 Found 11468 hits with Last Name = 'lu' and Initial = 'h'
Found 11468 hits with Last Name = 'lu' and Initial = 'h' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093582
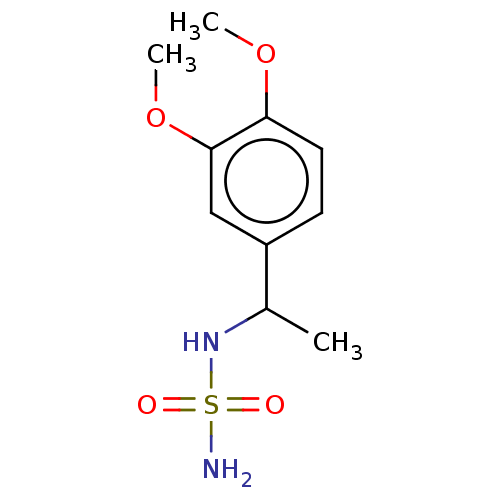
(CHEMBL3585782)Show InChI InChI=1S/C10H16N2O4S/c1-7(12-17(11,13)14)8-4-5-9(15-2)10(6-8)16-3/h4-7,12H,1-3H3,(H2,11,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 1 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093588
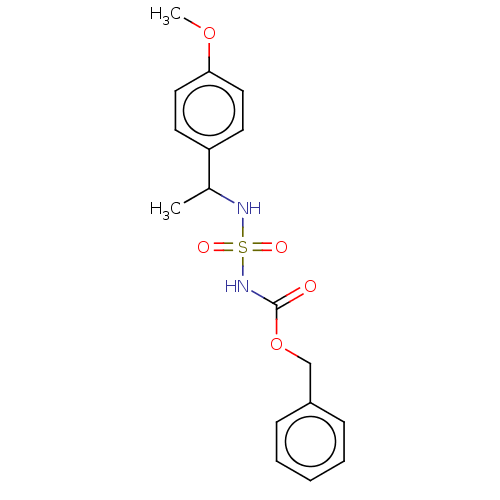
(CHEMBL3585776)Show InChI InChI=1S/C17H20N2O5S/c1-13(15-8-10-16(23-2)11-9-15)18-25(21,22)19-17(20)24-12-14-6-4-3-5-7-14/h3-11,13,18H,12H2,1-2H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093587
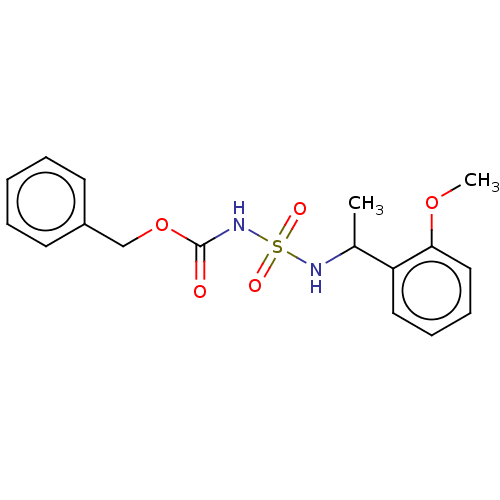
(CHEMBL3585777)Show SMILES COc1ccc(cc1OC)C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(15-9-10-16(24-2)17(11-15)25-3)19-27(22,23)20-18(21)26-12-14-7-5-4-6-8-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093589
(CHEMBL3585775)Show InChI InChI=1S/C17H20N2O5S/c1-13(15-10-6-7-11-16(15)23-2)18-25(21,22)19-17(20)24-12-14-8-4-3-5-9-14/h3-11,13,18H,12H2,1-2H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093583
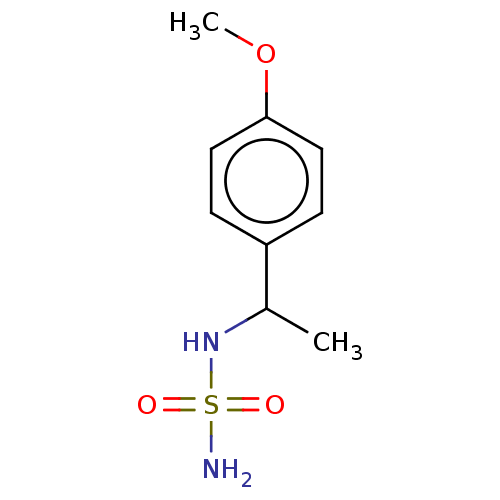
(CHEMBL3585781)Show InChI InChI=1S/C9H14N2O3S/c1-7(11-15(10,12)13)8-3-5-9(14-2)6-4-8/h3-7,11H,1-2H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093581
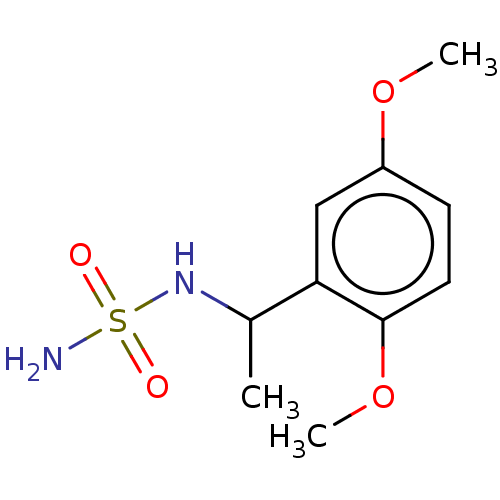
(CHEMBL3585783)Show InChI InChI=1S/C10H16N2O4S/c1-7(12-17(11,13)14)9-6-8(15-2)4-5-10(9)16-3/h4-7,12H,1-3H3,(H2,11,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388434
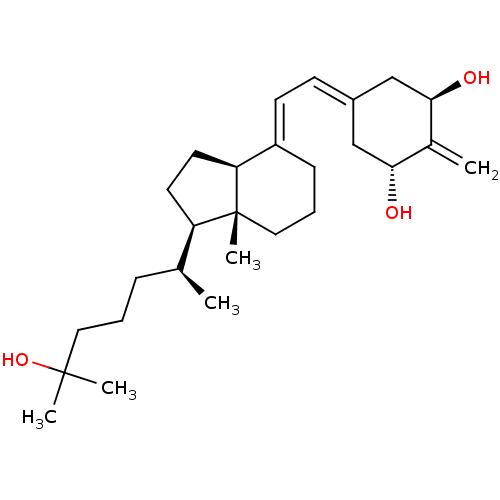
(CHEMBL605525)Show SMILES [#6]-[#6@@H](-[#6]-[#6]-[#6]C([#6])([#6])[#8])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1\[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)22-12-13-23-21(9-7-15-27(22,23)5)11-10-20-16-24(28)19(2)25(29)17-20/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b21-11+/t18-,22+,23-,24+,25+,27+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Activity at rat recombinant full length VDR expressed in rat ROS 17/2.8 cells transfected with 24-hydroxylase gene promoter assessed as transcription... |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093597
(CHEMBL3585784)Show InChI InChI=1S/C21H33NO2/c1-13(22-24)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h4,15-19,23-24H,5-12H2,1-3H3/b22-13-/t15?,16?,17-,18?,19?,20?,21?/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM25761
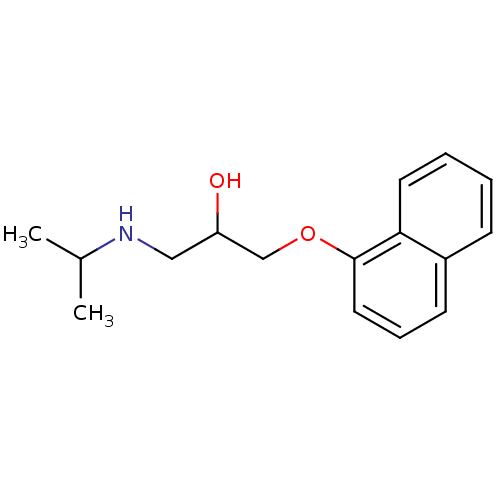
(Anapriline | Avlocardyl | CHEMBL27 | PROPANOLOL(-)...)Show InChI InChI=1S/C16H21NO2/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16/h3-9,12,14,17-18H,10-11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417515
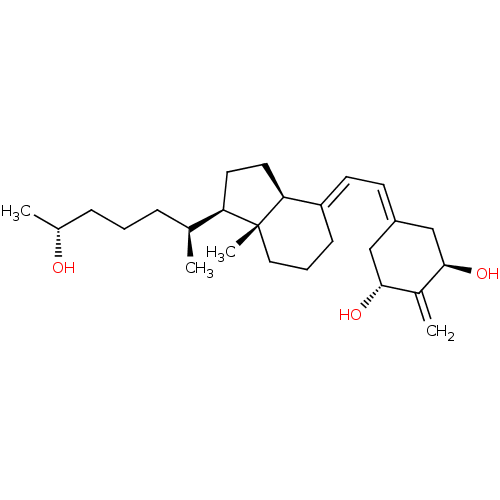
(CHEMBL1630755)Show SMILES [#6]-[#6@@H](-[#8])-[#6]-[#6]-[#6]-[#6@H](-[#6])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C26H42O3/c1-17(7-5-8-18(2)27)22-12-13-23-21(9-6-14-26(22,23)4)11-10-20-15-24(28)19(3)25(29)16-20/h10-11,17-18,22-25,27-29H,3,5-9,12-16H2,1-2,4H3/b21-11+/t17-,18+,22+,23-,24+,25+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Activity at rat recombinant full length VDR expressed in rat ROS 17/2.8 cells transfected with 24-hydroxylase gene promoter assessed as transcription... |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093585
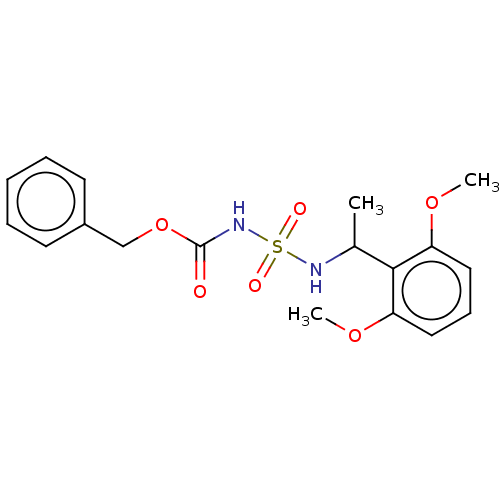
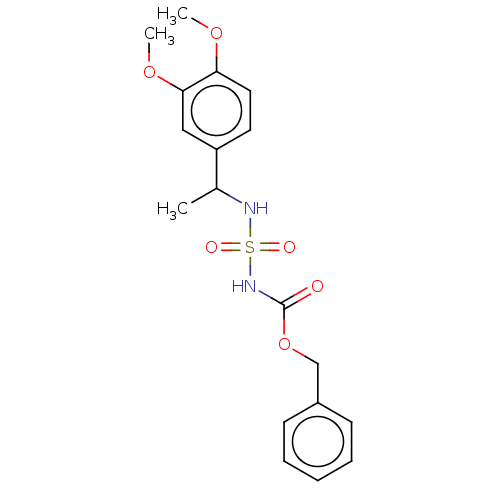
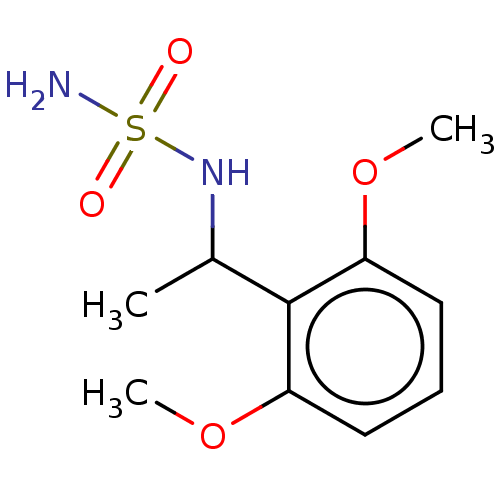
(CHEMBL3585779)Show SMILES COc1cccc(OC)c1C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(17-15(24-2)10-7-11-16(17)25-3)19-27(22,23)20-18(21)26-12-14-8-5-4-6-9-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 1 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM25761

(Anapriline | Avlocardyl | CHEMBL27 | PROPANOLOL(-)...)Show InChI InChI=1S/C16H21NO2/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16/h3-9,12,14,17-18H,10-11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50135183

(CHEMBL3745798)Show SMILES [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6]-[#6@H](-[#8])-[#6]-1)[#6@H](-[#6])-[#6]-[#6]C(F)(F)C([#6])([#6])[#8] |r| Show InChI InChI=1S/C26H42F2O3/c1-17(11-13-26(27,28)24(2,3)31)22-9-10-23-19(6-5-12-25(22,23)4)8-7-18-14-20(29)16-21(30)15-18/h7-8,17,20-23,29-31H,5-6,9-16H2,1-4H3/b19-8+/t17-,20-,21-,22-,23+,25-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50304585
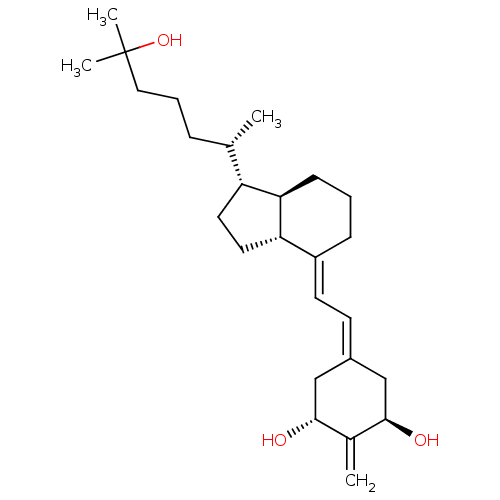
((20S)-1alpha,25-Dihydroxy-2-methylene-18,19-dinorv...)Show SMILES [#6]-[#6@@H](-[#6]-[#6]-[#6]C([#6])([#6])[#8])-[#6@H]-1-[#6]-[#6]-[#6@@H]2-[#6@@H]-1-[#6]-[#6]-[#6]\[#6]-2=[#6]/[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C26H42O3/c1-17(7-6-14-26(3,4)29)21-12-13-22-20(8-5-9-23(21)22)11-10-19-15-24(27)18(2)25(28)16-19/h10-11,17,21-25,27-29H,2,5-9,12-16H2,1,3-4H3/b20-11+/t17-,21+,22-,23+,24+,25+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled 1alpha,25-(OH)2D3 from rat recombinant full length VDR |
Bioorg Med Chem 17: 7658-69 (2009)
Article DOI: 10.1016/j.bmc.2009.09.047
BindingDB Entry DOI: 10.7270/Q26T0MQM |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417519
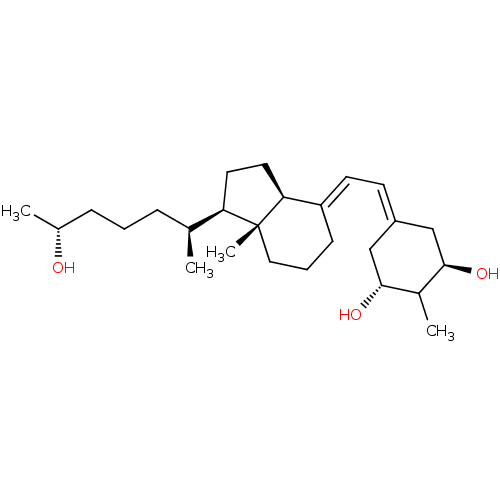
(CHEMBL1630759)Show SMILES C[C@@H](O)CCC[C@H](C)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1C[C@@H](O)C(C)[C@H](O)C1 |r,wU:22.24,11.10,16.18,8.7,6.6,1.1,wD:26.28,(1.96,-41.79,;.6,-42.52,;.6,-44.06,;-.7,-41.71,;-2.06,-42.44,;-3.37,-41.62,;-4.73,-42.35,;-6.07,-41.6,;-4.71,-43.89,;-3.82,-45.14,;-4.73,-46.37,;-6.18,-45.89,;-7.5,-46.66,;-8.85,-45.89,;-8.84,-44.35,;-7.5,-43.58,;-6.17,-44.36,;-6.18,-42.82,;-7.5,-48.2,;-8.84,-48.97,;-8.84,-50.51,;-7.51,-51.29,;-7.51,-52.83,;-6.17,-53.6,;-8.84,-53.59,;-8.84,-55.13,;-10.17,-52.83,;-11.51,-53.6,;-10.17,-51.29,)| Show InChI InChI=1S/C26H44O3/c1-17(7-5-8-18(2)27)22-12-13-23-21(9-6-14-26(22,23)4)11-10-20-15-24(28)19(3)25(29)16-20/h10-11,17-19,22-25,27-29H,5-9,12-16H2,1-4H3/b20-10-,21-11+/t17-,18+,19?,22+,23-,24+,25+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Activity at rat recombinant full length VDR expressed in rat ROS 17/2.8 cells transfected with 24-hydroxylase gene promoter assessed as transcription... |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388439
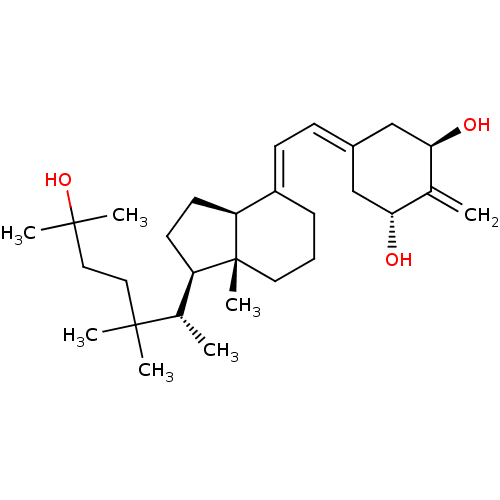
(CHEMBL2059272)Show SMILES [#6]-[#6@@H](-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)C([#6])([#6])[#6]-[#6]C([#6])([#6])[#8] |r| Show InChI InChI=1S/C29H48O3/c1-19-25(30)17-21(18-26(19)31)10-11-22-9-8-14-29(7)23(12-13-24(22)29)20(2)27(3,4)15-16-28(5,6)32/h10-11,20,23-26,30-32H,1,8-9,12-18H2,2-7H3/b22-11+/t20-,23+,24-,25+,26+,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093586
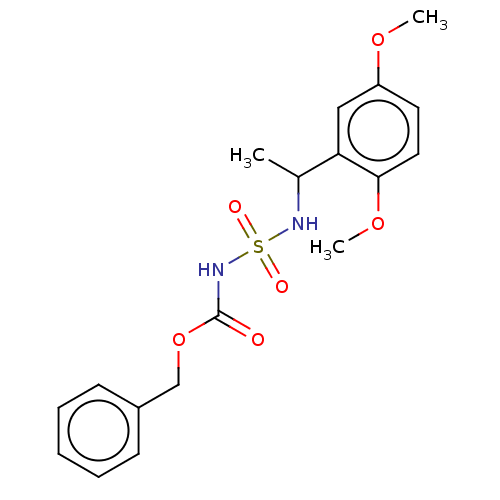
(CHEMBL3585778)Show SMILES COc1ccc(OC)c(c1)C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(16-11-15(24-2)9-10-17(16)25-3)19-27(22,23)20-18(21)26-12-14-7-5-4-6-8-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50135182
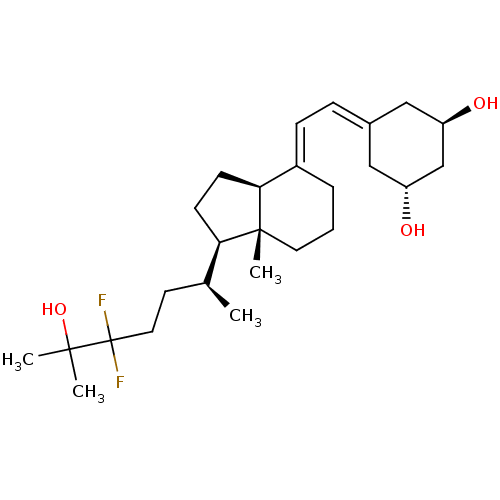
(CHEMBL3746107)Show SMILES [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6]-[#6@H](-[#8])-[#6]-1)[#6@@H](-[#6])-[#6]-[#6]C(F)(F)C([#6])([#6])[#8] |r| Show InChI InChI=1S/C26H42F2O3/c1-17(11-13-26(27,28)24(2,3)31)22-9-10-23-19(6-5-12-25(22,23)4)8-7-18-14-20(29)16-21(30)15-18/h7-8,17,20-23,29-31H,5-6,9-16H2,1-4H3/b19-8+/t17-,20+,21+,22+,23-,25+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50135184
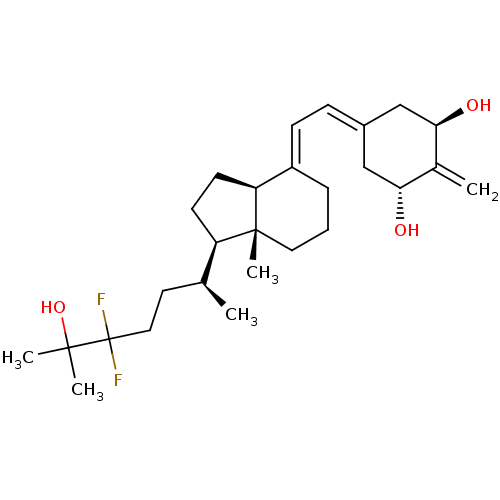
(CHEMBL3746058)Show SMILES [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)[#6@@H](-[#6])-[#6]-[#6]C(F)(F)C([#6])([#6])[#8] |r| Show InChI InChI=1S/C27H42F2O3/c1-17(12-14-27(28,29)25(3,4)32)21-10-11-22-20(7-6-13-26(21,22)5)9-8-19-15-23(30)18(2)24(31)16-19/h8-9,17,21-24,30-32H,2,6-7,10-16H2,1,3-5H3/b20-9+/t17-,21+,22-,23+,24+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50135186
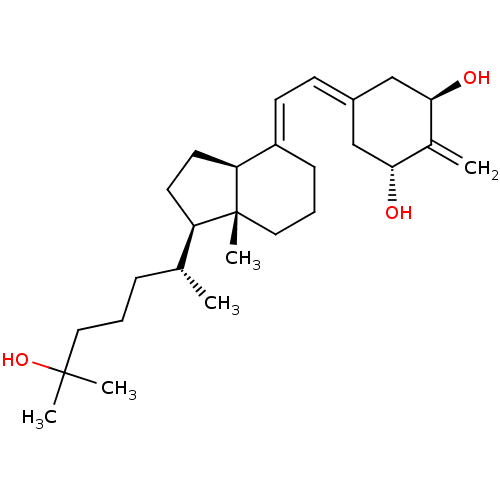
(CHEMBL1214620)Show SMILES [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)[#6@H](-[#6])-[#6]-[#6]-[#6]C([#6])([#6])[#8] |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)22-12-13-23-21(9-7-15-27(22,23)5)11-10-20-16-24(28)19(2)25(29)17-20/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b21-11+/t18-,22-,23+,24-,25-,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50093584
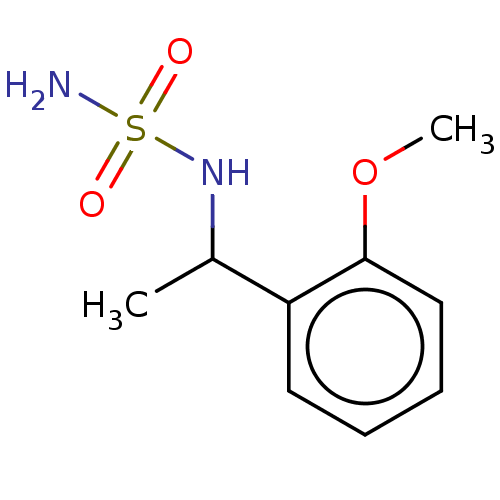
(CHEMBL3585780)Show InChI InChI=1S/C9H14N2O3S/c1-7(11-15(10,12)13)8-5-3-4-6-9(8)14-2/h3-7,11H,1-2H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50135185

(CHEMBL3746759)Show SMILES [H][C@@]1([#6]-[#6][C@@]2([H])\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)[#6@H](-[#6])-[#6]-[#6]C(F)(F)C([#6])([#6])[#8] |r| Show InChI InChI=1S/C27H42F2O3/c1-17(12-14-27(28,29)25(3,4)32)21-10-11-22-20(7-6-13-26(21,22)5)9-8-19-15-23(30)18(2)24(31)16-19/h8-9,17,21-24,30-32H,2,6-7,10-16H2,1,3-5H3/b20-9+/t17-,21-,22+,23-,24-,26-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182
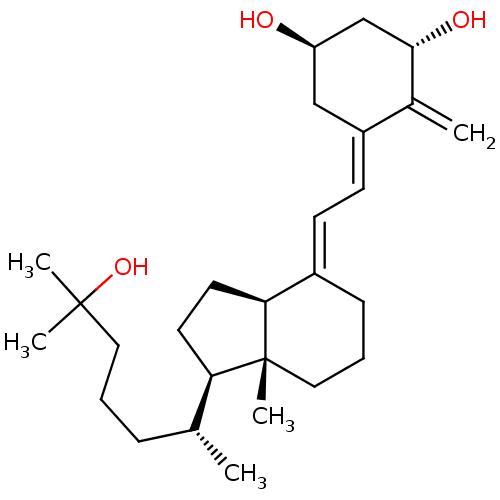
((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from full-length recombinant rat VDR by scintillation counter |
J Med Chem 58: 9731-41 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01564
BindingDB Entry DOI: 10.7270/Q2JW8GQS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388440

(CHEMBL2059269)Show SMILES [#6]-[#6@H](-[#6]-[#6]C([#6])([#6])[#8])-[#6@H](-[#6])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C28H46O3/c1-18(13-15-27(4,5)31)19(2)23-11-12-24-22(8-7-14-28(23,24)6)10-9-21-16-25(29)20(3)26(30)17-21/h9-10,18-19,23-26,29-31H,3,7-8,11-17H2,1-2,4-6H3/b22-10+/t18-,19+,23-,24+,25-,26-,28-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50093585

(CHEMBL3585779)Show SMILES COc1cccc(OC)c1C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(17-15(24-2)10-7-11-16(17)25-3)19-27(22,23)20-18(21)26-12-14-8-5-4-6-9-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50093587

(CHEMBL3585777)Show SMILES COc1ccc(cc1OC)C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(15-9-10-16(24-2)17(11-15)25-3)19-27(22,23)20-18(21)26-12-14-7-5-4-6-8-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of acetylcholinesterase (unknown origin) assessed as AChI hydrolysis using AChI and DTNB as substrate by Ellman's method |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50093586

(CHEMBL3585778)Show SMILES COc1ccc(OC)c(c1)C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(16-11-15(24-2)9-10-17(16)25-3)19-27(22,23)20-18(21)26-12-14-7-5-4-6-8-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
Bioorg Med Chem 16: 8563-73 (2008)
Article DOI: 10.1016/j.bmc.2008.08.011
BindingDB Entry DOI: 10.7270/Q20001X4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Francisella tularensis) | BDBM8726
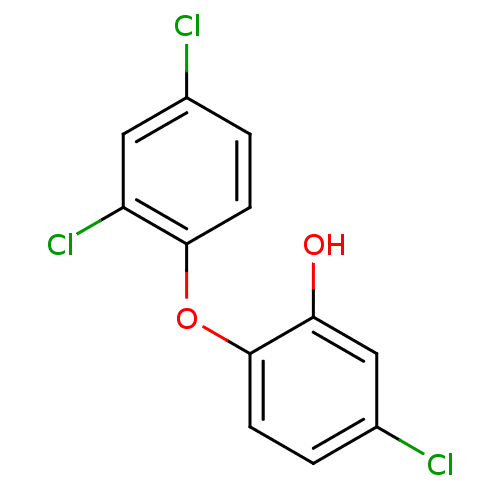
(5-chloro-2-(2,4-dichlorophenoxy)phenol | CHEMBL849...)Show InChI InChI=1S/C12H7Cl3O2/c13-7-1-3-11(9(15)5-7)17-12-4-2-8(14)6-10(12)16/h1-6,16H | PDB
MMDB
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0510 | -58.7 | n/a | n/a | n/a | n/a | n/a | 8.0 | 25 |
Stony Brook University
| Assay Description
Inhibition constant binding to E-NAD+ |
ACS Chem Biol 4: 221-31 (2009)
Article DOI: 10.1021/cb800306y
BindingDB Entry DOI: 10.7270/Q2T72FTC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50093589

(CHEMBL3585775)Show InChI InChI=1S/C17H20N2O5S/c1-13(15-10-6-7-11-16(15)23-2)18-25(21,22)19-17(20)24-12-14-8-4-3-5-9-14/h3-11,13,18H,12H2,1-2H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417520

(CHEMBL1630753)Show SMILES C[C@H](O)CCC[C@H](C)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C(=C)[C@H](O)C1=C |r| Show InChI InChI=1S/C27H42O3/c1-17(8-6-9-18(2)28)23-13-14-24-21(10-7-15-27(23,24)5)11-12-22-16-25(29)20(4)26(30)19(22)3/h11-12,17-18,23-26,28-30H,3-4,6-10,13-16H2,1-2,5H3/b21-11+,22-12-/t17-,18-,23+,24-,25+,26+,27+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Activity at rat recombinant full length VDR expressed in rat ROS 17/2.8 cells transfected with 24-hydroxylase gene promoter assessed as transcription... |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388438
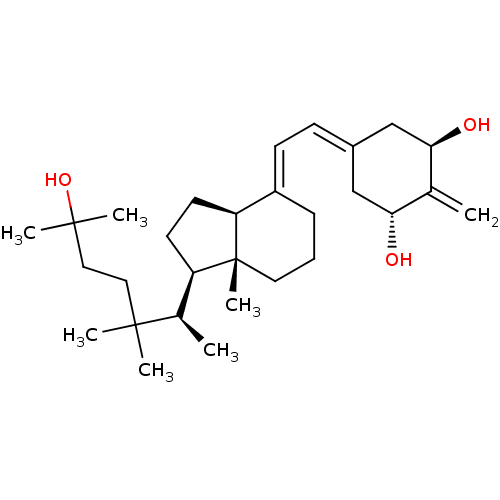
(CHEMBL2059271)Show SMILES [#6]-[#6@H](-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1)C([#6])([#6])[#6]-[#6]C([#6])([#6])[#8] |r| Show InChI InChI=1S/C29H48O3/c1-19-25(30)17-21(18-26(19)31)10-11-22-9-8-14-29(7)23(12-13-24(22)29)20(2)27(3,4)15-16-28(5,6)32/h10-11,20,23-26,30-32H,1,8-9,12-18H2,2-7H3/b22-11+/t20-,23-,24+,25-,26-,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417520

(CHEMBL1630753)Show SMILES C[C@H](O)CCC[C@H](C)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C(=C)[C@H](O)C1=C |r| Show InChI InChI=1S/C27H42O3/c1-17(8-6-9-18(2)28)23-13-14-24-21(10-7-15-27(23,24)5)11-12-22-16-25(29)20(4)26(30)19(22)3/h11-12,17-18,23-26,28-30H,3-4,6-10,13-16H2,1-2,5H3/b21-11+,22-12-/t17-,18-,23+,24-,25+,26+,27+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM85034

(BW-501 | BW-501C | BW501)Show InChI InChI=1S/C17H20ClN3O/c18-15-9-4-5-10-16(15)22-12-6-11-20-17(19)13-21-14-7-2-1-3-8-14/h1-5,7-10,21H,6,11-13H2,(H2,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417519

(CHEMBL1630759)Show SMILES C[C@@H](O)CCC[C@H](C)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1C[C@@H](O)C(C)[C@H](O)C1 |r,wU:22.24,11.10,16.18,8.7,6.6,1.1,wD:26.28,(1.96,-41.79,;.6,-42.52,;.6,-44.06,;-.7,-41.71,;-2.06,-42.44,;-3.37,-41.62,;-4.73,-42.35,;-6.07,-41.6,;-4.71,-43.89,;-3.82,-45.14,;-4.73,-46.37,;-6.18,-45.89,;-7.5,-46.66,;-8.85,-45.89,;-8.84,-44.35,;-7.5,-43.58,;-6.17,-44.36,;-6.18,-42.82,;-7.5,-48.2,;-8.84,-48.97,;-8.84,-50.51,;-7.51,-51.29,;-7.51,-52.83,;-6.17,-53.6,;-8.84,-53.59,;-8.84,-55.13,;-10.17,-52.83,;-11.51,-53.6,;-10.17,-51.29,)| Show InChI InChI=1S/C26H44O3/c1-17(7-5-8-18(2)27)22-12-13-23-21(9-6-14-26(22,23)4)11-10-20-15-24(28)19(3)25(29)16-20/h10-11,17-19,22-25,27-29H,5-9,12-16H2,1-4H3/b20-10-,21-11+/t17-,18+,19?,22+,23-,24+,25+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417517

(CHEMBL1630757)Show SMILES C[C@H](O)CCC[C@H](C)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1C[C@@H](O)C(C)[C@H](O)C1 |r,wU:22.24,11.10,16.18,8.7,6.6,wD:26.28,1.1,(14.49,-28.73,;13.13,-29.45,;13.13,-30.99,;11.83,-28.64,;10.47,-29.37,;9.16,-28.56,;7.8,-29.29,;6.46,-28.53,;7.82,-30.83,;8.71,-32.07,;7.8,-33.31,;6.35,-32.82,;5.03,-33.59,;3.68,-32.83,;3.69,-31.28,;5.03,-30.51,;6.36,-31.29,;6.35,-29.75,;5.03,-35.13,;3.69,-35.9,;3.69,-37.44,;5.02,-38.22,;5.02,-39.76,;6.36,-40.53,;3.69,-40.52,;3.69,-42.06,;2.36,-39.76,;1.03,-40.53,;2.36,-38.22,)| Show InChI InChI=1S/C26H44O3/c1-17(7-5-8-18(2)27)22-12-13-23-21(9-6-14-26(22,23)4)11-10-20-15-24(28)19(3)25(29)16-20/h10-11,17-19,22-25,27-29H,5-9,12-16H2,1-4H3/b20-10-,21-11+/t17-,18-,19?,22+,23-,24+,25+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Activity at rat recombinant full length VDR expressed in rat ROS 17/2.8 cells transfected with 24-hydroxylase gene promoter assessed as transcription... |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50093587

(CHEMBL3585777)Show SMILES COc1ccc(cc1OC)C(C)NS(=O)(=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C18H22N2O6S/c1-13(15-9-10-16(24-2)17(11-15)25-3)19-27(22,23)20-18(21)26-12-14-7-5-4-6-8-14/h4-11,13,19H,12H2,1-3H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0890 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AgriIbrahim£e£en University
Curated by ChEMBL
| Assay Description
Inhibition of esterase activity of carbonic anhydrase 2 in human erythrocytes using PNF as substrate by spectrophotometer analysis |
Bioorg Med Chem 23: 3592-602 (2015)
Article DOI: 10.1016/j.bmc.2015.04.019
BindingDB Entry DOI: 10.7270/Q2J104XP |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735
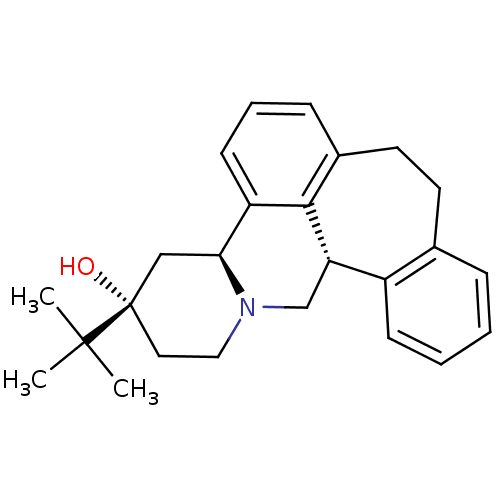
((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50417515

(CHEMBL1630755)Show SMILES [#6]-[#6@@H](-[#8])-[#6]-[#6]-[#6]-[#6@H](-[#6])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C26H42O3/c1-17(7-5-8-18(2)27)22-12-13-23-21(9-6-14-26(22,23)4)11-10-20-15-24(28)19(3)25(29)16-20/h10-11,17-18,22-25,27-29H,3,5-9,12-16H2,1-2,4H3/b21-11+/t17-,18+,22+,23-,24+,25+,26+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735

((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Warsaw
Curated by ChEMBL
| Assay Description
Displacement of radiolabelled 1alpha, 25-(OH)2D3 from recombinant rat VDR |
J Med Chem 54: 6832-42 (2011)
Article DOI: 10.1021/jm200743p
BindingDB Entry DOI: 10.7270/Q2K35V1M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50304583
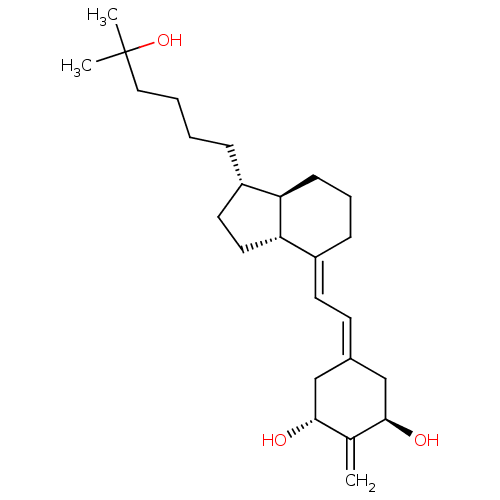
(1alpha,25-Dihydroxy-2-methylene-18,19,21-trinorvit...)Show SMILES [#6]C([#6])([#8])[#6]-[#6]-[#6]-[#6]-[#6@H]-1-[#6]-[#6]-[#6@@H]2-[#6@@H]-1-[#6]-[#6]-[#6]\[#6]-2=[#6]/[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C25H40O3/c1-17-23(26)15-18(16-24(17)27)10-11-20-8-6-9-21-19(12-13-22(20)21)7-4-5-14-25(2,3)28/h10-11,19,21-24,26-28H,1,4-9,12-16H2,2-3H3/b20-11+/t19-,21+,22-,23+,24+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled 1alpha,25-(OH)2D3 from rat recombinant full length VDR |
Bioorg Med Chem 17: 7658-69 (2009)
Article DOI: 10.1016/j.bmc.2009.09.047
BindingDB Entry DOI: 10.7270/Q26T0MQM |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
Bioorg Med Chem 17: 1747-63 (2009)
Article DOI: 10.1016/j.bmc.2008.11.082
BindingDB Entry DOI: 10.7270/Q2MK6CRR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Warsaw
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha25-(OH)2D3 from full length recombinant rat vitamin D receptor by scintillation counting analysis |
J Med Chem 58: 6237-47 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00795
BindingDB Entry DOI: 10.7270/Q2H133S9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM93062

(Vitamin D analog, 11 | Vitamin D analog, 15)Show SMILES C[C@@H](CCCCO)C1CCC2\C(CCC[C@]12C)=C\C=C1C[C@@H](O)C(C)[C@H](O)C1 |r,wU:23.25,25.27,15.17,1.0,wD:21.23,@@:23,(-.69,7.62,;.08,6.29,;1.59,6.61,;2.62,5.46,;4.13,5.78,;5.16,4.64,;6.66,4.96,;-.39,4.82,;.51,3.58,;-.39,2.33,;-1.86,2.81,;-3.19,2.04,;-4.52,2.81,;-4.52,4.35,;-3.19,5.12,;-1.86,4.35,;-1.86,5.89,;-3.19,.5,;-4.52,-.27,;-4.52,-1.81,;-5.86,-2.58,;-5.86,-4.13,;-7.19,-4.9,;-4.52,-4.9,;-4.52,-6.44,;-3.19,-4.13,;-1.86,-4.9,;-3.19,-2.58,)| Show InChI InChI=1S/C25H42O3/c1-17(7-4-5-14-26)21-11-12-22-20(8-6-13-25(21,22)3)10-9-19-15-23(27)18(2)24(28)16-19/h9-10,17-18,21-24,26-28H,4-8,11-16H2,1-3H3/b19-9-,20-10+/t17-,18?,21?,22?,23+,24+,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
| Assay Description
In vitro study, VDR binding assay were performed as previously described. |
Bioorg Chem 47: 9-16 (2013)
Article DOI: 10.1016/j.bioorg.2013.01.001
BindingDB Entry DOI: 10.7270/Q2FX782T |
More data for this
Ligand-Target Pair | |
Protachykinin-1
(Homo sapiens (Human)) | BDBM50001450
((SP)Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH...)Show SMILES [#6]-[#16]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C63H98N18O13S/c1-37(2)33-45(57(89)74-41(53(68)85)27-32-95-3)73-52(84)36-72-54(86)46(34-38-15-6-4-7-16-38)78-58(90)47(35-39-17-8-5-9-18-39)79-56(88)42(23-25-50(66)82)75-55(87)43(24-26-51(67)83)76-59(91)49-22-14-31-81(49)62(94)44(20-10-11-28-64)77-60(92)48-21-13-30-80(48)61(93)40(65)19-12-29-71-63(69)70/h4-9,15-18,37,40-49H,10-14,19-36,64-65H2,1-3H3,(H2,66,82)(H2,67,83)(H2,68,85)(H,72,86)(H,73,84)(H,74,89)(H,75,87)(H,76,91)(H,77,92)(H,78,90)(H,79,88)(H4,69,70,71)/t40-,41-,42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388434

(CHEMBL605525)Show SMILES [#6]-[#6@@H](-[#6]-[#6]-[#6]C([#6])([#6])[#8])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1\[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)22-12-13-23-21(9-7-15-27(22,23)5)11-10-20-16-24(28)19(2)25(29)17-20/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b21-11+/t18-,22+,23-,24+,25+,27+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50388436

(CHEMBL2059268)Show SMILES [#6]-[#6@@H](-[#6]-[#6]C([#6])([#6])[#8])-[#6@@H](-[#6])-[#6@H]1-[#6]-[#6]-[#6@H]2\[#6](-[#6]-[#6]-[#6][C@]12[#6])=[#6]\[#6]=[#6]-1/[#6]-[#6@@H](-[#8])-[#6](=[#6])-[#6@H](-[#8])-[#6]-1 |r| Show InChI InChI=1S/C28H46O3/c1-18(13-15-27(4,5)31)19(2)23-11-12-24-22(8-7-14-28(23,24)6)10-9-21-16-25(29)20(3)26(30)17-21/h9-10,18-19,23-26,29-31H,3,7-8,11-17H2,1-2,4-6H3/b22-10+/t18-,19+,23+,24-,25+,26+,28+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]-1alpha,25(OH)2D3 from recombinant rat VDR after overnight incubation by scintillation counting |
J Med Chem 55: 4352-66 (2012)
Article DOI: 10.1021/jm300187x
BindingDB Entry DOI: 10.7270/Q2PK0H71 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Rattus norvegicus) | BDBM50200182

((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...)Show SMILES C[C@H](CCCC(C)(C)O)[C@H]1CC[C@H]2\C(CCC[C@]12C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C |r| Show InChI InChI=1S/C27H44O3/c1-18(8-6-14-26(3,4)30)23-12-13-24-20(9-7-15-27(23,24)5)10-11-21-16-22(28)17-25(29)19(21)2/h10-11,18,22-25,28-30H,2,6-9,12-17H2,1,3-5H3/b20-10+,21-11-/t18-,22-,23-,24+,25+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Displacement of [3H]1alpha,25-(OH)2D3 from rat recombinant full length VDR |
J Med Chem 53: 8642-9 (2010)
Article DOI: 10.1021/jm1010447
BindingDB Entry DOI: 10.7270/Q23B61D2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data