 Found 713 hits with Last Name = 'krause' and Initial = 'j'
Found 713 hits with Last Name = 'krause' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573157
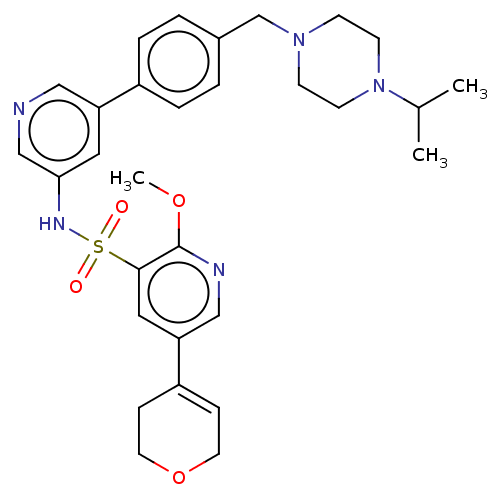
(CHEMBL4850297)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2CCN(CC2)C(C)C)cc1)C1=CCOCC1 |t:38| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573166
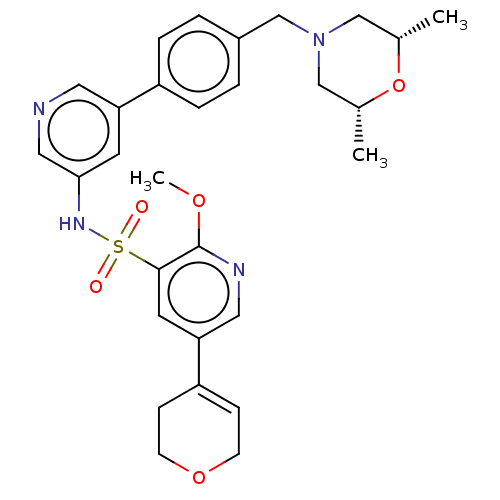
(CHEMBL4869783)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2C[C@H](C)O[C@H](C)C2)cc1)C1=CCOCC1 |r,t:37| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573177

(CHEMBL4167702)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)C)cc1F)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573167
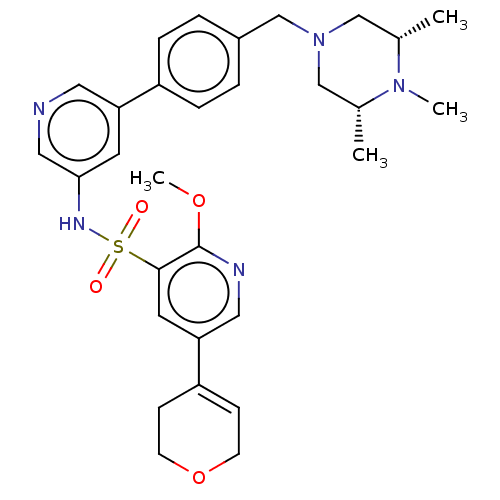
(CHEMBL4858875)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2C[C@H](C)N(C)[C@H](C)C2)cc1)C1=CCOCC1 |r,t:38| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573182
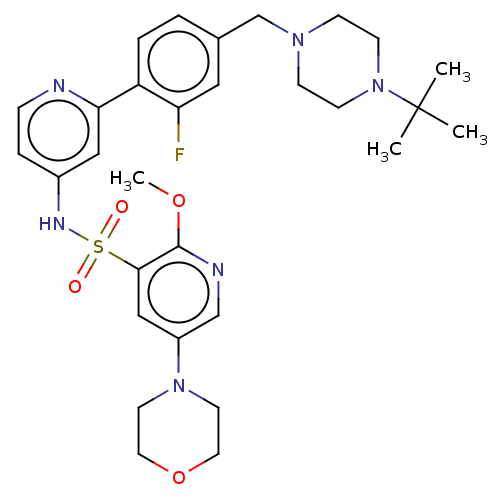
(CHEMBL4175571)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)(C)C)cc1F)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573181
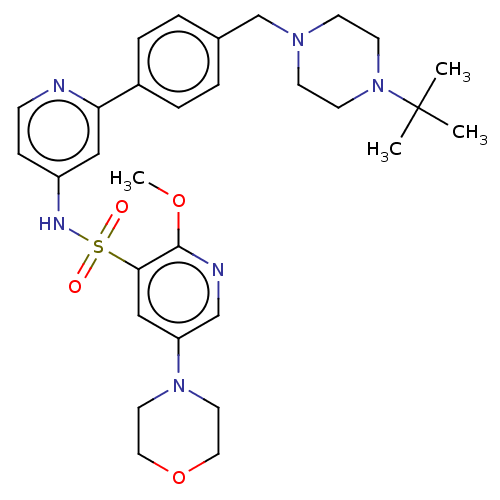
(CHEMBL4165185)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)(C)C)cc1)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573164

(CHEMBL4873390)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2CCN(CC2)C(C)C)nc1)C1=CCOCC1 |t:38| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573180
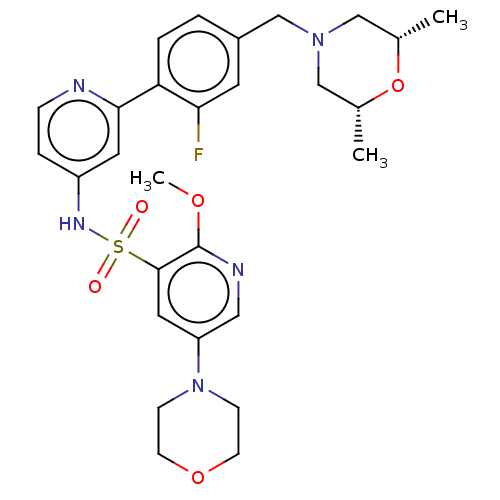
(CHEMBL4175737)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2C[C@H](C)O[C@H](C)C2)cc1F)N1CCOCC1 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573175
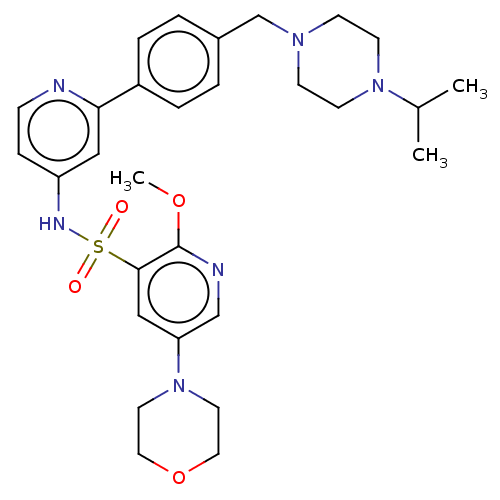
(CHEMBL4169192)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)C)cc1)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573176

(CHEMBL4173087)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)C)c(F)c1)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573179
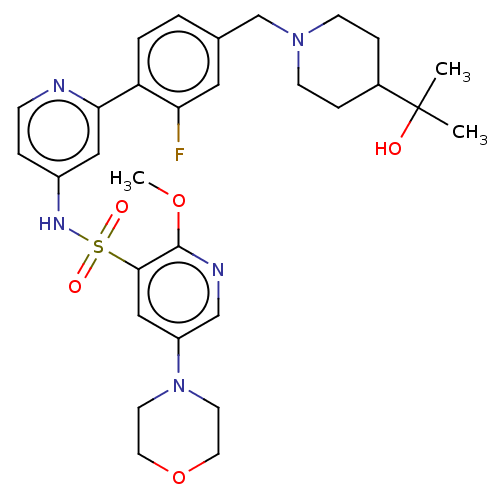
(CHEMBL4174874)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCC(CC2)C(C)(C)O)cc1F)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573178
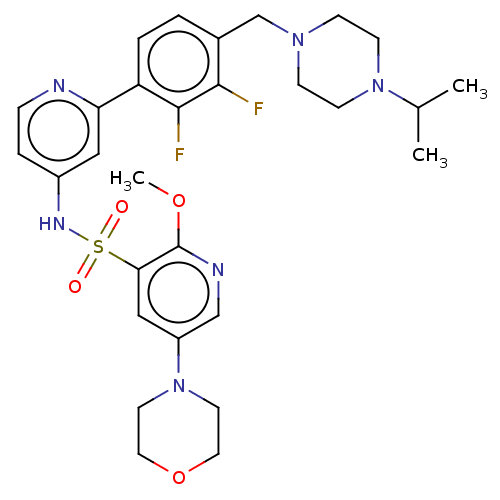
(CHEMBL4170075)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1ccnc(c1)-c1ccc(CN2CCN(CC2)C(C)C)c(F)c1F)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573170
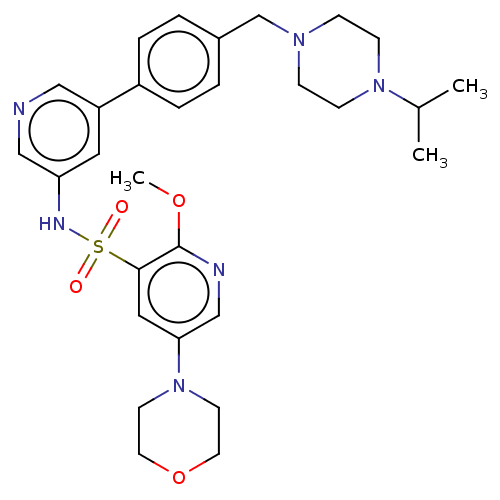
(CHEMBL4855234)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2CCN(CC2)C(C)C)cc1)N1CCOCC1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50573159
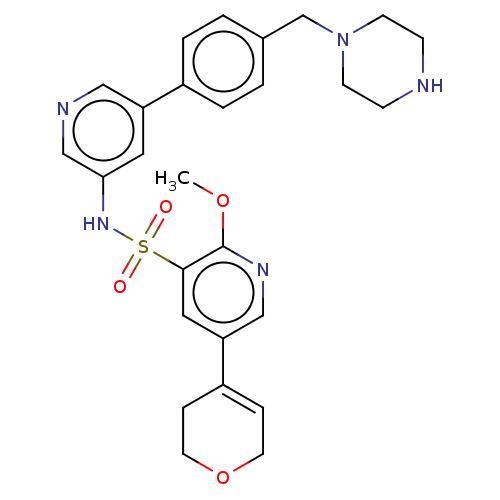
(CHEMBL4867203)Show SMILES COc1ncc(cc1S(=O)(=O)Nc1cncc(c1)-c1ccc(CN2CCNCC2)cc1)C1=CCOCC1 |t:35| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.126 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His6-tagged full length P110delta/full length untagged human p85alpha expressed in baculovirus infected Sf... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01102
BindingDB Entry DOI: 10.7270/Q2N87FKX |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50272695
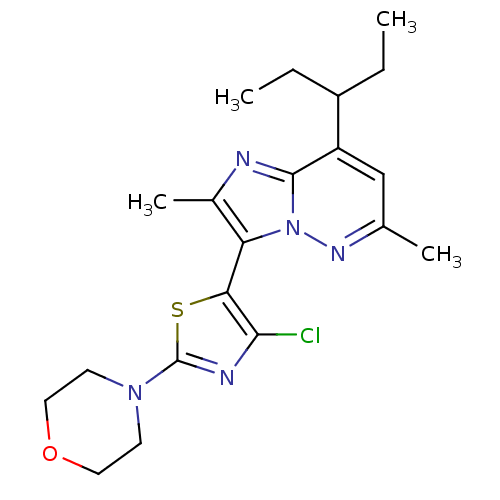
(3-(4-chloro-2-morpholinothiazol-5-yl)-2,6-dimethyl...)Show SMILES CCC(CC)c1cc(C)nn2c(c(C)nc12)-c1sc(nc1Cl)N1CCOCC1 |(8.98,-11.26,;8.98,-12.8,;7.65,-13.58,;6.32,-12.81,;4.99,-13.59,;7.66,-15.12,;6.34,-15.89,;6.33,-17.43,;5,-18.2,;7.67,-18.2,;9.01,-17.43,;10.49,-17.9,;11.4,-16.64,;12.94,-16.63,;10.48,-15.39,;9,-15.88,;10.98,-19.36,;12.45,-19.83,;12.46,-21.37,;11,-21.85,;10.09,-20.61,;8.55,-20.62,;13.71,-22.26,;13.55,-23.79,;14.79,-24.69,;16.2,-24.06,;16.36,-22.53,;15.11,-21.62,)| Show InChI InChI=1S/C20H26ClN5OS/c1-5-14(6-2)15-11-12(3)24-26-16(13(4)22-19(15)26)17-18(21)23-20(28-17)25-7-9-27-10-8-25/h11,14H,5-10H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from human CRF1 receptor expressed in IMR32 cells |
Bioorg Med Chem Lett 18: 4486-90 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.063
BindingDB Entry DOI: 10.7270/Q2NG4RJ4 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50272695

(3-(4-chloro-2-morpholinothiazol-5-yl)-2,6-dimethyl...)Show SMILES CCC(CC)c1cc(C)nn2c(c(C)nc12)-c1sc(nc1Cl)N1CCOCC1 |(8.98,-11.26,;8.98,-12.8,;7.65,-13.58,;6.32,-12.81,;4.99,-13.59,;7.66,-15.12,;6.34,-15.89,;6.33,-17.43,;5,-18.2,;7.67,-18.2,;9.01,-17.43,;10.49,-17.9,;11.4,-16.64,;12.94,-16.63,;10.48,-15.39,;9,-15.88,;10.98,-19.36,;12.45,-19.83,;12.46,-21.37,;11,-21.85,;10.09,-20.61,;8.55,-20.62,;13.71,-22.26,;13.55,-23.79,;14.79,-24.69,;16.2,-24.06,;16.36,-22.53,;15.11,-21.62,)| Show InChI InChI=1S/C20H26ClN5OS/c1-5-14(6-2)15-11-12(3)24-26-16(13(4)22-19(15)26)17-18(21)23-20(28-17)25-7-9-27-10-8-25/h11,14H,5-10H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CRF-1 receptor |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260806
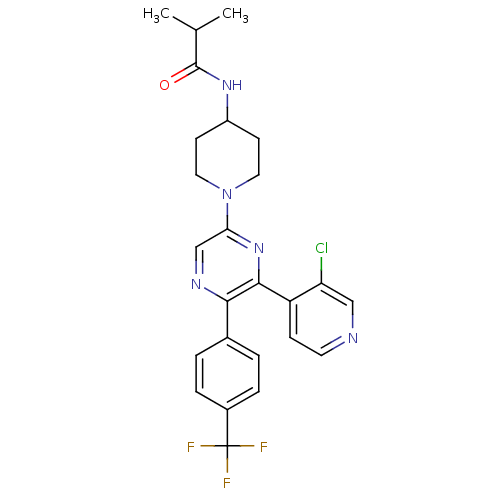
(CHEMBL497557 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(C)C(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C25H25ClF3N5O/c1-15(2)24(35)32-18-8-11-34(12-9-18)21-14-31-22(16-3-5-17(6-4-16)25(27,28)29)23(33-21)19-7-10-30-13-20(19)26/h3-7,10,13-15,18H,8-9,11-12H2,1-2H3,(H,32,35) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM21278

(5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl...)Show SMILES Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C22H21Cl3N4O/c1-14-20(22(30)27-28-11-3-2-4-12-28)26-29(19-10-9-17(24)13-18(19)25)21(14)15-5-7-16(23)8-6-15/h5-10,13H,2-4,11-12H2,1H3,(H,27,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260806

(CHEMBL497557 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(C)C(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C25H25ClF3N5O/c1-15(2)24(35)32-18-8-11-34(12-9-18)21-14-31-22(16-3-5-17(6-4-16)25(27,28)29)23(33-21)19-7-10-30-13-20(19)26/h3-7,10,13-15,18H,8-9,11-12H2,1-2H3,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM21278

(5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl...)Show SMILES Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C22H21Cl3N4O/c1-14-20(22(30)27-28-11-3-2-4-12-28)26-29(19-10-9-17(24)13-18(19)25)21(14)15-5-7-16(23)8-6-15/h5-10,13H,2-4,11-12H2,1H3,(H,27,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50202412
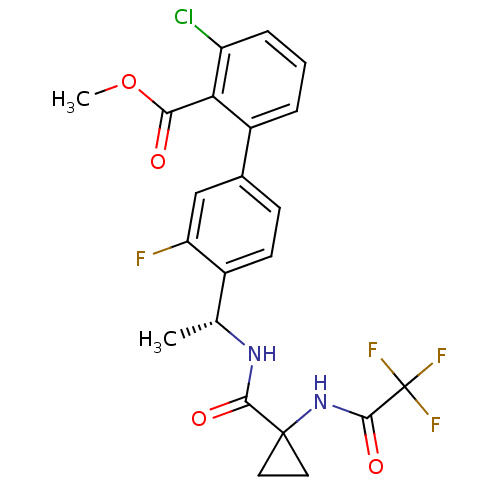
(3-Chloro-3'-fluoro-4'-((R)-1-{[1-(2,2,2-trifluoro-...)Show SMILES COC(=O)c1c(Cl)cccc1-c1ccc([C@@H](C)NC(=O)C2(CC2)NC(=O)C(F)(F)F)c(F)c1 |r| Show InChI InChI=1S/C22H19ClF4N2O4/c1-11(28-19(31)21(8-9-21)29-20(32)22(25,26)27)13-7-6-12(10-16(13)24)14-4-3-5-15(23)17(14)18(30)33-2/h3-7,10-11H,8-9H2,1-2H3,(H,28,31)(H,29,32)/t11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50127438
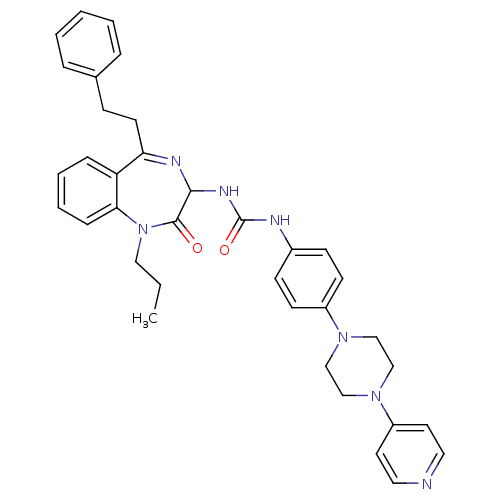
(1-(2-Oxo-5-phenethyl-1-propyl-2,3-dihydro-1H-benzo...)Show SMILES CCCN1c2ccccc2C(CCc2ccccc2)=NC(NC(=O)Nc2ccc(cc2)N2CCN(CC2)c2ccncc2)C1=O |c:20| Show InChI InChI=1S/C36H39N7O2/c1-2-22-43-33-11-7-6-10-31(33)32(17-12-27-8-4-3-5-9-27)39-34(35(43)44)40-36(45)38-28-13-15-29(16-14-28)41-23-25-42(26-24-41)30-18-20-37-21-19-30/h3-11,13-16,18-21,34H,2,12,17,22-26H2,1H3,(H2,38,40,45) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260767
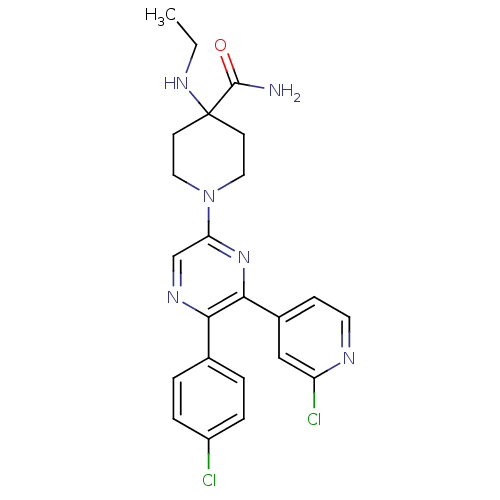
(1-(5-(4-chlorophenyl)-6-(2-chloropyridin-4-yl)pyra...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C23H24Cl2N6O/c1-2-29-23(22(26)32)8-11-31(12-9-23)19-14-28-20(15-3-5-17(24)6-4-15)21(30-19)16-7-10-27-18(25)13-16/h3-7,10,13-14,29H,2,8-9,11-12H2,1H3,(H2,26,32) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260805
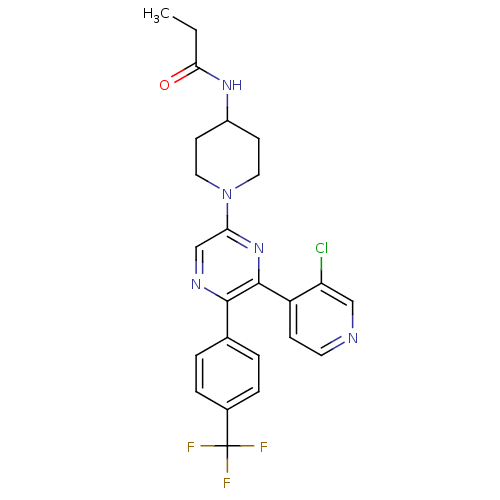
(CHEMBL524804 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CCC(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C24H23ClF3N5O/c1-2-21(34)31-17-8-11-33(12-9-17)20-14-30-22(15-3-5-16(6-4-15)24(26,27)28)23(32-20)18-7-10-29-13-19(18)25/h3-7,10,13-14,17H,2,8-9,11-12H2,1H3,(H,31,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260681
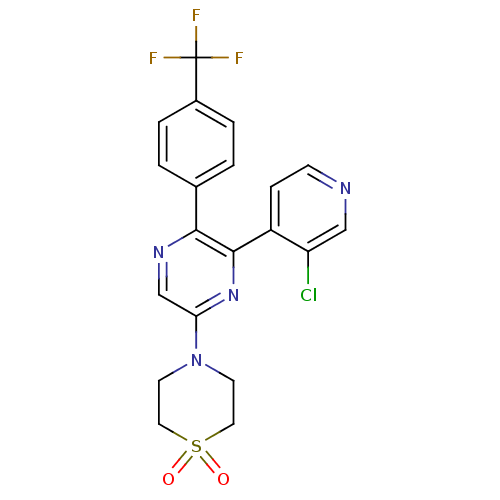
(4-[6-(3-Chloro-pyridin-4-yl)-5-(4-trifluoromethyl-...)Show SMILES FC(F)(F)c1ccc(cc1)-c1ncc(nc1-c1ccncc1Cl)N1CCS(=O)(=O)CC1 Show InChI InChI=1S/C20H16ClF3N4O2S/c21-16-11-25-6-5-15(16)19-18(13-1-3-14(4-2-13)20(22,23)24)26-12-17(27-19)28-7-9-31(29,30)10-8-28/h1-6,11-12H,7-10H2 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260681

(4-[6-(3-Chloro-pyridin-4-yl)-5-(4-trifluoromethyl-...)Show SMILES FC(F)(F)c1ccc(cc1)-c1ncc(nc1-c1ccncc1Cl)N1CCS(=O)(=O)CC1 Show InChI InChI=1S/C20H16ClF3N4O2S/c21-16-11-25-6-5-15(16)19-18(13-1-3-14(4-2-13)20(22,23)24)26-12-17(27-19)28-7-9-31(29,30)10-8-28/h1-6,11-12H,7-10H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260768
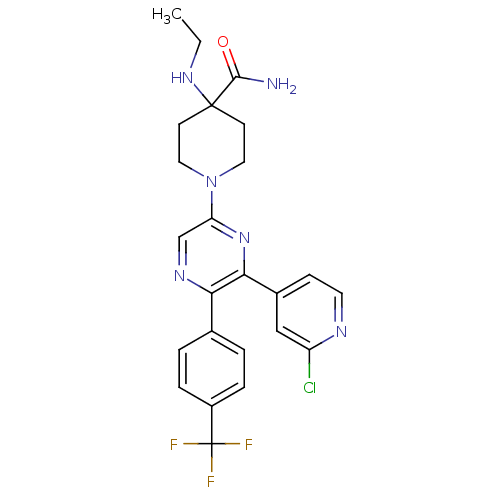
(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C24H24ClF3N6O/c1-2-32-23(22(29)35)8-11-34(12-9-23)19-14-31-20(15-3-5-17(6-4-15)24(26,27)28)21(33-19)16-7-10-30-18(25)13-16/h3-7,10,13-14,32H,2,8-9,11-12H2,1H3,(H2,29,35) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50132951
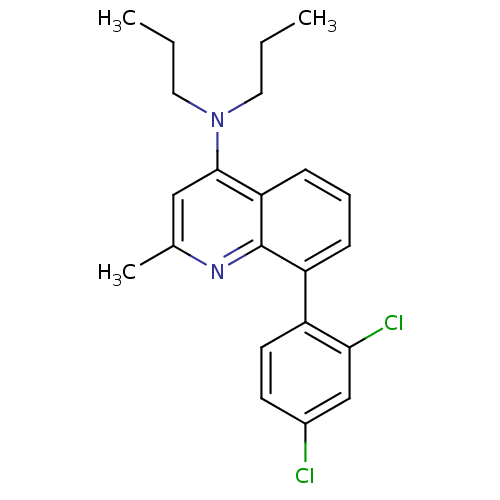
(8-(2,4-dichlorophenyl)-2-methyl-N,N-dipropylquinol...)Show SMILES CCCN(CCC)c1cc(C)nc2c(cccc12)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C22H24Cl2N2/c1-4-11-26(12-5-2)21-13-15(3)25-22-18(7-6-8-19(21)22)17-10-9-16(23)14-20(17)24/h6-10,13-14H,4-5,11-12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from CRF1 receptor expressed in human IMR32 cells |
Bioorg Med Chem Lett 18: 891-6 (2008)
Article DOI: 10.1016/j.bmcl.2007.12.050
BindingDB Entry DOI: 10.7270/Q28S4PPS |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50132951

(8-(2,4-dichlorophenyl)-2-methyl-N,N-dipropylquinol...)Show SMILES CCCN(CCC)c1cc(C)nc2c(cccc12)-c1ccc(Cl)cc1Cl Show InChI InChI=1S/C22H24Cl2N2/c1-4-11-26(12-5-2)21-13-15(3)25-22-18(7-6-8-19(21)22)17-10-9-16(23)14-20(17)24/h6-10,13-14H,4-5,11-12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CRF-1 receptor |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260682
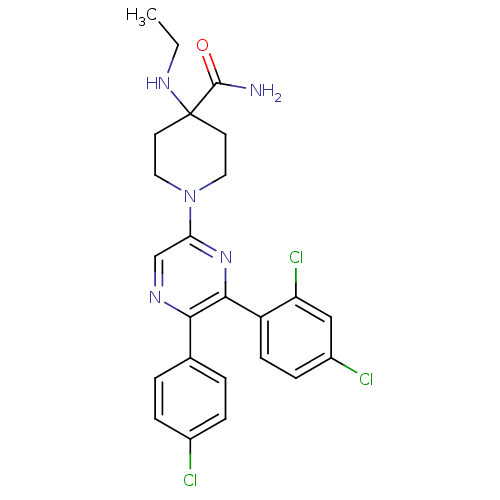
(1-(5-(4-chlorophenyl)-6-(2,4-dichlorophenyl)pyrazi...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccc(Cl)cc1Cl)C(N)=O Show InChI InChI=1S/C24H24Cl3N5O/c1-2-30-24(23(28)33)9-11-32(12-10-24)20-14-29-21(15-3-5-16(25)6-4-15)22(31-20)18-8-7-17(26)13-19(18)27/h3-8,13-14,30H,2,9-12H2,1H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(RAT) | BDBM50127438

(1-(2-Oxo-5-phenethyl-1-propyl-2,3-dihydro-1H-benzo...)Show SMILES CCCN1c2ccccc2C(CCc2ccccc2)=NC(NC(=O)Nc2ccc(cc2)N2CCN(CC2)c2ccncc2)C1=O |c:20| Show InChI InChI=1S/C36H39N7O2/c1-2-22-43-33-11-7-6-10-31(33)32(17-12-27-8-4-3-5-9-27)39-34(35(43)44)40-36(45)38-28-13-15-29(16-14-28)41-23-25-42(26-24-41)30-18-20-37-21-19-30/h3-11,13-16,18-21,34H,2,12,17,22-26H2,1H3,(H2,38,40,45) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.920 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348782
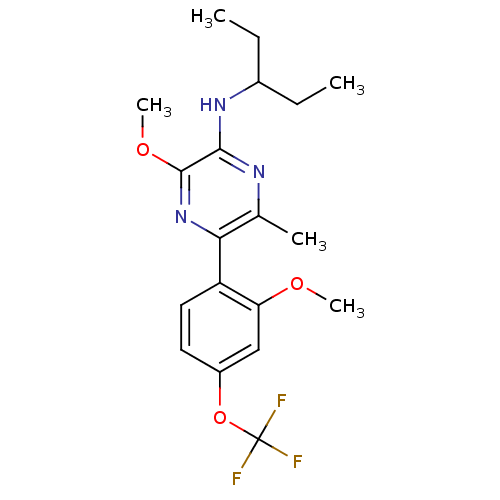
(CHEMBL1807053)Show SMILES CCC(CC)Nc1nc(C)c(nc1OC)-c1ccc(OC(F)(F)F)cc1OC Show InChI InChI=1S/C19H24F3N3O3/c1-6-12(7-2)24-17-18(27-5)25-16(11(3)23-17)14-9-8-13(10-15(14)26-4)28-19(20,21)22/h8-10,12H,6-7H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348778
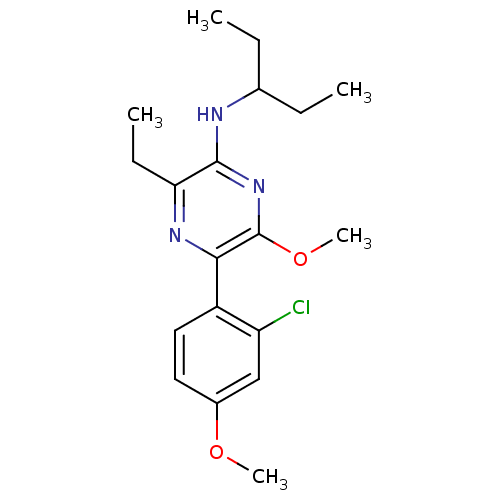
(CHEMBL1807058)Show InChI InChI=1S/C19H26ClN3O2/c1-6-12(7-2)21-18-16(8-3)22-17(19(23-18)25-5)14-10-9-13(24-4)11-15(14)20/h9-12H,6-8H2,1-5H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260803
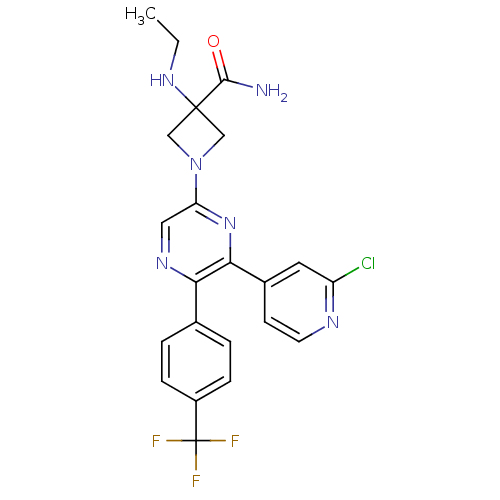
(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CN(C1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C22H20ClF3N6O/c1-2-30-21(20(27)33)11-32(12-21)17-10-29-18(13-3-5-15(6-4-13)22(24,25)26)19(31-17)14-7-8-28-16(23)9-14/h3-10,30H,2,11-12H2,1H3,(H2,27,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348829
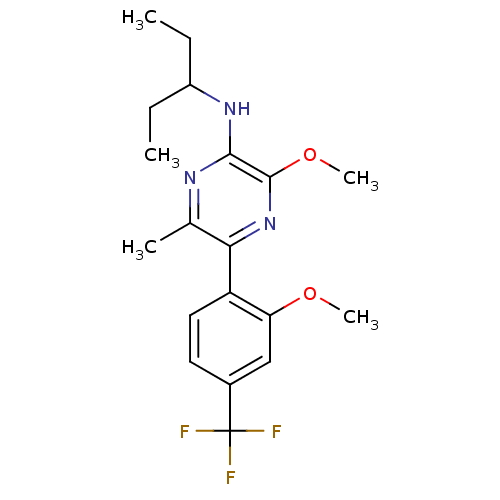
(CHEMBL1807054)Show SMILES CCC(CC)Nc1nc(C)c(nc1OC)-c1ccc(cc1OC)C(F)(F)F Show InChI InChI=1S/C19H24F3N3O2/c1-6-13(7-2)24-17-18(27-5)25-16(11(3)23-17)14-9-8-12(19(20,21)22)10-15(14)26-4/h8-10,13H,6-7H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348783
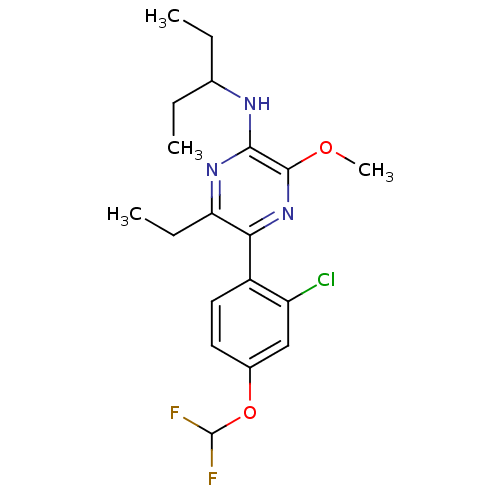
(CHEMBL1807052)Show SMILES CCC(CC)Nc1nc(CC)c(nc1OC)-c1ccc(OC(F)F)cc1Cl Show InChI InChI=1S/C19H24ClF2N3O2/c1-5-11(6-2)23-17-18(26-4)25-16(15(7-3)24-17)13-9-8-12(10-14(13)20)27-19(21)22/h8-11,19H,5-7H2,1-4H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260718

(1-(6-(2-chlorophenyl)-5-(4-chlorophenyl)pyrazin-2-...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccccc1Cl)C(N)=O Show InChI InChI=1S/C24H25Cl2N5O/c1-2-29-24(23(27)32)11-13-31(14-12-24)20-15-28-21(16-7-9-17(25)10-8-16)22(30-20)18-5-3-4-6-19(18)26/h3-10,15,29H,2,11-14H2,1H3,(H2,27,32) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.66 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260717
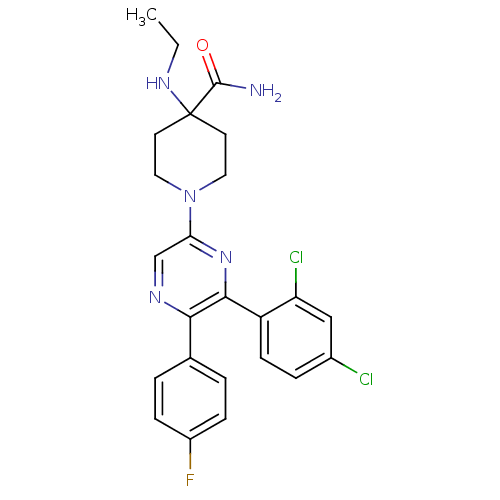
(1-(6-(2,4-dichlorophenyl)-5-(4-fluorophenyl)pyrazi...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(F)cc2)c(n1)-c1ccc(Cl)cc1Cl)C(N)=O Show InChI InChI=1S/C24H24Cl2FN5O/c1-2-30-24(23(28)33)9-11-32(12-10-24)20-14-29-21(15-3-6-17(27)7-4-15)22(31-20)18-8-5-16(25)13-19(18)26/h3-8,13-14,30H,2,9-12H2,1H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348786
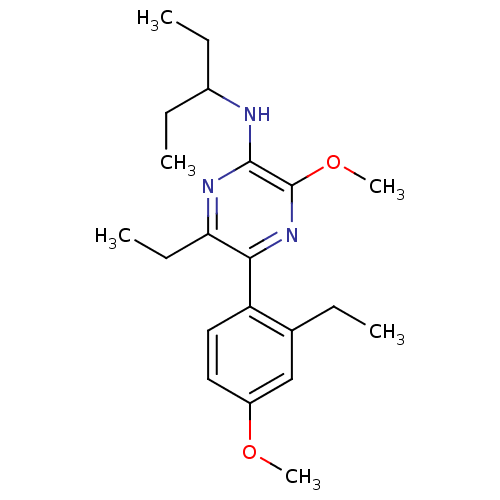
(CHEMBL1807049)Show InChI InChI=1S/C21H31N3O2/c1-7-14-13-16(25-5)11-12-17(14)19-18(10-4)23-20(21(24-19)26-6)22-15(8-2)9-3/h11-13,15H,7-10H2,1-6H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260804
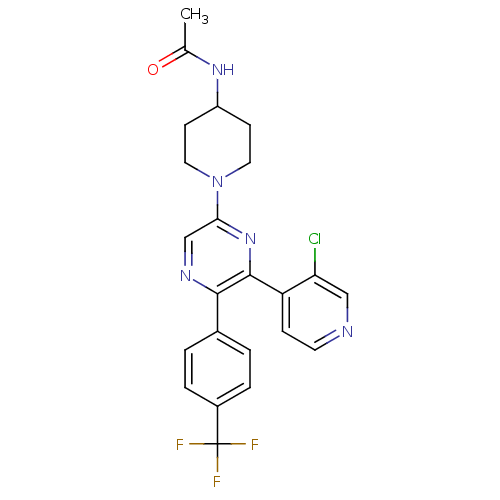
(CHEMBL497556 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C23H21ClF3N5O/c1-14(33)30-17-7-10-32(11-8-17)20-13-29-21(15-2-4-16(5-3-15)23(25,26)27)22(31-20)18-6-9-28-12-19(18)24/h2-6,9,12-13,17H,7-8,10-11H2,1H3,(H,30,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.97 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50084875
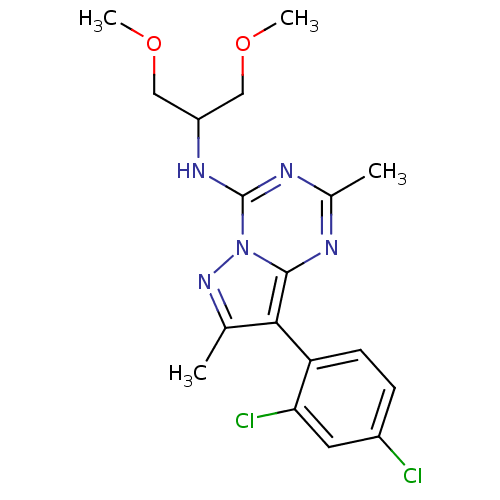
(8-(2,4-dichlorophenyl)-N-(1,3-dimethoxypropan-2-yl...)Show SMILES COCC(COC)Nc1nc(C)nc2c(c(C)nn12)-c1ccc(Cl)cc1Cl |(-3.04,-6.69,;-3.03,-8.23,;-4.37,-9,;-4.37,-10.54,;-3.03,-11.31,;-1.7,-10.54,;-1.7,-9.01,;-5.7,-11.31,;-5.7,-12.85,;-7.03,-13.63,;-7.03,-15.17,;-8.36,-15.94,;-5.7,-15.93,;-4.37,-15.17,;-2.91,-15.64,;-2,-14.4,;-.46,-14.4,;-2.91,-13.15,;-4.37,-13.63,;-2.18,-17.01,;-3.01,-18.3,;-2.29,-19.67,;-.75,-19.73,;-.03,-21.08,;.06,-18.41,;-.65,-17.05,;.16,-15.75,)| Show InChI InChI=1S/C18H21Cl2N5O2/c1-10-16(14-6-5-12(19)7-15(14)20)17-21-11(2)22-18(25(17)24-10)23-13(8-26-3)9-27-4/h5-7,13H,8-9H2,1-4H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CRF-1 receptor |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348828
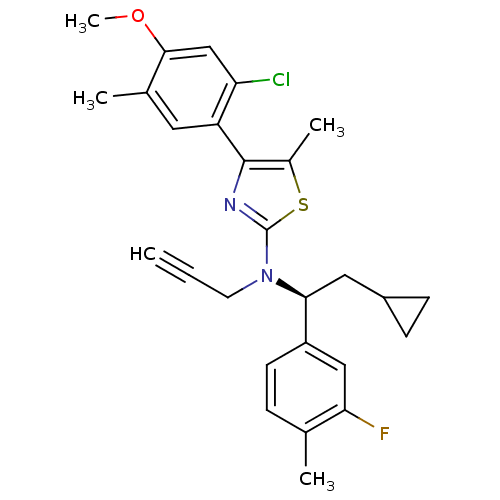
(CHEMBL1628268 | SR-125543A)Show SMILES COc1cc(Cl)c(cc1C)-c1nc(sc1C)N(CC#C)[C@@H](CC1CC1)c1ccc(C)c(F)c1 |r| Show InChI InChI=1S/C27H28ClFN2OS/c1-6-11-31(24(13-19-8-9-19)20-10-7-16(2)23(29)14-20)27-30-26(18(4)33-27)21-12-17(3)25(32-5)15-22(21)28/h1,7,10,12,14-15,19,24H,8-9,11,13H2,2-5H3/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from human CRF1 receptor expressed in IMR32 cells |
Bioorg Med Chem Lett 18: 4486-90 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.063
BindingDB Entry DOI: 10.7270/Q2NG4RJ4 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM29442
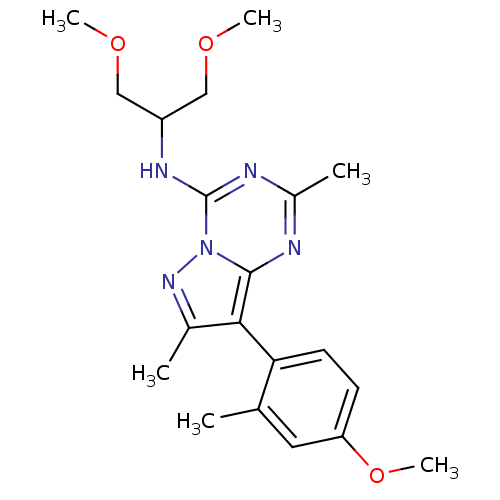
(CHEMBL524616 | Pyrazolo[1,5-a]-1,3,5-triazine, 12-...)Show SMILES COCC(COC)Nc1nc(C)nc2c(c(C)nn12)-c1ccc(OC)cc1C |(2.32,11.47,;1.92,9.99,;.43,9.59,;-.06,8.13,;.95,6.97,;2.44,7.37,;3.53,6.28,;-1.57,7.83,;-2.07,6.37,;-3.58,6.06,;-4.07,4.61,;-5.58,4.3,;-3.05,3.45,;-1.54,3.75,;-.31,2.83,;.95,3.72,;2.41,3.23,;.49,5.19,;-1.05,5.21,;-.33,1.29,;-1.66,.52,;-1.66,-1.02,;-.33,-1.79,;-.33,-3.33,;1.01,-4.1,;1.01,-1.02,;1.01,.52,;2.34,1.29,)| Show InChI InChI=1S/C20H27N5O3/c1-12-9-16(28-6)7-8-17(12)18-13(2)24-25-19(18)21-14(3)22-20(25)23-15(10-26-4)11-27-5/h7-9,15H,10-11H2,1-6H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from human CRF1 receptor expressed in IMR32 cells |
Bioorg Med Chem Lett 18: 4486-90 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.063
BindingDB Entry DOI: 10.7270/Q2NG4RJ4 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50272638
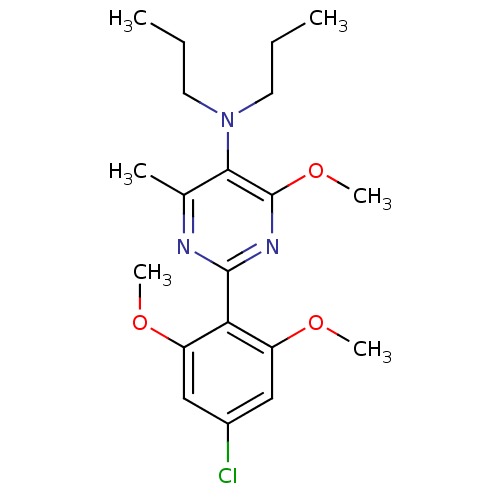
(2-(4-chloro-2,6-dimethoxyphenyl)-4-methoxy-6-methy...)Show SMILES CCCN(CCC)c1c(C)nc(nc1OC)-c1c(OC)cc(Cl)cc1OC Show InChI InChI=1S/C20H28ClN3O3/c1-7-9-24(10-8-2)18-13(3)22-19(23-20(18)27-6)17-15(25-4)11-14(21)12-16(17)26-5/h11-12H,7-10H2,1-6H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from human CRF1 receptor expressed in IMR32 cells |
Bioorg Med Chem Lett 18: 4486-90 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.063
BindingDB Entry DOI: 10.7270/Q2NG4RJ4 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348828

(CHEMBL1628268 | SR-125543A)Show SMILES COc1cc(Cl)c(cc1C)-c1nc(sc1C)N(CC#C)[C@@H](CC1CC1)c1ccc(C)c(F)c1 |r| Show InChI InChI=1S/C27H28ClFN2OS/c1-6-11-31(24(13-19-8-9-19)20-10-7-16(2)23(29)14-20)27-30-26(18(4)33-27)21-12-17(3)25(32-5)15-22(21)28/h1,7,10,12,14-15,19,24H,8-9,11,13H2,2-5H3/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CRF-1 receptor |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50054245
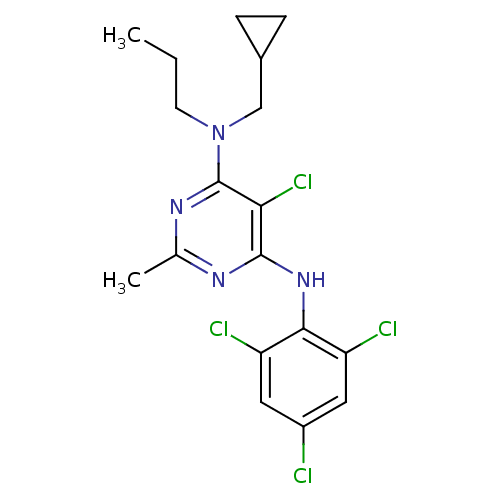
(5-Chloro-N-cyclopropylmethyl-2-methyl-N-propyl-N''...)Show SMILES CCCN(CC1CC1)c1nc(C)nc(Nc2c(Cl)cc(Cl)cc2Cl)c1Cl Show InChI InChI=1S/C18H20Cl4N4/c1-3-6-26(9-11-4-5-11)18-15(22)17(23-10(2)24-18)25-16-13(20)7-12(19)8-14(16)21/h7-8,11H,3-6,9H2,1-2H3,(H,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CRF-1 receptor |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260803

(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CN(C1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C22H20ClF3N6O/c1-2-30-21(20(27)33)11-32(12-21)17-10-29-18(13-3-5-15(6-4-13)22(24,25)26)19(31-17)14-7-8-28-16(23)9-14/h3-10,30H,2,11-12H2,1H3,(H2,27,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.06 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348784
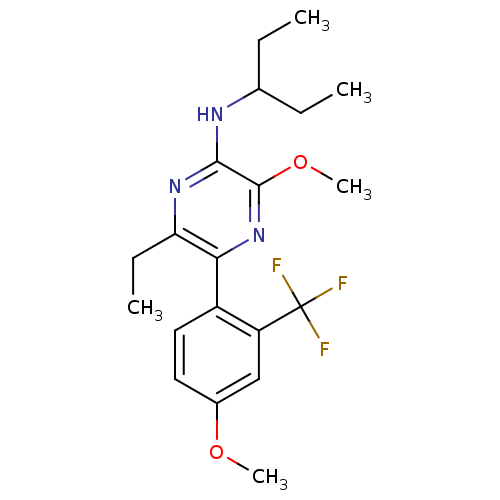
(CHEMBL1807051)Show SMILES CCC(CC)Nc1nc(CC)c(nc1OC)-c1ccc(OC)cc1C(F)(F)F Show InChI InChI=1S/C20H26F3N3O2/c1-6-12(7-2)24-18-19(28-5)26-17(16(8-3)25-18)14-10-9-13(27-4)11-15(14)20(21,22)23/h9-12H,6-8H2,1-5H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50348777

(CHEMBL1807059)Show SMILES CCC(CC)Nc1nc(OC)c(nc1CC)-c1ccc(OC(F)F)cc1Cl Show InChI InChI=1S/C19H24ClF2N3O2/c1-5-11(6-2)23-17-15(7-3)24-16(18(25-17)26-4)13-9-8-12(10-14(13)20)27-19(21)22/h8-11,19H,5-7H2,1-4H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-sauvagine from human CRF-1 receptor expressed in human IMR-32 cells after 2 hrs by scintillation counting |
J Med Chem 54: 4187-206 (2011)
Article DOI: 10.1021/jm200365y
BindingDB Entry DOI: 10.7270/Q2XS5VR1 |
More data for this
Ligand-Target Pair | |
Corticotropin-releasing factor receptor 1
(Homo sapiens (Human)) | BDBM50058163
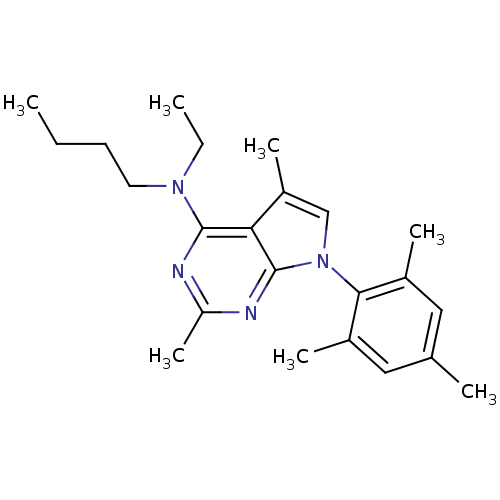
(Butyl-[2,5-dimethyl-7-(2,4,6-trimethyl-phenyl)-7H-...)Show SMILES CCCCN(CC)c1nc(C)nc2n(cc(C)c12)-c1c(C)cc(C)cc1C |(-10.62,2.69,;-9.29,3.46,;-7.95,2.7,;-6.62,3.48,;-5.28,2.71,;-3.95,3.49,;-3.96,5.03,;-5.27,1.17,;-6.6,.4,;-6.6,-1.14,;-7.94,-1.91,;-5.27,-1.91,;-3.92,-1.14,;-2.44,-1.61,;-1.54,-.35,;-2.46,.9,;-1.99,2.37,;-3.93,.41,;-1.96,-3.07,;-2.99,-4.22,;-4.49,-3.91,;-2.5,-5.68,;-.99,-5.99,;-.51,-7.46,;.03,-4.83,;-.46,-3.38,;.56,-2.22,)| Show InChI InChI=1S/C23H32N4/c1-8-10-11-26(9-2)22-20-18(6)14-27(23(20)25-19(7)24-22)21-16(4)12-15(3)13-17(21)5/h12-14H,8-11H2,1-7H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]sauvagine from CRF1 receptor expressed in human IMR32 cells |
Bioorg Med Chem Lett 18: 891-6 (2008)
Article DOI: 10.1016/j.bmcl.2007.12.050
BindingDB Entry DOI: 10.7270/Q28S4PPS |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data