

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16254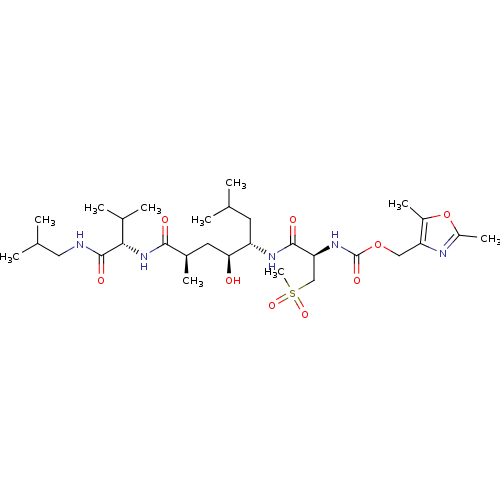 ((2,5-dimethyl-1,3-oxazol-4-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.120 | -58.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50231938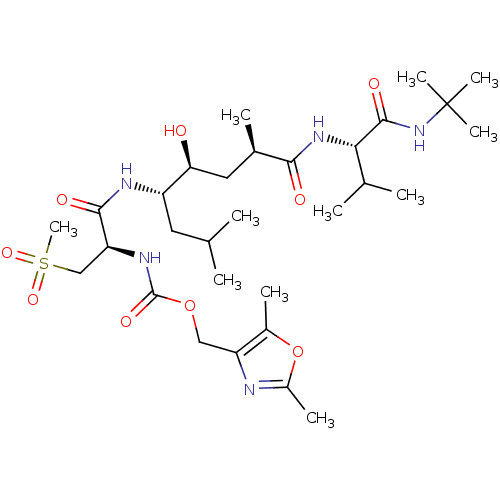 ((2,5-dimethyloxazol-4-yl)methyl (R)-1-((4S,5S,7R)-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Binding affinity to recombinant memapsin 2 | Bioorg Med Chem Lett 18: 1031-6 (2008) Article DOI: 10.1016/j.bmcl.2007.12.028 BindingDB Entry DOI: 10.7270/Q2513XZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16253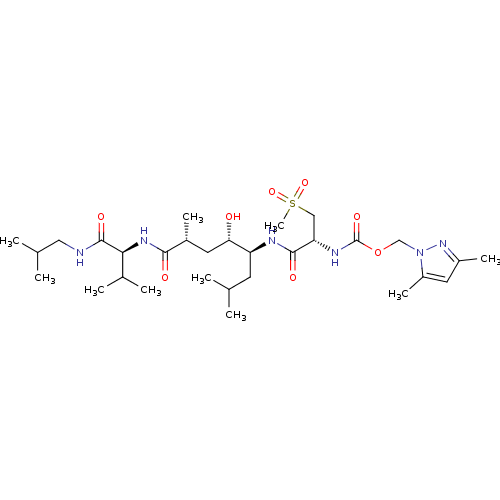 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16259 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 1.10 | -53.2 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16250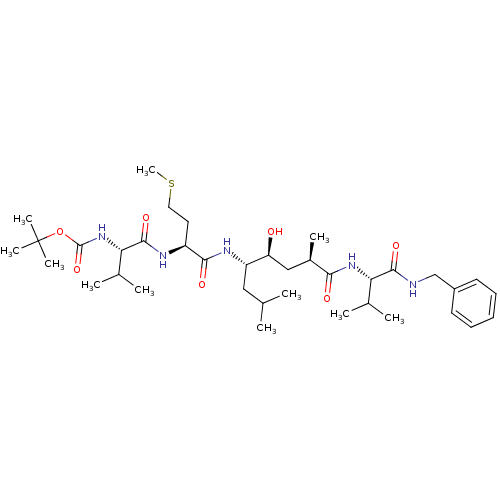 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.20 | -53.0 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16047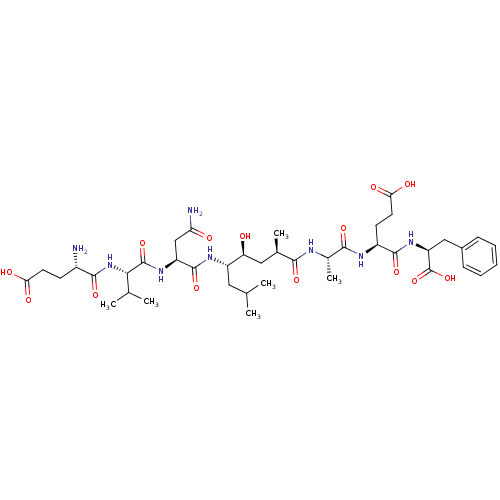 ((4S)-4-[(2S)-2-[(2R,4S,5S)-5-[(2S)-2-[(2S)-2-[(4S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 1.60 | -52.2 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16047 ((4S)-4-[(2S)-2-[(2R,4S,5S)-5-[(2S)-2-[(2S)-2-[(4S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Binding affinity to recombinant memapsin 2 | Bioorg Med Chem Lett 18: 1031-6 (2008) Article DOI: 10.1016/j.bmcl.2007.12.028 BindingDB Entry DOI: 10.7270/Q2513XZC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16257 (1-N-[(2,5-dimethyl-1,3-oxazol-4-yl)methyl]-3-N-[(1...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.80 | -51.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50231937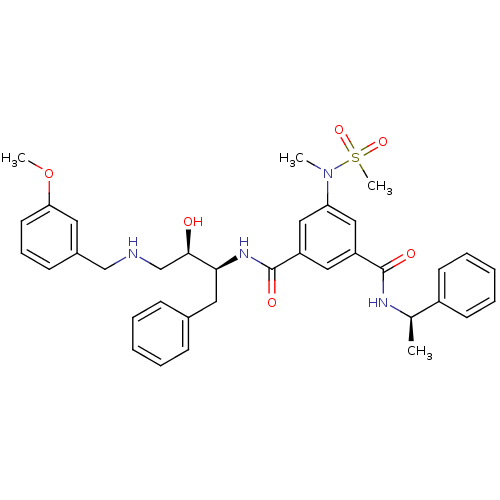 (CHEMBL403268 | N-{(1S,2R)-1-benzyl-2-hydroxy-3-[(3...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Binding affinity to recombinant memapsin 2 | Bioorg Med Chem Lett 18: 1031-6 (2008) Article DOI: 10.1016/j.bmcl.2007.12.028 BindingDB Entry DOI: 10.7270/Q2513XZC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240554 (CHEMBL4081377) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.5 | -51.1 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16252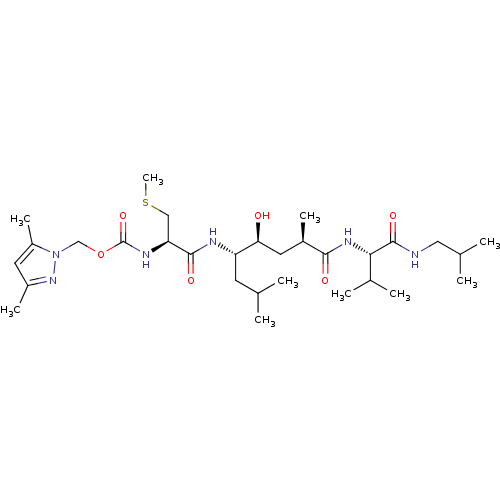 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem Similars | Article PubMed | 4.40 | -49.6 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50462838 (CHEMBL4239092) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant BACE1 (unknown origin) using fluorogenic substrate | Bioorg Med Chem Lett 28: 2605-2610 (2018) Article DOI: 10.1016/j.bmcl.2018.06.045 BindingDB Entry DOI: 10.7270/Q2571FNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240565 (CHEMBL4092310) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM120272 (US8703947, GRL- 0849AL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16251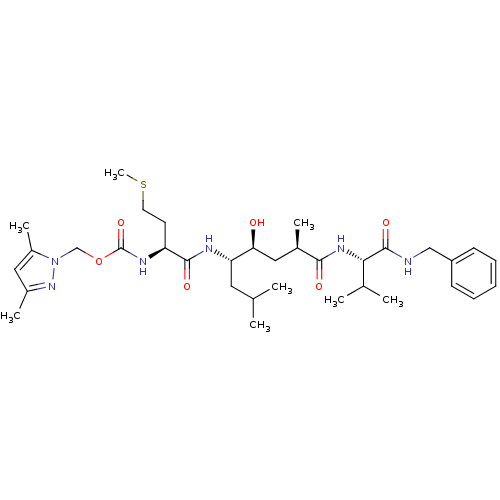 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1S)-1-{[(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 14 | -46.6 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16252 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem Similars | Article PubMed | 15 | -46.5 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240555 (CHEMBL4089241) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240569 (CHEMBL4067893) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16251 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1S)-1-{[(...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 25 | -45.1 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240572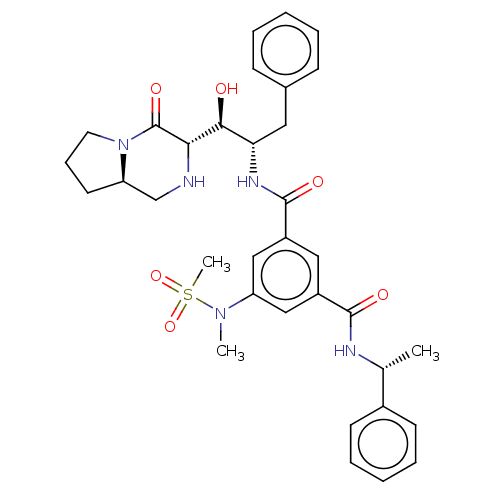 (CHEMBL4078781) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50231932 (CHEMBL253930 | N1-((2S,3R)-3-hydroxy-4-((1-(methyl...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Binding affinity to recombinant memapsin 2 | Bioorg Med Chem Lett 18: 1031-6 (2008) Article DOI: 10.1016/j.bmcl.2007.12.028 BindingDB Entry DOI: 10.7270/Q2513XZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16262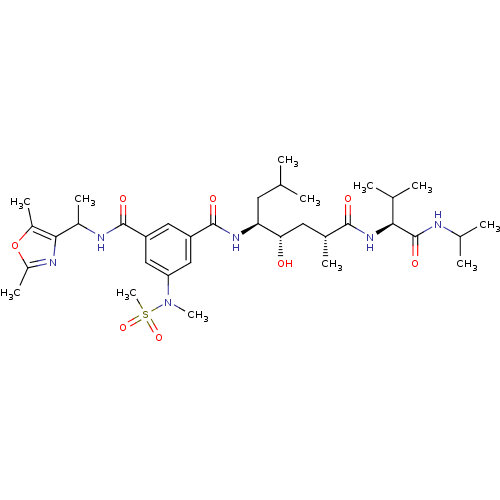 (1-N-[1-(2,5-dimethyl-1,3-oxazol-4-yl)ethyl]-3-N-[(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 27 | -44.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50462836 (CHEMBL4239838) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant BACE1 (unknown origin) using fluorogenic substrate | Bioorg Med Chem Lett 28: 2605-2610 (2018) Article DOI: 10.1016/j.bmcl.2018.06.045 BindingDB Entry DOI: 10.7270/Q2571FNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16259 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 31 | -44.6 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16259 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 41 | -43.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16256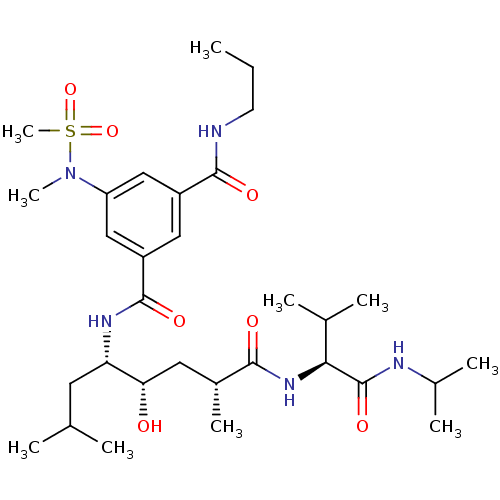 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 52.5 | -43.2 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240557 (CHEMBL4065655) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM120273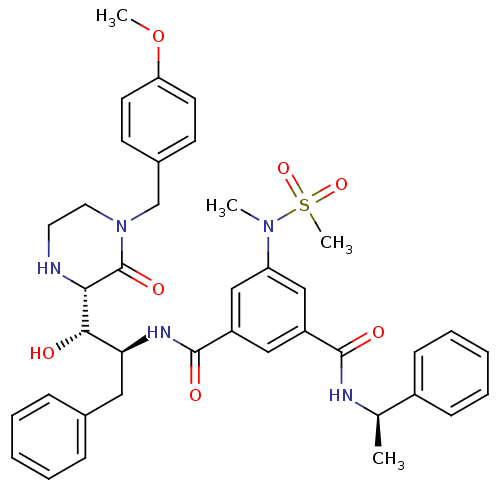 (US8703947, GRL- 0889AL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50462840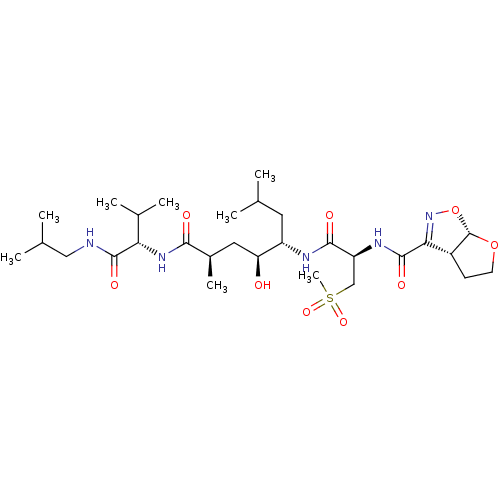 (CHEMBL4247466) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 111 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant BACE1 (unknown origin) using fluorogenic substrate | Bioorg Med Chem Lett 28: 2605-2610 (2018) Article DOI: 10.1016/j.bmcl.2018.06.045 BindingDB Entry DOI: 10.7270/Q2571FNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM120276 (US8703947, GRL- 1079AL) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16253 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 131 | -40.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM50240554 (CHEMBL4081377) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 134 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of BACE2 (unknown origin) | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16258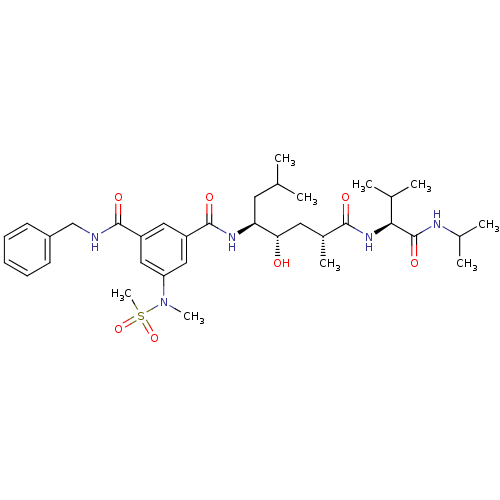 (1-N-benzyl-3-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 136 | -40.8 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240556 (CHEMBL4079215) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 144 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16252 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem Similars | Article PubMed | 161 | -40.3 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240559 (CHEMBL4087999) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 176 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50231933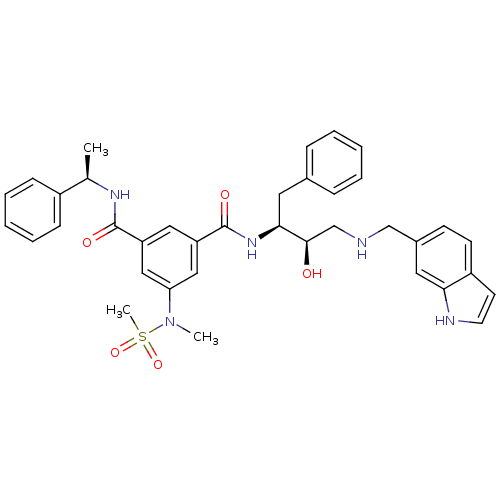 (CHEMBL253929 | N1-((2S,3R)-4-((1H-indol-6-yl)methy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Binding affinity to recombinant memapsin 2 | Bioorg Med Chem Lett 18: 1031-6 (2008) Article DOI: 10.1016/j.bmcl.2007.12.028 BindingDB Entry DOI: 10.7270/Q2513XZC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16034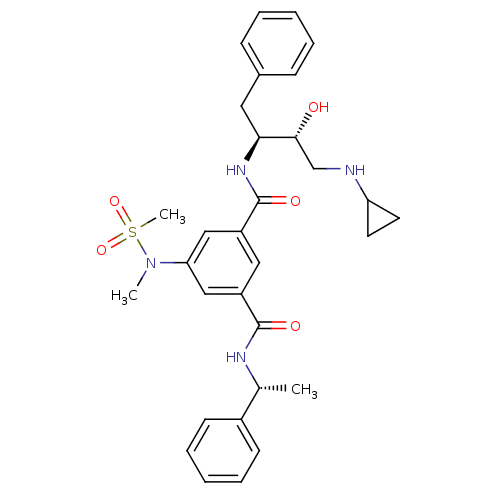 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 233 | -39.4 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16034 (1-N-[(2S,3R)-4-(cyclopropylamino)-3-hydroxy-1-phen...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 240 | -39.3 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16257 (1-N-[(2,5-dimethyl-1,3-oxazol-4-yl)methyl]-3-N-[(1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 245 | -39.3 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50462844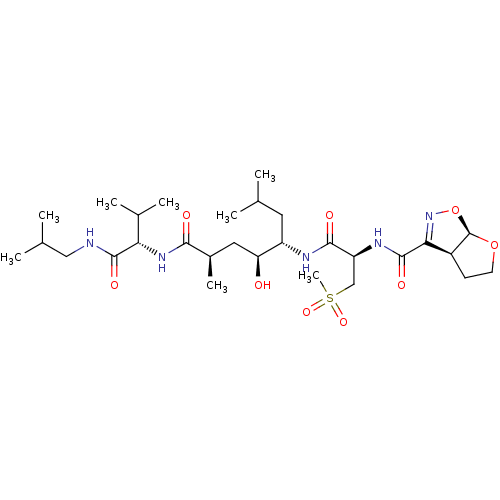 (CHEMBL4251397) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant BACE1 (unknown origin) using fluorogenic substrate | Bioorg Med Chem Lett 28: 2605-2610 (2018) Article DOI: 10.1016/j.bmcl.2018.06.045 BindingDB Entry DOI: 10.7270/Q2571FNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240562 (CHEMBL4059708) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM16254 ((2,5-dimethyl-1,3-oxazol-4-yl)methyl N-[(1R)-1-{[(...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 304 | -38.7 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16260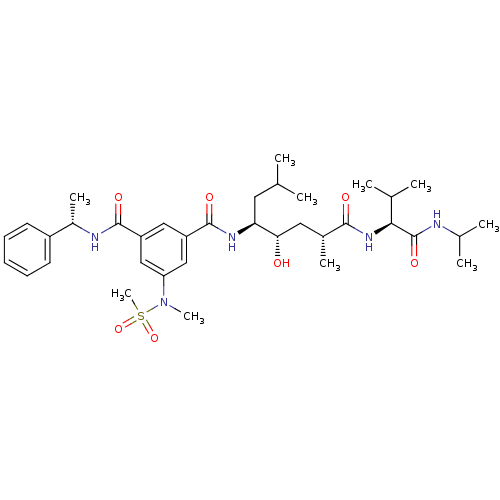 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 315 | -38.6 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50240566 (CHEMBL4064628) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 (Ala8P to Ala326 residues) expressed in Escherichia coli BL21(DE3) using Arg-Glu(EDANS)-Glu-Val-Asn-Leu-Asp-Ala... | Bioorg Med Chem Lett 27: 2432-2438 (2017) Article DOI: 10.1016/j.bmcl.2017.04.011 BindingDB Entry DOI: 10.7270/Q2R213JX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16256 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 340 | -38.4 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16253 ((3,5-dimethyl-1H-pyrazol-1-yl)methyl N-[(1R)-1-{[(...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 356 | -38.3 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16261 (1-N-[(1R,3S,4S)-3-hydroxy-1,6-dimethyl-1-{[(1S)-2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 397 | -38.0 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 2 (Homo sapiens (Human)) | BDBM16262 (1-N-[1-(2,5-dimethyl-1,3-oxazol-4-yl)ethyl]-3-N-[(...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 408 | -37.9 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 50: 2399-407 (2007) Article DOI: 10.1021/jm061338s BindingDB Entry DOI: 10.7270/Q2348HNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 89 total ) | Next | Last >> |