 Found 290 hits with Last Name = 'huband' and Initial = 'm'
Found 290 hits with Last Name = 'huband' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human norepinephrine transporter |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Macrophage metalloelastase
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.35E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP12 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP13 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP9 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP14 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP3 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrilysin
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP7 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP2 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP1 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human TACE |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-26
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP26 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-25
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP25 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-24
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP24 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-20
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP20 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-16
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP16 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-15
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP15 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Stromelysin-2
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP10 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Neutrophil collagenase
(Homo sapiens (Human)) | BDBM50365344

(CHEMBL1956115)Show SMILES C[C@@](CCc1ccc(cc1)-c1ccccc1)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C18H21NO4S/c1-18(17(20)19-21,24(2,22)23)13-12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,21H,12-13H2,1-2H3,(H,19,20)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human MMP8 |
J Med Chem 55: 914-23 (2012)
Article DOI: 10.1021/jm2014748
BindingDB Entry DOI: 10.7270/Q2VX0H0W |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205450
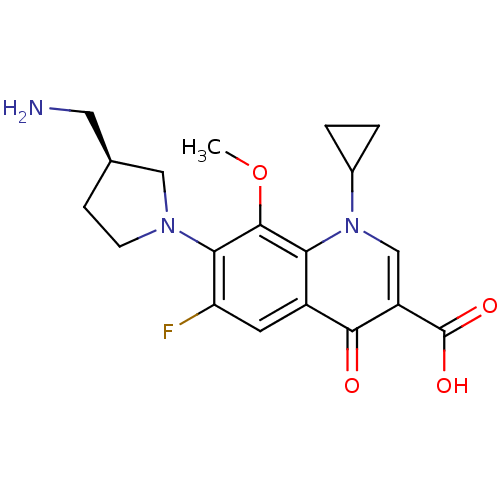
((S)-7-(3-(aminomethyl)pyrrolidin-1-yl)-1-cycloprop...)Show SMILES COc1c(N2CC[C@@H](CN)C2)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C19H22FN3O4/c1-27-18-15-12(6-14(20)16(18)22-5-4-10(7-21)8-22)17(24)13(19(25)26)9-23(15)11-2-3-11/h6,9-11H,2-5,7-8,21H2,1H3,(H,25,26)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205452
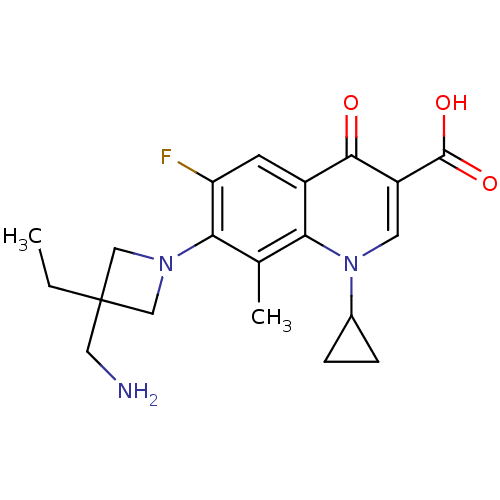
(7-(3-(aminomethyl)-3-ethylazetidin-1-yl)-1-cyclopr...)Show SMILES CCC1(CN)CN(C1)c1c(F)cc2c(c1C)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C20H24FN3O3/c1-3-20(8-22)9-23(10-20)17-11(2)16-13(6-15(17)21)18(25)14(19(26)27)7-24(16)12-4-5-12/h6-7,12H,3-5,8-10,22H2,1-2H3,(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205465
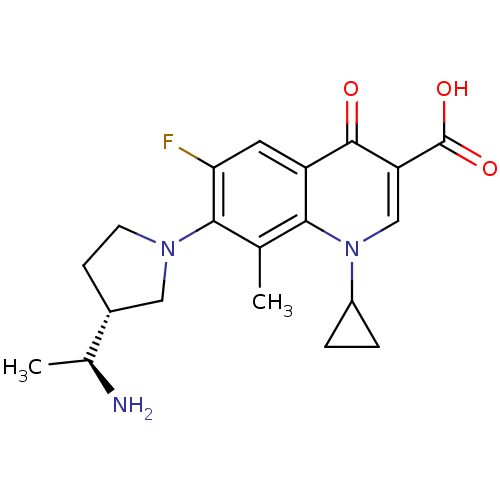
(7-((R)-3-((S)-1-aminoethyl)pyrrolidin-1-yl)-1-cycl...)Show SMILES C[C@H](N)[C@@H]1CCN(C1)c1c(F)cc2c(c1C)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C20H24FN3O3/c1-10-17-14(19(25)15(20(26)27)9-24(17)13-3-4-13)7-16(21)18(10)23-6-5-12(8-23)11(2)22/h7,9,11-13H,3-6,8,22H2,1-2H3,(H,26,27)/t11-,12+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50116067
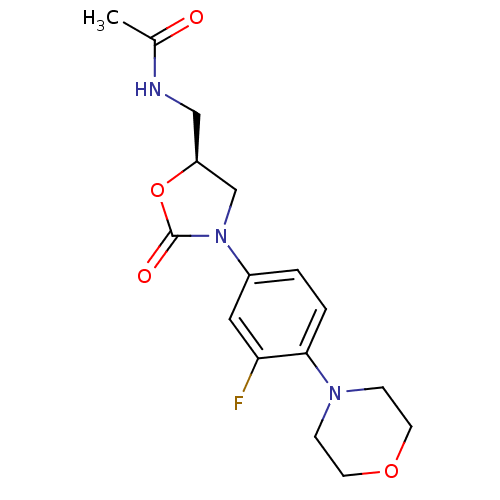
((Linezolid)N-[3-(3-Fluoro-4-morpholin-4-yl-phenyl)...)Show SMILES CC(=O)NC[C@H]1CN(C(=O)O1)c1ccc(N2CCOCC2)c(F)c1 |r| Show InChI InChI=1S/C16H20FN3O4/c1-11(21)18-9-13-10-20(16(22)24-13)12-2-3-15(14(17)8-12)19-4-6-23-7-5-19/h2-3,8,13H,4-7,9-10H2,1H3,(H,18,21)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50226485

((R)-3-(3-fluoro-4-(tetrahydro-2H-pyran-4-yl)phenyl...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1ccc(C2CCOCC2)c(F)c1 Show InChI InChI=1S/C15H17FN2O4/c16-12-7-10(18-8-13(14(17)19)22-15(18)20)1-2-11(12)9-3-5-21-6-4-9/h1-2,7,9,13H,3-6,8H2,(H2,17,19)/t13-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205451
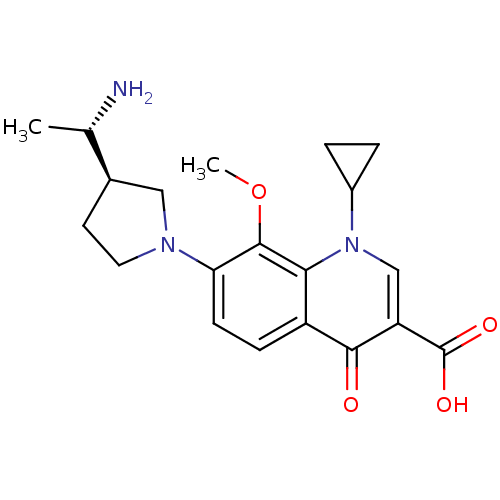
(7-((R)-3-((S)-1-aminoethyl)pyrrolidin-1-yl)-1-cycl...)Show SMILES COc1c(ccc2c1n(cc(C(O)=O)c2=O)C1CC1)N1CC[C@H](C1)[C@H](C)N Show InChI InChI=1S/C20H25N3O4/c1-11(21)12-7-8-22(9-12)16-6-5-14-17(19(16)27-2)23(13-3-4-13)10-15(18(14)24)20(25)26/h5-6,10-13H,3-4,7-9,21H2,1-2H3,(H,25,26)/t11-,12+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50226479
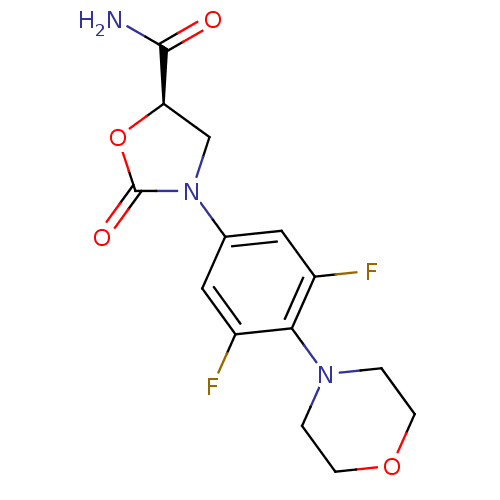
((R)-3-(3,5-difluoro-4-morpholinophenyl)-2-oxooxazo...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1cc(F)c(N2CCOCC2)c(F)c1 Show InChI InChI=1S/C14H15F2N3O4/c15-9-5-8(19-7-11(13(17)20)23-14(19)21)6-10(16)12(9)18-1-3-22-4-2-18/h5-6,11H,1-4,7H2,(H2,17,20)/t11-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205466
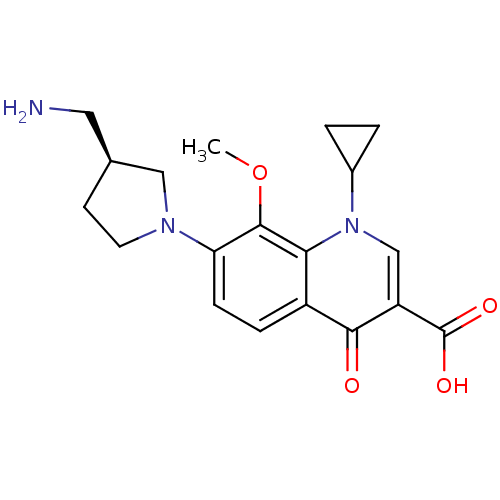
((S)-7-(3-(aminomethyl)pyrrolidin-1-yl)-1-cycloprop...)Show SMILES COc1c(ccc2c1n(cc(C(O)=O)c2=O)C1CC1)N1CC[C@@H](CN)C1 Show InChI InChI=1S/C19H23N3O4/c1-26-18-15(21-7-6-11(8-20)9-21)5-4-13-16(18)22(12-2-3-12)10-14(17(13)23)19(24)25/h4-5,10-12H,2-3,6-9,20H2,1H3,(H,24,25)/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205457

(7-((R)-3-((S)-1-(2-cyanoethylamino)ethyl)pyrrolidi...)Show SMILES C[C@H](NCCC#N)[C@@H]1CCN(C1)c1c(F)cc2c(c1C)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C23H27FN4O3/c1-13-20-17(22(29)18(23(30)31)12-28(20)16-4-5-16)10-19(24)21(13)27-9-6-15(11-27)14(2)26-8-3-7-25/h10,12,14-16,26H,3-6,8-9,11H2,1-2H3,(H,30,31)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205453
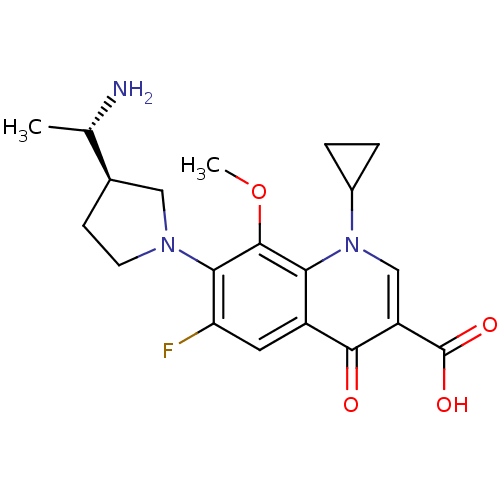
(7-((R)-3-((S)-1-aminoethyl)pyrrolidin-1-yl)-1-cycl...)Show SMILES COc1c(N2CC[C@H](C2)[C@H](C)N)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C20H24FN3O4/c1-10(22)11-5-6-23(8-11)17-15(21)7-13-16(19(17)28-2)24(12-3-4-12)9-14(18(13)25)20(26)27/h7,9-12H,3-6,8,22H2,1-2H3,(H,26,27)/t10-,11+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205446
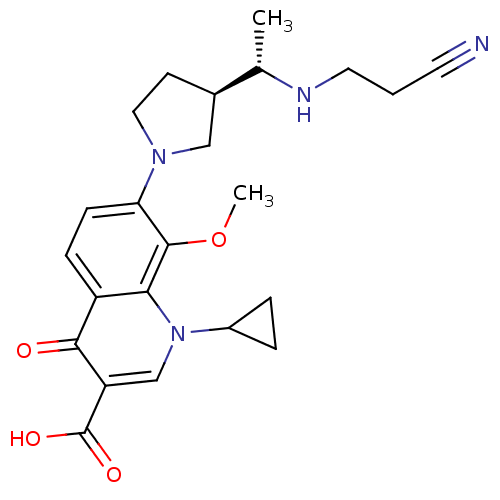
(7-((R)-3-((S)-1-(2-cyanoethylamino)ethyl)pyrrolidi...)Show SMILES COc1c(ccc2c1n(cc(C(O)=O)c2=O)C1CC1)N1CC[C@H](C1)[C@H](C)NCCC#N Show InChI InChI=1S/C23H28N4O4/c1-14(25-10-3-9-24)15-8-11-26(12-15)19-7-6-17-20(22(19)31-2)27(16-4-5-16)13-18(21(17)28)23(29)30/h6-7,13-16,25H,3-5,8,10-12H2,1-2H3,(H,29,30)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205461
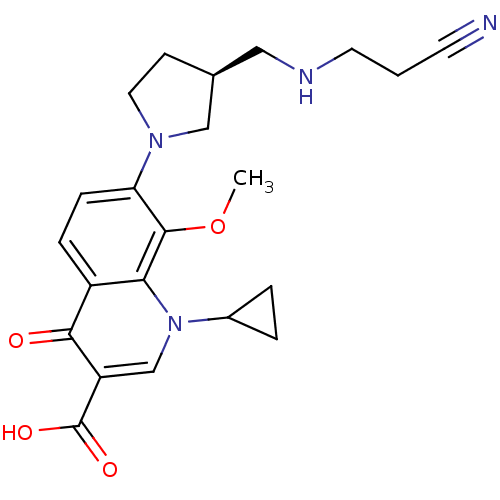
((S)-7-(3-((2-cyanoethylamino)methyl)pyrrolidin-1-y...)Show SMILES COc1c(ccc2c1n(cc(C(O)=O)c2=O)C1CC1)N1CC[C@@H](CNCCC#N)C1 Show InChI InChI=1S/C22H26N4O4/c1-30-21-18(25-10-7-14(12-25)11-24-9-2-8-23)6-5-16-19(21)26(15-3-4-15)13-17(20(16)27)22(28)29/h5-6,13-15,24H,2-4,7,9-12H2,1H3,(H,28,29)/t14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM21690
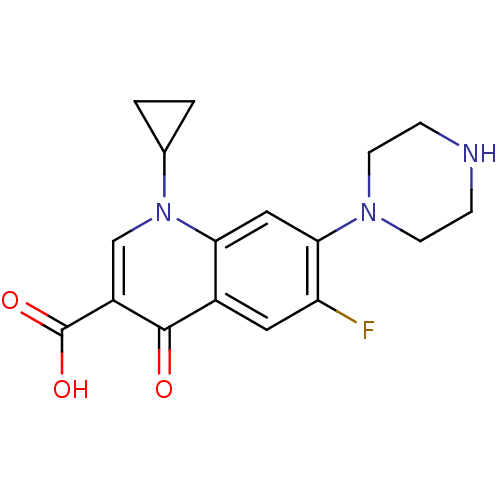
(1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,...)Show InChI InChI=1S/C17H18FN3O3/c18-13-7-11-14(8-15(13)20-5-3-19-4-6-20)21(10-1-2-10)9-12(16(11)22)17(23)24/h7-10,19H,1-6H2,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205447

(10-((R)-3-((S)-1-aminoethyl)pyrrolidin-1-yl)-9-flu...)Show SMILES C[C@H](N)[C@@H]1CCN(C1)c1c(F)cc2c3c1OCC(C)n3cc(C(O)=O)c2=O |w:17.19| Show InChI InChI=1S/C19H22FN3O4/c1-9-8-27-18-15-12(17(24)13(19(25)26)7-23(9)15)5-14(20)16(18)22-4-3-11(6-22)10(2)21/h5,7,9-11H,3-4,6,8,21H2,1-2H3,(H,25,26)/t9?,10-,11+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50226484
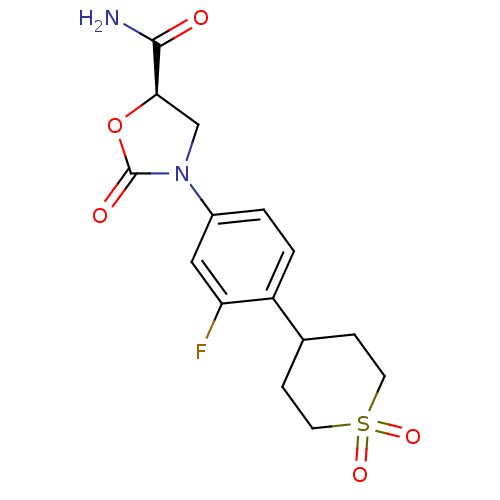
((5R)-3-[4-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1ccc(C2CCS(=O)(=O)CC2)c(F)c1 Show InChI InChI=1S/C15H17FN2O5S/c16-12-7-10(18-8-13(14(17)19)23-15(18)20)1-2-11(12)9-3-5-24(21,22)6-4-9/h1-2,7,9,13H,3-6,8H2,(H2,17,19)/t13-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.05E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205459
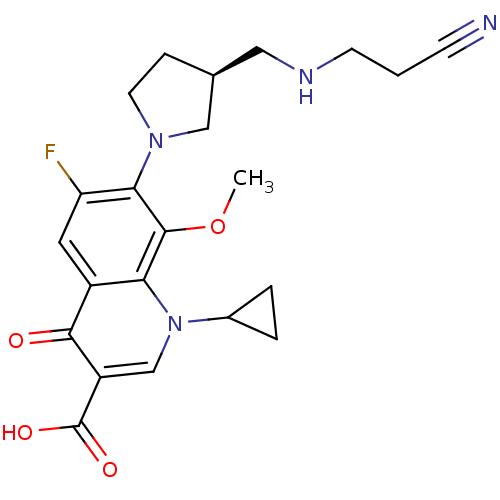
((S)-7-(3-(aminomethyl)pyrrolidin-1-yl)-1-cycloprop...)Show SMILES COc1c(N2CC[C@@H](CNCCC#N)C2)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C22H25FN4O4/c1-31-21-18-15(20(28)16(22(29)30)12-27(18)14-3-4-14)9-17(23)19(21)26-8-5-13(11-26)10-25-7-2-6-24/h9,12-14,25H,2-5,7-8,10-11H2,1H3,(H,29,30)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.06E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
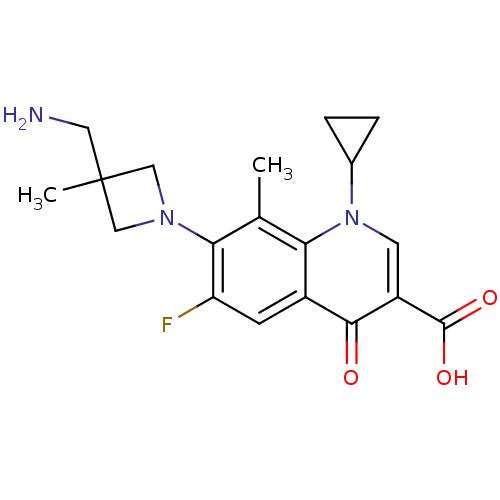
(Homo sapiens (Human)) | BDBM50205455
(7-(3-(aminomethyl)-3-methylazetidin-1-yl)-1-cyclop...)Show SMILES Cc1c(N2CC(C)(CN)C2)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C19H22FN3O3/c1-10-15-12(5-14(20)16(10)22-8-19(2,7-21)9-22)17(24)13(18(25)26)6-23(15)11-3-4-11/h5-6,11H,3-4,7-9,21H2,1-2H3,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.12E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
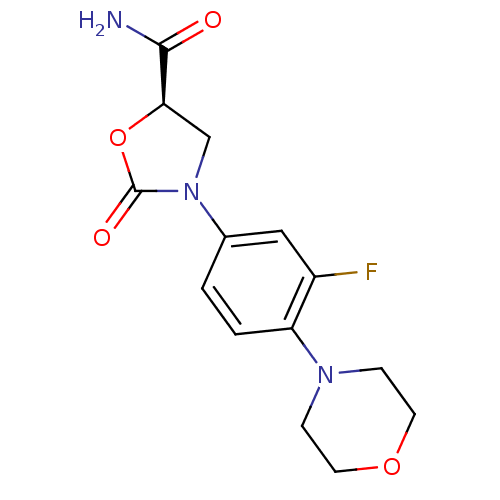
(Homo sapiens (Human)) | BDBM50226480
((R)-3-(3-fluoro-4-morpholinophenyl)-2-oxooxazolidi...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1ccc(N2CCOCC2)c(F)c1 Show InChI InChI=1S/C14H16FN3O4/c15-10-7-9(18-8-12(13(16)19)22-14(18)20)1-2-11(10)17-3-5-21-6-4-17/h1-2,7,12H,3-6,8H2,(H2,16,19)/t12-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.46E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
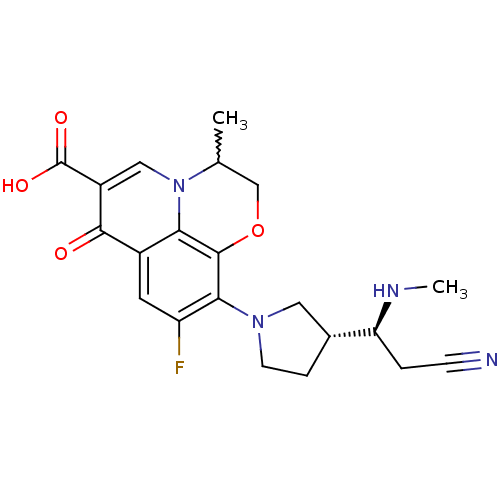
(Homo sapiens (Human)) | BDBM50205454
(10-((R)-3-((S)-2-cyano-1-(methylamino)ethyl)pyrrol...)Show SMILES CN[C@@H](CC#N)[C@@H]1CCN(C1)c1c(F)cc2c3c1OCC(C)n3cc(C(O)=O)c2=O |w:20.22| Show InChI InChI=1S/C21H23FN4O4/c1-11-10-30-20-17-13(19(27)14(21(28)29)9-26(11)17)7-15(22)18(20)25-6-4-12(8-25)16(24-2)3-5-23/h7,9,11-12,16,24H,3-4,6,8,10H2,1-2H3,(H,28,29)/t11?,12-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205456
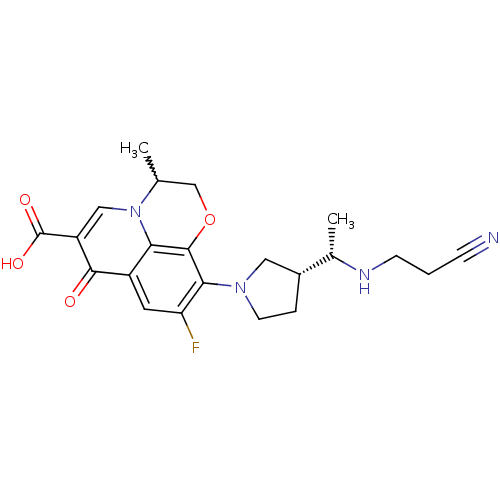
(10-((R)-3-((S)-1-(2-cyanoethylamino)ethyl)pyrrolid...)Show SMILES C[C@H](NCCC#N)[C@@H]1CCN(C1)c1c(F)cc2c3c1OCC(C)n3cc(C(O)=O)c2=O |w:21.23| Show InChI InChI=1S/C22H25FN4O4/c1-12-11-31-21-18-15(20(28)16(22(29)30)10-27(12)18)8-17(23)19(21)26-7-4-14(9-26)13(2)25-6-3-5-24/h8,10,12-14,25H,3-4,6-7,9,11H2,1-2H3,(H,29,30)/t12?,13-,14+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205462
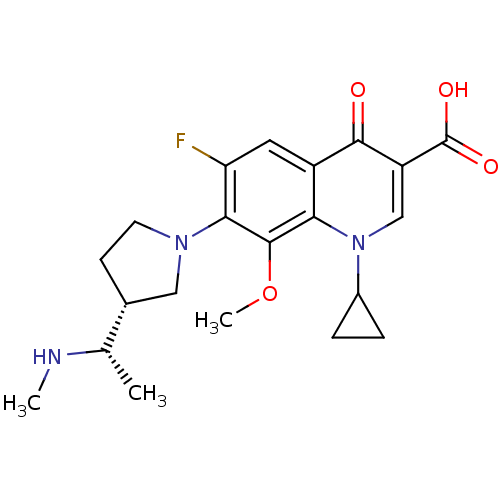
(1-cyclopropyl-6-fluoro-8-methoxy-7-((R)-3-((S)-1-(...)Show SMILES CN[C@@H](C)[C@@H]1CCN(C1)c1c(F)cc2c(c1OC)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C21H26FN3O4/c1-11(23-2)12-6-7-24(9-12)18-16(22)8-14-17(20(18)29-3)25(13-4-5-13)10-15(19(14)26)21(27)28/h8,10-13,23H,4-7,9H2,1-3H3,(H,27,28)/t11-,12+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205463
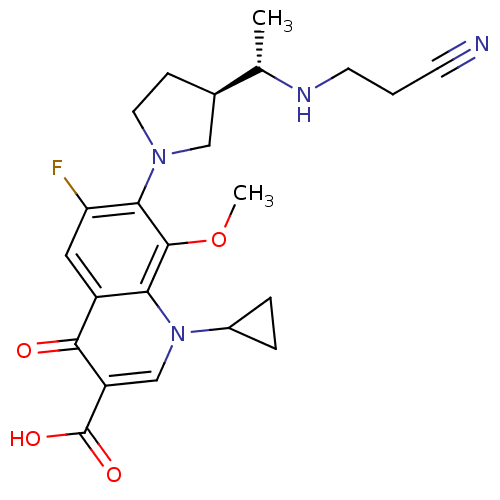
(7-((R)-3-((S)-1-(2-cyanoethylamino)ethyl)pyrrolidi...)Show SMILES COc1c(N2CC[C@H](C2)[C@H](C)NCCC#N)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C23H27FN4O4/c1-13(26-8-3-7-25)14-6-9-27(11-14)20-18(24)10-16-19(22(20)32-2)28(15-4-5-15)12-17(21(16)29)23(30)31/h10,12-15,26H,3-6,8-9,11H2,1-2H3,(H,30,31)/t13-,14+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205467
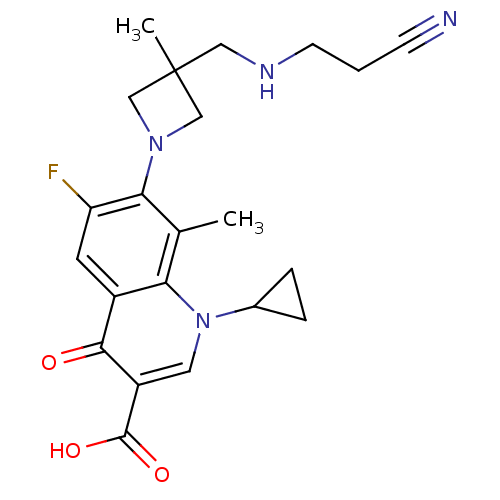
(7-(3-((2-cyanoethylamino)methyl)-3-methylazetidin-...)Show SMILES Cc1c(N2CC(C)(CNCCC#N)C2)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C22H25FN4O3/c1-13-18-15(20(28)16(21(29)30)9-27(18)14-4-5-14)8-17(23)19(13)26-11-22(2,12-26)10-25-7-3-6-24/h8-9,14,25H,3-5,7,10-12H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205464
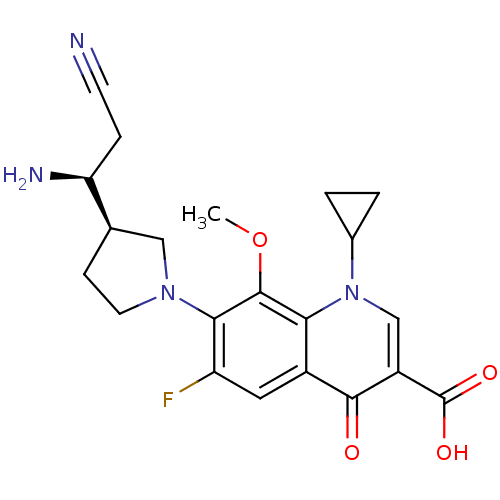
(7-((R)-3-((S)-1-amino-2-cyanoethyl)pyrrolidin-1-yl...)Show SMILES COc1c(N2CC[C@H](C2)[C@@H](N)CC#N)c(F)cc2c1n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C21H23FN4O4/c1-30-20-17-13(19(27)14(21(28)29)10-26(17)12-2-3-12)8-15(22)18(20)25-7-5-11(9-25)16(24)4-6-23/h8,10-12,16H,2-5,7,9,24H2,1H3,(H,28,29)/t11-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205460
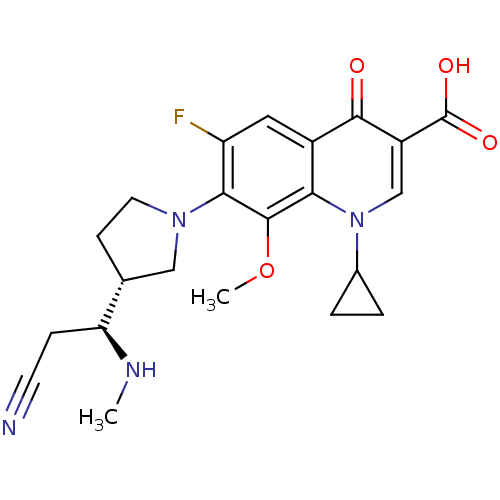
(7-((R)-3-((S)-2-cyano-1-(methylamino)ethyl)pyrroli...)Show SMILES CN[C@@H](CC#N)[C@@H]1CCN(C1)c1c(F)cc2c(c1OC)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C22H25FN4O4/c1-25-17(5-7-24)12-6-8-26(10-12)19-16(23)9-14-18(21(19)31-2)27(13-3-4-13)11-15(20(14)28)22(29)30/h9,11-13,17,25H,3-6,8,10H2,1-2H3,(H,29,30)/t12-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205448
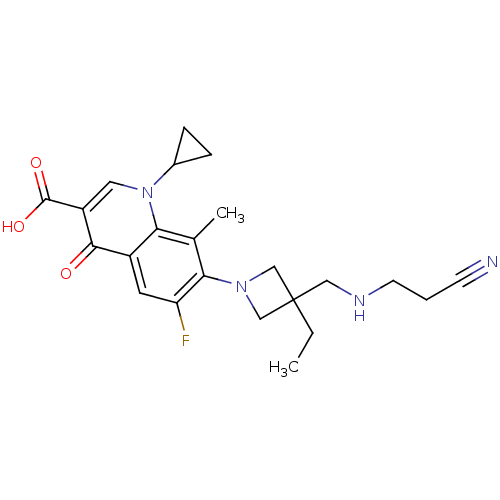
(7-(3-((2-cyanoethylamino)methyl)-3-ethylazetidin-1...)Show SMILES CCC1(CNCCC#N)CN(C1)c1c(F)cc2c(c1C)n(cc(C(O)=O)c2=O)C1CC1 Show InChI InChI=1S/C23H27FN4O3/c1-3-23(11-26-8-4-7-25)12-27(13-23)20-14(2)19-16(9-18(20)24)21(29)17(22(30)31)10-28(19)15-5-6-15/h9-10,15,26H,3-6,8,11-13H2,1-2H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205458
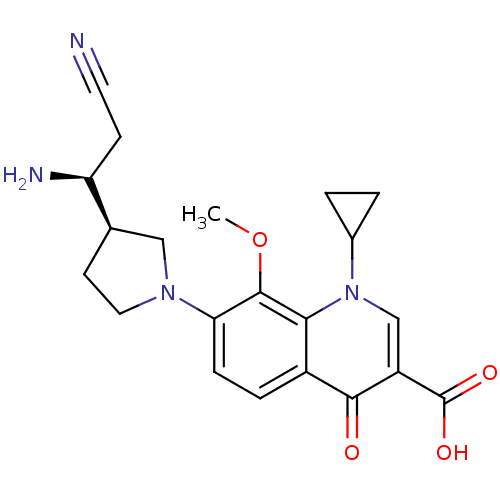
(7-((R)-3-((S)-1-amino-2-cyanoethyl)pyrrolidin-1-yl...)Show SMILES COc1c(ccc2c1n(cc(C(O)=O)c2=O)C1CC1)N1CC[C@H](C1)[C@@H](N)CC#N Show InChI InChI=1S/C21H24N4O4/c1-29-20-17(24-9-7-12(10-24)16(23)6-8-22)5-4-14-18(20)25(13-2-3-13)11-15(19(14)26)21(27)28/h4-5,11-13,16H,2-3,6-7,9-10,23H2,1H3,(H,27,28)/t12-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50131445
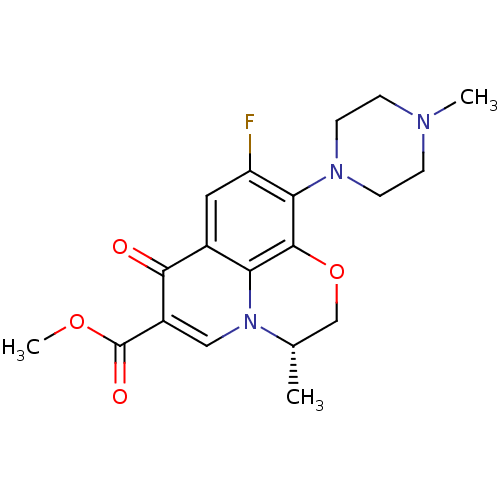
((3S)-9-fluoro-3-methyl-10-(4-methylpiperazin-1-yl)...)Show SMILES COC(=O)c1cn2[C@@H](C)COc3c(N4CCN(C)CC4)c(F)cc(c23)c1=O Show InChI InChI=1S/C19H22FN3O4/c1-11-10-27-18-15-12(17(24)13(9-23(11)15)19(25)26-3)8-14(20)16(18)22-6-4-21(2)5-7-22/h8-9,11H,4-7,10H2,1-3H3/t11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50205449
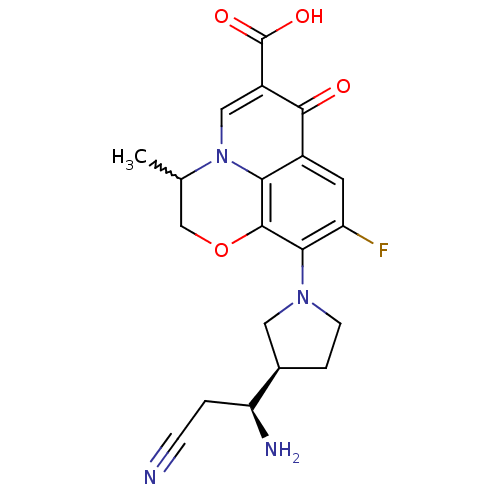
(10-((R)-3-((S)-1-amino-2-cyanoethyl)pyrrolidin-1-y...)Show SMILES CC1COc2c(N3CC[C@H](C3)[C@@H](N)CC#N)c(F)cc3c2n1cc(C(O)=O)c3=O |w:1.0| Show InChI InChI=1S/C20H21FN4O4/c1-10-9-29-19-16-12(18(26)13(20(27)28)8-25(10)16)6-14(21)17(19)24-5-3-11(7-24)15(23)2-4-22/h6,8,10-11,15H,2-3,5,7,9,23H2,1H3,(H,27,28)/t10?,11-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG by fliter binding assay |
Bioorg Med Chem Lett 17: 2150-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.090
BindingDB Entry DOI: 10.7270/Q2GM86Z7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50226481
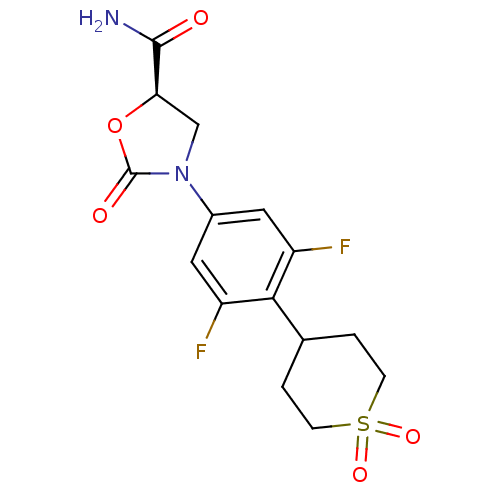
((5R)-3-[4-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1cc(F)c(C2CCS(=O)(=O)CC2)c(F)c1 Show InChI InChI=1S/C15H16F2N2O5S/c16-10-5-9(19-7-12(14(18)20)24-15(19)21)6-11(17)13(10)8-1-3-25(22,23)4-2-8/h5-6,8,12H,1-4,7H2,(H2,18,20)/t12-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.93E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM50226482
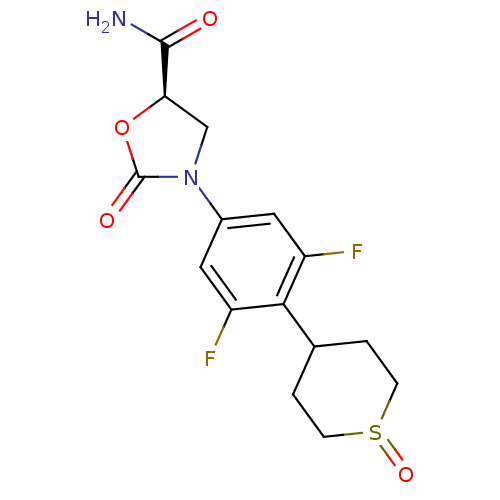
((5R)-3-[3,5-difluoro-4-(1-oxidotetrahydro-2H-thiop...)Show SMILES NC(=O)[C@H]1CN(C(=O)O1)c1cc(F)c(C2CCS(=O)CC2)c(F)c1 Show InChI InChI=1S/C15H16F2N2O4S/c16-10-5-9(19-7-12(14(18)20)23-15(19)21)6-11(17)13(10)8-1-3-24(22)4-2-8/h5-6,8,12H,1-4,7H2,(H2,18,20)/t8?,12-,24?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.55E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAOA |
J Med Chem 50: 5886-9 (2007)
Article DOI: 10.1021/jm070708p
BindingDB Entry DOI: 10.7270/Q22J6BM7 |
More data for this
Ligand-Target Pair | |
UDP-3-O-acyl-N-acetylglucosamine deacetylase
(Pseudomonas aeruginosa) | BDBM50485077
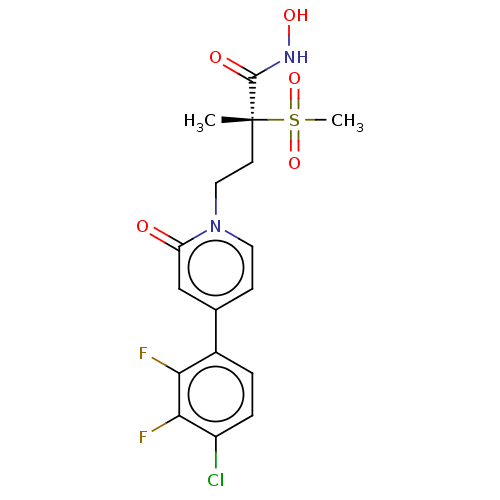
(CHEMBL2023517)Show SMILES C[C@@](CCn1ccc(cc1=O)-c1ccc(Cl)c(F)c1F)(C(=O)NO)S(C)(=O)=O |r| Show InChI InChI=1S/C17H17ClF2N2O5S/c1-17(16(24)21-25,28(2,26)27)6-8-22-7-5-10(9-13(22)23)11-3-4-12(18)15(20)14(11)19/h3-5,7,9,25H,6,8H2,1-2H3,(H,21,24)/t17-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.520 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa LpxC expressed in Escherichia coli using UDP-3-O-N-acetylglucosamine substrate measured after 30 mins by mass sp... |
J Med Chem 55: 1662-70 (2012)
Article DOI: 10.1021/jm2014875
BindingDB Entry DOI: 10.7270/Q2W38050 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data