

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Androgen receptor (Homo sapiens (Human)) | BDBM50157823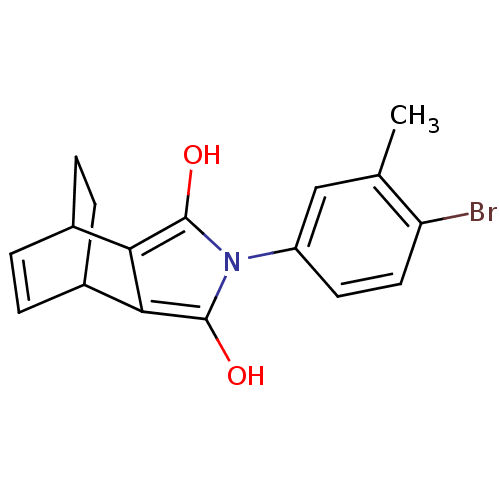 ((2R,6S)-4-(4-Bromo-3-methyl-phenyl)-4-aza-tricyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity for mutant T877A Androgen receptor in human LNCaP cells | Bioorg Med Chem Lett 15: 271-6 (2004) Article DOI: 10.1016/j.bmcl.2004.10.085 BindingDB Entry DOI: 10.7270/Q2N58KVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29321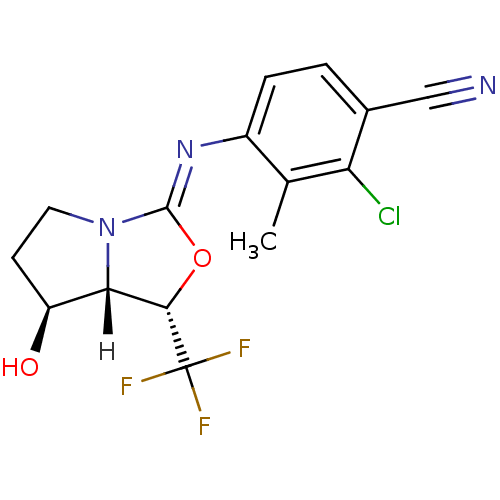 (oxazolidin-2-imine, 6d) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.200 | -54.8 | n/a | n/a | 19 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18161 ((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.300 | -53.8 | n/a | n/a | 2.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29319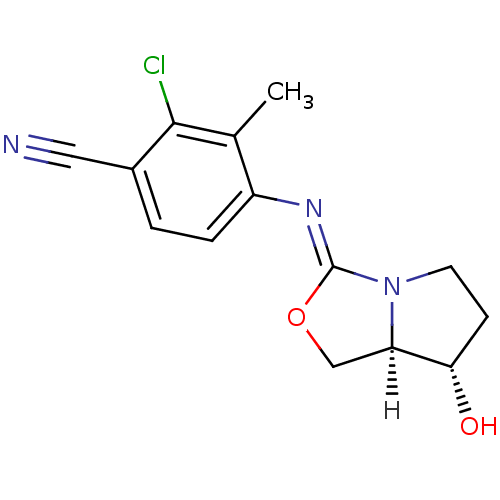 (oxazolidin-2-imine, 6b) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.300 | -53.8 | n/a | n/a | 14 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29323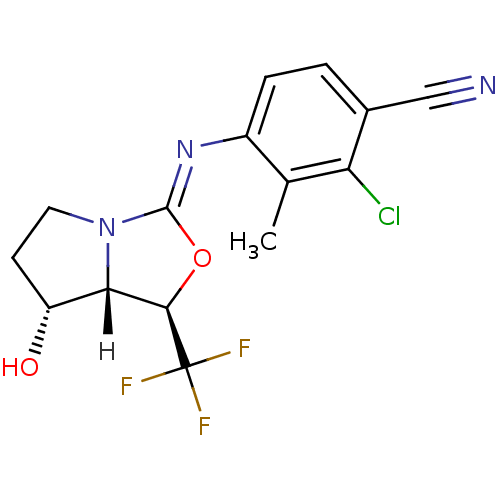 (oxazolidin-2-imine, 6f) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.300 | -53.8 | n/a | n/a | 1.40 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29320 (BMS-665139 | oxazolidin-2-imine, 6c) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.300 | -53.8 | n/a | n/a | 0.200 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18188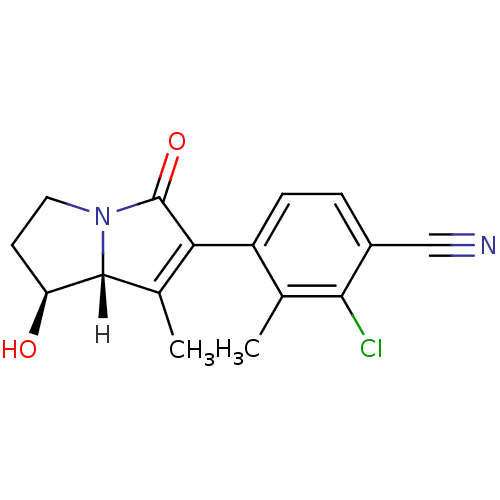 (4-[(1S,7aR)-1-hydroxy-7-methyl-5-oxo-2,3,5,7a-tetr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents | Article PubMed | 0.5 | -52.6 | n/a | n/a | 2 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50157826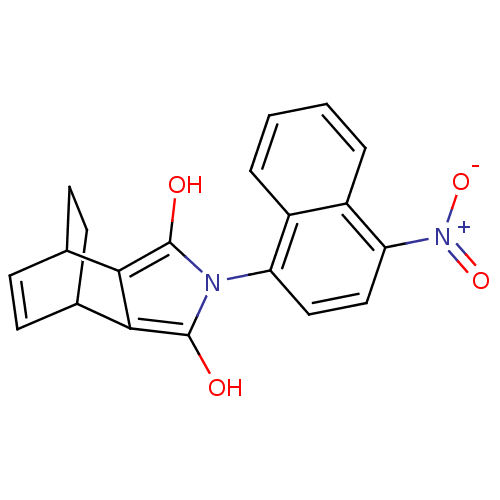 ((2S,6R)-4-(4-Nitro-naphthalen-1-yl)-4-aza-tricyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity for mutant T877A Androgen receptor in human LNCaP cells | Bioorg Med Chem Lett 15: 271-6 (2004) Article DOI: 10.1016/j.bmcl.2004.10.085 BindingDB Entry DOI: 10.7270/Q2N58KVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29324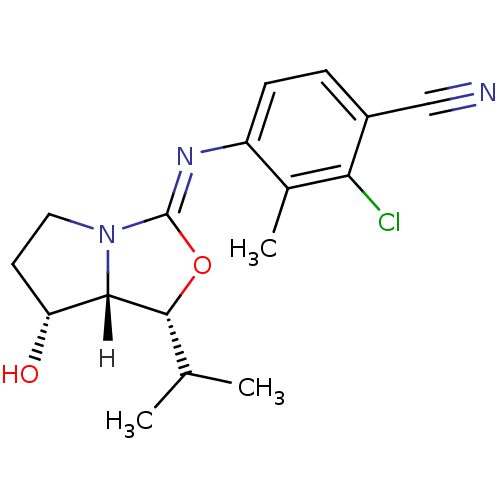 (oxazolidin-2-imine, 6g) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | -51.7 | n/a | n/a | 3.70 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205111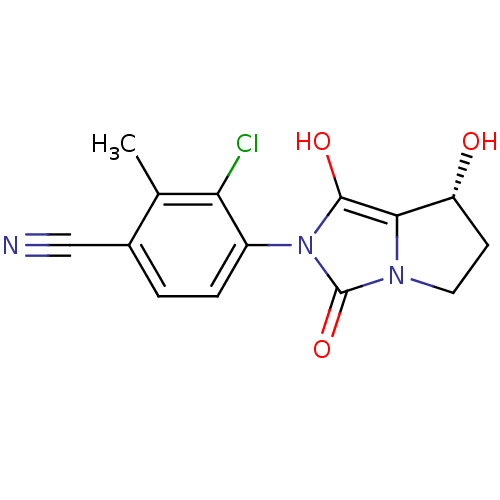 (3-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18183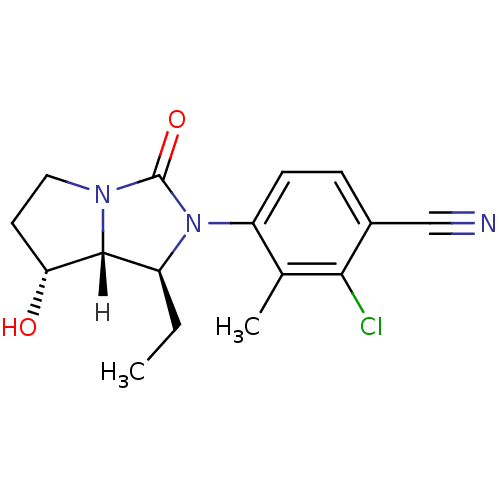 (4-[(1S,7R,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | -51.7 | n/a | n/a | 2.60 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29318 (oxazolidin-2-imine, 6a) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.800 | -51.4 | n/a | n/a | 4.80 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18177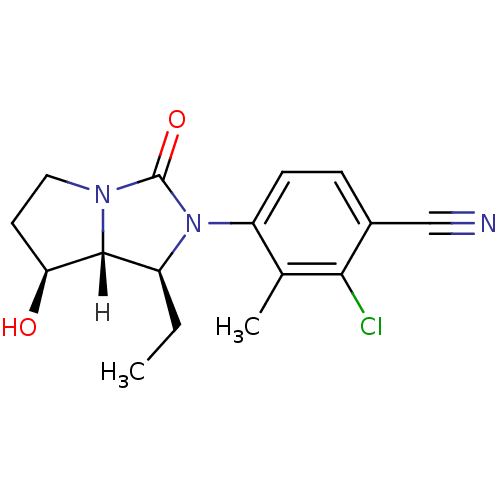 (4-[(1S,7S,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.900 | -51.1 | n/a | n/a | 1.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18178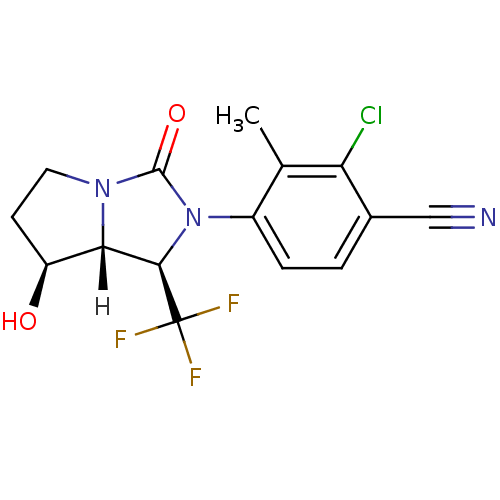 (4-[(1R,7S,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.900 | -51.1 | n/a | n/a | 2.5 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18162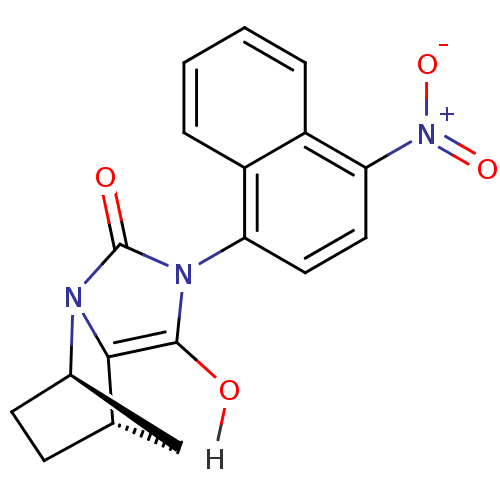 ((1R,6R,7S)-4-(4-nitronaphthalen-1-yl)-2,4-diazatri...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Displacement of [3H]-DHT from androgen receptor of human MDA-453 cells | Bioorg Med Chem Lett 14: 6107-11 (2004) Article DOI: 10.1016/j.bmcl.2004.09.049 BindingDB Entry DOI: 10.7270/Q2T1533W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18162 ((1R,6R,7S)-4-(4-nitronaphthalen-1-yl)-2,4-diazatri...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | -50.9 | n/a | n/a | 385 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50372090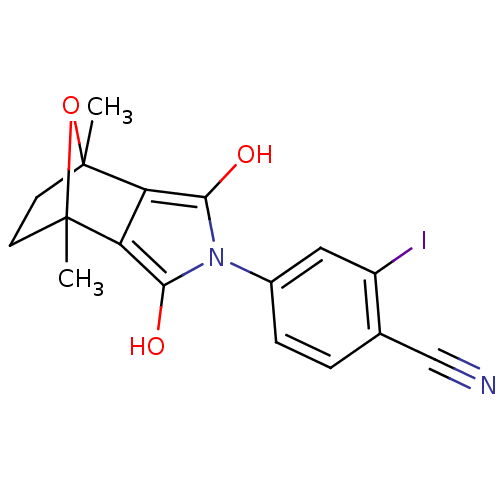 (CHEMBL403668) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]DHT from human androgen receptor in MDA453 cells | Bioorg Med Chem Lett 18: 1910-5 (2008) Article DOI: 10.1016/j.bmcl.2008.02.006 BindingDB Entry DOI: 10.7270/Q2DJ5GGT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50122646 (CHEMBL3623125) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol Myers-Squibb Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysis | ACS Med Chem Lett 6: 908-12 (2015) Article DOI: 10.1021/acsmedchemlett.5b00173 BindingDB Entry DOI: 10.7270/Q2QV3P96 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18184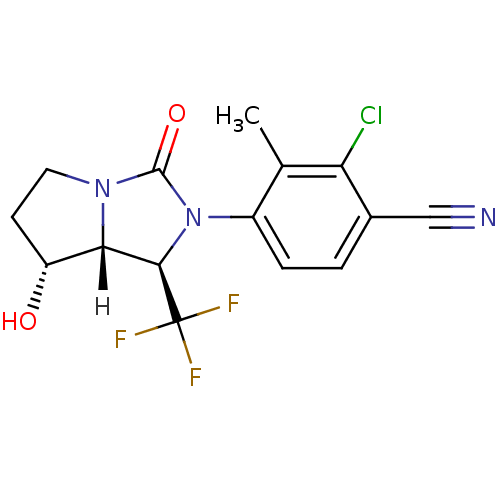 (4-[(1R,7R,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | -50.9 | n/a | n/a | 2.90 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205116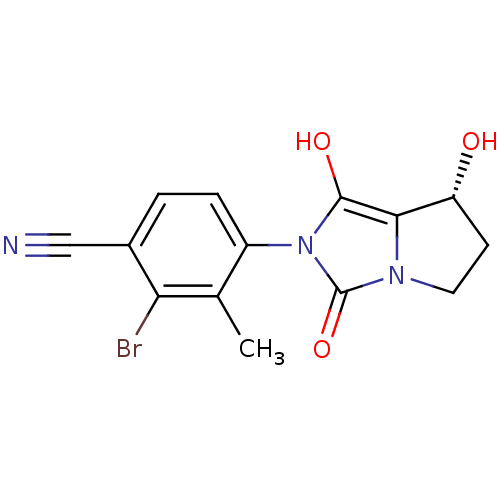 (2-bromo-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205114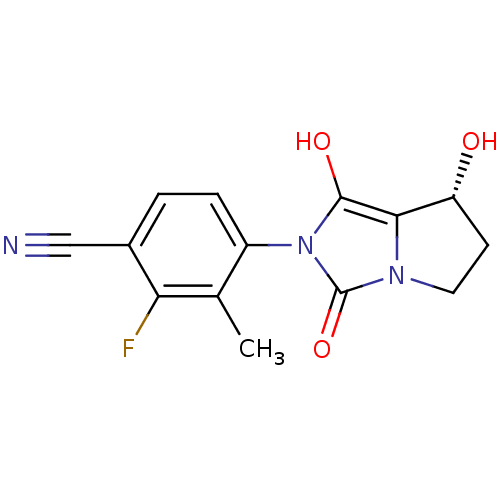 (2-fluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18173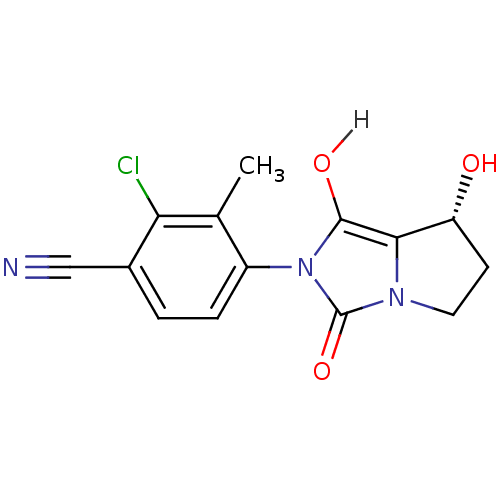 (4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 1.40 | -50.0 | n/a | n/a | 0.700 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18176 (4-[(1S,7S,7aR)-7-hydroxy-1-methyl-3-oxo-hexahydro-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.5 | -49.9 | n/a | n/a | 5.20 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18165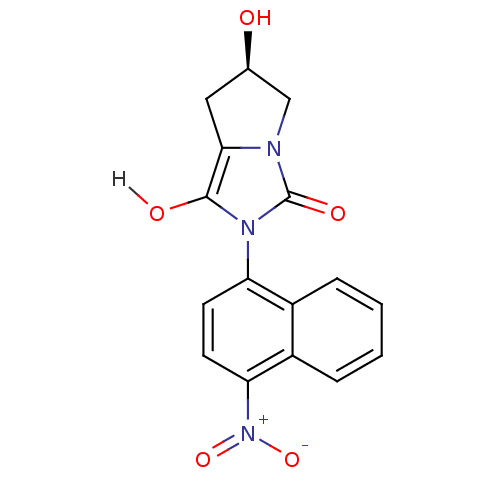 ((6R)-6-hydroxy-2-(4-nitronaphthalen-1-yl)-hexahydr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.5 | -49.9 | n/a | n/a | 320 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205096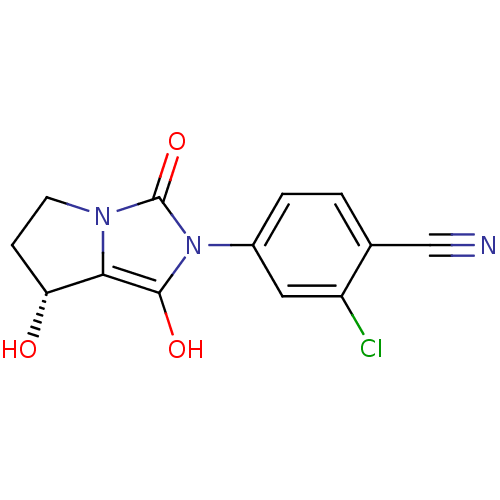 (2-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18169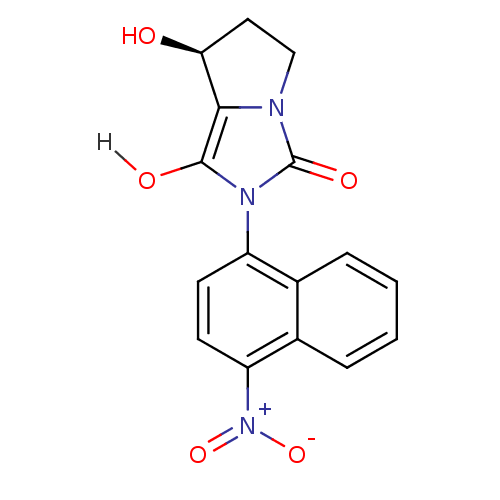 ((7S,7aR)-7-hydroxy-2-(4-nitronaphthalen-1-yl)-hexa...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.5 | -49.9 | n/a | n/a | 281 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18180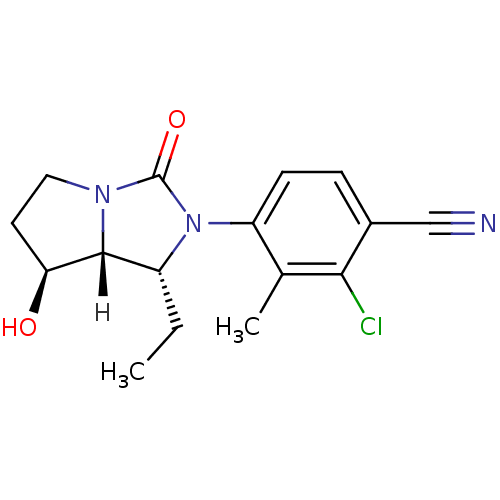 (4-[(1R,7S,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.60 | -49.7 | n/a | n/a | 7.10 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18176 (4-[(1S,7S,7aR)-7-hydroxy-1-methyl-3-oxo-hexahydro-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.60 | -49.7 | n/a | n/a | 5.40 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50122650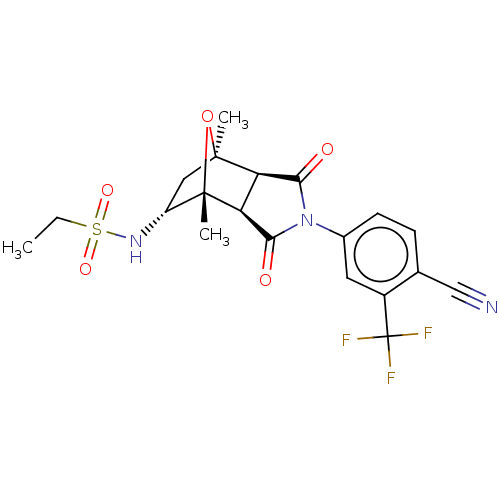 (CHEMBL3623127) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol Myers-Squibb Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysis | ACS Med Chem Lett 6: 908-12 (2015) Article DOI: 10.1021/acsmedchemlett.5b00173 BindingDB Entry DOI: 10.7270/Q2QV3P96 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18164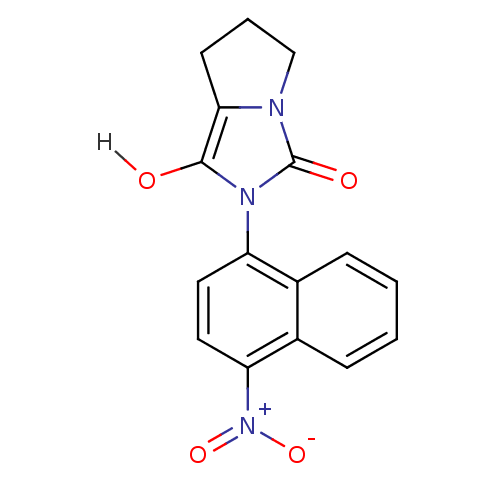 (2-(4-nitronaphthalen-1-yl)-hexahydro-1H-pyrrolo[1,...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.70 | -49.6 | n/a | n/a | 270 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29326 (guanidine derivative, 12) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.90 | -49.3 | n/a | n/a | 44 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18181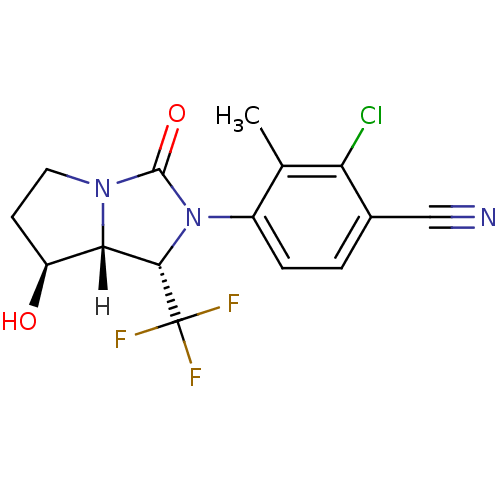 (4-[(1S,7S,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1.90 | -49.3 | n/a | n/a | 48 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18174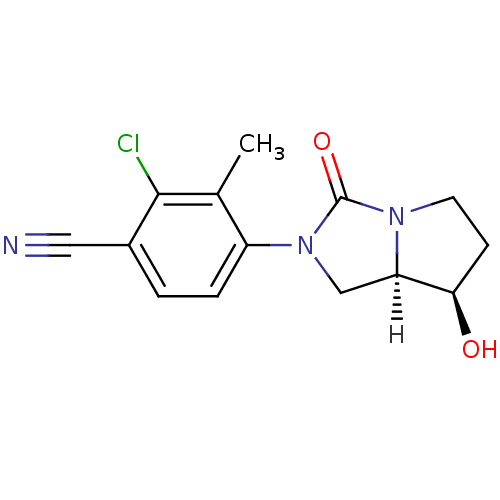 (4-[(7R,7aR)-7-hydroxy-3-oxo-hexahydro-1H-pyrrolo[1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | -49.2 | n/a | n/a | 15 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM35909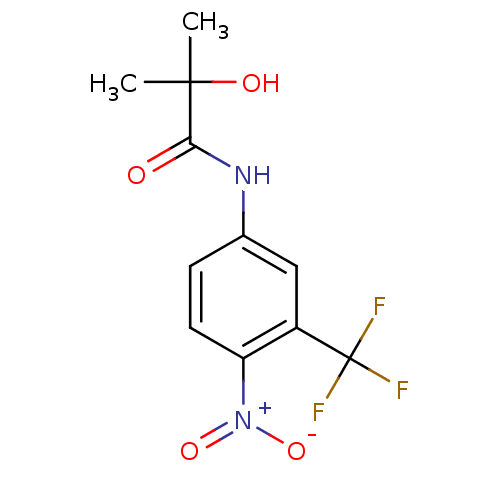 (2-Hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-ph...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity for mutant T877A Androgen receptor in human LNCaP cells | Bioorg Med Chem Lett 15: 271-6 (2004) Article DOI: 10.1016/j.bmcl.2004.10.085 BindingDB Entry DOI: 10.7270/Q2N58KVH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50157825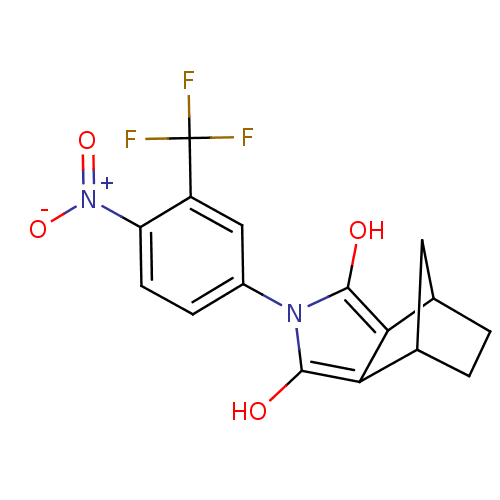 ((2R,6S)-4-(4-Nitro-3-trifluoromethyl-phenyl)-4-aza...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity for mutant T877A Androgen receptor in human LNCaP cells | Bioorg Med Chem Lett 15: 271-6 (2004) Article DOI: 10.1016/j.bmcl.2004.10.085 BindingDB Entry DOI: 10.7270/Q2N58KVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50122647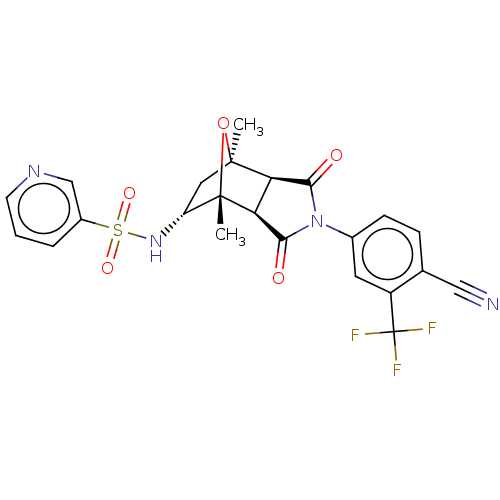 (CHEMBL3623126) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol Myers-Squibb Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]-DHT from androgen receptor in human MDA-MB-453 cells after 90 mins by TopCount analysis | ACS Med Chem Lett 6: 908-12 (2015) Article DOI: 10.1021/acsmedchemlett.5b00173 BindingDB Entry DOI: 10.7270/Q2QV3P96 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18173 (4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18168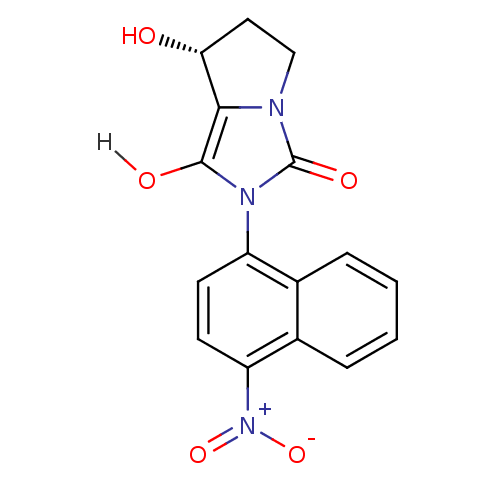 ((7R,7aS)-7-hydroxy-2-(4-nitronaphthalen-1-yl)-hexa...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2.10 | -49.0 | n/a | n/a | 1.5 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 49: 7596-9 (2006) Article DOI: 10.1021/jm061101w BindingDB Entry DOI: 10.7270/Q2862DQ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18175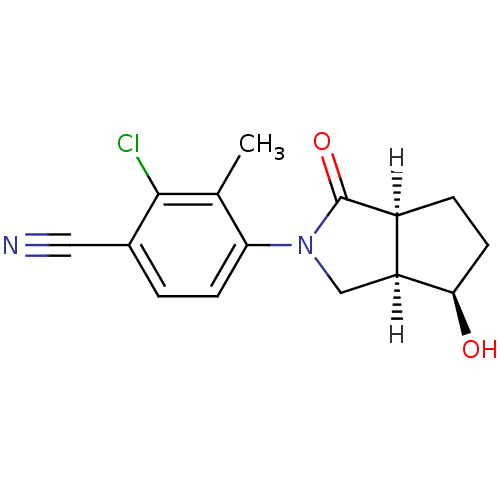 (4-[(3aR,4R,6aS)-4-hydroxy-1-oxo-octahydrocyclopent...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents | Article PubMed | 2.20 | -48.9 | n/a | n/a | 20 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18186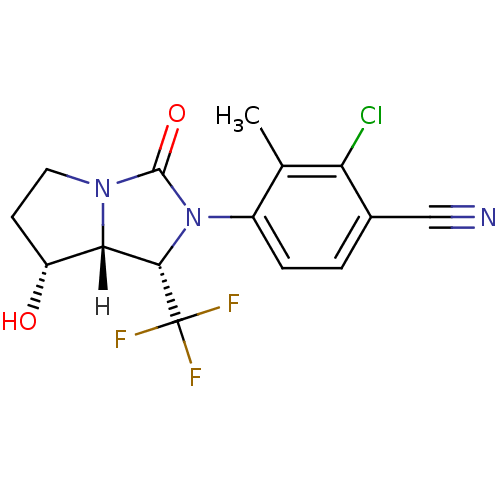 (4-[(1S,7R,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2.20 | -48.9 | n/a | n/a | 2.10 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM29322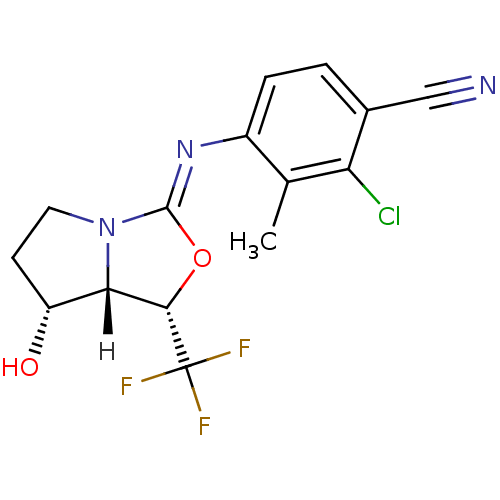 (oxazolidin-2-imine, 6e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2.30 | -48.8 | n/a | n/a | 1.10 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company | Assay Description Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... | J Med Chem 52: 2794-8 (2009) Article DOI: 10.1021/jm801583j BindingDB Entry DOI: 10.7270/Q2W66J30 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18185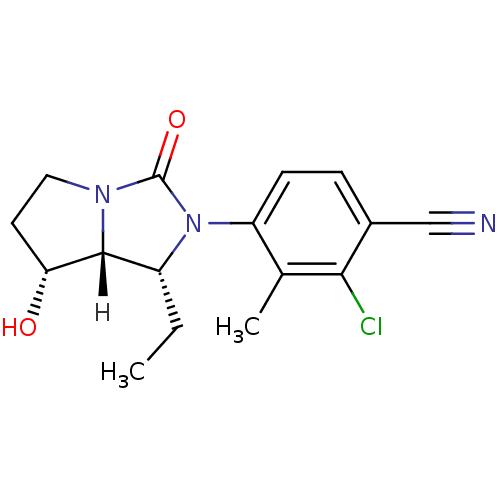 (4-[(1R,7R,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2.30 | -48.8 | n/a | n/a | 7.40 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50372078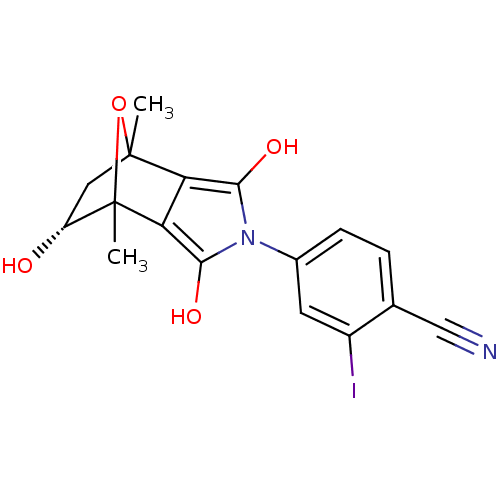 (CHEMBL272574) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]DHT from human androgen receptor in MDA453 cells | Bioorg Med Chem Lett 18: 1910-5 (2008) Article DOI: 10.1016/j.bmcl.2008.02.006 BindingDB Entry DOI: 10.7270/Q2DJ5GGT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205100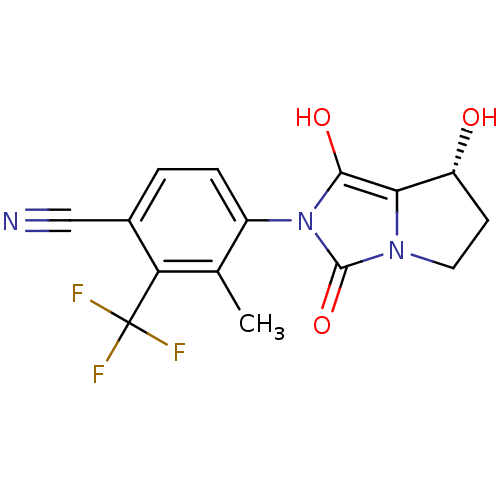 (4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM18182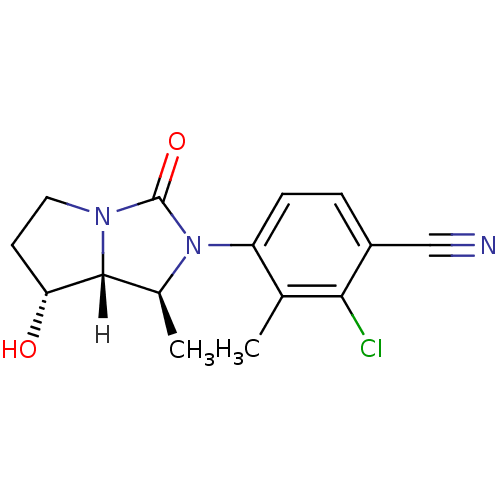 (4-[(1S,7R,7aR)-7-hydroxy-1-methyl-3-oxo-hexahydro-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2.90 | -48.2 | n/a | n/a | 13 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute | Assay Description Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... | J Med Chem 50: 3015-3025 (2007) Article DOI: 10.1021/jm070312d BindingDB Entry DOI: 10.7270/Q24F1P1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50157824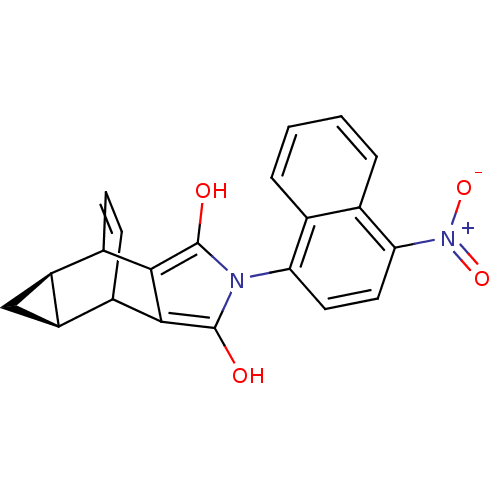 ((3a alpha,4beta,8beta,8a alpha)-4,5,6,7,8,8a-hexah...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity for androgen receptor in human MDA-453 cells | Bioorg Med Chem Lett 15: 271-6 (2004) Article DOI: 10.1016/j.bmcl.2004.10.085 BindingDB Entry DOI: 10.7270/Q2N58KVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50372103 (CHEMBL402807) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]DHT from human androgen receptor in MDA453 cells | Bioorg Med Chem Lett 18: 1910-5 (2008) Article DOI: 10.1016/j.bmcl.2008.02.006 BindingDB Entry DOI: 10.7270/Q2DJ5GGT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50372089 (CHEMBL272509) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]DHT from human androgen receptor in MDA453 cells | Bioorg Med Chem Lett 18: 1910-5 (2008) Article DOI: 10.1016/j.bmcl.2008.02.006 BindingDB Entry DOI: 10.7270/Q2DJ5GGT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50205113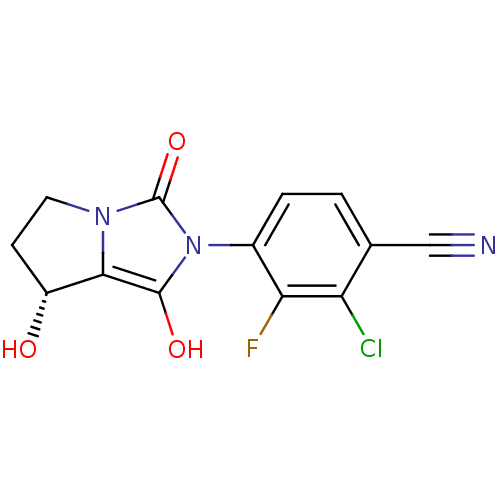 (2-chloro-3-fluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute Curated by ChEMBL | Assay Description Binding affinity to human AR | Bioorg Med Chem Lett 17: 1860-4 (2007) Article DOI: 10.1016/j.bmcl.2007.01.076 BindingDB Entry DOI: 10.7270/Q2S46RN1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen receptor (Homo sapiens (Human)) | BDBM50372088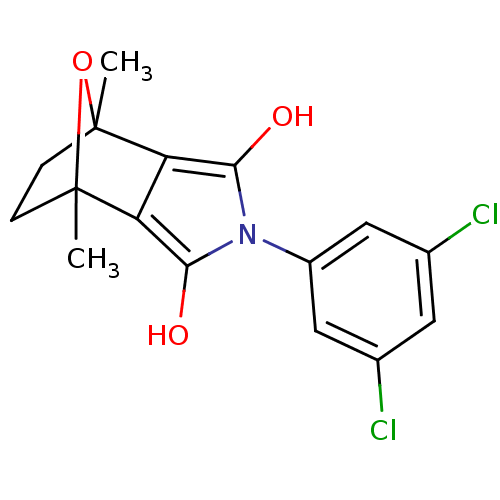 (CHEMBL257668) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]DHT from human androgen receptor in MDA453 cells | Bioorg Med Chem Lett 18: 1910-5 (2008) Article DOI: 10.1016/j.bmcl.2008.02.006 BindingDB Entry DOI: 10.7270/Q2DJ5GGT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 634 total ) | Next | Last >> |