

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50079527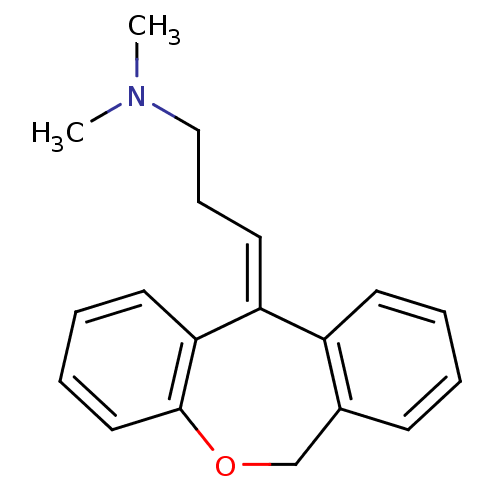 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.178 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
VU University Amsterdam Curated by ChEMBL | Assay Description Displacement of [3H]mepyramine from human histamine H1 receptor expressed in HEK293 cells after 1 to 1.5 hrs by scintillation counting | J Med Chem 54: 8195-206 (2011) Article DOI: 10.1021/jm2011589 BindingDB Entry DOI: 10.7270/Q2QF8T85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50017698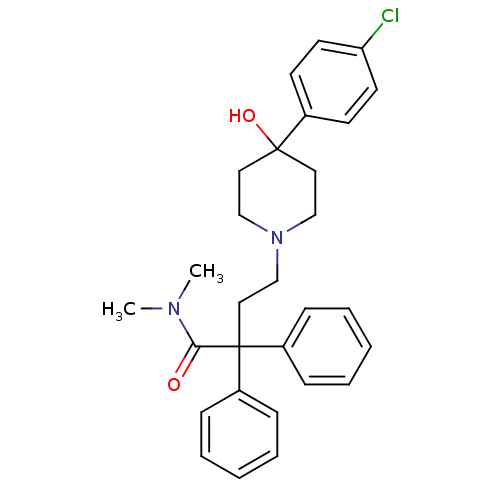 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.268 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50585129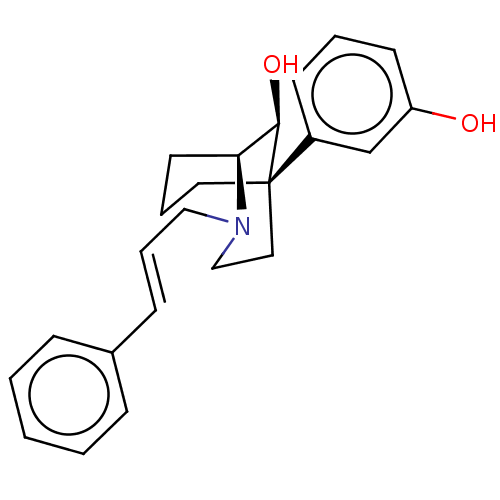 (CHEMBL5077645) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.377 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50364063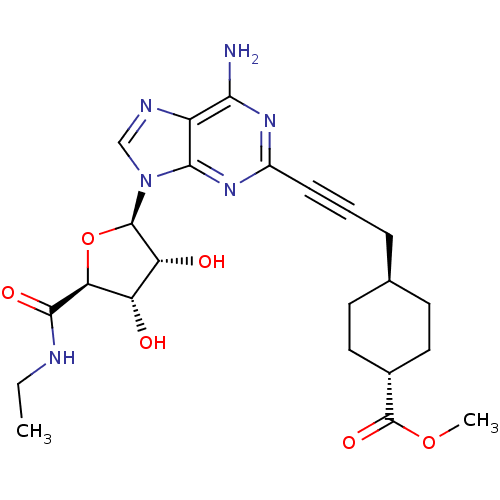 (CHEMBL1950649) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [D138N] (Homo sapiens (Human)) | BDBM50159165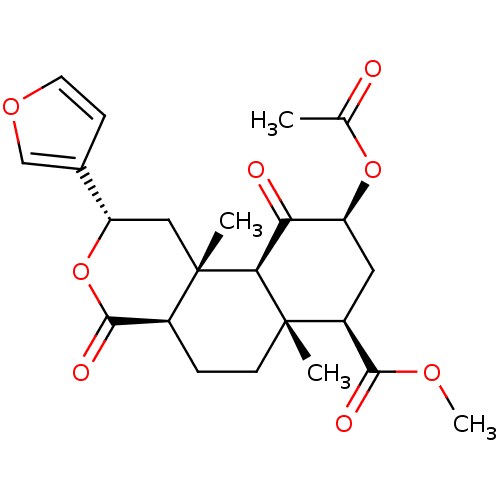 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.590 | -52.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50585128 (CHEMBL5080654) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.633 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50585107 (CHEMBL5085974) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.832 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50585112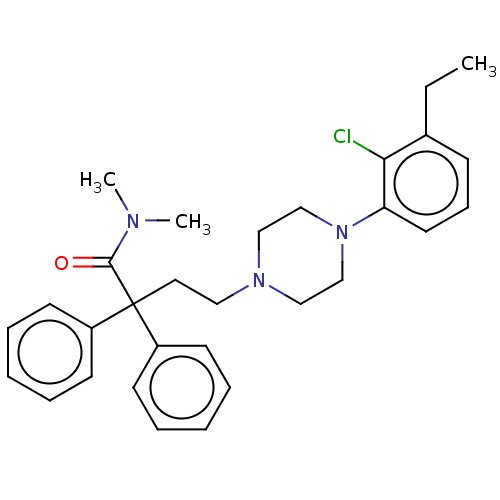 (CHEMBL5088070) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.842 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(4) dopamine receptor (Homo sapiens (Human)) | BDBM21398 (4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D4 receptor expressed in HEK293 cell membranes incubated for 60 mins by microbeta scintill... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM50389797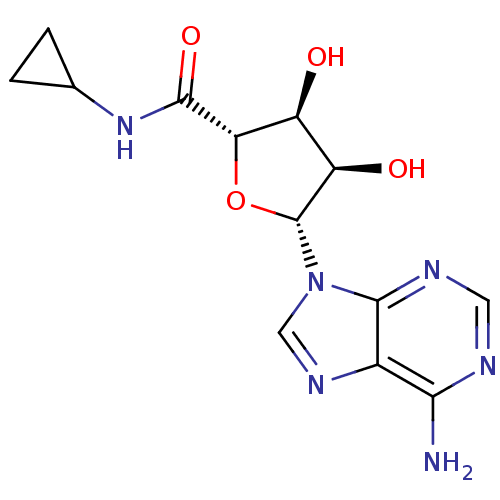 (CHEMBL261482) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of 3[H]R-PIA from human A1 adenosine receptor expressed in CHO cells after 60 mins by Liquid scintillation analysis | J Med Chem 55: 4297-308 (2012) Article DOI: 10.1021/jm300095s BindingDB Entry DOI: 10.7270/Q21C1XXC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens) | BDBM50515233 (CHEMBL4551160) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibition of acetylcholinesterase in Rattus norvegicus (rat) cortex by Ellman method | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | CHEMBL5273118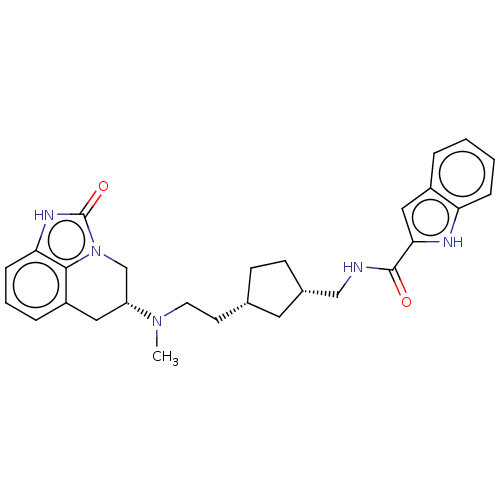 | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL CHEMBL PC cid PC sid UniChem Similars | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibition of acetylcholinesterase in Rattus norvegicus (rat) cortex by Ellman method | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22567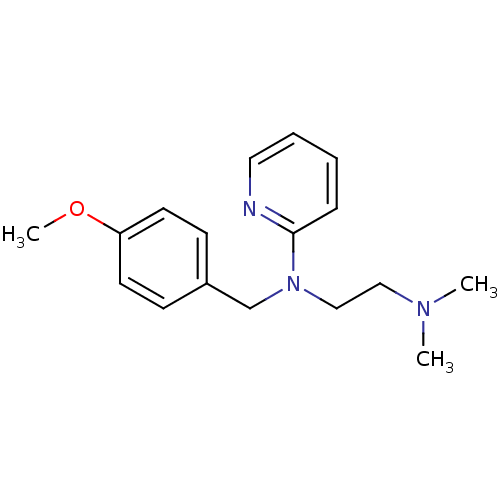 (3H]pyrilamine | CHEMBL511 | Dorantamin | Mepyramin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 2.09 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
VU University Amsterdam Curated by ChEMBL | Assay Description Displacement of [3H]mepyramine from human histamine H1 receptor expressed in HEK293 cells after 1 to 1.5 hrs by scintillation counting | J Med Chem 54: 8195-206 (2011) Article DOI: 10.1021/jm2011589 BindingDB Entry DOI: 10.7270/Q2QF8T85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K227A] (Homo sapiens (Human)) | BDBM214798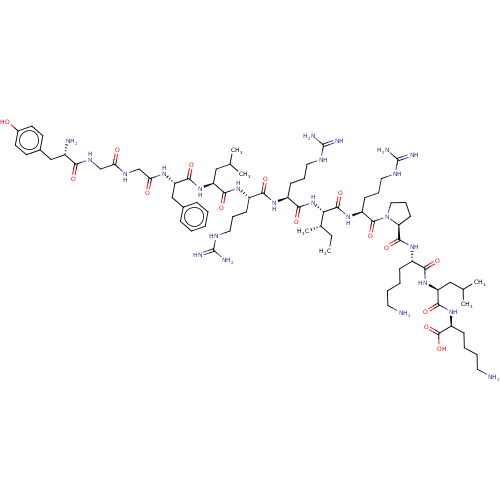 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 2.20 | -49.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens) | BDBM50585122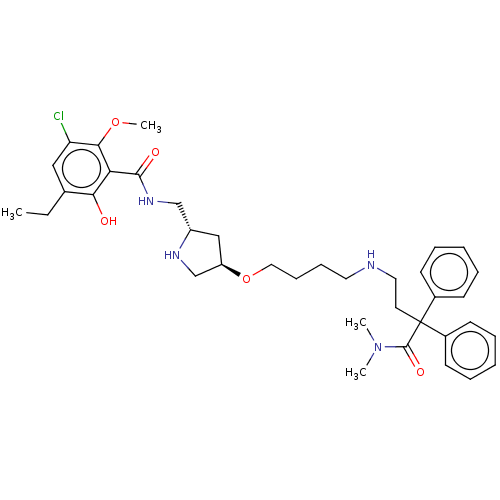 (CHEMBL5081136) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor expressed in HEK293 cell membranes incubated for 60 mins by microbeta scintill... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM21220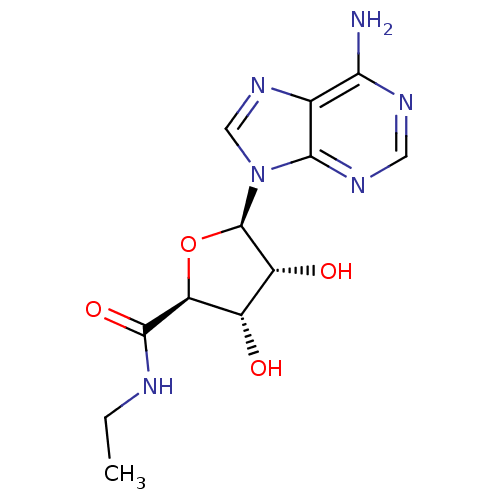 ((2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-N-ethyl-3,...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50364061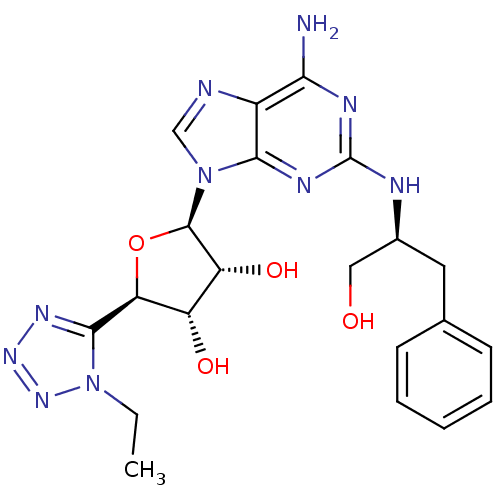 (CHEMBL1950652) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to human A2A adenosine receptor expressed in CHO cells | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K227A] (Homo sapiens (Human)) | BDBM21130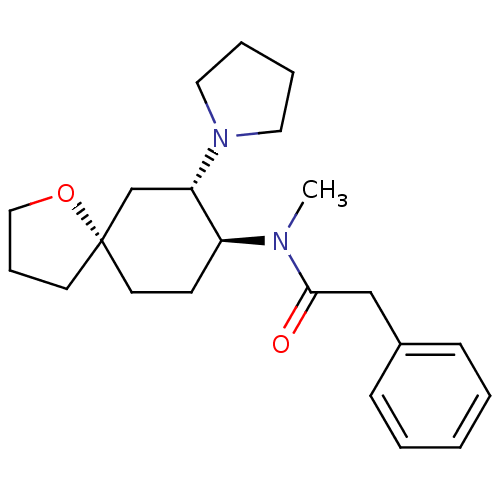 (N-methyl-2-phenyl-N-[(5R,7S,8S)-7-(pyrrolidin-1-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 2.5 | -49.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM21220 ((2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-N-ethyl-3,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of 3[H]R-PIA from human A1 adenosine receptor expressed in CHO cells after 60 mins by Liquid scintillation analysis | J Med Chem 55: 4297-308 (2012) Article DOI: 10.1021/jm300095s BindingDB Entry DOI: 10.7270/Q21C1XXC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K212A] (Homo sapiens (Human)) | BDBM21130 (N-methyl-2-phenyl-N-[(5R,7S,8S)-7-(pyrrolidin-1-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 3.30 | -48.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [D138A] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.5 | -48.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | BDBM21398 (4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D2 receptor expressed in HEK293 cell membranes incubated for 60 mins by microbeta scintill... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50364044 (CHEMBL260203) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [I294A] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.90 | -48.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50316212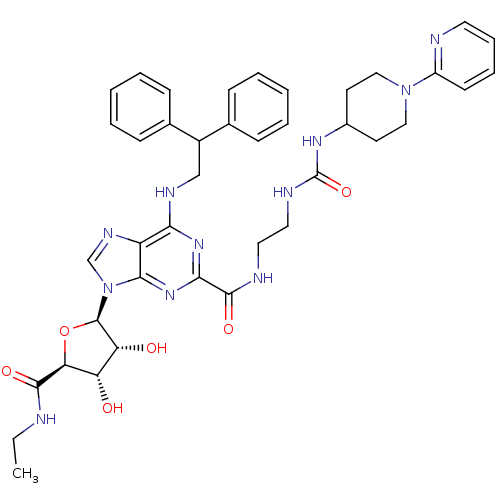 (6-(2,2-diphenylethylamino)-9-((2R,3R,4S,5S)-5-(eth...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Kappa-type opioid receptor [D138N] (Homo sapiens (Human)) | BDBM214799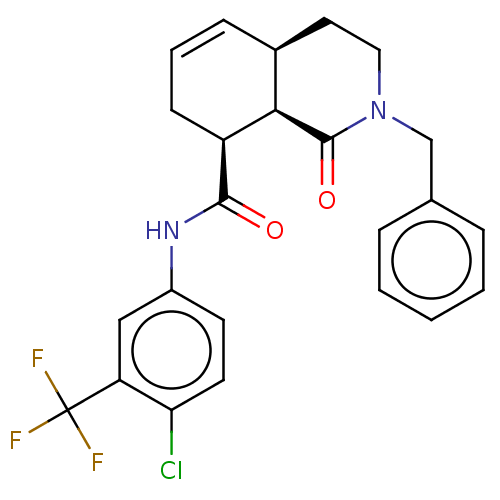 (1xx) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4 | -47.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K227A] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.40 | -47.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens) | BDBM21398 (4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor expressed in HEK293 cell membranes incubated for 60 mins by microbeta scintill... | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | CHEMBL5273776 | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL CHEMBL PC cid PC sid UniChem Similars | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibition of acetylcholinesterase in Rattus norvegicus (rat) cortex by Ellman method | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K200A] (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 4.80 | -47.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50008984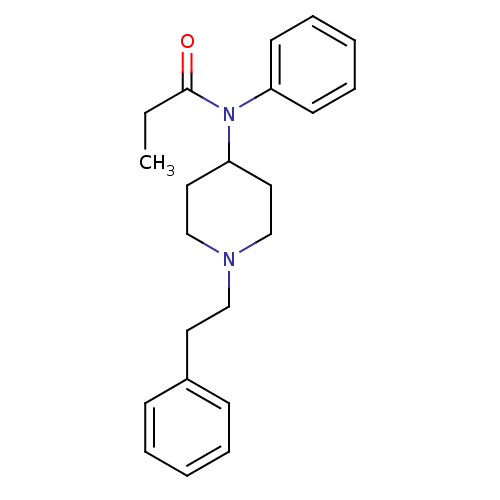 (4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-ph...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in HEK293 cell membrane incubated for 60 mins by radioligand binding assay | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00611 BindingDB Entry DOI: 10.7270/Q2N301V5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D(3) dopamine receptor (Homo sapiens) | BDBM50515233 (CHEMBL4551160) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Binding affinity to bovine serum albumin by circular dichroic probe technique | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [K212A] (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 5.40 | -47.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM50389798 (CHEMBL2070507) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of 3[H]R-PIA from human A1 adenosine receptor expressed in CHO cells after 60 mins by Liquid scintillation analysis | J Med Chem 55: 4297-308 (2012) Article DOI: 10.1021/jm300095s BindingDB Entry DOI: 10.7270/Q21C1XXC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens) | CHEMBL5267958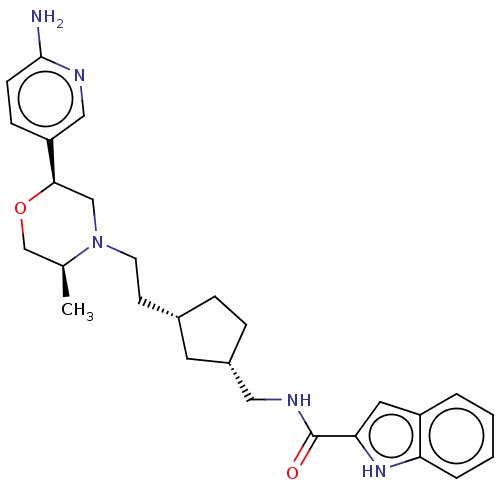 | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL CHEMBL PC cid PC sid UniChem Similars | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibition of acetylcholinesterase in Rattus norvegicus (rat) cortex by Ellman method | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50119168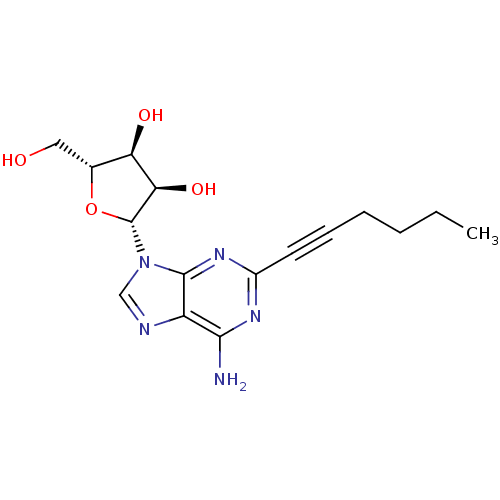 ((2R,3R,4S,5R)-2-(6-Amino-2-hex-1-ynyl-purin-9-yl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Rattus norvegicus (rat)) | BDBM50364060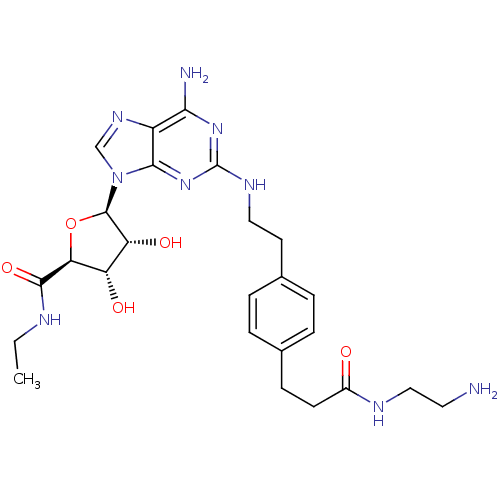 (CHEMBL1950647) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of [3H]NECA from A2A adenosine receptor expressed in rat striatal membranes after 30 mins by liquid scintillation counting | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [I135A] (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 6.20 | -46.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50358532 (CHEMBL1923521) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
VU University Amsterdam Curated by ChEMBL | Assay Description Displacement of [3H]mepyramine from human histamine H1 receptor expressed in HEK293 cells after 1 to 1.5 hrs by scintillation counting | J Med Chem 54: 8195-206 (2011) Article DOI: 10.1021/jm2011589 BindingDB Entry DOI: 10.7270/Q2QF8T85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50009263 ((2S,3S,4R,5R)-5-(6-amino-2-(hex-1-ynyl)-9H-purin-9...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50389797 (CHEMBL261482) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Displacement of 3[H]R-PIA from rat A1 adenosine receptor expressed in CHO cells after 60 min by Liquid scintillation analysis | J Med Chem 55: 4297-308 (2012) Article DOI: 10.1021/jm300095s BindingDB Entry DOI: 10.7270/Q21C1XXC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [Y320A] (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 6.60 | -46.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [M226A] (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 7.10 | -46.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50339076 ((2R,3R,4S)-2-(6-amino-2-(hex-1-ynyl)-9H-purin-9-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Binding affinity to A2A adenosine receptor | J Med Chem 55: 538-52 (2012) Article DOI: 10.1021/jm201461q BindingDB Entry DOI: 10.7270/Q22Z160R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [M226A] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 7.20 | -46.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [E297A] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 7.20 | -46.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens) | CHEMBL5288831 | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL CHEMBL PC cid PC sid UniChem Similars | 7.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibition of acetylcholinesterase in Rattus norvegicus (rat) cortex by Ellman method | Citation and Details | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [H291F] (Homo sapiens (Human)) | BDBM21130 (N-methyl-2-phenyl-N-[(5R,7S,8S)-7-(pyrrolidin-1-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 7.60 | -46.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor [H291F] (Homo sapiens (Human)) | BDBM50159165 ((2S,4aR,6aR,7R,9S,10aS,10bR)-9-(acetyloxy)-2-(fura...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 7.80 | -46.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM214798 (Dynorphin A (1-17) | YGGFLRRIRPKLK) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 8 | -46.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
University of North Carolina School of Medicine | Assay Description Radioligand binding assays were performed as previously detailed [Yan et al., Biochemistry, 48:6898-6908]. In brief, crude cell membranes were prepar... | J Biol Chem 288: 34470-83 (2013) Article DOI: 10.1074/jbc.M113.515668 BindingDB Entry DOI: 10.7270/Q2P849RP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 774 total ) | Next | Last >> |