 Found 30228 hits with Last Name = 'lu' and Initial = 'm'
Found 30228 hits with Last Name = 'lu' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glutamyl aminopeptidase
(Sus scrofa) | BDBM50078120
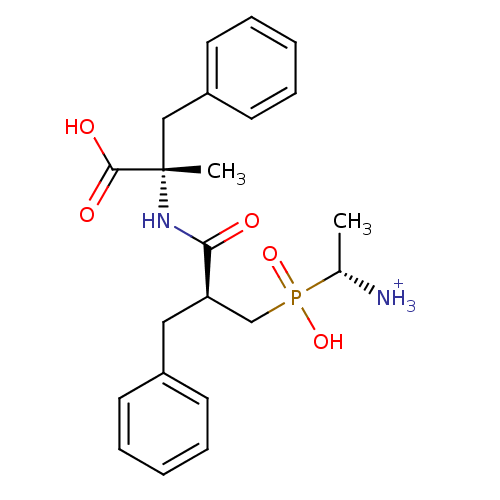
((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for A+B stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50078120

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for C+D stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase B
(Mus musculus) | BDBM50078122
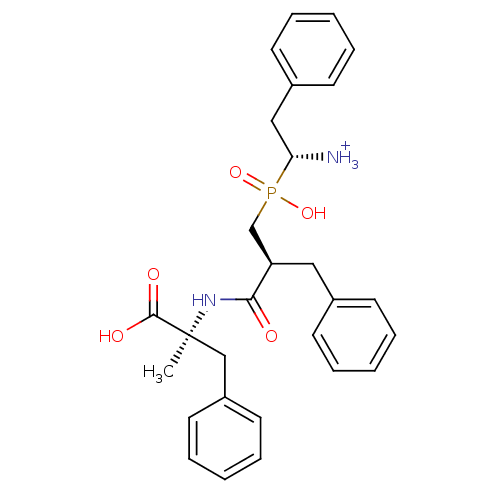
((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase B using Arg p.NA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Glutamyl aminopeptidase
(Sus scrofa) | BDBM50078122

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase using GluNA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase B
(Mus musculus) | BDBM50078120

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase B using Arg p.NA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50078122

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for C+D stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433523
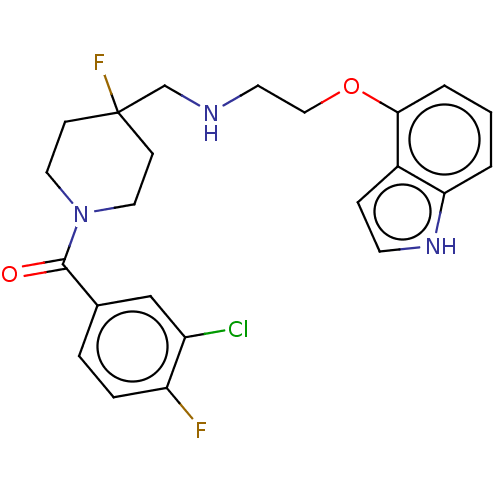
(US10562853, Compound 44)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2cccc3[nH]ccc23)CC1 Show InChI InChI=1S/C23H24ClF2N3O2/c24-18-14-16(4-5-19(18)25)22(30)29-11-7-23(26,8-12-29)15-27-10-13-31-21-3-1-2-20-17(21)6-9-28-20/h1-6,9,14,27-28H,7-8,10-13,15H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433523

(US10562853, Compound 44)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2cccc3[nH]ccc23)CC1 Show InChI InChI=1S/C23H24ClF2N3O2/c24-18-14-16(4-5-19(18)25)22(30)29-11-7-23(26,8-12-29)15-27-10-13-31-21-3-1-2-20-17(21)6-9-28-20/h1-6,9,14,27-28H,7-8,10-13,15H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50493155
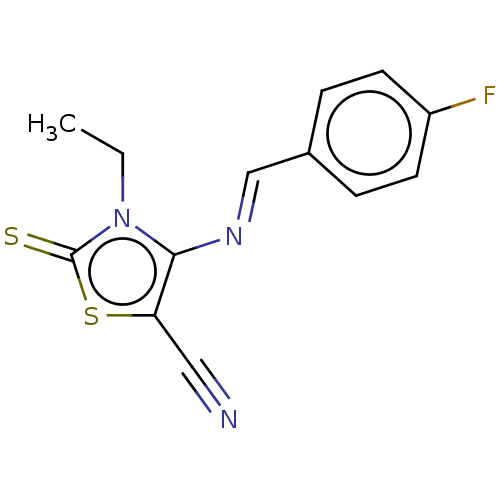
(CHEMBL2419148)Show InChI InChI=1S/C13H10FN3S2/c1-2-17-12(11(7-15)19-13(17)18)16-8-9-3-5-10(14)6-4-9/h3-6,8H,2H2,1H3/b16-8+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPCPX from human A1 receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50493144
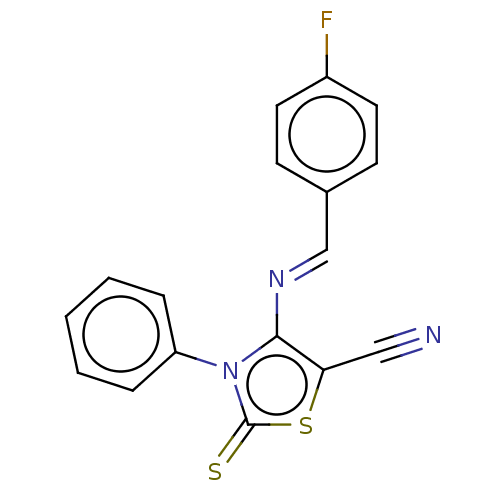
(CHEMBL2419144)Show InChI InChI=1S/C17H10FN3S2/c18-13-8-6-12(7-9-13)11-20-16-15(10-19)23-17(22)21(16)14-4-2-1-3-5-14/h1-9,11H/b20-11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPCPX from human A1 receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Homo sapiens (Human)) | BDBM50493151
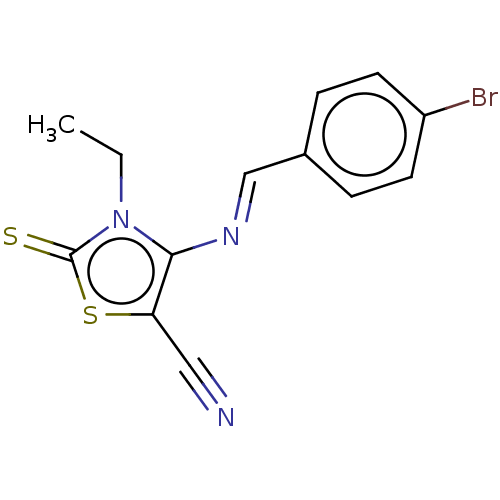
(CHEMBL2419139)Show InChI InChI=1S/C13H10BrN3S2/c1-2-17-12(11(7-15)19-13(17)18)16-8-9-3-5-10(14)6-4-9/h3-6,8H,2H2,1H3/b16-8+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPCPX from human A1 receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50434903
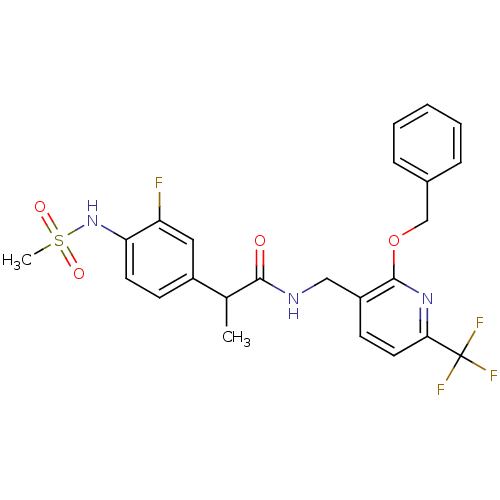
(CHEMBL2385223)Show SMILES CC(C(=O)NCc1ccc(nc1OCc1ccccc1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 Show InChI InChI=1S/C24H23F4N3O4S/c1-15(17-8-10-20(19(25)12-17)31-36(2,33)34)22(32)29-13-18-9-11-21(24(26,27)28)30-23(18)35-14-16-6-4-3-5-7-16/h3-12,15,31H,13-14H2,1-2H3,(H,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-arachidonoyldopamine-induced activity after 5 mins by FLIPR a... |
Eur J Med Chem 64: 589-602 (2013)
Article DOI: 10.1016/j.ejmech.2013.04.003
BindingDB Entry DOI: 10.7270/Q2BZ67FC |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096105
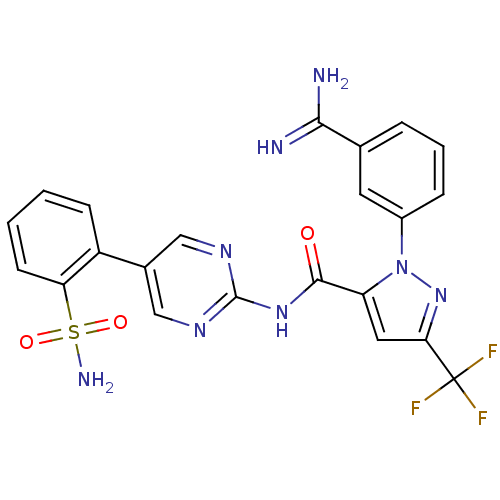
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ncc(cn1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C22H17F3N8O3S/c23-22(24,25)18-9-16(33(32-18)14-5-3-4-12(8-14)19(26)27)20(34)31-21-29-10-13(11-30-21)15-6-1-2-7-17(15)37(28,35)36/h1-11H,(H3,26,27)(H2,28,35,36)(H,29,30,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 4
(RAT) | BDBM82300
(CAS_59763-91-6 | NSC_41735 | PP, rat)Show SMILES CCC(C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=[NH2+])NC(=O)[C@H](CCCNC(N)=[NH2+])NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=[NH2+])NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](Cc1c[nH+]c[nH]1)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCSC)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]1CCCN1C(=O)C(C)N)C(C)O)C(C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C195H298N58O57S/c1-17-96(8)151(182(302)243-135(88-145(200)266)177(297)249-155(103(15)257)186(306)241-130(81-95(6)7)175(295)248-154(102(14)256)184(304)234-125(31-22-73-217-195(210)211)188(308)252-76-25-34-140(252)179(299)230-118(30-21-72-216-194(208)209)161(281)235-127(156(201)276)82-104-36-46-110(258)47-37-104)245-176(296)133(85-107-42-52-113(261)53-43-107)238-162(282)117(29-20-71-215-193(206)207)225-160(280)116(28-19-70-214-192(204)205)226-170(290)128(79-93(2)3)236-166(286)121(58-64-144(199)265)232-183(303)153(101(13)255)247-168(288)123(60-66-148(270)271)229-172(292)132(84-106-40-50-112(260)51-41-106)237-165(285)119(56-62-142(197)263)223-157(277)98(10)220-159(279)115(27-18-69-213-191(202)203)224-163(283)120(57-63-143(198)264)227-164(284)122(59-65-147(268)269)228-173(293)134(87-109-90-212-92-219-109)242-185(305)152(100(12)254)246-158(278)99(11)221-169(289)131(83-105-38-48-111(259)49-39-105)239-174(294)136(89-150(274)275)222-146(267)91-218-178(298)138-32-23-75-251(138)190(310)137(86-108-44-54-114(262)55-45-108)244-167(287)124(68-78-311-16)231-180(300)141-35-26-77-253(141)189(309)126(61-67-149(272)273)233-171(291)129(80-94(4)5)240-181(301)139-33-24-74-250(139)187(307)97(9)196/h36-55,90,92-103,115-141,151-155,254-262H,17-35,56-89,91,196H2,1-16H3,(H2,197,263)(H2,198,264)(H2,199,265)(H2,200,266)(H2,201,276)(H,212,219)(H,218,298)(H,220,279)(H,221,289)(H,222,267)(H,223,277)(H,224,283)(H,225,280)(H,226,290)(H,227,284)(H,228,293)(H,229,292)(H,230,299)(H,231,300)(H,232,303)(H,233,291)(H,234,304)(H,235,281)(H,236,286)(H,237,285)(H,238,282)(H,239,294)(H,240,301)(H,241,306)(H,242,305)(H,243,302)(H,244,287)(H,245,296)(H,246,278)(H,247,288)(H,248,295)(H,249,297)(H,268,269)(H,270,271)(H,272,273)(H,274,275)(H4,202,203,213)(H4,204,205,214)(H4,206,207,215)(H4,208,209,216)(H4,210,211,217)/p+4/t96?,97?,98-,99-,100?,101?,102+,103?,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,141-,151-,152-,153-,154-,155-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University
Curated by PDSP Ki Database
| |
Regul Pept 29-37 (1998)
Article DOI: 10.1016/s0167-0115(98)00050-0
BindingDB Entry DOI: 10.7270/Q2DV1HDX |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 4
(RAT) | BDBM82286

(CAS_59763-91-6 | PP, human | PP,SALMON)Show SMILES [#6]-[#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7+]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7+])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6](-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6](-[#6])-[#7])-[#6](-[#6])-[#6])-[#6](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#16]-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccc(-[#8])cc1)-[#6](-[#7])=O Show InChI InChI=1S/C185H287N53O54S2/c1-21-91(10)143(175(286)228-125(84-137(190)248)164(275)213-114(63-74-294-20)158(269)221-120(77-89(6)7)167(278)232-144(98(17)239)176(287)217-115(34-25-67-203-185(198)199)178(289)235-69-28-37-131(235)170(281)214-110(33-24-66-202-184(196)197)154(265)218-117(146(191)257)78-100-40-48-104(241)49-41-100)231-168(279)122(80-102-44-52-106(243)53-45-102)224-155(266)109(32-23-65-201-183(194)195)210-153(264)108(31-22-64-200-182(192)193)211-161(272)118(75-87(2)3)222-165(276)126(85-140(253)254)219-149(260)94(13)204-147(258)93(12)206-159(270)121(79-101-42-50-105(242)51-43-101)223-156(267)111(56-59-134(187)245)209-148(259)95(14)205-152(263)113(62-73-293-19)212-163(274)124(83-136(189)247)225-157(268)112(57-60-138(249)250)215-171(282)132-38-30-72-238(132)181(292)145(99(18)240)233-151(262)97(16)207-160(271)123(82-135(188)246)226-166(277)127(86-141(255)256)220-150(261)96(15)208-169(280)129-35-27-71-237(129)180(291)128(81-103-46-54-107(244)55-47-103)229-174(285)142(90(8)9)230-173(284)133-39-29-70-236(133)179(290)116(58-61-139(251)252)216-162(273)119(76-88(4)5)227-172(283)130-36-26-68-234(130)177(288)92(11)186/h40-55,87-99,108-133,142-145,239-244H,21-39,56-86,186H2,1-20H3,(H2,187,245)(H2,188,246)(H2,189,247)(H2,190,248)(H2,191,257)(H,204,258)(H,205,263)(H,206,270)(H,207,271)(H,208,280)(H,209,259)(H,210,264)(H,211,272)(H,212,274)(H,213,275)(H,214,281)(H,215,282)(H,216,273)(H,217,287)(H,218,265)(H,219,260)(H,220,261)(H,221,269)(H,222,276)(H,223,267)(H,224,266)(H,225,268)(H,226,277)(H,227,283)(H,228,286)(H,229,285)(H,230,284)(H,231,279)(H,232,278)(H,233,262)(H,249,250)(H,251,252)(H,253,254)(H,255,256)(H4,192,193,200)(H4,194,195,201)(H4,196,197,202)(H4,198,199,203)/p+2/t91?,92?,93-,94-,95-,96?,97-,98+,99?,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,142-,143-,144-,145-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University
Curated by PDSP Ki Database
| |
Regul Pept 29-37 (1998)
Article DOI: 10.1016/s0167-0115(98)00050-0
BindingDB Entry DOI: 10.7270/Q2DV1HDX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50493145
(CHEMBL2419149)Show InChI InChI=1S/C17H10BrN3S2/c18-13-8-6-12(7-9-13)11-20-16-15(10-19)23-17(22)21(16)14-4-2-1-3-5-14/h1-9,11H/b20-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM241385 from human A2A receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50493144

(CHEMBL2419144)Show InChI InChI=1S/C17H10FN3S2/c18-13-8-6-12(7-9-13)11-20-16-15(10-19)23-17(22)21(16)14-4-2-1-3-5-14/h1-9,11H/b20-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM241385 from human A2A receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377655
(CHEMBL260160)Show SMILES COc1cc(Cl)cc(C(=O)Nc2ccc(Cl)cn2)c1NC(=O)c1ccc(cc1F)-n1ccccc1=O Show InChI InChI=1S/C25H17Cl2FN4O4/c1-36-20-11-15(27)10-18(25(35)30-21-8-5-14(26)13-29-21)23(20)31-24(34)17-7-6-16(12-19(17)28)32-9-3-2-4-22(32)33/h2-13H,1H3,(H,31,34)(H,29,30,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2845-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.03.092
BindingDB Entry DOI: 10.7270/Q2611169 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096099
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2F)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21FN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 4
(RAT) | BDBM82297

(CAS_59763-91-6 | PP, BOVINE)Show SMILES [#6]-[#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7+]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7+])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6](-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6](-[#6])-[#7])-[#6](-[#6])-[#6])-[#6](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#16]-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccc(-[#8])cc1)-[#6](-[#7])=O Show InChI InChI=1S/C186H289N53O54S2/c1-21-92(10)144(176(287)229-126(85-138(191)249)165(276)215-116(65-76-295-20)160(271)223-122(79-90(6)7)168(279)233-145(99(17)240)177(288)219-117(34-25-69-204-186(199)200)179(290)236-71-28-37-132(236)171(282)216-111(33-24-68-203-185(197)198)155(266)220-119(147(192)258)80-101-40-48-105(242)49-41-101)232-169(280)124(82-103-44-52-107(244)53-45-103)226-156(267)110(32-23-67-202-184(195)196)211-154(265)109(31-22-66-201-183(193)194)212-163(274)120(77-88(2)3)224-166(277)127(86-141(254)255)221-150(261)95(13)205-148(259)94(12)207-161(272)123(81-102-42-50-106(243)51-43-102)225-159(270)112(56-60-135(188)246)210-149(260)96(14)206-153(264)115(64-75-294-19)214-157(268)113(57-61-136(189)247)213-158(269)114(58-62-139(250)251)217-172(283)133-38-30-74-239(133)182(293)146(100(18)241)234-152(263)98(16)208-162(273)125(84-137(190)248)227-167(278)128(87-142(256)257)222-151(262)97(15)209-170(281)130-35-27-73-238(130)181(292)129(83-104-46-54-108(245)55-47-104)230-175(286)143(91(8)9)231-174(285)134-39-29-72-237(134)180(291)118(59-63-140(252)253)218-164(275)121(78-89(4)5)228-173(284)131-36-26-70-235(131)178(289)93(11)187/h40-55,88-100,109-134,143-146,240-245H,21-39,56-87,187H2,1-20H3,(H2,188,246)(H2,189,247)(H2,190,248)(H2,191,249)(H2,192,258)(H,205,259)(H,206,264)(H,207,272)(H,208,273)(H,209,281)(H,210,260)(H,211,265)(H,212,274)(H,213,269)(H,214,268)(H,215,276)(H,216,282)(H,217,283)(H,218,275)(H,219,288)(H,220,266)(H,221,261)(H,222,262)(H,223,271)(H,224,277)(H,225,270)(H,226,267)(H,227,278)(H,228,284)(H,229,287)(H,230,286)(H,231,285)(H,232,280)(H,233,279)(H,234,263)(H,250,251)(H,252,253)(H,254,255)(H,256,257)(H4,193,194,201)(H4,195,196,202)(H4,197,198,203)(H4,199,200,204)/p+2/t92?,93?,94-,95-,96-,97?,98-,99+,100?,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,143-,144-,145-,146-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University
Curated by PDSP Ki Database
| |
Regul Pept 29-37 (1998)
Article DOI: 10.1016/s0167-0115(98)00050-0
BindingDB Entry DOI: 10.7270/Q2DV1HDX |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 4
(RAT) | BDBM85378
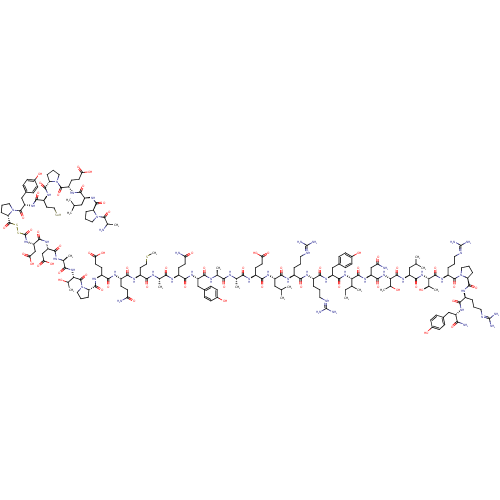
(PP, guinea pig)Show SMILES [#6]-[#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7+]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7+])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#16]-[#16]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6][Se])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6](-[#6])-[#7])-[#6](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccc(-[#8])cc1)-[#6](-[#7])=O Show InChI InChI=1S/C183H281N51O56S3Se/c1-18-89(8)140(170(281)223-123(83-134(187)244)165(276)228-142(96(15)236)172(283)222-120(78-88(6)7)163(274)227-141(95(14)235)171(282)215-115(31-22-68-200-182(195)196)174(285)231-70-24-33-128(231)166(277)211-108(30-21-67-199-181(193)194)151(262)216-117(144(188)255)79-98-37-45-102(238)46-38-98)226-164(275)122(81-100-41-49-104(240)50-42-100)219-152(263)107(29-20-66-198-180(191)192)207-150(261)106(28-19-65-197-179(189)190)208-160(271)118(76-86(2)3)217-156(267)111(55-60-135(245)246)206-146(257)92(11)201-145(256)91(10)203-158(269)121(80-99-39-47-103(239)48-40-99)218-155(266)109(53-58-132(185)242)205-147(258)93(12)202-149(260)113(63-74-291-17)210-153(264)110(54-59-133(186)243)209-154(265)112(56-61-136(247)248)212-168(279)130-35-26-72-233(130)177(288)143(97(16)237)229-148(259)94(13)204-159(270)124(84-138(251)252)220-162(273)125(85-139(253)254)225-183(290)293-292-178(289)131-36-27-73-234(131)176(287)126(82-101-43-51-105(241)52-44-101)224-157(268)114(64-75-294)213-167(278)129-34-25-71-232(129)175(286)116(57-62-137(249)250)214-161(272)119(77-87(4)5)221-169(280)127-32-23-69-230(127)173(284)90(9)184/h37-52,86-97,106-131,140-143,235-241,294H,18-36,53-85,184H2,1-17H3,(H2,185,242)(H2,186,243)(H2,187,244)(H2,188,255)(H,201,256)(H,202,260)(H,203,269)(H,204,270)(H,205,258)(H,206,257)(H,207,261)(H,208,271)(H,209,265)(H,210,264)(H,211,277)(H,212,279)(H,213,278)(H,214,272)(H,215,282)(H,216,262)(H,217,267)(H,218,266)(H,219,263)(H,220,273)(H,221,280)(H,222,283)(H,223,281)(H,224,268)(H,225,290)(H,226,275)(H,227,274)(H,228,276)(H,229,259)(H,245,246)(H,247,248)(H,249,250)(H,251,252)(H,253,254)(H4,189,190,197)(H4,191,192,198)(H4,193,194,199)(H4,195,196,200)/p+2/t89?,90?,91-,92-,93-,94-,95+,96?,97?,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,140-,141-,142-,143-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University
Curated by PDSP Ki Database
| |
Regul Pept 29-37 (1998)
Article DOI: 10.1016/s0167-0115(98)00050-0
BindingDB Entry DOI: 10.7270/Q2DV1HDX |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096101
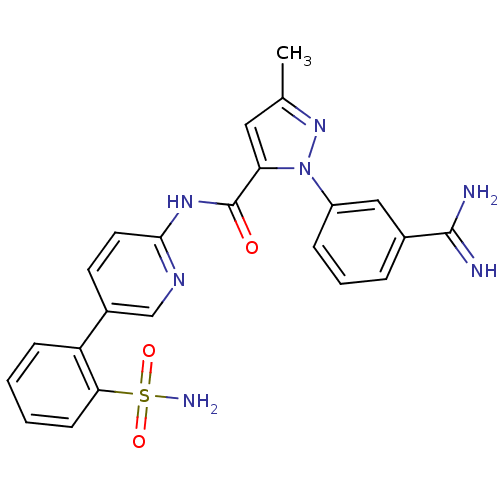
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cn2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C23H21N7O3S/c1-14-11-19(30(29-14)17-6-4-5-15(12-17)22(24)25)23(31)28-21-10-9-16(13-27-21)18-7-2-3-8-20(18)34(26,32)33/h2-13H,1H3,(H3,24,25)(H2,26,32,33)(H,27,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096110
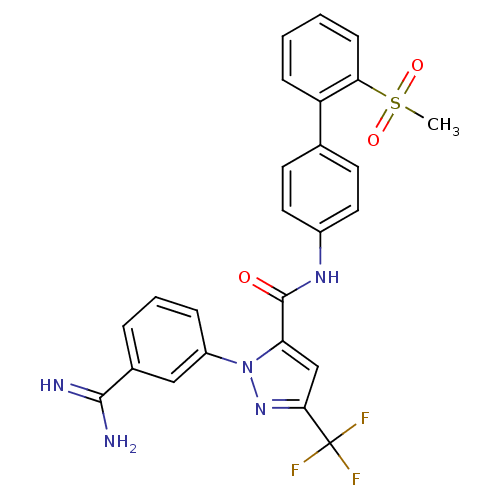
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(NC(=O)c2cc(nn2-c2cccc(c2)C(N)=N)C(F)(F)F)cc1 Show InChI InChI=1S/C25H20F3N5O3S/c1-37(35,36)21-8-3-2-7-19(21)15-9-11-17(12-10-15)31-24(34)20-14-22(25(26,27)28)32-33(20)18-6-4-5-16(13-18)23(29)30/h2-14H,1H3,(H3,29,30)(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096091
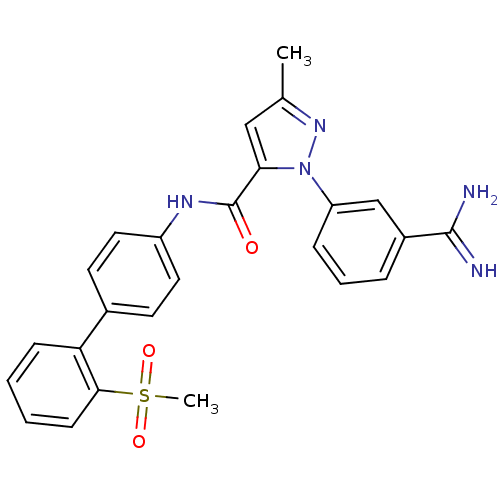
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(C)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C25H23N5O3S/c1-16-14-22(30(29-16)20-7-5-6-18(15-20)24(26)27)25(31)28-19-12-10-17(11-13-19)21-8-3-4-9-23(21)34(2,32)33/h3-15H,1-2H3,(H3,26,27)(H,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50315548
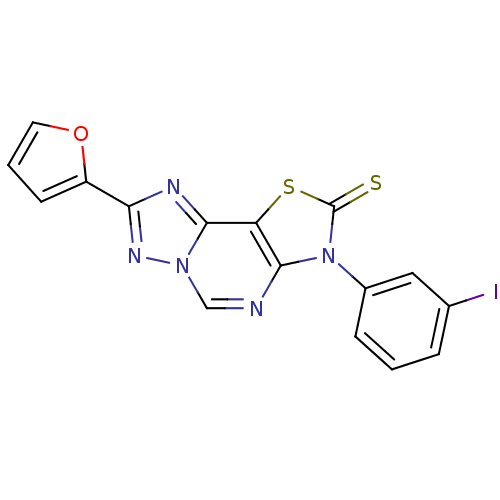
(8-(2-Thioxo-7(3-m-iodophenyl)-2-(2-furyl)thiazolo[...)Show InChI InChI=1S/C16H8IN5OS2/c17-9-3-1-4-10(7-9)22-14-12(25-16(22)24)15-19-13(11-5-2-6-23-11)20-21(15)8-18-14/h1-8H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM241385 from human adenosine A2A receptor expressed in HEK293 cells after 60 mins by rapid filtration assay |
Bioorg Med Chem 18: 2491-500 (2010)
Article DOI: 10.1016/j.bmc.2010.02.048
BindingDB Entry DOI: 10.7270/Q27W6D57 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433537
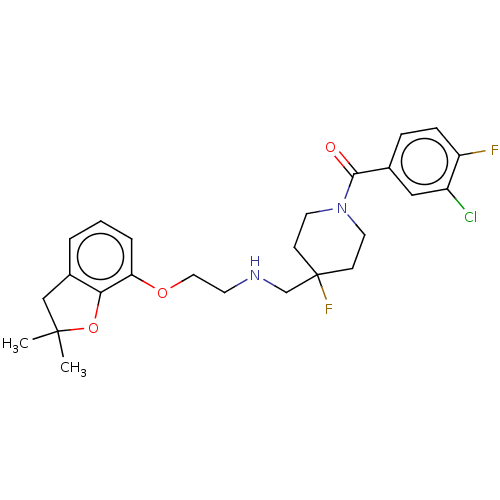
(US10562853, Compound 60)Show SMILES CC1(C)Cc2cccc(OCCNCC3(F)CCN(CC3)C(=O)c3ccc(F)c(Cl)c3)c2O1 Show InChI InChI=1S/C25H29ClF2N2O3/c1-24(2)15-18-4-3-5-21(22(18)33-24)32-13-10-29-16-25(28)8-11-30(12-9-25)23(31)17-6-7-20(27)19(26)14-17/h3-7,14,29H,8-13,15-16H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00851 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433537

(US10562853, Compound 60)Show SMILES CC1(C)Cc2cccc(OCCNCC3(F)CCN(CC3)C(=O)c3ccc(F)c(Cl)c3)c2O1 Show InChI InChI=1S/C25H29ClF2N2O3/c1-24(2)15-18-4-3-5-21(22(18)33-24)32-13-10-29-16-25(28)8-11-30(12-9-25)23(31)17-6-7-20(27)19(26)14-17/h3-7,14,29H,8-13,15-16H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00851 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096108
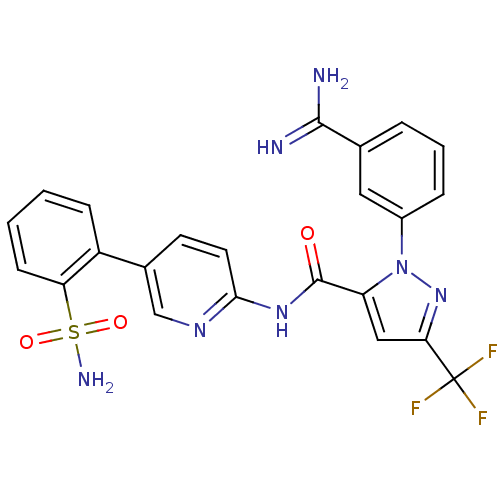
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cn1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C23H18F3N7O3S/c24-23(25,26)19-11-17(33(32-19)15-5-3-4-13(10-15)21(27)28)22(34)31-20-9-8-14(12-30-20)16-6-1-2-7-18(16)37(29,35)36/h1-12H,(H3,27,28)(H2,29,35,36)(H,30,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096085
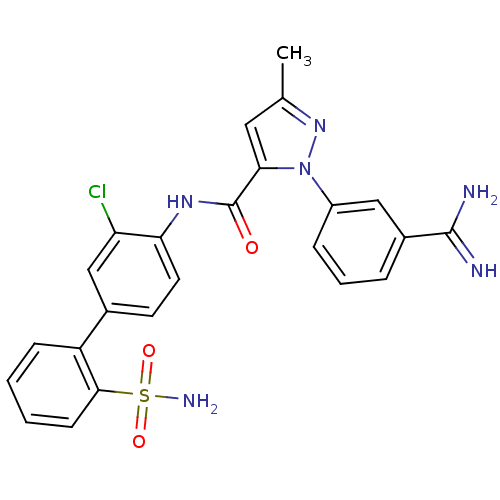
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2Cl)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21ClN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50398494
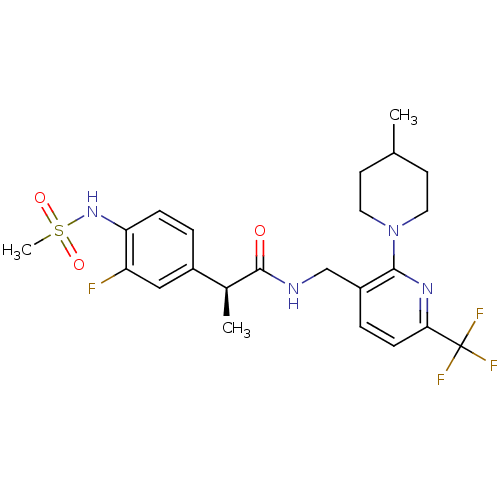
(CHEMBL2177429)Show SMILES C[C@H](C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay |
J Med Chem 55: 8392-408 (2012)
Article DOI: 10.1021/jm300780p
BindingDB Entry DOI: 10.7270/Q2TX3GH1 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096098
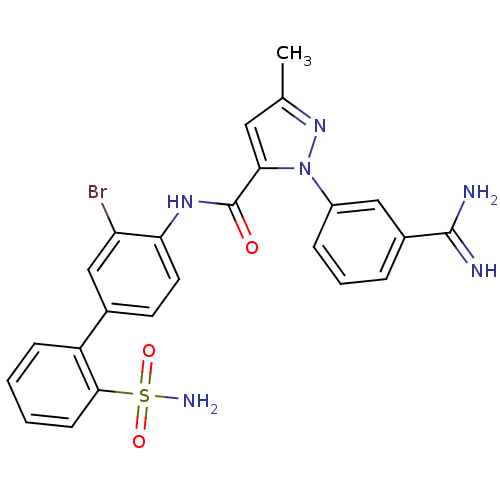
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2Br)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21BrN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50493151

(CHEMBL2419139)Show InChI InChI=1S/C13H10BrN3S2/c1-2-17-12(11(7-15)19-13(17)18)16-8-9-3-5-10(14)6-4-9/h3-6,8H,2H2,1H3/b16-8+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM241385 from human A2A receptor expressed in HEK293 cell membranes after 60 mins by scintillation counting |
Bioorg Med Chem 21: 6077-83 (2013)
Article DOI: 10.1016/j.bmc.2013.07.005
BindingDB Entry DOI: 10.7270/Q2CC13NZ |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Homo sapiens (Human)) | BDBM50398494

(CHEMBL2177429)Show SMILES C[C@H](C(=O)NCc1ccc(nc1N1CCC(C)CC1)C(F)(F)F)c1ccc(NS(C)(=O)=O)c(F)c1 |r| Show InChI InChI=1S/C23H28F4N4O3S/c1-14-8-10-31(11-9-14)21-17(5-7-20(29-21)23(25,26)27)13-28-22(32)15(2)16-4-6-19(18(24)12-16)30-35(3,33)34/h4-7,12,14-15,30H,8-11,13H2,1-3H3,(H,28,32)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay |
Bioorg Med Chem 21: 6657-64 (2013)
Article DOI: 10.1016/j.bmc.2013.08.015
BindingDB Entry DOI: 10.7270/Q26Q1ZPN |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433539
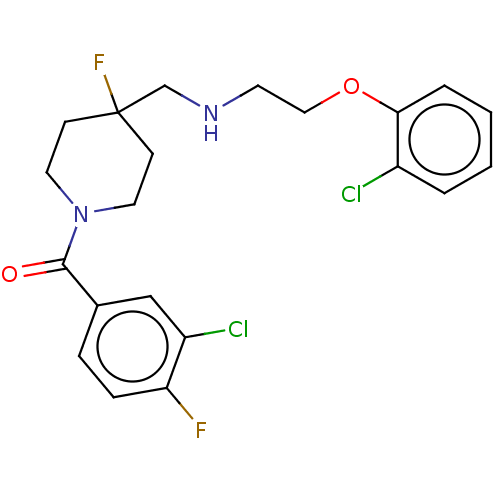
(US10562853, Compound 62)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2ccccc2Cl)CC1 Show InChI InChI=1S/C21H22Cl2F2N2O2/c22-16-3-1-2-4-19(16)29-12-9-26-14-21(25)7-10-27(11-8-21)20(28)15-5-6-18(24)17(23)13-15/h1-6,13,26H,7-12,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433539

(US10562853, Compound 62)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2ccccc2Cl)CC1 Show InChI InChI=1S/C21H22Cl2F2N2O2/c22-16-3-1-2-4-19(16)29-12-9-26-14-21(25)7-10-27(11-8-21)20(28)15-5-6-18(24)17(23)13-15/h1-6,13,26H,7-12,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377635
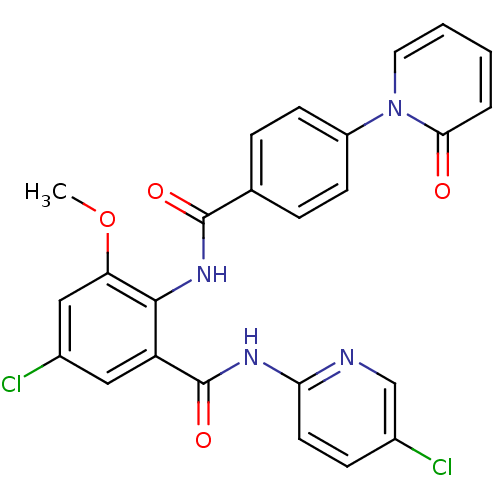
(CHEMBL402980)Show SMILES COc1cc(Cl)cc(C(=O)Nc2ccc(Cl)cn2)c1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C25H18Cl2N4O4/c1-35-20-13-17(27)12-19(25(34)29-21-10-7-16(26)14-28-21)23(20)30-24(33)15-5-8-18(9-6-15)31-11-3-2-4-22(31)32/h2-14H,1H3,(H,30,33)(H,28,29,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2845-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.03.092
BindingDB Entry DOI: 10.7270/Q2611169 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374877
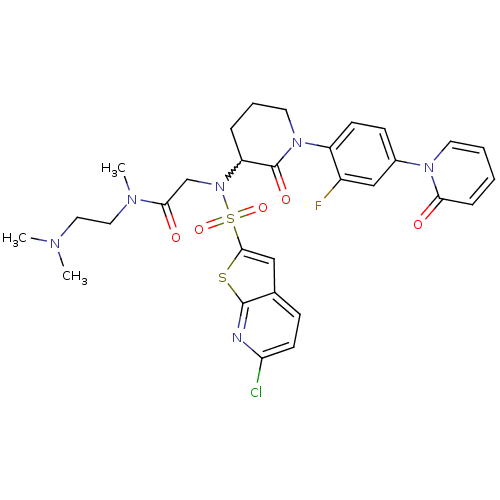
(CHEMBL270221)Show SMILES CN(C)CCN(C)C(=O)CN(C1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:11.10| Show InChI InChI=1S/C30H32ClFN6O5S2/c1-34(2)15-16-35(3)27(40)19-38(45(42,43)28-17-20-9-12-25(31)33-29(20)44-28)24-7-6-14-37(30(24)41)23-11-10-21(18-22(23)32)36-13-5-4-8-26(36)39/h4-5,8-13,17-18,24H,6-7,14-16,19H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Tested in vitro for inhibition of human Coagulation factor X |
J Med Chem 46: 5298-315 (2003)
Article DOI: 10.1021/jm030212h
BindingDB Entry DOI: 10.7270/Q2ZW1MP2 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | -61.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | -61.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were determined ... |
J Med Chem 46: 4405-18 (2003)
Article DOI: 10.1021/jm020578e
BindingDB Entry DOI: 10.7270/Q2TT4P78 |
More data for this
Ligand-Target Pair | |
Pancreatic polypeptide receptor 1
(GUINEA PIG) | BDBM82297

(CAS_59763-91-6 | PP, BOVINE)Show SMILES [#6]-[#6]-[#6](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7+]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7+])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6](-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6](-[#6])-[#7])-[#6](-[#6])-[#6])-[#6](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#16]-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccc(-[#8])cc1)-[#6](-[#7])=O Show InChI InChI=1S/C186H289N53O54S2/c1-21-92(10)144(176(287)229-126(85-138(191)249)165(276)215-116(65-76-295-20)160(271)223-122(79-90(6)7)168(279)233-145(99(17)240)177(288)219-117(34-25-69-204-186(199)200)179(290)236-71-28-37-132(236)171(282)216-111(33-24-68-203-185(197)198)155(266)220-119(147(192)258)80-101-40-48-105(242)49-41-101)232-169(280)124(82-103-44-52-107(244)53-45-103)226-156(267)110(32-23-67-202-184(195)196)211-154(265)109(31-22-66-201-183(193)194)212-163(274)120(77-88(2)3)224-166(277)127(86-141(254)255)221-150(261)95(13)205-148(259)94(12)207-161(272)123(81-102-42-50-106(243)51-43-102)225-159(270)112(56-60-135(188)246)210-149(260)96(14)206-153(264)115(64-75-294-19)214-157(268)113(57-61-136(189)247)213-158(269)114(58-62-139(250)251)217-172(283)133-38-30-74-239(133)182(293)146(100(18)241)234-152(263)98(16)208-162(273)125(84-137(190)248)227-167(278)128(87-142(256)257)222-151(262)97(15)209-170(281)130-35-27-73-238(130)181(292)129(83-104-46-54-108(245)55-47-104)230-175(286)143(91(8)9)231-174(285)134-39-29-72-237(134)180(291)118(59-63-140(252)253)218-164(275)121(78-89(4)5)228-173(284)131-36-26-70-235(131)178(289)93(11)187/h40-55,88-100,109-134,143-146,240-245H,21-39,56-87,187H2,1-20H3,(H2,188,246)(H2,189,247)(H2,190,248)(H2,191,249)(H2,192,258)(H,205,259)(H,206,264)(H,207,272)(H,208,273)(H,209,281)(H,210,260)(H,211,265)(H,212,274)(H,213,269)(H,214,268)(H,215,276)(H,216,282)(H,217,283)(H,218,275)(H,219,288)(H,220,266)(H,221,261)(H,222,262)(H,223,271)(H,224,277)(H,225,270)(H,226,267)(H,227,278)(H,228,284)(H,229,287)(H,230,286)(H,231,285)(H,232,280)(H,233,279)(H,234,263)(H,250,251)(H,252,253)(H,254,255)(H,256,257)(H4,193,194,201)(H4,195,196,202)(H4,197,198,203)(H4,199,200,204)/p+2/t92?,93?,94-,95-,96-,97?,98-,99+,100?,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,143-,144-,145-,146-/m0/s1 | Reactome pathway
KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University
Curated by PDSP Ki Database
| |
Regul Pept 29-37 (1998)
Article DOI: 10.1016/s0167-0115(98)00050-0
BindingDB Entry DOI: 10.7270/Q2DV1HDX |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50555494
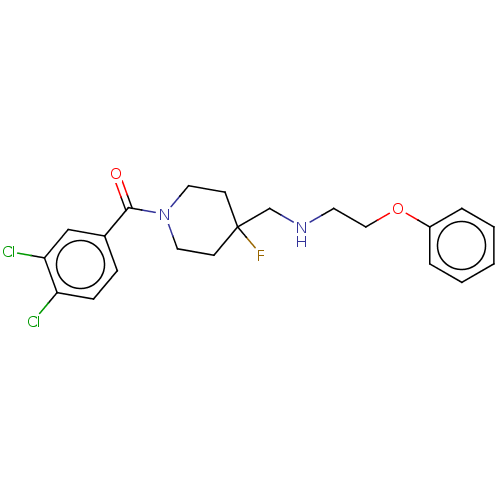
(CHEMBL4742112)Show SMILES FC1(CNCCOc2ccccc2)CCN(CC1)C(=O)c1ccc(Cl)c(Cl)c1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0148 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from recombinant human 5HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by microbeta2 sc... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.9b00062
BindingDB Entry DOI: 10.7270/Q23F4T9H |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096111
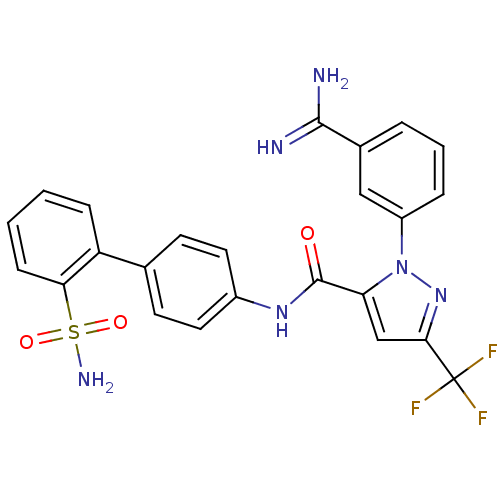
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3N6O3S/c25-24(26,27)21-13-19(33(32-21)17-5-3-4-15(12-17)22(28)29)23(34)31-16-10-8-14(9-11-16)18-6-1-2-7-20(18)37(30,35)36/h1-13H,(H3,28,29)(H,31,34)(H2,30,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433538
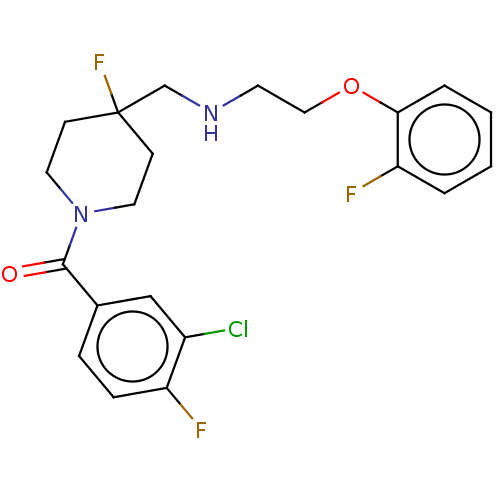
(US10562853, Compound 61)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2ccccc2F)CC1 Show InChI InChI=1S/C21H22ClF3N2O2/c22-16-13-15(5-6-17(16)23)20(28)27-10-7-21(25,8-11-27)14-26-9-12-29-19-4-2-1-3-18(19)24/h1-6,13,26H,7-12,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433538

(US10562853, Compound 61)Show SMILES Fc1ccc(cc1Cl)C(=O)N1CCC(F)(CNCCOc2ccccc2F)CC1 Show InChI InChI=1S/C21H22ClF3N2O2/c22-16-13-15(5-6-17(16)23)20(28)27-10-7-21(25,8-11-27)14-26-9-12-29-19-4-2-1-3-18(19)24/h1-6,13,26H,7-12,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50315541

(8-(2-Thioxo-7(3-allyl)-2-(2-furyl)thiazole[4,3-e]1...)Show InChI InChI=1S/C13H9N5OS2/c1-2-5-17-11-9(21-13(17)20)12-15-10(8-4-3-6-19-8)16-18(12)7-14-11/h2-4,6-7H,1,5H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Delhi
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM241385 from human adenosine A2A receptor expressed in HEK293 cells after 60 mins by rapid filtration assay |
Bioorg Med Chem 18: 2491-500 (2010)
Article DOI: 10.1016/j.bmc.2010.02.048
BindingDB Entry DOI: 10.7270/Q27W6D57 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374879
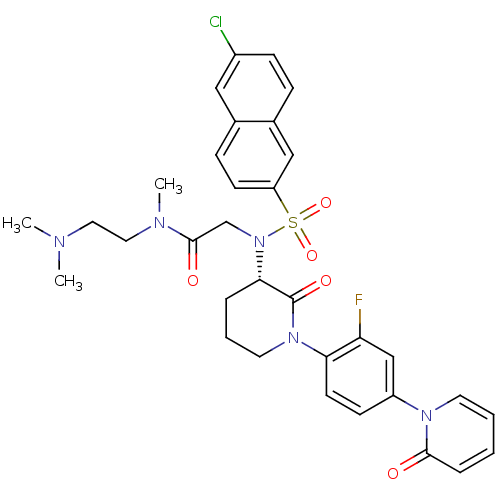
(CHEMBL401958)Show SMILES CN(C)CCN(C)C(=O)CN([C@H]1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 Show InChI InChI=1S/C33H35ClFN5O5S/c1-36(2)17-18-37(3)32(42)22-40(46(44,45)27-13-10-23-19-25(34)11-9-24(23)20-27)30-7-6-16-39(33(30)43)29-14-12-26(21-28(29)35)38-15-5-4-8-31(38)41/h4-5,8-15,19-21,30H,6-7,16-18,22H2,1-3H3/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433479
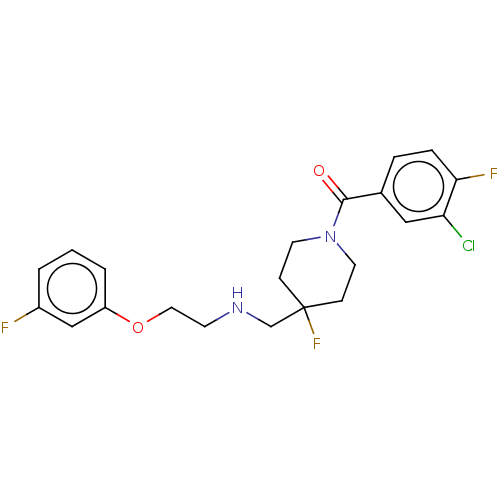
(US10562853, Compound 5)Show SMILES Fc1cccc(OCCNCC2(F)CCN(CC2)C(=O)c2ccc(F)c(Cl)c2)c1 Show InChI InChI=1S/C21H22ClF3N2O2/c22-18-12-15(4-5-19(18)24)20(28)27-9-6-21(25,7-10-27)14-26-8-11-29-17-3-1-2-16(23)13-17/h1-5,12-13,26H,6-11,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0166 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM433479

(US10562853, Compound 5)Show SMILES Fc1cccc(OCCNCC2(F)CCN(CC2)C(=O)c2ccc(F)c(Cl)c2)c1 Show InChI InChI=1S/C21H22ClF3N2O2/c22-18-12-15(4-5-19(18)24)20(28)27-9-6-21(25,7-10-27)14-26-8-11-29-17-3-1-2-16(23)13-17/h1-5,12-13,26H,6-11,14H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0166 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5-HT1A receptor stably expressed in CHO-K1 cell membranes measured after 60 mins by Microbeta2 scintillation... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00814
BindingDB Entry DOI: 10.7270/Q24T6NZT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data