 Found 15 hits with Last Name = 'hudson' and Initial = 'sa'
Found 15 hits with Last Name = 'hudson' and Initial = 'sa' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50169700
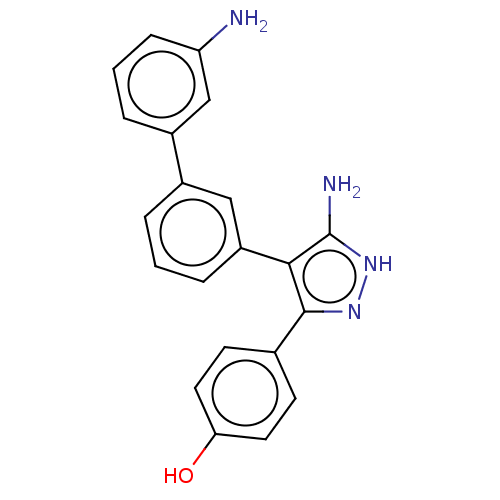
(CHEMBL3806115)Show SMILES Nc1[nH]nc(c1-c1cccc(c1)-c1cccc(N)c1)-c1ccc(O)cc1 Show InChI InChI=1S/C21H18N4O/c22-17-6-2-4-15(12-17)14-3-1-5-16(11-14)19-20(24-25-21(19)23)13-7-9-18(26)10-8-13/h1-12,26H,22H2,(H3,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50169702
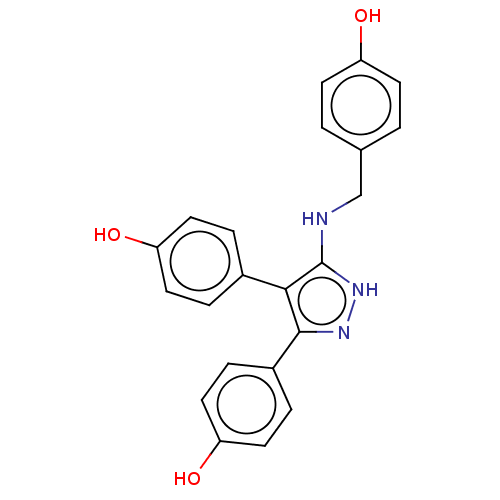
(CHEMBL3805279)Show SMILES Oc1ccc(CNc2[nH]nc(c2-c2ccc(O)cc2)-c2ccc(O)cc2)cc1 Show InChI InChI=1S/C22H19N3O3/c26-17-7-1-14(2-8-17)13-23-22-20(15-3-9-18(27)10-4-15)21(24-25-22)16-5-11-19(28)12-6-16/h1-12,26-28H,13H2,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using tolbutamide as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50169698
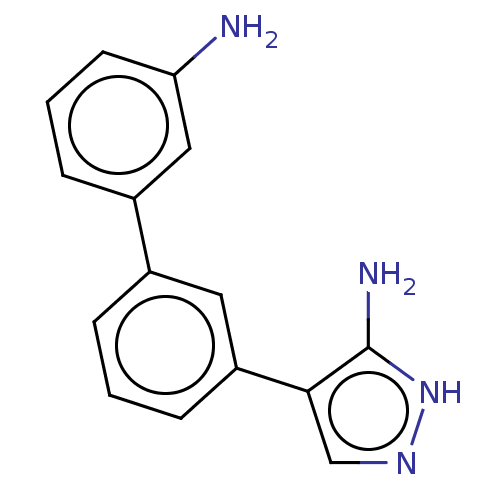
(CHEMBL3806191)Show InChI InChI=1S/C15H14N4/c16-13-6-2-4-11(8-13)10-3-1-5-12(7-10)14-9-18-19-15(14)17/h1-9H,16H2,(H3,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using mephenytoin as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50169700

(CHEMBL3806115)Show SMILES Nc1[nH]nc(c1-c1cccc(c1)-c1cccc(N)c1)-c1ccc(O)cc1 Show InChI InChI=1S/C21H18N4O/c22-17-6-2-4-15(12-17)14-3-1-5-16(11-14)19-20(24-25-21(19)23)13-7-9-18(26)10-8-13/h1-12,26H,22H2,(H3,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using mephenytoin as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50169702

(CHEMBL3805279)Show SMILES Oc1ccc(CNc2[nH]nc(c2-c2ccc(O)cc2)-c2ccc(O)cc2)cc1 Show InChI InChI=1S/C22H19N3O3/c26-17-7-1-14(2-8-17)13-23-22-20(15-3-9-18(27)10-4-15)21(24-25-22)16-5-11-19(28)12-6-16/h1-12,26-28H,13H2,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using mephenytoin as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50169698

(CHEMBL3806191)Show InChI InChI=1S/C15H14N4/c16-13-6-2-4-11(8-13)10-3-1-5-12(7-10)14-9-18-19-15(14)17/h1-9H,16H2,(H3,17,18,19) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50169700

(CHEMBL3806115)Show SMILES Nc1[nH]nc(c1-c1cccc(c1)-c1cccc(N)c1)-c1ccc(O)cc1 Show InChI InChI=1S/C21H18N4O/c22-17-6-2-4-15(12-17)14-3-1-5-16(11-14)19-20(24-25-21(19)23)13-7-9-18(26)10-8-13/h1-12,26H,22H2,(H3,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50169702

(CHEMBL3805279)Show SMILES Oc1ccc(CNc2[nH]nc(c2-c2ccc(O)cc2)-c2ccc(O)cc2)cc1 Show InChI InChI=1S/C22H19N3O3/c26-17-7-1-14(2-8-17)13-23-22-20(15-3-9-18(27)10-4-15)21(24-25-22)16-5-11-19(28)12-6-16/h1-12,26-28H,13H2,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50169698

(CHEMBL3806191)Show InChI InChI=1S/C15H14N4/c16-13-6-2-4-11(8-13)10-3-1-5-12(7-10)14-9-18-19-15(14)17/h1-9H,16H2,(H3,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using tolbutamide as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50169700

(CHEMBL3806115)Show SMILES Nc1[nH]nc(c1-c1cccc(c1)-c1cccc(N)c1)-c1ccc(O)cc1 Show InChI InChI=1S/C21H18N4O/c22-17-6-2-4-15(12-17)14-3-1-5-16(11-14)19-20(24-25-21(19)23)13-7-9-18(26)10-8-13/h1-12,26H,22H2,(H3,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using tolbutamide as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50169698

(CHEMBL3806191)Show InChI InChI=1S/C15H14N4/c16-13-6-2-4-11(8-13)10-3-1-5-12(7-10)14-9-18-19-15(14)17/h1-9H,16H2,(H3,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50169702

(CHEMBL3805279)Show SMILES Oc1ccc(CNc2[nH]nc(c2-c2ccc(O)cc2)-c2ccc(O)cc2)cc1 Show InChI InChI=1S/C22H19N3O3/c26-17-7-1-14(2-8-17)13-23-22-20(15-3-9-18(27)10-4-15)21(24-25-22)16-5-11-19(28)12-6-16/h1-12,26-28H,13H2,(H2,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate by LC-MS/MS analysis |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Mycocyclosin synthase
(Mycobacterium tuberculosis) | BDBM31772
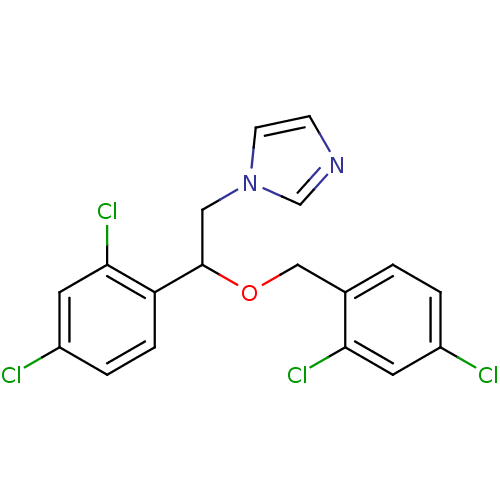
(1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...)Show SMILES Clc1ccc(COC(Cn2ccnc2)c2ccc(Cl)cc2Cl)c(Cl)c1 Show InChI InChI=1S/C18H14Cl4N2O/c19-13-2-1-12(16(21)7-13)10-25-18(9-24-6-5-23-11-24)15-4-3-14(20)8-17(15)22/h1-8,11,18H,9-10H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Binding affinity to Mycobacterium tuberculosis H37Rv wild type CYP121 by titration assay |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Mycocyclosin synthase
(Mycobacterium tuberculosis) | BDBM31773
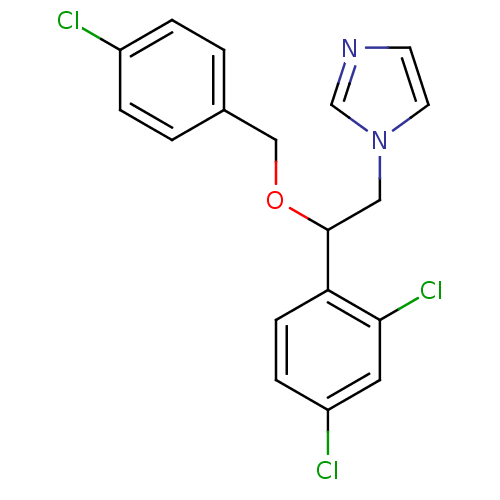
(ECONAZOLE | Econazole nitrate | Gyno-pevaryl | Pev...)Show InChI InChI=1S/C18H15Cl3N2O/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21/h1-9,12,18H,10-11H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Binding affinity to Mycobacterium tuberculosis H37Rv wild type CYP121 by titration assay |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |
Mycocyclosin synthase
(Mycobacterium tuberculosis) | BDBM31774
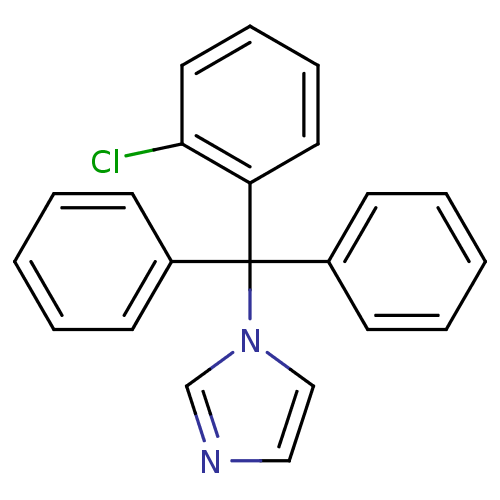
(CHEMBL104 | Canesten | Clotrimazole | Lotrimin | M...)Show InChI InChI=1S/C22H17ClN2/c23-21-14-8-7-13-20(21)22(25-16-15-24-17-25,18-9-3-1-4-10-18)19-11-5-2-6-12-19/h1-17H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a |
University of Cambridge
Curated by ChEMBL
| Assay Description
Binding affinity to Mycobacterium tuberculosis H37Rv wild type CYP121 by titration assay |
J Med Chem 59: 3272-302 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00007
BindingDB Entry DOI: 10.7270/Q2MS3VN4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data