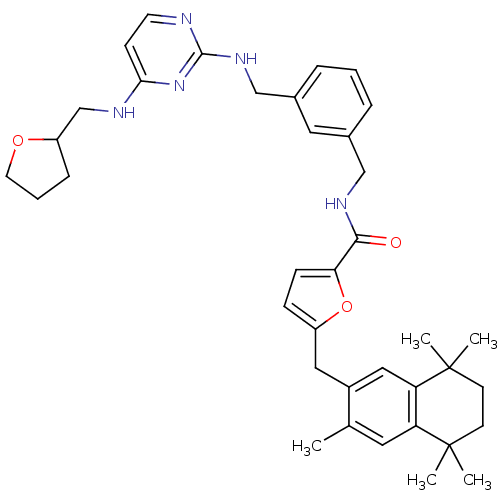
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3nccc(NCC4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-22-26-8-6-9-27(19-26)23-42-36-39-16-13-34(43-36)40-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053065
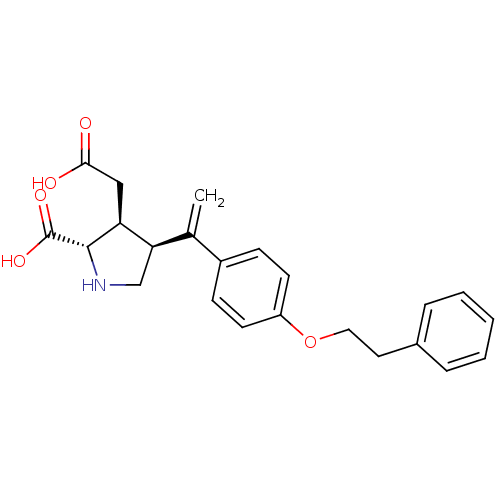
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-phenethyloxy-ph...)Show SMILES OC(=O)C[C@H]1[C@H](CN[C@@H]1C(O)=O)C(=C)c1ccc(OCCc2ccccc2)cc1 Show InChI InChI=1S/C23H25NO5/c1-15(20-14-24-22(23(27)28)19(20)13-21(25)26)17-7-9-18(10-8-17)29-12-11-16-5-3-2-4-6-16/h2-10,19-20,22,24H,1,11-14H2,(H,25,26)(H,27,28)/t19-,20+,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50596777

(CHEMBL5197564)Show SMILES NS(=O)(=O)c1ccc(NC(=O)c2cc(nc3ccccc23)-c2ccc3OCOc3c2)cc1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50596801
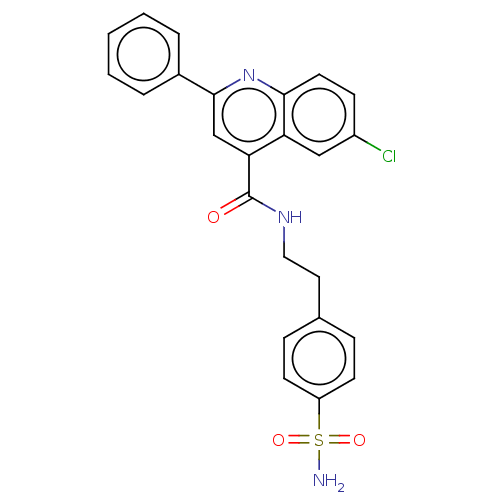
(CHEMBL5169617)Show SMILES NS(=O)(=O)c1ccc(CCNC(=O)c2cc(nc3ccc(Cl)cc23)-c2ccccc2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 6.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50596800

(CHEMBL5204736)Show SMILES NS(=O)(=O)c1ccc(CCNC(=O)c2cc(nc3ccccc23)-c2cccc(c2)C(F)(F)F)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 6.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50088223

(CHEMBL61751 | E-2-Amino-4-(3-naphthalen-1-yl-allyl...)Show SMILES N[C@@H](C[C@@H](C\C=C\c1cccc2ccccc12)C(O)=O)C(O)=O Show InChI InChI=1S/C18H19NO4/c19-16(18(22)23)11-14(17(20)21)9-4-8-13-7-3-6-12-5-1-2-10-15(12)13/h1-8,10,14,16H,9,11,19H2,(H,20,21)(H,22,23)/b8-4+/t14-,16+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly, S.A.
Curated by ChEMBL
| Assay Description
Binding affinity against human ionotropic glutamate receptor kainate 1 in HK293 cells using [3H]-kainate as radioligand |
J Med Chem 43: 1958-68 (2000)
BindingDB Entry DOI: 10.7270/Q2FX78QB |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121462
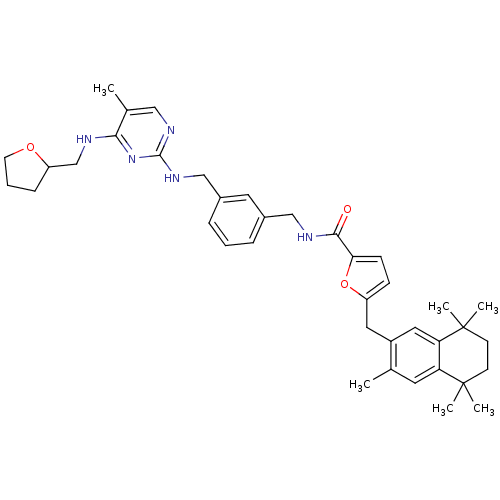
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cnc(NCc2cccc(CNC(=O)c3ccc(Cc4cc5c(cc4C)C(C)(C)CCC5(C)C)o3)c2)nc1NCC1CCCO1 Show InChI InChI=1S/C39H49N5O3/c1-25-17-32-33(39(5,6)15-14-38(32,3)4)20-29(25)19-30-12-13-34(47-30)36(45)41-22-27-9-7-10-28(18-27)23-43-37-42-21-26(2)35(44-37)40-24-31-11-8-16-46-31/h7,9-10,12-13,17-18,20-21,31H,8,11,14-16,19,22-24H2,1-6H3,(H,41,45)(H2,40,42,43,44) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM50060628

((S)-2-Amino-3-(6-chloro-3,5-dioxo-4,5-dihydro-3H-[...)Show InChI InChI=1S/C6H7ClN4O4/c7-3-4(12)9-6(15)11(10-3)1-2(8)5(13)14/h2H,1,8H2,(H,13,14)(H,9,12,15)/t2-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Displacement of [3H]AMPA from human Ionotropic glutamate receptor AMPA 1 expressed in HEK293 cells |
J Med Chem 40: 3645-50 (1997)
Article DOI: 10.1021/jm9702387
BindingDB Entry DOI: 10.7270/Q28G8MCB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50596777

(CHEMBL5197564)Show SMILES NS(=O)(=O)c1ccc(NC(=O)c2cc(nc3ccccc23)-c2ccc3OCOc3c2)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50121466
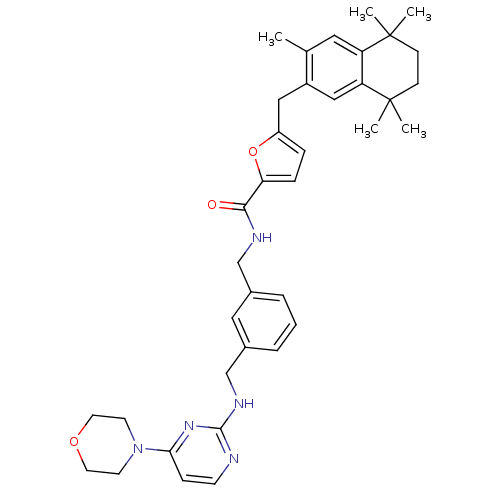
(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3nccc(n3)N3CCOCC3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C37H45N5O3/c1-25-19-30-31(37(4,5)13-12-36(30,2)3)22-28(25)21-29-9-10-32(45-29)34(43)39-23-26-7-6-8-27(20-26)24-40-35-38-14-11-33(41-35)42-15-17-44-18-16-42/h6-11,14,19-20,22H,12-13,15-18,21,23-24H2,1-5H3,(H,39,43)(H,38,40,41) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards rat gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Rattus norvegicus) | BDBM50053089
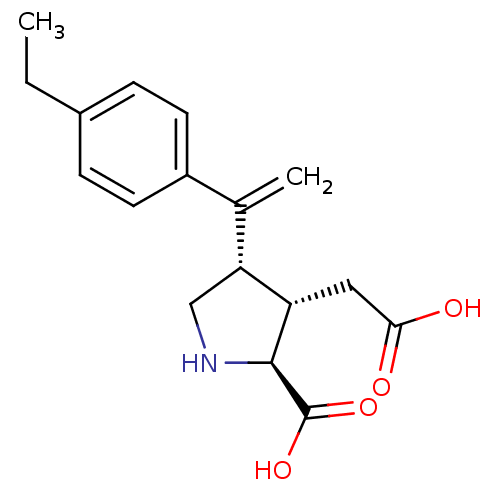
((2S,3S,4S)-3-Carboxymethyl-4-[1-(4-ethyl-phenyl)-v...)Show SMILES CCc1ccc(cc1)C(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O Show InChI InChI=1S/C17H21NO4/c1-3-11-4-6-12(7-5-11)10(2)14-9-18-16(17(21)22)13(14)8-15(19)20/h4-7,13-14,16,18H,2-3,8-9H2,1H3,(H,19,20)(H,21,22)/t13-,14+,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-kainate binding at rat forebrain ionotropic glutamate receptor kainate 2 |
J Med Chem 39: 3617-24 (1996)
Article DOI: 10.1021/jm960155a
BindingDB Entry DOI: 10.7270/Q2WW7GRZ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50060630

((S)-2-Amino-3-(6-bromo-3,5-dioxo-4,5-dihydro-3H-[1...)Show InChI InChI=1S/C6H7BrN4O4/c7-3-4(12)9-6(15)11(10-3)1-2(8)5(13)14/h2H,1,8H2,(H,13,14)(H,9,12,15)/t2-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Displacement of [3H]AMPA from human Ionotropic glutamate receptor AMPA 4 expressed in HEK293 cells |
J Med Chem 40: 3645-50 (1997)
Article DOI: 10.1021/jm9702387
BindingDB Entry DOI: 10.7270/Q28G8MCB |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50121472

(5-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Cc1ccc(o1)C(=O)NCc1cccc(CNc3nccc(NC[C@@H]4CCCO4)n3)c1)C(C)(C)CCC2(C)C Show InChI InChI=1S/C38H47N5O3/c1-25-18-31-32(38(4,5)15-14-37(31,2)3)21-28(25)20-29-11-12-33(46-29)35(44)41-22-26-8-6-9-27(19-26)23-42-36-39-16-13-34(43-36)40-24-30-10-7-17-45-30/h6,8-9,11-13,16,18-19,21,30H,7,10,14-15,17,20,22-24H2,1-5H3,(H,41,44)(H2,39,40,42,43)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development-La Jolla/Agouron Pharmaceuticals
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards human gonadotropin-releasing hormone receptor |
Bioorg Med Chem Lett 12: 3635-9 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8DXZ |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50088197
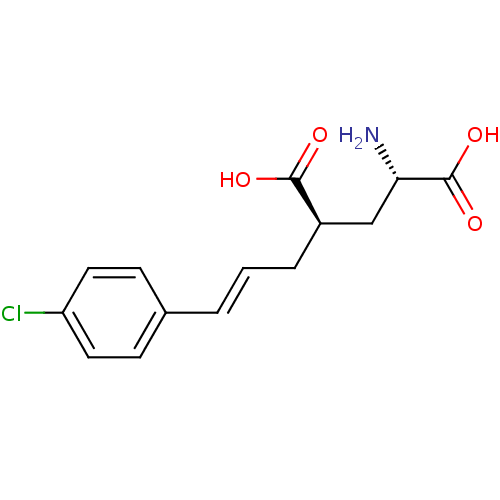
(CHEMBL58590 | E-2-Amino-4-[3-(4-chloro-phenyl)-all...)Show SMILES N[C@@H](C[C@@H](C\C=C\c1ccc(Cl)cc1)C(O)=O)C(O)=O Show InChI InChI=1S/C14H16ClNO4/c15-11-6-4-9(5-7-11)2-1-3-10(13(17)18)8-12(16)14(19)20/h1-2,4-7,10,12H,3,8,16H2,(H,17,18)(H,19,20)/b2-1+/t10-,12+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly, S.A.
Curated by ChEMBL
| Assay Description
Binding affinity against human ionotropic glutamate receptor kainate 1 in HK293 cells using [3H]-kainate as radioligand |
J Med Chem 43: 1958-68 (2000)
BindingDB Entry DOI: 10.7270/Q2FX78QB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM50002369

((2S-(2alpha,3beta,4beta))-2-carboxy-4-(1-methyleth...)Show SMILES CC(=C)[C@H]1CN[C@@H]([C@H]1CC(O)=O)C(O)=O |r| Show InChI InChI=1S/C10H15NO4/c1-5(2)7-4-11-9(10(14)15)6(7)3-8(12)13/h6-7,9,11H,1,3-4H2,2H3,(H,12,13)(H,14,15)/t6-,7+,9-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly, S.A.
Curated by ChEMBL
| Assay Description
Ability of the compound to bind to Ionotropic glutamate receptor kainate (kainate 2) was evaluated. |
J Med Chem 43: 1958-68 (2000)
BindingDB Entry DOI: 10.7270/Q2FX78QB |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 1
(Homo sapiens (Human)) | BDBM50088197

(CHEMBL58590 | E-2-Amino-4-[3-(4-chloro-phenyl)-all...)Show SMILES N[C@@H](C[C@@H](C\C=C\c1ccc(Cl)cc1)C(O)=O)C(O)=O Show InChI InChI=1S/C14H16ClNO4/c15-11-6-4-9(5-7-11)2-1-3-10(13(17)18)8-12(16)14(19)20/h1-2,4-7,10,12H,3,8,16H2,(H,17,18)(H,19,20)/b2-1+/t10-,12+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly, S.A.
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against Ionotropic glutamate receptor ionotropic kainate 1 in HK293 cells using [3H]-kainate as radioligand;... |
J Med Chem 43: 1958-68 (2000)
BindingDB Entry DOI: 10.7270/Q2FX78QB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50596812
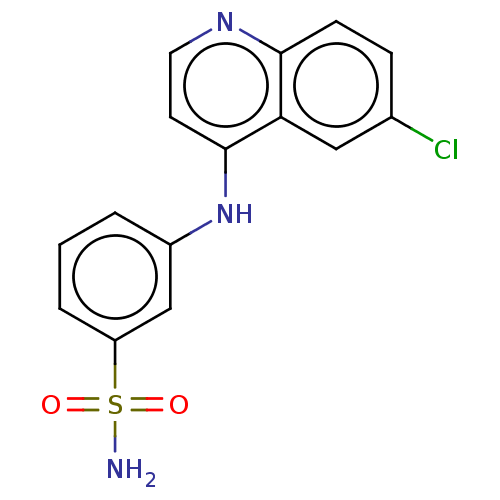
(CHEMBL5178258) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 8.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50517111
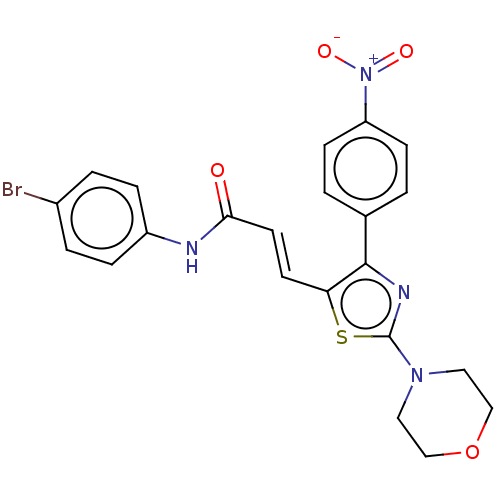
(CHEMBL4466660)Show SMILES [O-][N+](=O)c1ccc(cc1)-c1nc(sc1\C=C\C(=O)Nc1ccc(Br)cc1)N1CCOCC1 Show InChI InChI=1S/C22H19BrN4O4S/c23-16-3-5-17(6-4-16)24-20(28)10-9-19-21(15-1-7-18(8-2-15)27(29)30)25-22(32-19)26-11-13-31-14-12-26/h1-10H,11-14H2,(H,24,28)/b10-9+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 8.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114247
BindingDB Entry DOI: 10.7270/Q2MC941H |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data