

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826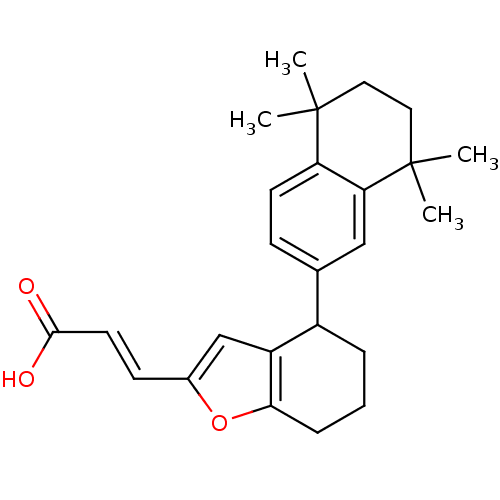 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143827 ((E)-3-[4-(3-Methoxy-5,5,8,8-tetramethyl-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143825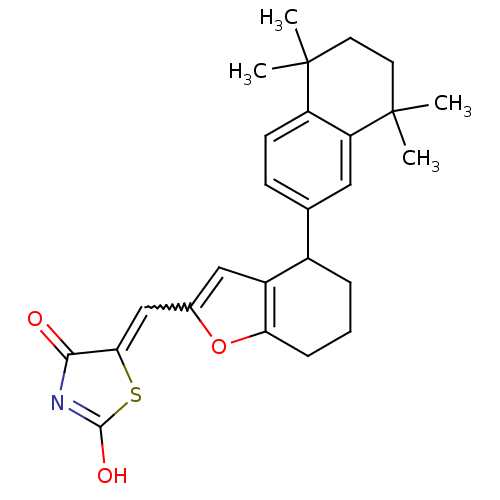 (5-[1-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143832 ((E)-3-[4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143833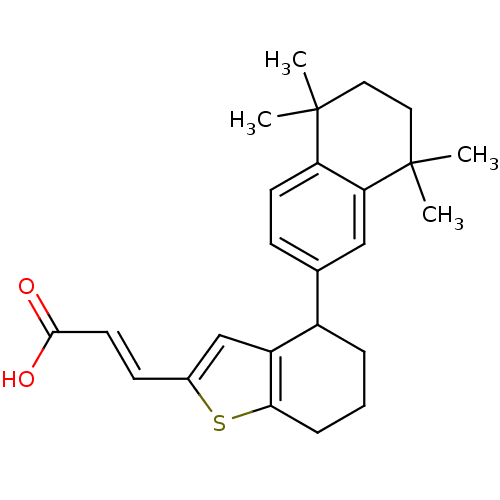 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50409928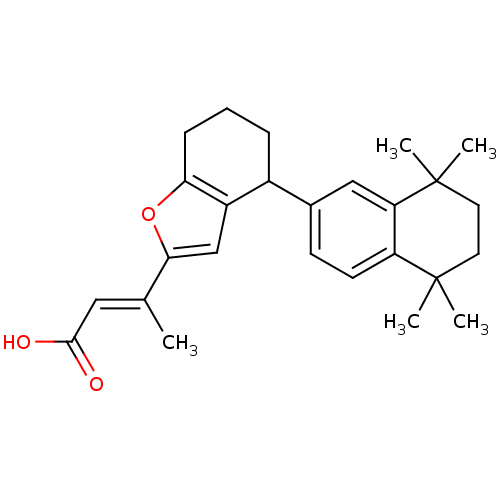 (CHEMBL2113737) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50409929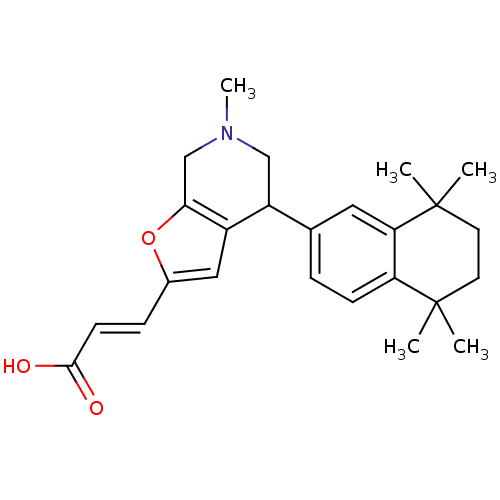 (CHEMBL2113736) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143824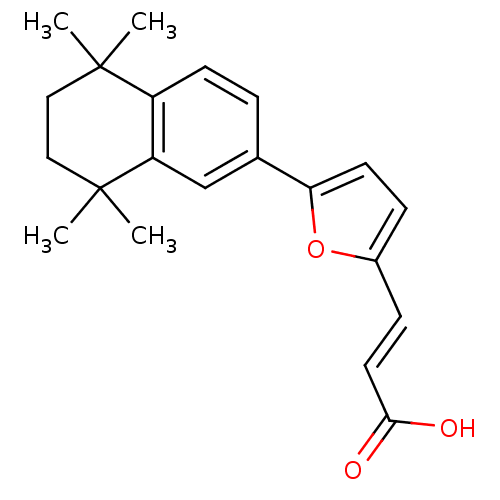 ((E)-3-[5-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143821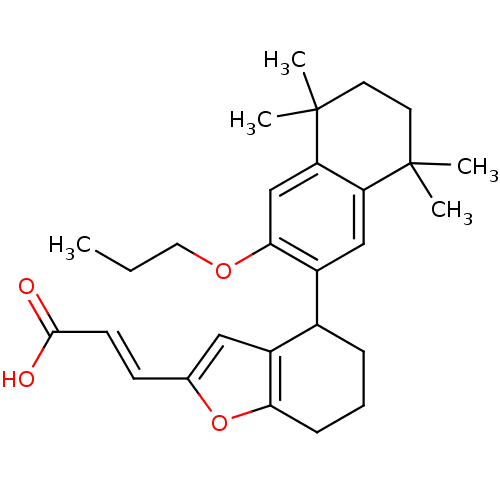 ((E)-3-[4-(5,5,8,8-Tetramethyl-3-propoxy-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143835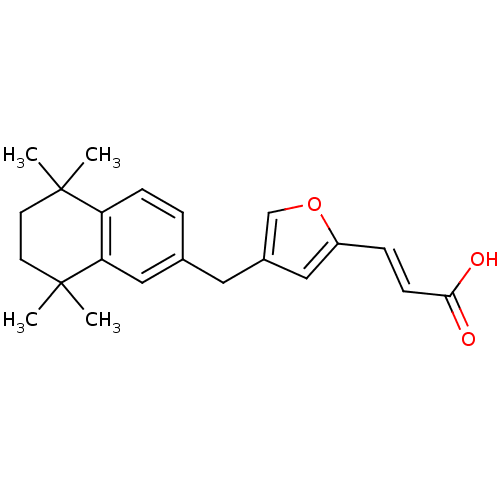 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 138 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143823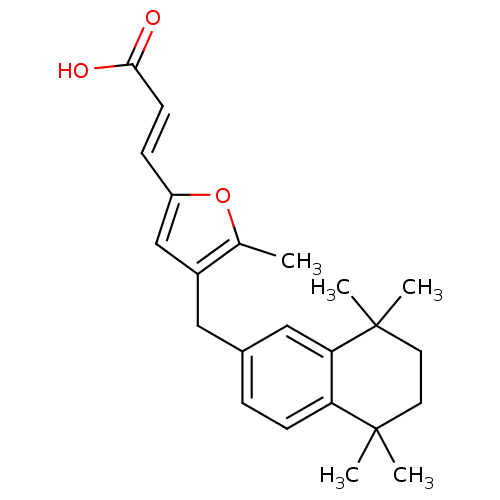 ((E)-3-[5-Methyl-4-(5,5,8,8-tetramethyl-5,6,7,8-tet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143831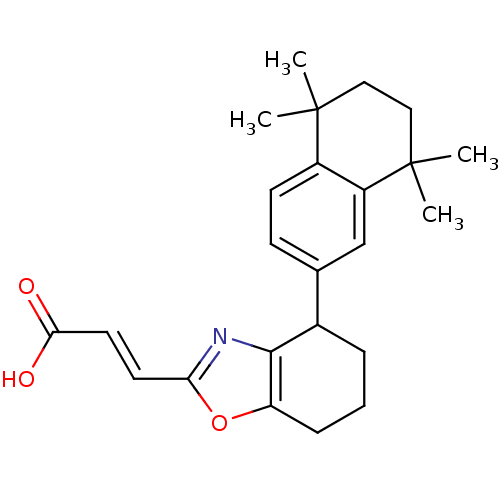 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143828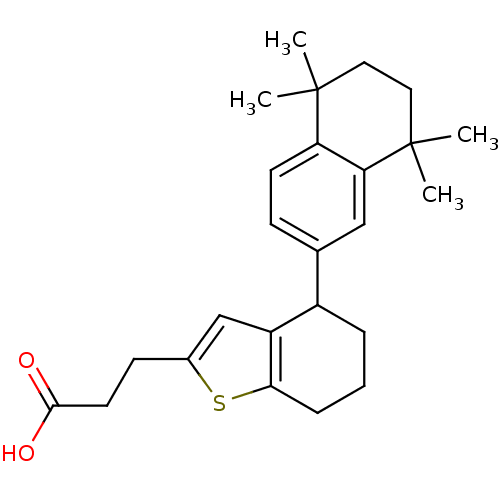 (3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-napht...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143830 (4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143825 (5-[1-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143823 ((E)-3-[5-Methyl-4-(5,5,8,8-tetramethyl-5,6,7,8-tet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143827 ((E)-3-[4-(3-Methoxy-5,5,8,8-tetramethyl-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143824 ((E)-3-[5-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143833 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143828 (3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-napht...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50409928 (CHEMBL2113737) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50409929 (CHEMBL2113736) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143821 ((E)-3-[4-(5,5,8,8-Tetramethyl-3-propoxy-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143831 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143835 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143832 ((E)-3-[4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143834 (5-[1-[5-(3-Bromo-phenyl)-furan-2-yl]-meth-(Z)-ylid...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143830 (4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143836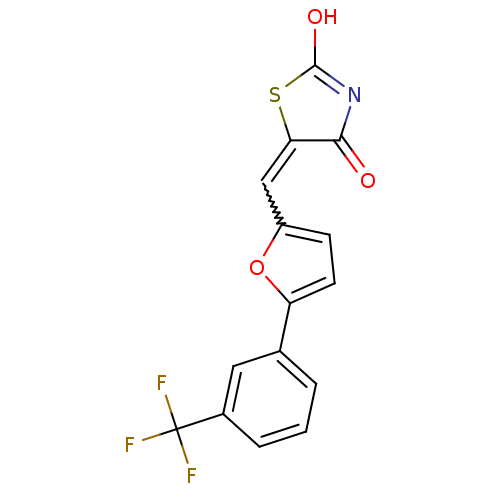 (5-[1-[5-(3-Trifluoromethyl-phenyl)-furan-2-yl]-met...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143821 ((E)-3-[4-(5,5,8,8-Tetramethyl-3-propoxy-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Inhibitory concentration for lipogenesis induced by retinoic acid receptor alpha in C3H10T1/2 clone 8 fibroblast cells | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50099485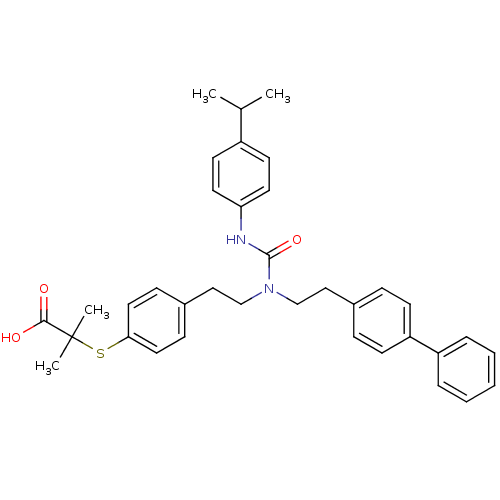 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(4-isopropyl-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR alpha ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50099486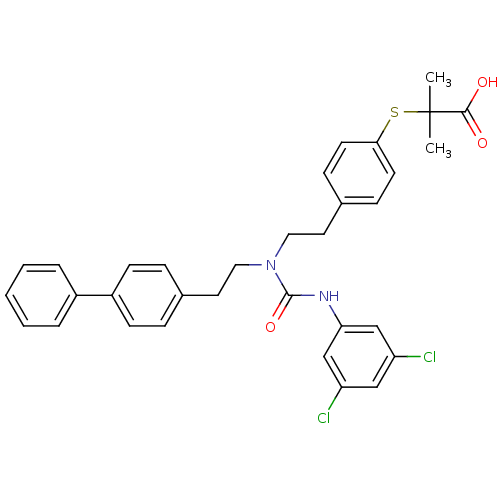 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(3,5-dichloro...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50099485 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(4-isopropyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 8.50E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50099485 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(4-isopropyl-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50099487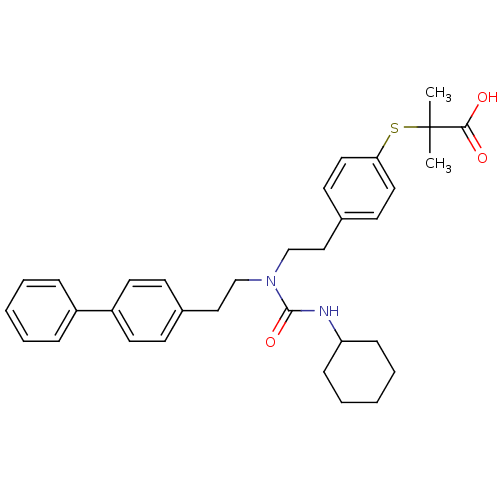 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-cyclohexyl-ur...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50099484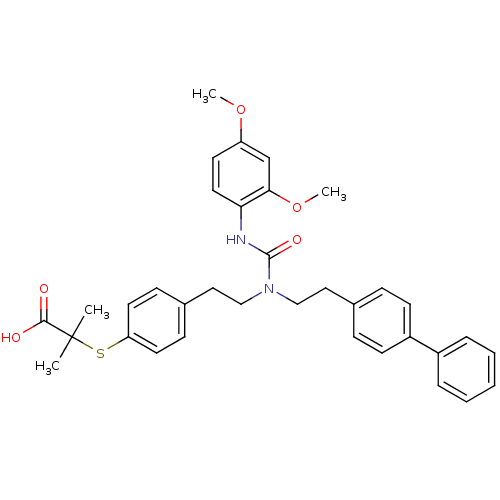 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(2,4-dimethox...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50099488 (2-(4-{2-[3-(3-Cyano-phenyl)-1-(4-cyclohexyl-butyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 9 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR alpha ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50099489 (2-(4-{2-[1-(4-Cyclohexyl-butyl)-3-(2-methoxy-pheny...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR alpha ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50099487 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-cyclohexyl-ur...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Mus musculus) | BDBM50099491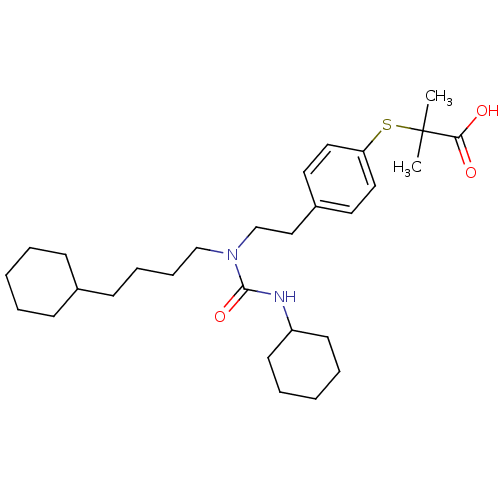 (2-(4-(2-(3-cyclohexyl-1-(4-cyclohexylbutyl)ureido)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Activation of murine PPAR alpha ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50099490 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(2-methoxy-5-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50099491 (2-(4-(2-(3-cyclohexyl-1-(4-cyclohexylbutyl)ureido)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 6.20E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM50099484 (2-(4-{2-[1-(2-Biphenyl-4-yl-ethyl)-3-(2,4-dimethox...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor delta (Homo sapiens (Human)) | BDBM28799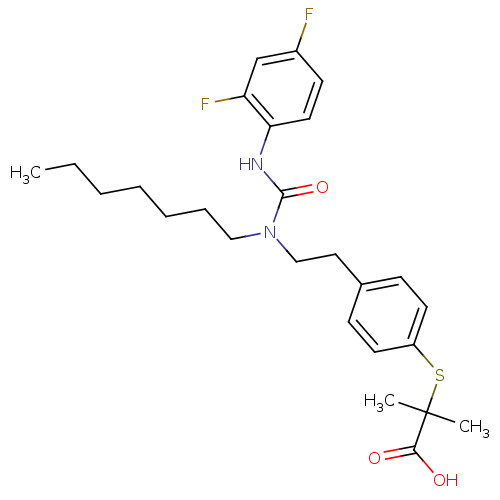 (2-{[4-(2-{[(2,4-difluorophenyl)carbamoyl](heptyl)a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR delta ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Mus musculus) | BDBM50099491 (2-(4-(2-(3-cyclohexyl-1-(4-cyclohexylbutyl)ureido)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Activation of murine PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28799 (2-{[4-(2-{[(2,4-difluorophenyl)carbamoyl](heptyl)a...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description In vitro transcriptional activation in CV-1 cells expressing human Gal4-PPAR gamma ligand binding domain | Bioorg Med Chem Lett 11: 1225-7 (2001) BindingDB Entry DOI: 10.7270/Q2Z31XX0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 124 total ) | Next | Last >> |