 Found 1477 hits with Last Name = 'liao' and Initial = 'y'
Found 1477 hits with Last Name = 'liao' and Initial = 'y' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093352
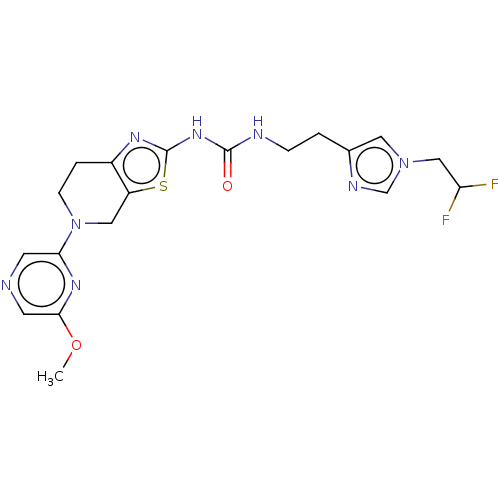
(CHEMBL3586678)Show SMILES COc1cncc(n1)N1CCc2nc(NC(=O)NCCc3cn(CC(F)F)cn3)sc2C1 Show InChI InChI=1S/C19H22F2N8O2S/c1-31-17-7-22-6-16(26-17)29-5-3-13-14(9-29)32-19(25-13)27-18(30)23-4-2-12-8-28(11-24-12)10-15(20)21/h6-8,11,15H,2-5,9-10H2,1H3,(H2,23,25,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093351
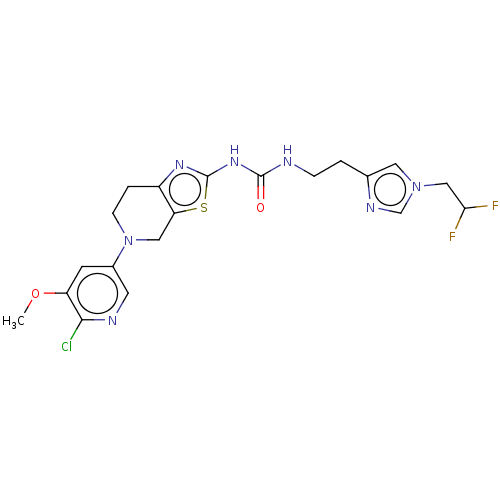
(CHEMBL3585362)Show SMILES COc1cc(cnc1Cl)N1CCc2nc(NC(=O)NCCc3cn(CC(F)F)cn3)sc2C1 Show InChI InChI=1S/C20H22ClF2N7O2S/c1-32-15-6-13(7-25-18(15)21)30-5-3-14-16(9-30)33-20(27-14)28-19(31)24-4-2-12-8-29(11-26-12)10-17(22)23/h6-8,11,17H,2-5,9-10H2,1H3,(H2,24,27,28,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093355
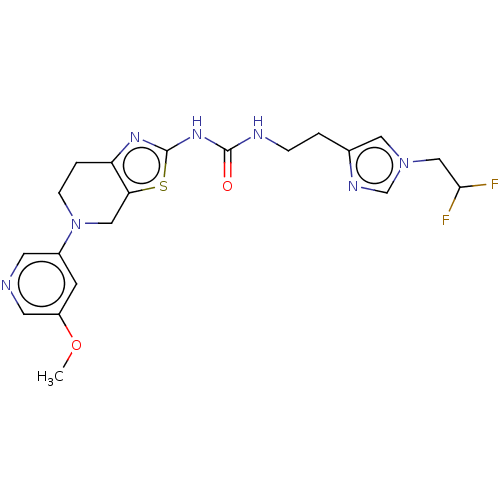
(CHEMBL3586677)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(CC(F)F)cn3)sc2C1 Show InChI InChI=1S/C20H23F2N7O2S/c1-31-15-6-14(7-23-8-15)29-5-3-16-17(10-29)32-20(26-16)27-19(30)24-4-2-13-9-28(12-25-13)11-18(21)22/h6-9,12,18H,2-5,10-11H2,1H3,(H2,24,26,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093356
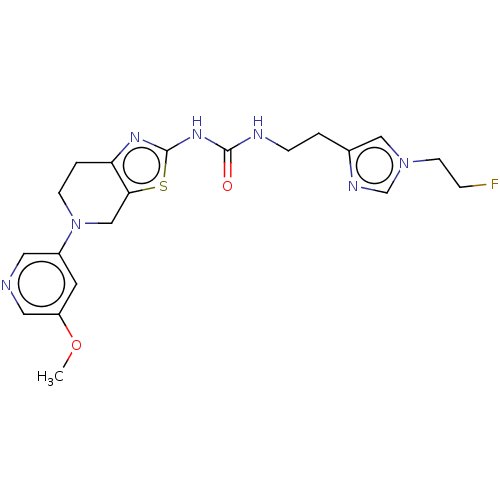
(CHEMBL3586676)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(CCF)cn3)sc2C1 Show InChI InChI=1S/C20H24FN7O2S/c1-30-16-8-15(9-22-10-16)28-6-3-17-18(12-28)31-20(25-17)26-19(29)23-5-2-14-11-27(7-4-21)13-24-14/h8-11,13H,2-7,12H2,1H3,(H2,23,25,26,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093354

(CHEMBL3586679)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(CC(F)(F)F)cn3)sc2C1 Show InChI InChI=1S/C20H22F3N7O2S/c1-32-15-6-14(7-24-8-15)30-5-3-16-17(10-30)33-19(27-16)28-18(31)25-4-2-13-9-29(12-26-13)11-20(21,22)23/h6-9,12H,2-5,10-11H2,1H3,(H2,25,27,28,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093395
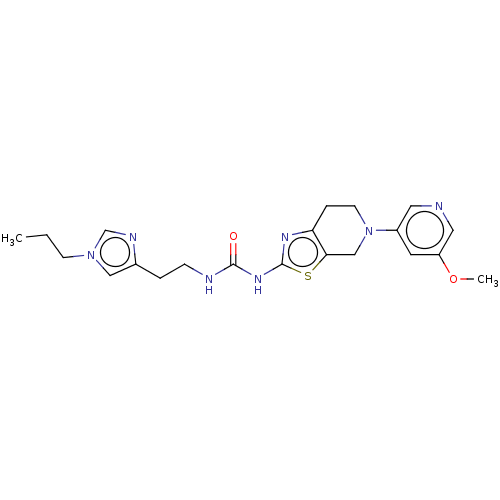
(CHEMBL3586674)Show SMILES CCCn1cnc(CCNC(=O)Nc2nc3CCN(Cc3s2)c2cncc(OC)c2)c1 Show InChI InChI=1S/C21H27N7O2S/c1-3-7-27-12-15(24-14-27)4-6-23-20(29)26-21-25-18-5-8-28(13-19(18)31-21)16-9-17(30-2)11-22-10-16/h9-12,14H,3-8,13H2,1-2H3,(H2,23,25,26,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093417
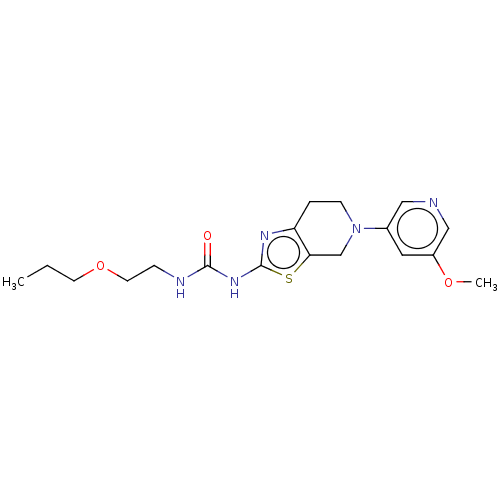
(CHEMBL3586672)Show InChI InChI=1S/C18H25N5O3S/c1-3-7-26-8-5-20-17(24)22-18-21-15-4-6-23(12-16(15)27-18)13-9-14(25-2)11-19-10-13/h9-11H,3-8,12H2,1-2H3,(H2,20,21,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
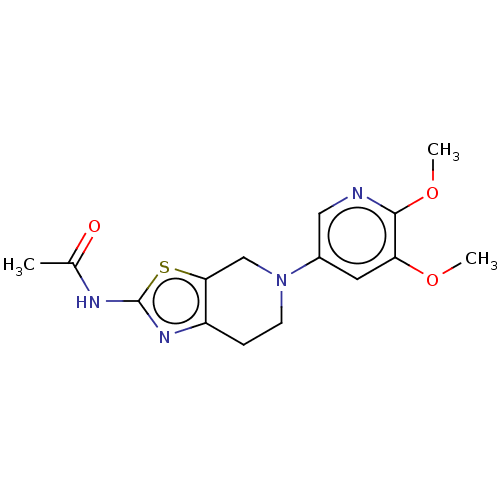
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093437
(CHEMBL3586668)Show InChI InChI=1S/C20H32O2/c1-19-7-5-14(21)10-13(19)3-4-15-16(19)6-8-20(2)17(15)9-12-11-22-18(12)20/h12-18,21H,3-11H2,1-2H3/t12-,13+,14-,15-,16+,17+,18+,19+,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093399
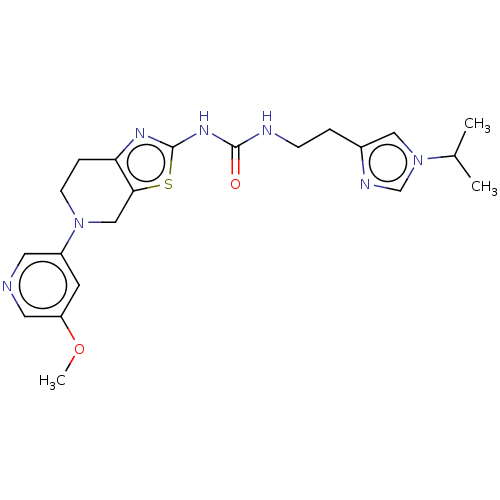
(CHEMBL3586673)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(cn3)C(C)C)sc2C1 Show InChI InChI=1S/C21H27N7O2S/c1-14(2)28-11-15(24-13-28)4-6-23-20(29)26-21-25-18-5-7-27(12-19(18)31-21)16-8-17(30-3)10-22-9-16/h8-11,13-14H,4-7,12H2,1-3H3,(H2,23,25,26,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
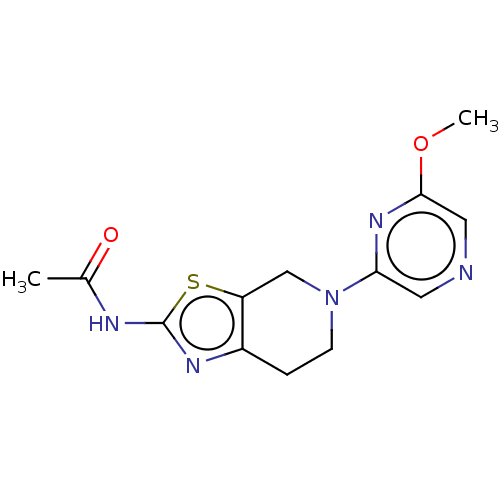
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093434
(CHEMBL3586670)Show InChI InChI=1S/C12H11NO8S2/c1-6(14)13-10-4-8(22(16,17)18)2-7-3-9(23(19,20)21)5-11(15)12(7)10/h2-5,15H,1H3,(H,13,14)(H,16,17,18)(H,19,20,21)/p-2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
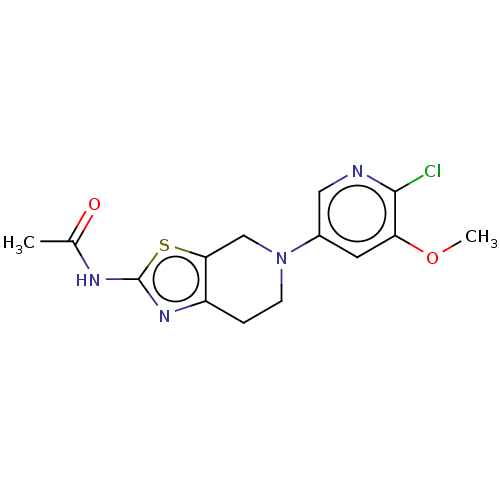
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093436
(CHEMBL3586669)Show InChI InChI=1S/C22H18O12/c23-13-5-1-11(9-15(13)25)3-7-17(27)33-19(21(29)30)20(22(31)32)34-18(28)8-4-12-2-6-14(24)16(26)10-12/h1-10,19-20,23-26H,(H,29,30)(H,31,32)/p-2/b7-3+,8-4+/t19-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
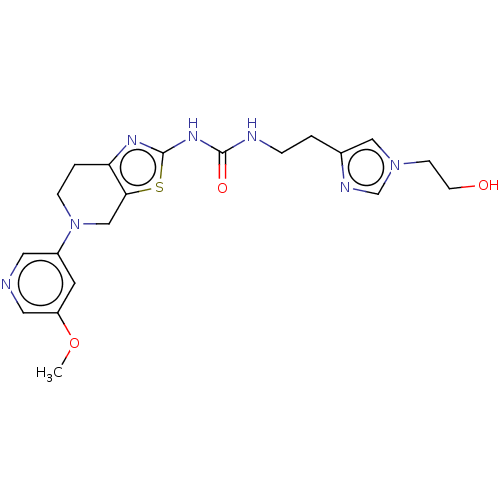
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093353
(CHEMBL3586680)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(CCO)cn3)sc2C1 Show InChI InChI=1S/C20H25N7O3S/c1-30-16-8-15(9-21-10-16)27-5-3-17-18(12-27)31-20(24-17)25-19(29)22-4-2-14-11-26(6-7-28)13-23-14/h8-11,13,28H,2-7,12H2,1H3,(H2,22,24,25,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
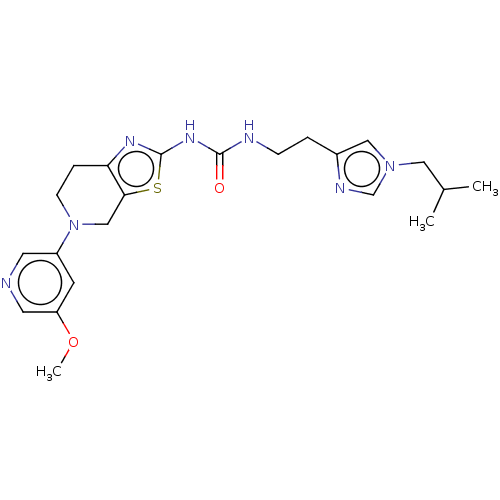
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093391
(CHEMBL3586675)Show SMILES COc1cncc(c1)N1CCc2nc(NC(=O)NCCc3cn(CC(C)C)cn3)sc2C1 Show InChI InChI=1S/C22H29N7O2S/c1-15(2)11-28-12-16(25-14-28)4-6-24-21(30)27-22-26-19-5-7-29(13-20(19)32-22)17-8-18(31-3)10-23-9-17/h8-10,12,14-15H,4-7,11,13H2,1-3H3,(H2,24,26,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093439
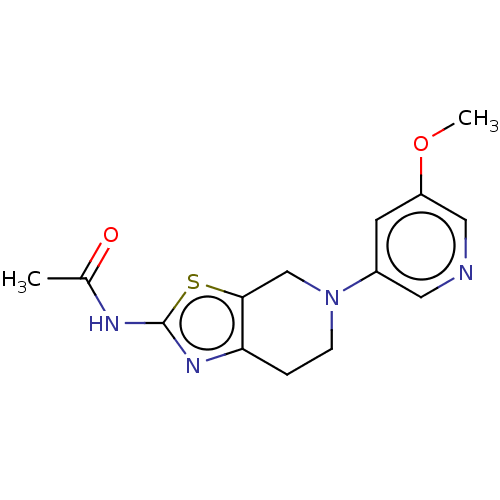
(CHEMBL3586666)Show InChI InChI=1S/C19H30O2/c1-18-7-5-12(20)9-11(18)3-4-13-14(18)6-8-19(2)15(13)10-16-17(19)21-16/h11-17,20H,3-10H2,1-2H3/t11-,12+,13+,14-,15-,16+,17+,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093419
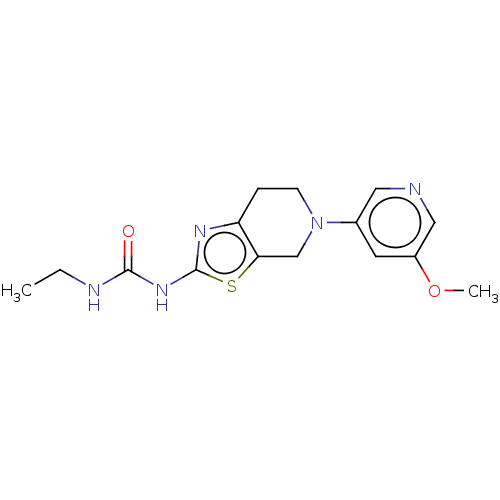
(CHEMBL3586671)Show InChI InChI=1S/C15H19N5O2S/c1-3-17-14(21)19-15-18-12-4-5-20(9-13(12)23-15)10-6-11(22-2)8-16-7-10/h6-8H,3-5,9H2,1-2H3,(H2,17,18,19,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093440
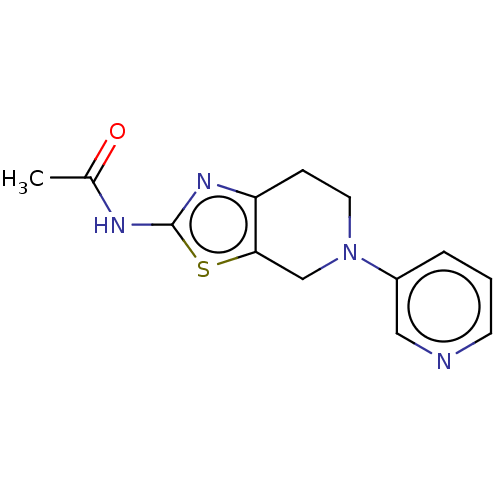
(CHEMBL3586665)Show InChI InChI=1S/C21H32O3/c1-12(22)16-6-7-17-15-5-4-13-10-14(23)8-9-20(13,2)19(15)18(24)11-21(16,17)3/h13-17,19,23H,4-11H2,1-3H3/t13?,14-,15?,16-,17?,19?,20+,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.157 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50093438
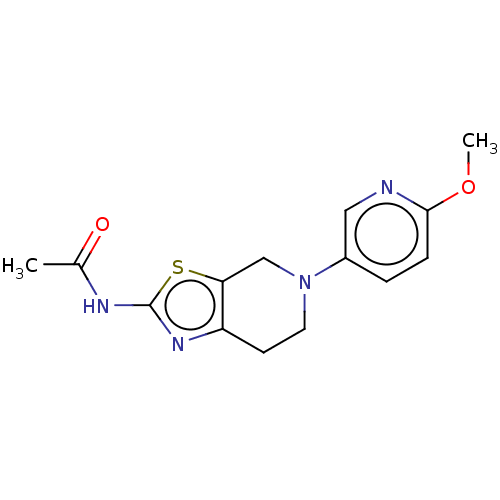
(CHEMBL3586667)Show InChI InChI=1S/C20H31NO/c1-19-9-7-15(22)11-13(19)3-5-16-17-6-4-14(12-21)20(17,2)10-8-18(16)19/h13-18,22H,3-11H2,1-2H3/t13-,14-,15+,16-,17-,18-,19-,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using [33P]ATP and PIP2 incubated for 15 mins by liquid scintillation counting method |
J Med Chem 58: 5684-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00498
BindingDB Entry DOI: 10.7270/Q2Z89F53 |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
EMD-Serono Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity at human prostaglandin EP4 receptor |
Bioorg Med Chem Lett 17: 6572-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.074
BindingDB Entry DOI: 10.7270/Q29887V9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50044287
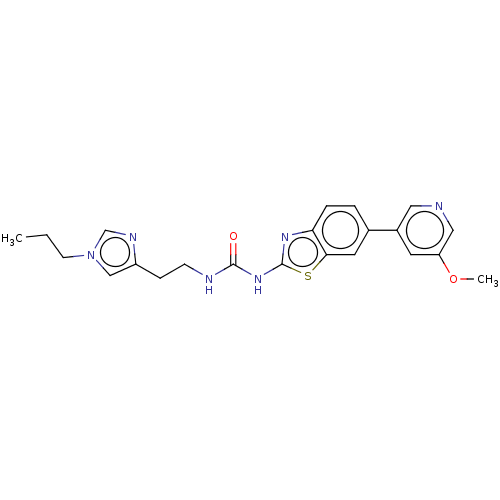
(CHEMBL3356900)Show SMILES CCCn1cnc(CCNC(=O)Nc2nc3ccc(cc3s2)-c2cncc(OC)c2)c1 Show InChI InChI=1S/C22H24N6O2S/c1-3-8-28-13-17(25-14-28)6-7-24-21(29)27-22-26-19-5-4-15(10-20(19)31-22)16-9-18(30-2)12-23-11-16/h4-5,9-14H,3,6-8H2,1-2H3,(H2,24,26,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274571
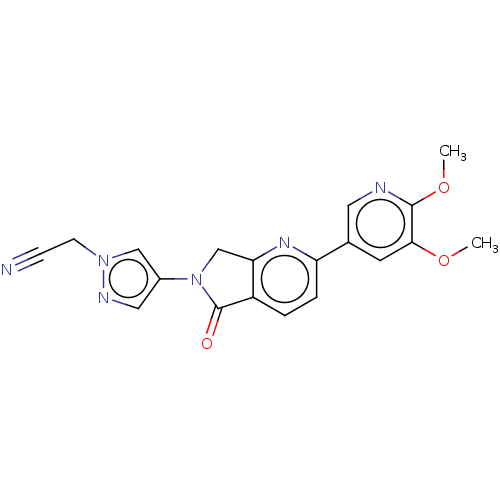
(CHEMBL4127784)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N(Cc2n1)c1cnn(CC#N)c1 Show InChI InChI=1S/C19H16N6O3/c1-27-17-7-12(8-21-18(17)28-2)15-4-3-14-16(23-15)11-25(19(14)26)13-9-22-24(10-13)6-5-20/h3-4,7-10H,6,11H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM496902
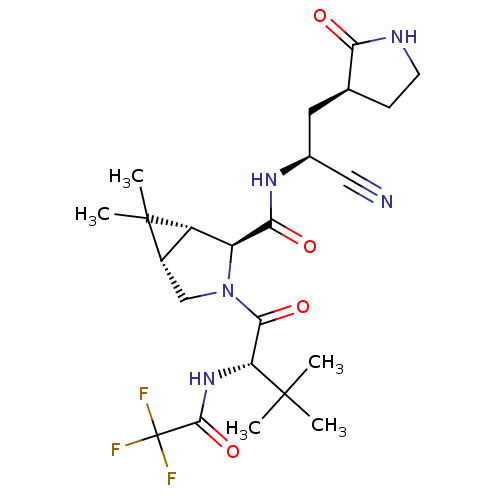
(CVD-0018409 | PF-07321332 | US11351149, Example 13...)Show SMILES CC(C)(C)[C@H](NC(=O)C(F)(F)F)C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)N[C@@H](C[C@@H]1CCNC1=O)C#N)C2(C)C Show InChI InChI=1S/C23H32F3N5O4/c1-21(2,3)16(30-20(35)23(24,25)26)19(34)31-10-13-14(22(13,4)5)15(31)18(33)29-12(9-27)8-11-6-7-28-17(11)32/h11-16H,6-8,10H2,1-5H3,(H,28,32)(H,29,33)(H,30,35)/t11-,12-,13-,14-,15-,16+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00954
BindingDB Entry DOI: 10.7270/Q2R49VTB |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM50066910
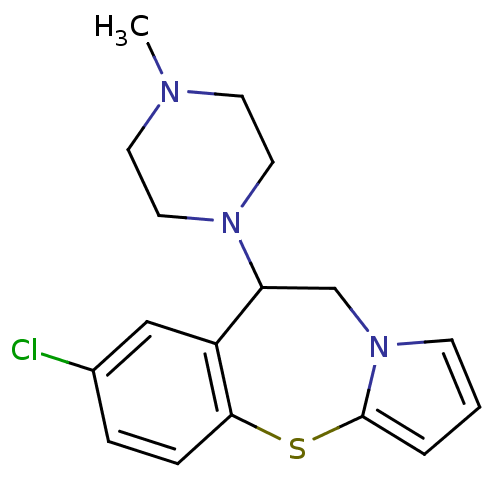
((+/-)7-Chloro-9-(4-methyl-piperazin-1-yl)-9,10-dih...)Show InChI InChI=1S/C17H20ClN3S/c1-19-7-9-20(10-8-19)15-12-21-6-2-3-17(21)22-16-5-4-13(18)11-14(15)16/h2-6,11,15H,7-10,12H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' di Siena
Curated by ChEMBL
| Assay Description
Binding affinity which represents concentration giving half-maximal inhibition of [3H]spiperone (Dopamine receptor D2) binding to rat tissue homogena... |
J Med Chem 41: 3763-72 (1998)
Article DOI: 10.1021/jm9706832
BindingDB Entry DOI: 10.7270/Q2JH3MVJ |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24615
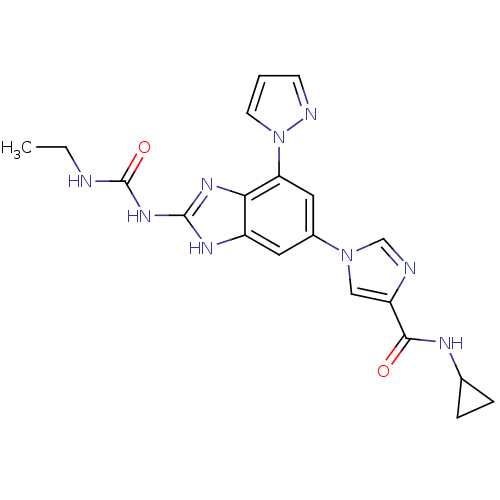
(Benzimidazole urea analogue, 19 | N-cyclopropyl-1-...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-n1cnc(c1)C(=O)NC1CC1)-n1cccn1 Show InChI InChI=1S/C20H21N9O2/c1-2-21-20(31)27-19-25-14-8-13(9-16(17(14)26-19)29-7-3-6-23-29)28-10-15(22-11-28)18(30)24-12-4-5-12/h3,6-12H,2,4-5H2,1H3,(H,24,30)(H3,21,25,26,27,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24601
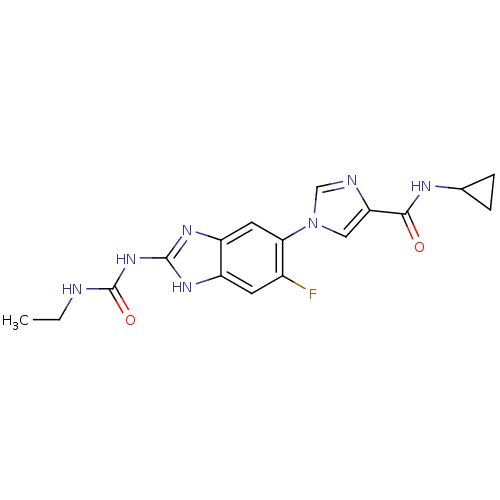
(Benzimidazole urea analogue, 5 | N-cyclopropyl-1-{...)Show SMILES CCNC(=O)Nc1nc2cc(c(F)cc2[nH]1)-n1cnc(c1)C(=O)NC1CC1 Show InChI InChI=1S/C17H18FN7O2/c1-2-19-17(27)24-16-22-11-5-10(18)14(6-12(11)23-16)25-7-13(20-8-25)15(26)21-9-3-4-9/h5-9H,2-4H2,1H3,(H,21,26)(H3,19,22,23,24,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24606
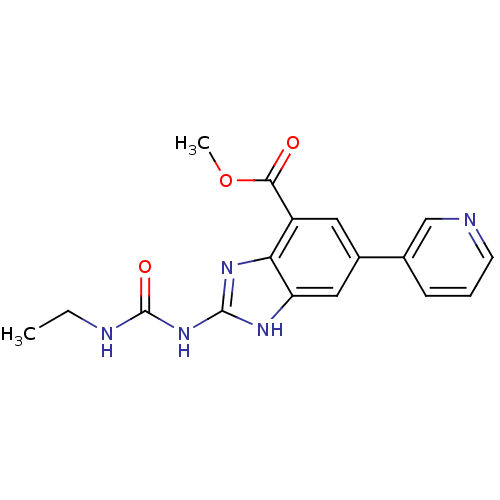
(Benzimidazole urea analogue, 10 | methyl 2-[(ethyl...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)C(=O)OC Show InChI InChI=1S/C17H17N5O3/c1-3-19-17(24)22-16-20-13-8-11(10-5-4-6-18-9-10)7-12(14(13)21-16)15(23)25-2/h4-9H,3H2,1-2H3,(H3,19,20,21,22,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24608
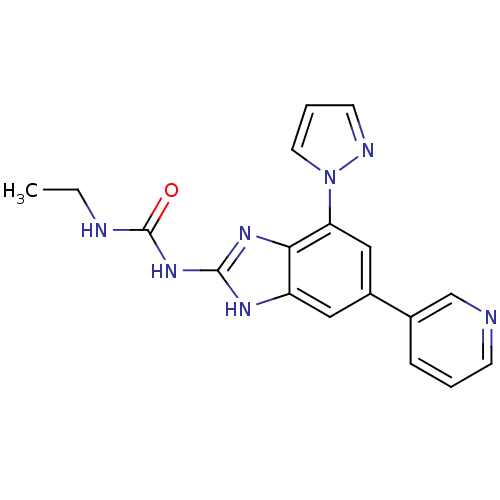
(3-ethyl-1-[7-(1H-pyrazol-1-yl)-5-(pyridin-3-yl)-1H...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)-n1cccn1 Show InChI InChI=1S/C18H17N7O/c1-2-20-18(26)24-17-22-14-9-13(12-5-3-6-19-11-12)10-15(16(14)23-17)25-8-4-7-21-25/h3-11H,2H2,1H3,(H3,20,22,23,24,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24616
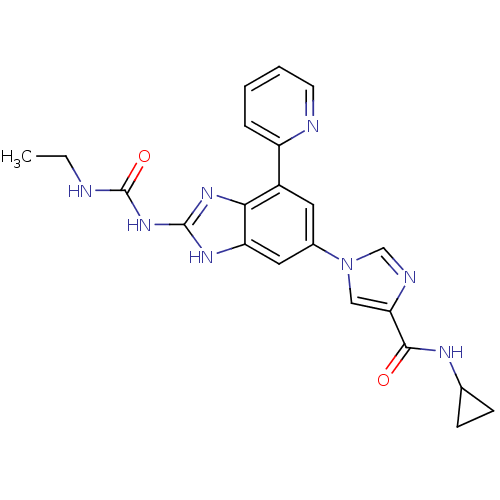
(1-(6-(4-(Cyclopropylcarbamoyl)-1H-imidazol-1-yl)-4...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-n1cnc(c1)C(=O)NC1CC1)-c1ccccn1 Show InChI InChI=1S/C22H22N8O2/c1-2-23-22(32)29-21-27-17-10-14(9-15(19(17)28-21)16-5-3-4-8-24-16)30-11-18(25-12-30)20(31)26-13-6-7-13/h3-5,8-13H,2,6-7H2,1H3,(H,26,31)(H3,23,27,28,29,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
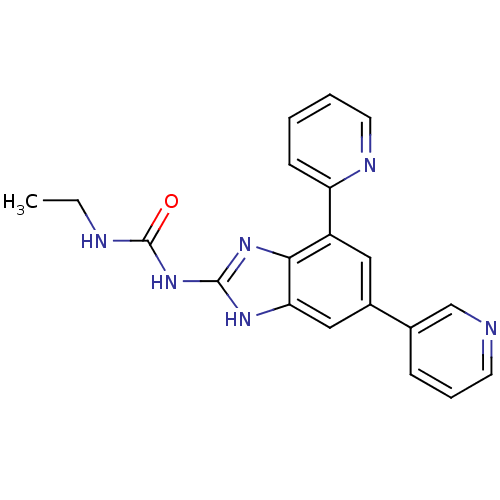
(Escherichia coli (strain K12)) | BDBM24609
(3-ethyl-1-[7-(pyridin-2-yl)-5-(pyridin-3-yl)-1H-1,...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)-c1ccccn1 Show InChI InChI=1S/C20H18N6O/c1-2-22-20(27)26-19-24-17-11-14(13-6-5-8-21-12-13)10-15(18(17)25-19)16-7-3-4-9-23-16/h3-12H,2H2,1H3,(H3,22,24,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24617
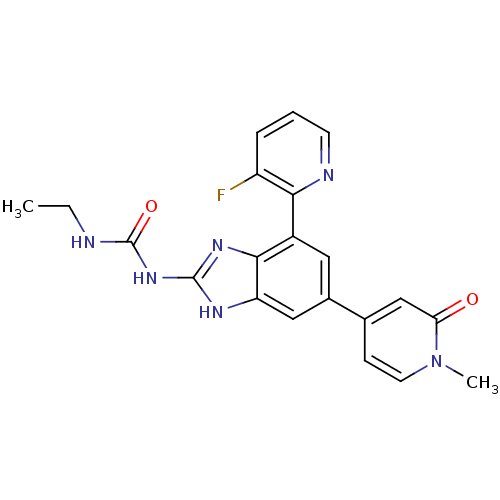
(3-ethyl-1-[7-(3-fluoropyridin-2-yl)-5-(1-methyl-2-...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1ccn(C)c(=O)c1)-c1ncccc1F Show InChI InChI=1S/C21H19FN6O2/c1-3-23-21(30)27-20-25-16-10-13(12-6-8-28(2)17(29)11-12)9-14(19(16)26-20)18-15(22)5-4-7-24-18/h4-11H,3H2,1-2H3,(H3,23,25,26,27,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24611
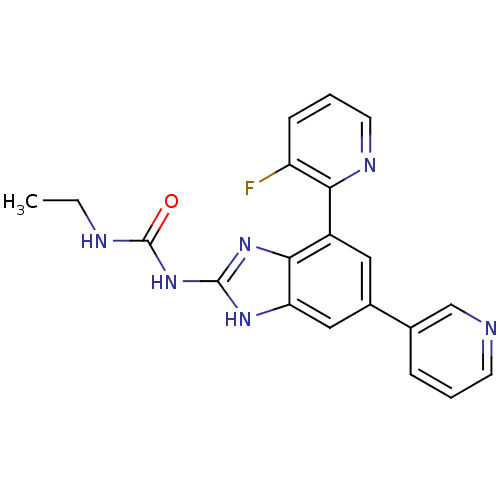
(3-ethyl-1-[7-(3-fluoropyridin-2-yl)-5-(pyridin-3-y...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)-c1ncccc1F Show InChI InChI=1S/C20H17FN6O/c1-2-23-20(28)27-19-25-16-10-13(12-5-3-7-22-11-12)9-14(18(16)26-19)17-15(21)6-4-8-24-17/h3-11H,2H2,1H3,(H3,23,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50497604
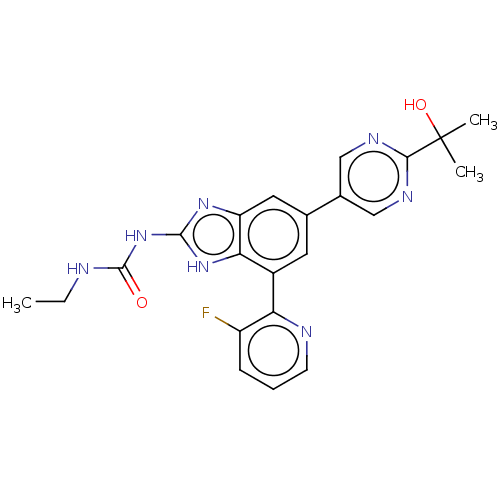
(CHEMBL3264033)Show SMILES CCNC(=O)Nc1nc2cc(cc(-c3ncccc3F)c2[nH]1)-c1cnc(nc1)C(C)(C)O Show InChI InChI=1S/C22H22FN7O2/c1-4-24-21(31)30-20-28-16-9-12(13-10-26-19(27-11-13)22(2,3)32)8-14(18(16)29-20)17-15(23)6-5-7-25-17/h5-11,32H,4H2,1-3H3,(H3,24,28,29,30,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of aureus Escherichia coli DNA gyrase A2B2 using pBR322 plasmid DNA as substrate by coupled enzyme reaction assay |
J Med Chem 57: 8792-816 (2014)
Article DOI: 10.1021/jm500563g
BindingDB Entry DOI: 10.7270/Q2TF01BF |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24614
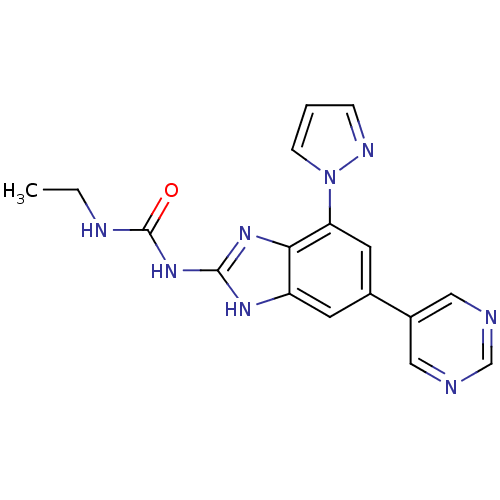
(3-ethyl-1-[7-(1H-pyrazol-1-yl)-5-(pyrimidin-5-yl)-...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cncnc1)-n1cccn1 Show InChI InChI=1S/C17H16N8O/c1-2-20-17(26)24-16-22-13-6-11(12-8-18-10-19-9-12)7-14(15(13)23-16)25-5-3-4-21-25/h3-10H,2H2,1H3,(H3,20,22,23,24,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| <4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274537
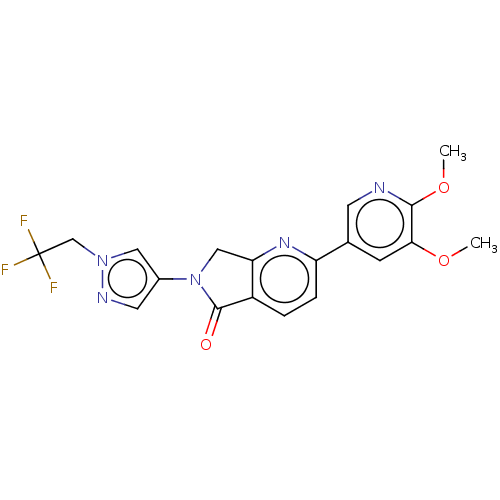
(CHEMBL4129974)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N(Cc2n1)c1cnn(CC(F)(F)F)c1 Show InChI InChI=1S/C19H16F3N5O3/c1-29-16-5-11(6-23-17(16)30-2)14-4-3-13-15(25-14)9-27(18(13)28)12-7-24-26(8-12)10-19(20,21)22/h3-8H,9-10H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274538
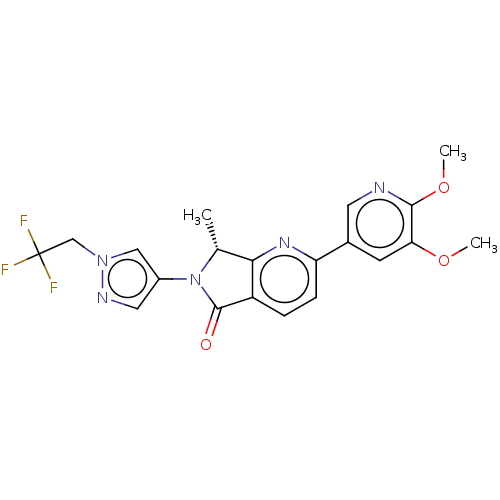
(CHEMBL4126773)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N([C@H](C)c2n1)c1cnn(CC(F)(F)F)c1 |r| Show InChI InChI=1S/C20H18F3N5O3/c1-11-17-14(19(29)28(11)13-8-25-27(9-13)10-20(21,22)23)4-5-15(26-17)12-6-16(30-2)18(31-3)24-7-12/h4-9,11H,10H2,1-3H3/t11-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Rattus norvegicus (Rat)) | BDBM50066910

((+/-)7-Chloro-9-(4-methyl-piperazin-1-yl)-9,10-dih...)Show InChI InChI=1S/C17H20ClN3S/c1-19-7-9-20(10-8-19)15-12-21-6-2-3-17(21)22-16-5-4-13(18)11-14(15)16/h2-6,11,15H,7-10,12H2,1H3 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universita' di Siena
Curated by ChEMBL
| Assay Description
Binding affinity which represents concentration giving half-maximal inhibition of [3H]7-OH-DPAT binding to Dopamine receptor D3 in rat tissue homogen... |
J Med Chem 41: 3763-72 (1998)
Article DOI: 10.1021/jm9706832
BindingDB Entry DOI: 10.7270/Q2JH3MVJ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 4.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
EMD-Serono Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity at human prostaglandin EP2 receptor |
Bioorg Med Chem Lett 17: 6572-5 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.074
BindingDB Entry DOI: 10.7270/Q29887V9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24607
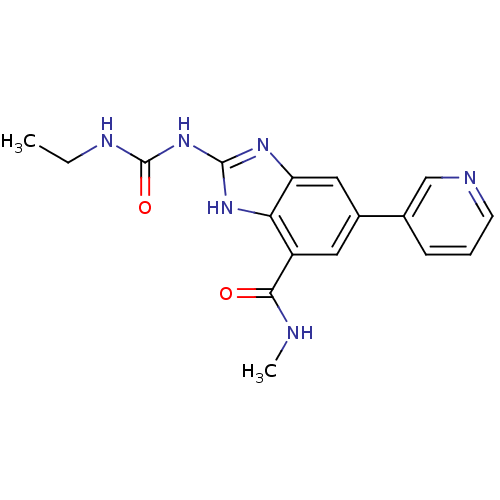
(2-[(ethylcarbamoyl)amino]-N-methyl-5-(pyridin-3-yl...)Show SMILES CCNC(=O)Nc1nc2cc(cc(C(=O)NC)c2[nH]1)-c1cccnc1 Show InChI InChI=1S/C17H18N6O2/c1-3-20-17(25)23-16-21-13-8-11(10-5-4-6-19-9-10)7-12(14(13)22-16)15(24)18-2/h4-9H,3H2,1-2H3,(H,18,24)(H3,20,21,22,23,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50497603
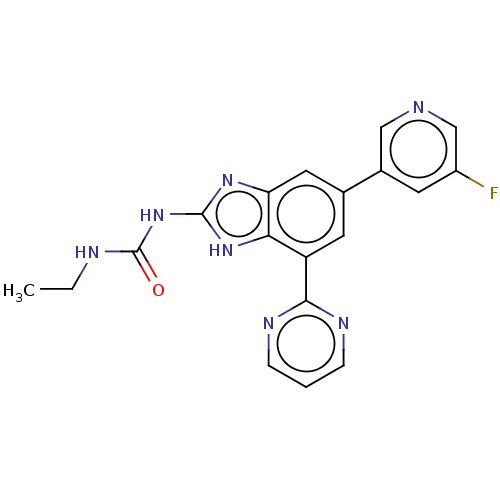
(CHEMBL3356986)Show SMILES CCNC(=O)Nc1nc2cc(cc(-c3ncccn3)c2[nH]1)-c1cncc(F)c1 Show InChI InChI=1S/C19H16FN7O/c1-2-22-19(28)27-18-25-15-8-11(12-6-13(20)10-21-9-12)7-14(16(15)26-18)17-23-4-3-5-24-17/h3-10H,2H2,1H3,(H3,22,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of aureus Escherichia coli DNA gyrase A2B2 using pBR322 plasmid DNA as substrate by coupled enzyme reaction assay |
J Med Chem 57: 8792-816 (2014)
Article DOI: 10.1021/jm500563g
BindingDB Entry DOI: 10.7270/Q2TF01BF |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM50497602
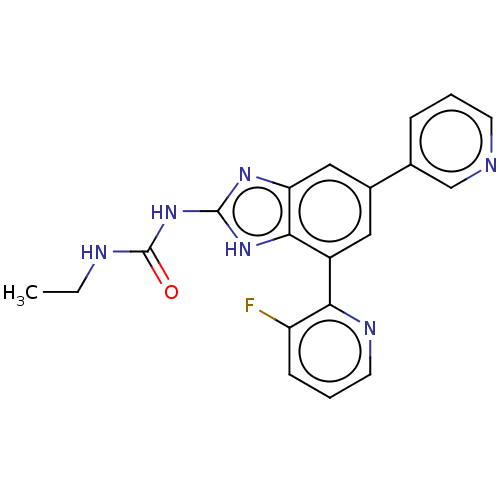
(CHEMBL222333 | VRT-752586)Show SMILES CCNC(=O)Nc1nc2cc(cc(-c3ncccc3F)c2[nH]1)-c1cccnc1 Show InChI InChI=1S/C20H17FN6O/c1-2-23-20(28)27-19-25-16-10-13(12-5-3-7-22-11-12)9-14(18(16)26-19)17-15(21)6-4-8-24-17/h3-11H,2H2,1H3,(H3,23,25,26,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of aureus Escherichia coli DNA gyrase A2B2 using pBR322 plasmid DNA as substrate by coupled enzyme reaction assay |
J Med Chem 57: 8792-816 (2014)
Article DOI: 10.1021/jm500563g
BindingDB Entry DOI: 10.7270/Q2TF01BF |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274572
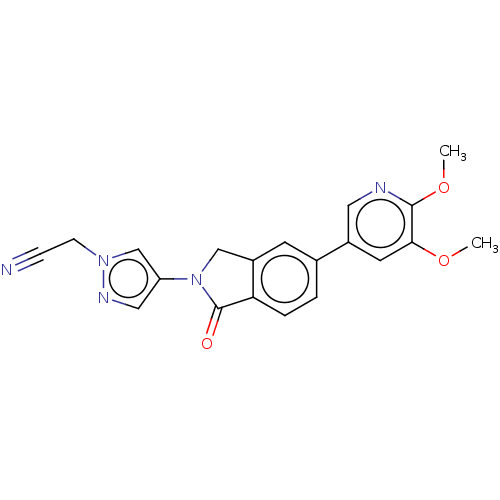
(CHEMBL4129180)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N(Cc2c1)c1cnn(CC#N)c1 Show InChI InChI=1S/C20H17N5O3/c1-27-18-8-14(9-22-19(18)28-2)13-3-4-17-15(7-13)11-25(20(17)26)16-10-23-24(12-16)6-5-21/h3-4,7-10,12H,6,11H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Escherichia coli (strain K12)) | BDBM24610
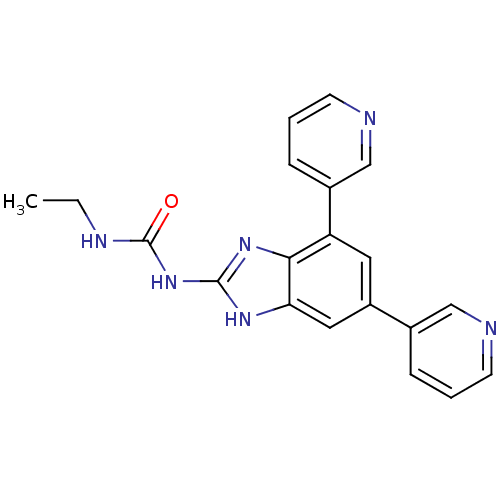
(1-[5,7-bis(pyridin-3-yl)-1H-1,3-benzodiazol-2-yl]-...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)-c1cccnc1 Show InChI InChI=1S/C20H18N6O/c1-2-23-20(27)26-19-24-17-10-15(13-5-3-7-21-11-13)9-16(18(17)25-19)14-6-4-8-22-12-14/h3-12H,2H2,1H3,(H3,23,24,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM24609

(3-ethyl-1-[7-(pyridin-2-yl)-5-(pyridin-3-yl)-1H-1,...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)-c1ccccn1 Show InChI InChI=1S/C20H18N6O/c1-2-22-20(27)26-19-24-17-11-14(13-6-5-8-21-12-13)10-15(18(17)25-19)16-7-3-4-9-23-16/h3-12H,2H2,1H3,(H3,22,24,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7 | -47.3 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274541
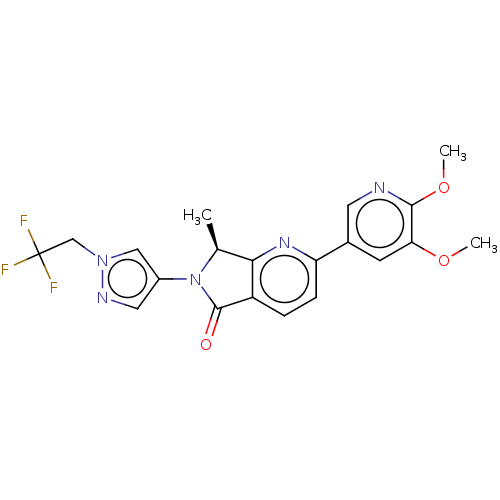
(CHEMBL4130036)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N([C@@H](C)c2n1)c1cnn(CC(F)(F)F)c1 |r| Show InChI InChI=1S/C20H18F3N5O3/c1-11-17-14(19(29)28(11)13-8-25-27(9-13)10-20(21,22)23)4-5-15(26-17)12-6-16(30-2)18(31-3)24-7-12/h4-9,11H,10H2,1-3H3/t11-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
DNA topoisomerase 4 subunit A/B
(Escherichia coli (strain K12)) | BDBM24616

(1-(6-(4-(Cyclopropylcarbamoyl)-1H-imidazol-1-yl)-4...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-n1cnc(c1)C(=O)NC1CC1)-c1ccccn1 Show InChI InChI=1S/C22H22N8O2/c1-2-23-22(32)29-21-27-17-10-14(9-15(19(17)28-21)16-5-3-4-8-24-16)30-11-18(25-12-30)20(31)26-13-6-7-13/h3-5,8-13H,2,6-7H2,1H3,(H,26,31)(H3,23,27,28,29,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274559

(CHEMBL4126707)Show SMILES CCn1cc(cn1)N1Cc2nc(ccc2C1=O)-c1cnc(OC)c(OC)c1 Show InChI InChI=1S/C19H19N5O3/c1-4-23-10-13(9-21-23)24-11-16-14(19(24)25)5-6-15(22-16)12-7-17(26-2)18(27-3)20-8-12/h5-10H,4,11H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM24606

(Benzimidazole urea analogue, 10 | methyl 2-[(ethyl...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-c1cccnc1)C(=O)OC Show InChI InChI=1S/C17H17N5O3/c1-3-19-17(24)22-16-20-13-8-11(10-5-4-6-18-9-10)7-12(14(13)21-16)15(23)25-2/h4-9H,3H2,1-2H3,(H3,19,20,21,22,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 8 | -47.0 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274543

(CHEMBL4129251)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N(Cc2n1)c1cnn(CC(F)F)c1 Show InChI InChI=1S/C19H17F2N5O3/c1-28-16-5-11(6-22-18(16)29-2)14-4-3-13-15(24-14)9-26(19(13)27)12-7-23-25(8-12)10-17(20)21/h3-8,17H,9-10H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM50393079
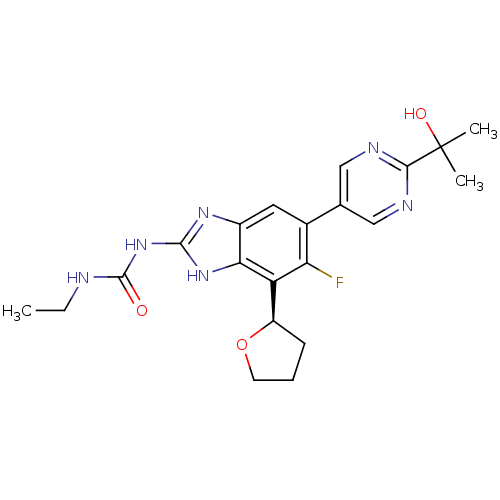
(CHEMBL2152855 | US9040542, 23)Show SMILES CCNC(=O)Nc1nc2cc(c(F)c([C@H]3CCCO3)c2[nH]1)-c1cnc(nc1)C(C)(C)O |r| Show InChI InChI=1S/C21H25FN6O3/c1-4-23-20(29)28-19-26-13-8-12(11-9-24-18(25-10-11)21(2,3)30)16(22)15(17(13)27-19)14-6-5-7-31-14/h8-10,14,30H,4-7H2,1-3H3,(H3,23,26,27,28,29)/t14-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus DNA gyrase using pBR322 plasmid DNA as substrate by coupled enzyme reaction assay |
J Med Chem 57: 8792-816 (2014)
Article DOI: 10.1021/jm500563g
BindingDB Entry DOI: 10.7270/Q2TF01BF |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50274542

(CHEMBL4127853)Show SMILES COc1cc(cnc1OC)-c1ccc2C(=O)N(Cc2n1)c1cnn(CC#C)c1 Show InChI InChI=1S/C20H17N5O3/c1-4-7-24-11-14(10-22-24)25-12-17-15(20(25)26)5-6-16(23-17)13-8-18(27-2)19(28-3)21-9-13/h1,5-6,8-11H,7,12H2,2-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate after 15 mins in presence of [33P-ATP] by liquid scintillation counting method |
J Med Chem 61: 5245-5256 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00085
BindingDB Entry DOI: 10.7270/Q2V98BK8 |
More data for this
Ligand-Target Pair | |
DNA gyrase subunit A/B
(Staphylococcus aureus) | BDBM24615

(Benzimidazole urea analogue, 19 | N-cyclopropyl-1-...)Show SMILES CCNC(=O)Nc1nc2c(cc(cc2[nH]1)-n1cnc(c1)C(=O)NC1CC1)-n1cccn1 Show InChI InChI=1S/C20H21N9O2/c1-2-21-20(31)27-19-25-14-8-13(9-16(17(14)26-19)29-7-3-6-23-29)28-10-15(22-11-28)18(30)24-12-4-5-12/h3,6-12H,2,4-5H2,1H3,(H,24,30)(H3,21,25,26,27,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 10 | -46.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
Vertex
| Assay Description
Enzymatic hydrolysis of ATP to ADP was coupled to the conversion of NADH to NAD+. The decrease in NADH absorbance was monitored at 340 nm for 20 min ... |
J Med Chem 51: 5243-63 (2008)
Article DOI: 10.1021/jm800318d
BindingDB Entry DOI: 10.7270/Q2J67F7T |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data