 Found 2803 hits with Last Name = 'luettgen' and Initial = 'jm'
Found 2803 hits with Last Name = 'luettgen' and Initial = 'jm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096105
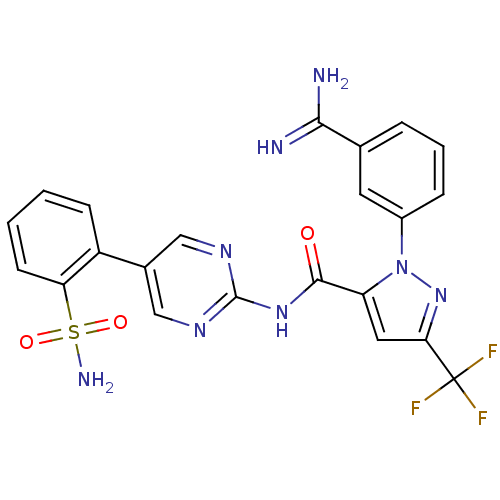
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ncc(cn1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C22H17F3N8O3S/c23-22(24,25)18-9-16(33(32-18)14-5-3-4-12(8-14)19(26)27)20(34)31-21-29-10-13(11-30-21)15-6-1-2-7-17(15)37(28,35)36/h1-11H,(H3,26,27)(H2,28,35,36)(H,29,30,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377655
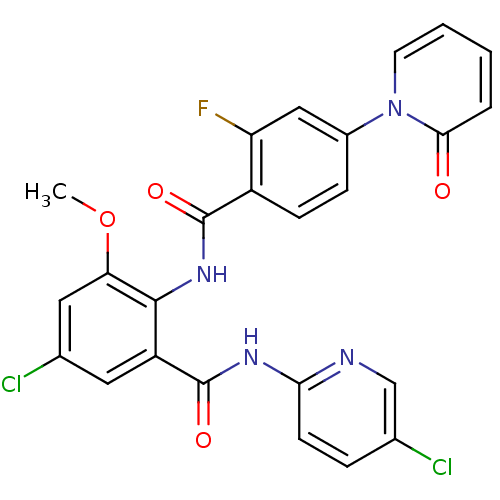
(CHEMBL260160)Show SMILES COc1cc(Cl)cc(C(=O)Nc2ccc(Cl)cn2)c1NC(=O)c1ccc(cc1F)-n1ccccc1=O Show InChI InChI=1S/C25H17Cl2FN4O4/c1-36-20-11-15(27)10-18(25(35)30-21-8-5-14(26)13-29-21)23(20)31-24(34)17-7-6-16(12-19(17)28)32-9-3-2-4-22(32)33/h2-13H,1H3,(H,31,34)(H,29,30,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2845-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.03.092
BindingDB Entry DOI: 10.7270/Q2611169 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096099
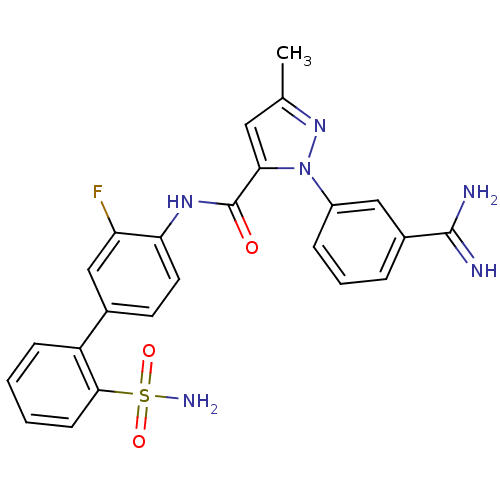
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2F)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21FN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096101
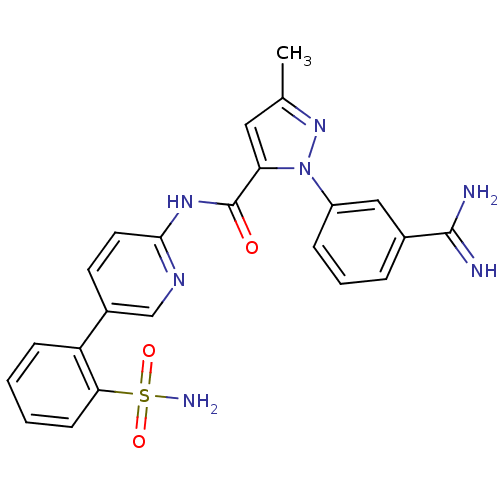
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cn2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C23H21N7O3S/c1-14-11-19(30(29-14)17-6-4-5-15(12-17)22(24)25)23(31)28-21-10-9-16(13-27-21)18-7-2-3-8-20(18)34(26,32)33/h2-13H,1H3,(H3,24,25)(H2,26,32,33)(H,27,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096091
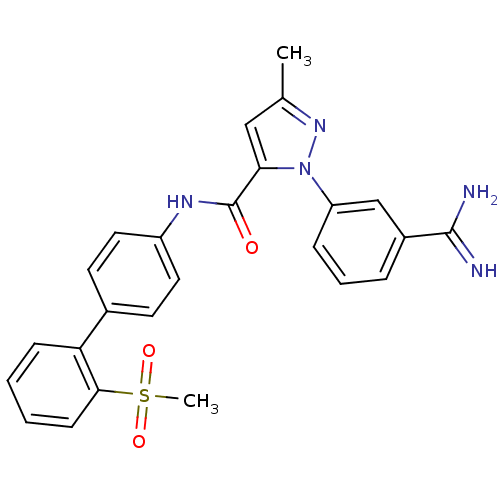
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(C)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C25H23N5O3S/c1-16-14-22(30(29-16)20-7-5-6-18(15-20)24(26)27)25(31)28-19-12-10-17(11-13-19)21-8-3-4-9-23(21)34(2,32)33/h3-15H,1-2H3,(H3,26,27)(H,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096110
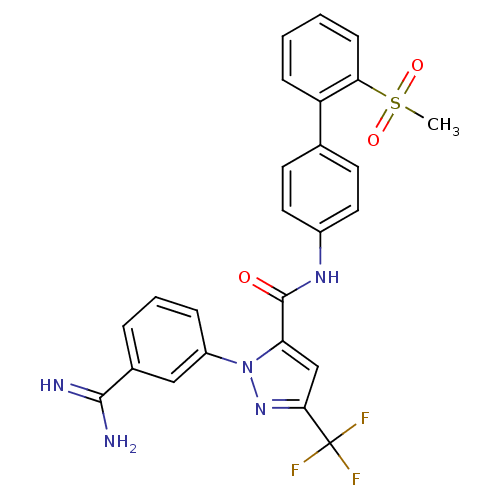
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(NC(=O)c2cc(nn2-c2cccc(c2)C(N)=N)C(F)(F)F)cc1 Show InChI InChI=1S/C25H20F3N5O3S/c1-37(35,36)21-8-3-2-7-19(21)15-9-11-17(12-10-15)31-24(34)20-14-22(25(26,27)28)32-33(20)18-6-4-5-16(13-18)23(29)30/h2-14H,1H3,(H3,29,30)(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096085
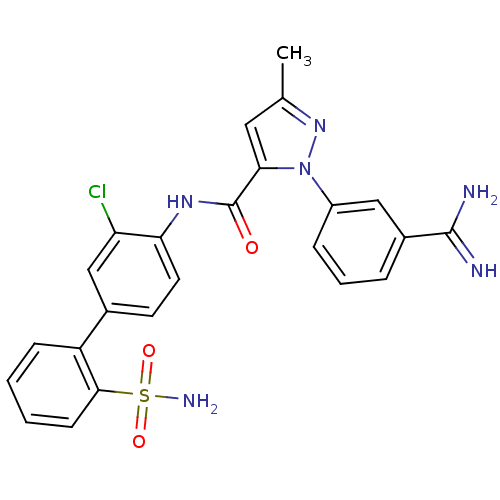
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2Cl)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21ClN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096108
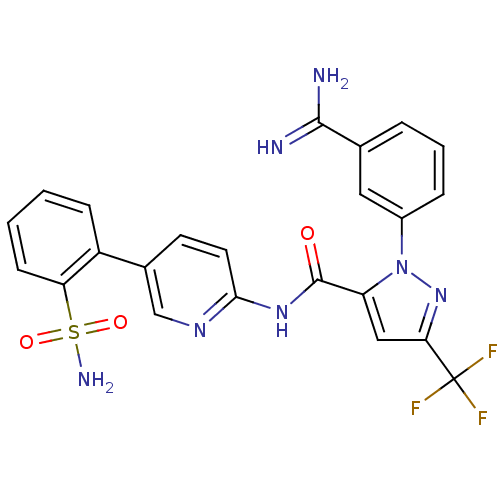
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cn1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C23H18F3N7O3S/c24-23(25,26)19-11-17(33(32-19)15-5-3-4-13(10-15)21(27)28)22(34)31-20-9-8-14(12-30-20)16-6-1-2-7-18(16)37(29,35)36/h1-12H,(H3,27,28)(H2,29,35,36)(H,30,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096098
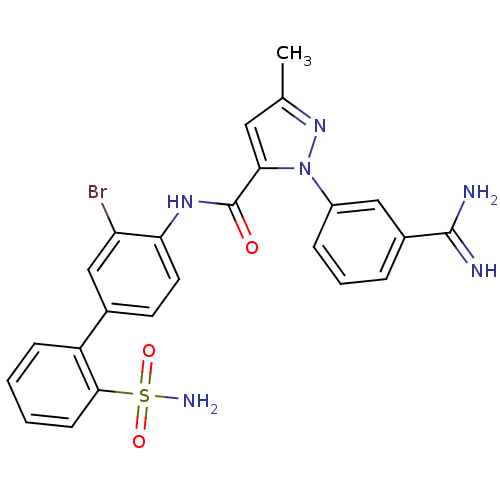
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2Br)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H21BrN6O3S/c1-14-11-21(31(30-14)17-6-4-5-16(12-17)23(26)27)24(32)29-20-10-9-15(13-19(20)25)18-7-2-3-8-22(18)35(28,33)34/h2-13H,1H3,(H3,26,27)(H,29,32)(H2,28,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374877
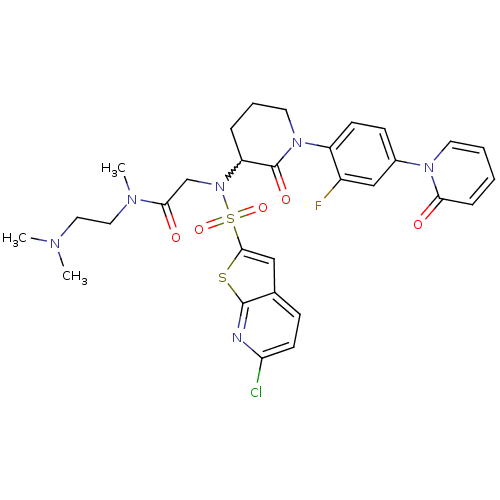
(CHEMBL270221)Show SMILES CN(C)CCN(C)C(=O)CN(C1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:11.10| Show InChI InChI=1S/C30H32ClFN6O5S2/c1-34(2)15-16-35(3)27(40)19-38(45(42,43)28-17-20-9-12-25(31)33-29(20)44-28)24-7-6-14-37(30(24)41)23-11-10-21(18-22(23)32)36-13-5-4-8-26(36)39/h4-5,8-13,17-18,24H,6-7,14-16,19H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377635
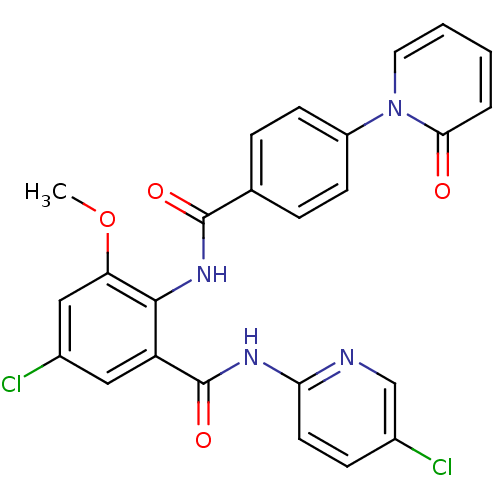
(CHEMBL402980)Show SMILES COc1cc(Cl)cc(C(=O)Nc2ccc(Cl)cn2)c1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C25H18Cl2N4O4/c1-35-20-13-17(27)12-19(25(34)29-21-10-7-16(26)14-28-21)23(20)30-24(33)15-5-8-18(9-6-15)31-11-3-2-4-22(31)32/h2-14H,1H3,(H,30,33)(H,28,29,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2845-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.03.092
BindingDB Entry DOI: 10.7270/Q2611169 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | -61.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were determined ... |
J Med Chem 46: 4405-18 (2003)
Article DOI: 10.1021/jm020578e
BindingDB Entry DOI: 10.7270/Q2TT4P78 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | -61.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Tested in vitro for inhibition of human Coagulation factor X |
J Med Chem 46: 5298-315 (2003)
Article DOI: 10.1021/jm030212h
BindingDB Entry DOI: 10.7270/Q2ZW1MP2 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096111
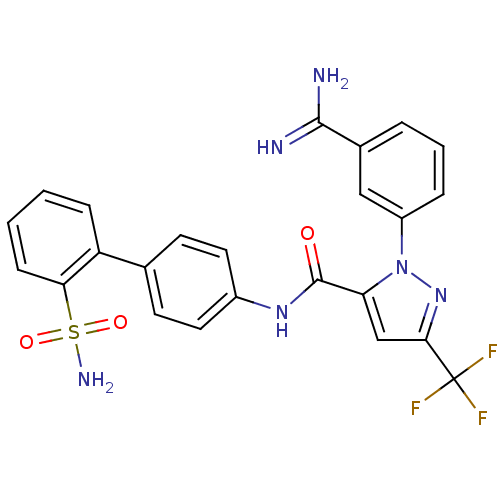
(2-(3-Carbamimidoyl-phenyl)-5-trifluoromethyl-2H-py...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1)-c1ccccc1S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3N6O3S/c25-24(26,27)21-13-19(33(32-21)17-5-3-4-15(12-17)22(28)29)23(34)31-16-10-8-14(9-11-16)18-6-1-2-7-20(18)37(30,35)36/h1-13H,(H3,28,29)(H,31,34)(H2,30,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374879
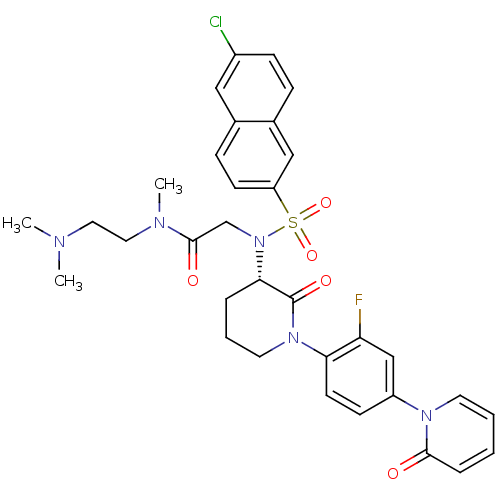
(CHEMBL401958)Show SMILES CN(C)CCN(C)C(=O)CN([C@H]1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 Show InChI InChI=1S/C33H35ClFN5O5S/c1-36(2)17-18-37(3)32(42)22-40(46(44,45)27-13-10-23-19-25(34)11-9-24(23)20-27)30-7-6-16-39(33(30)43)29-14-12-26(21-28(29)35)38-15-5-4-8-31(38)41/h4-5,8-15,19-21,30H,6-7,16-18,22H2,1-3H3/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50260646
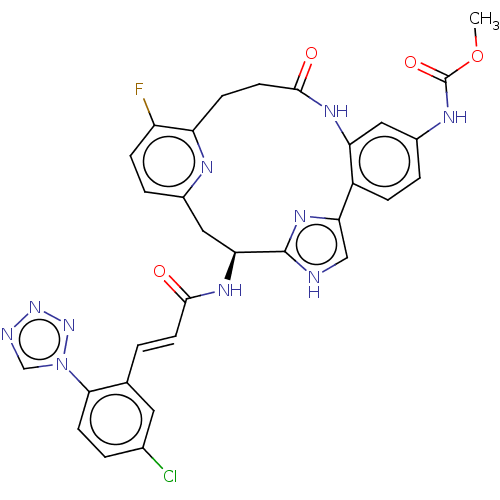
(CHEMBL4096251)Show SMILES COC(=O)Nc1ccc2-c3c[nH]c(n3)[C@H](Cc3ccc(F)c(CCC(=O)Nc2c1)n3)NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1 |r| Show InChI InChI=1S/C31H26ClFN10O4/c1-47-31(46)37-20-4-6-21-24(13-20)38-29(45)11-8-23-22(33)7-5-19(36-23)14-25(30-34-15-26(21)40-30)39-28(44)10-2-17-12-18(32)3-9-27(17)43-16-35-41-42-43/h2-7,9-10,12-13,15-16,25H,8,11,14H2,1H3,(H,34,40)(H,37,46)(H,38,45)(H,39,44)/b10-2+/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company, Research and Development, 350 Carter Road, Hopewell, NJ 08540 United States.
Curated by ChEMBL
| Assay Description
Inhibition of human F11a using peptide substrate by spectrophotometry |
Bioorg Med Chem Lett 27: 4056-4060 (2017)
Article DOI: 10.1016/j.bmcl.2017.07.048
BindingDB Entry DOI: 10.7270/Q2TB19B3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12852
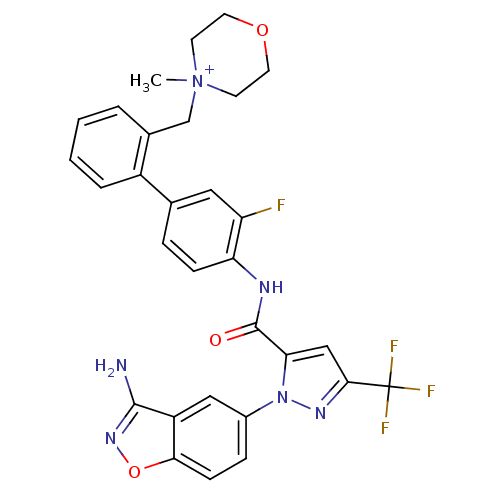
(4-{[2-(4-{[1-(3-amino-1,2-benzoxazol-5-yl)-3-(trif...)Show SMILES C[N+]1(Cc2ccccc2-c2ccc(NC(=O)c3cc(nn3-c3ccc4onc(N)c4c3)C(F)(F)F)c(F)c2)CCOCC1 Show InChI InChI=1S/C30H26F4N6O3/c1-40(10-12-42-13-11-40)17-19-4-2-3-5-21(19)18-6-8-24(23(31)14-18)36-29(41)25-16-27(30(32,33)34)37-39(25)20-7-9-26-22(15-20)28(35)38-43-26/h2-9,14-16H,10-13,17H2,1H3,(H2-,35,36,38,41)/p+1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | -60.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 1795-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.010
BindingDB Entry DOI: 10.7270/Q2FB515N |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374878
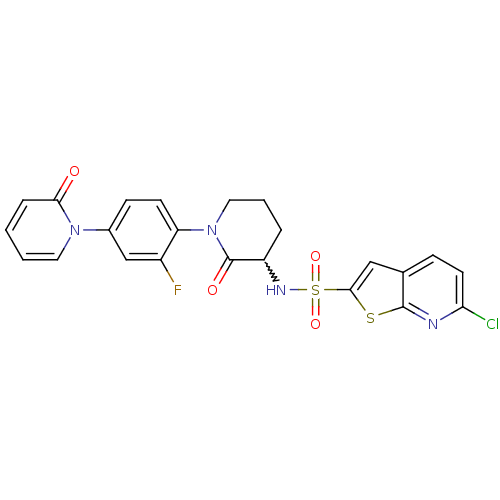
(CHEMBL270862)Show SMILES Fc1cc(ccc1N1CCCC(NS(=O)(=O)c2cc3ccc(Cl)nc3s2)C1=O)-n1ccccc1=O |w:11.12| Show InChI InChI=1S/C23H18ClFN4O4S2/c24-19-9-6-14-12-21(34-22(14)26-19)35(32,33)27-17-4-3-11-29(23(17)31)18-8-7-15(13-16(18)25)28-10-2-1-5-20(28)30/h1-2,5-10,12-13,17,27H,3-4,11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374879

(CHEMBL401958)Show SMILES CN(C)CCN(C)C(=O)CN([C@H]1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 Show InChI InChI=1S/C33H35ClFN5O5S/c1-36(2)17-18-37(3)32(42)22-40(46(44,45)27-13-10-23-19-25(34)11-9-24(23)20-27)30-7-6-16-39(33(30)43)29-14-12-26(21-28(29)35)38-15-5-4-8-31(38)41/h4-5,8-15,19-21,30H,6-7,16-18,22H2,1-3H3/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50266214
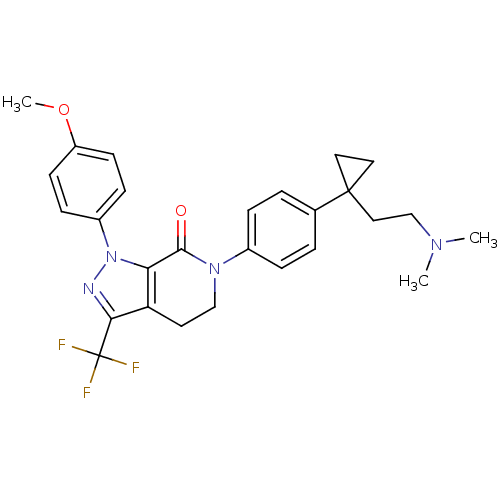
(6-(4-(1-(2-(dimethylamino)ethyl)cyclopropyl)phenyl...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CCN(C)C)CC1)C(F)(F)F Show InChI InChI=1S/C27H29F3N4O2/c1-32(2)17-15-26(13-14-26)18-4-6-19(7-5-18)33-16-12-22-23(25(33)35)34(31-24(22)27(28,29)30)20-8-10-21(36-3)11-9-20/h4-11H,12-17H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50266215

(1-(4-methoxyphenyl)-6-(4-(1-(2-(pyrrolidin-1-yl)et...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CCN2CCCC2)CC1)C(F)(F)F Show InChI InChI=1S/C29H31F3N4O2/c1-38-23-10-8-22(9-11-23)36-25-24(26(33-36)29(30,31)32)12-18-35(27(25)37)21-6-4-20(5-7-21)28(13-14-28)15-19-34-16-2-3-17-34/h4-11H,2-3,12-19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50266249

(6-(4-(1-(4,5-dihydro-1H-imidazol-2-yl)cyclopropyl)...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CC1)C1=NCCN1)C(F)(F)F |t:32| Show InChI InChI=1S/C26H24F3N5O2/c1-36-19-8-6-18(7-9-19)34-21-20(22(32-34)26(27,28)29)10-15-33(23(21)35)17-4-2-16(3-5-17)25(11-12-25)24-30-13-14-31-24/h2-9H,10-15H2,1H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243592
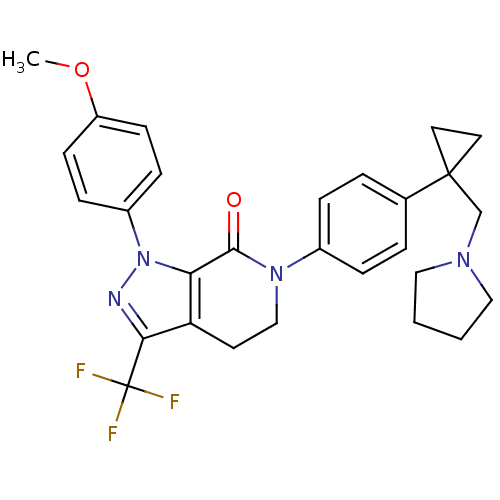
(1-(4-methoxyphenyl)-6-(4-(1-(pyrrolidin-1-ylmethyl...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CN2CCCC2)CC1)C(F)(F)F Show InChI InChI=1S/C28H29F3N4O2/c1-37-22-10-8-21(9-11-22)35-24-23(25(32-35)28(29,30)31)12-17-34(26(24)36)20-6-4-19(5-7-20)27(13-14-27)18-33-15-2-3-16-33/h4-11H,2-3,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096096
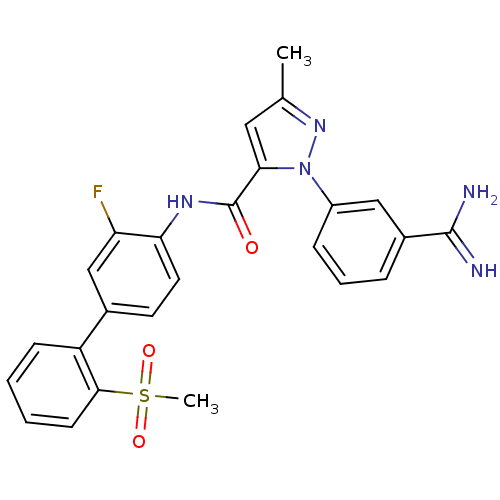
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2F)-c2ccccc2S(C)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C25H22FN5O3S/c1-15-12-22(31(30-15)18-7-5-6-17(13-18)24(27)28)25(32)29-21-11-10-16(14-20(21)26)19-8-3-4-9-23(19)35(2,33)34/h3-14H,1-2H3,(H3,27,28)(H,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243592

(1-(4-methoxyphenyl)-6-(4-(1-(pyrrolidin-1-ylmethyl...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CN2CCCC2)CC1)C(F)(F)F Show InChI InChI=1S/C28H29F3N4O2/c1-37-22-10-8-21(9-11-22)35-24-23(25(32-35)28(29,30)31)12-17-34(26(24)36)20-6-4-19(5-7-20)27(13-14-27)18-33-15-2-3-16-33/h4-11H,2-3,12-18H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 4118-23 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.095
BindingDB Entry DOI: 10.7270/Q20Z7322 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12754
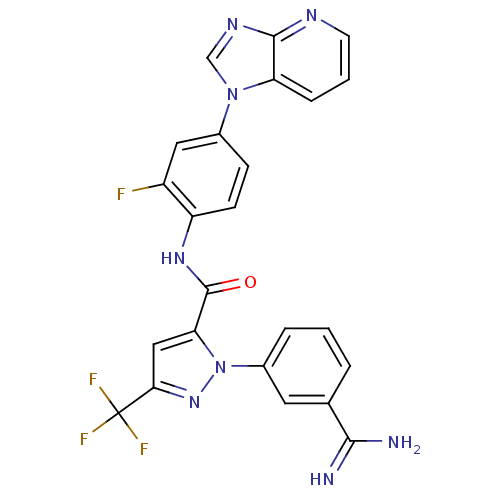
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{1H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2ncccc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-10-14(35-12-32-22-18(35)5-2-8-31-22)6-7-17(16)33-23(37)19-11-20(24(26,27)28)34-36(19)15-4-1-3-13(9-15)21(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0240 | -60.0 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243668
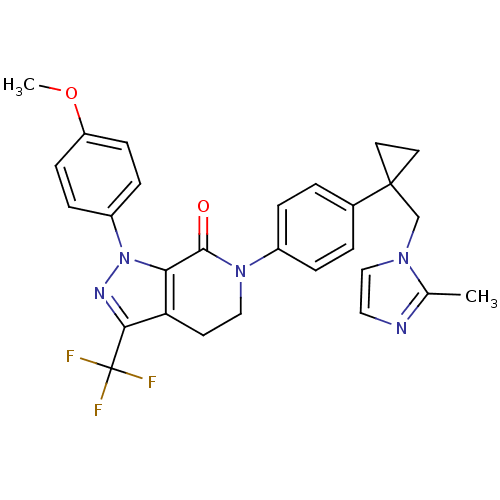
(1-(4-methoxyphenyl)-6-(4-(1-((2-methyl-1H-imidazol...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(Cn2ccnc2C)CC1)C(F)(F)F Show InChI InChI=1S/C28H26F3N5O2/c1-18-32-14-16-34(18)17-27(12-13-27)19-3-5-20(6-4-19)35-15-11-23-24(26(35)37)36(33-25(23)28(29,30)31)21-7-9-22(38-2)10-8-21/h3-10,14,16H,11-13,15,17H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243668

(1-(4-methoxyphenyl)-6-(4-(1-((2-methyl-1H-imidazol...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(Cn2ccnc2C)CC1)C(F)(F)F Show InChI InChI=1S/C28H26F3N5O2/c1-18-32-14-16-34(18)17-27(12-13-27)19-3-5-20(6-4-19)35-15-11-23-24(26(35)37)36(33-25(23)28(29,30)31)21-7-9-22(38-2)10-8-21/h3-10,14,16H,11-13,15,17H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 4118-23 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.095
BindingDB Entry DOI: 10.7270/Q20Z7322 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50266047
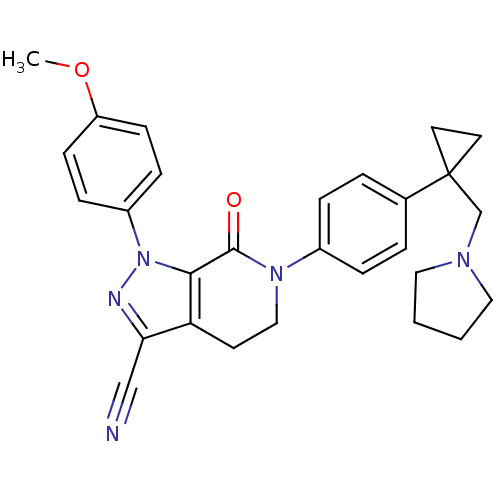
(1-(4-methoxyphenyl)-7-oxo-6-(4-(1-(pyrrolidin-1-yl...)Show SMILES COc1ccc(cc1)-n1nc(C#N)c2CCN(C(=O)c12)c1ccc(cc1)C1(CN2CCCC2)CC1 Show InChI InChI=1S/C28H29N5O2/c1-35-23-10-8-22(9-11-23)33-26-24(25(18-29)30-33)12-17-32(27(26)34)21-6-4-20(5-7-21)28(13-14-28)19-31-15-2-3-16-31/h4-11H,2-3,12-17,19H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50377637
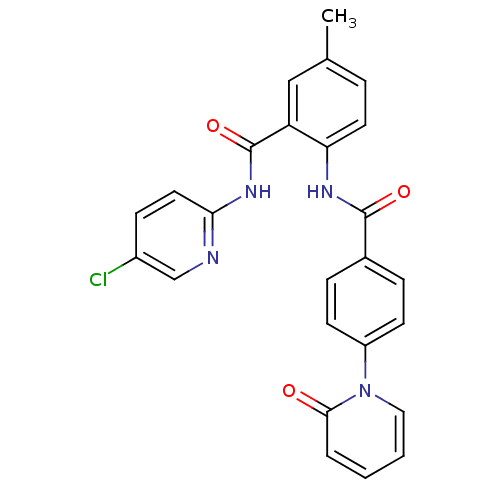
(CHEMBL257398)Show SMILES Cc1ccc(NC(=O)c2ccc(cc2)-n2ccccc2=O)c(c1)C(=O)Nc1ccc(Cl)cn1 Show InChI InChI=1S/C25H19ClN4O3/c1-16-5-11-21(20(14-16)25(33)29-22-12-8-18(26)15-27-22)28-24(32)17-6-9-19(10-7-17)30-13-3-2-4-23(30)31/h2-15H,1H3,(H,28,32)(H,27,29,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2845-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.03.092
BindingDB Entry DOI: 10.7270/Q2611169 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12757
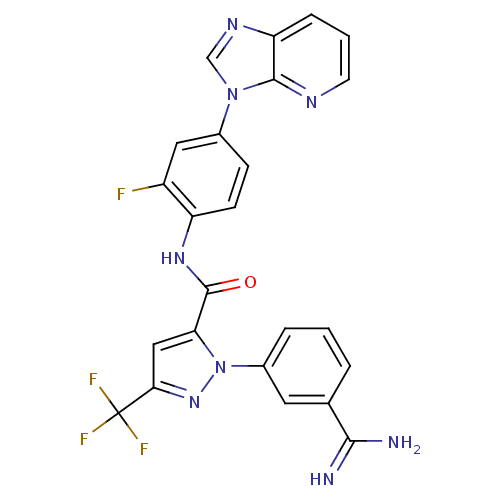
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{3H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cccnc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-10-14(35-12-32-18-5-2-8-31-22(18)35)6-7-17(16)33-23(37)19-11-20(24(26,27)28)34-36(19)15-4-1-3-13(9-15)21(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0280 | -59.6 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50230326
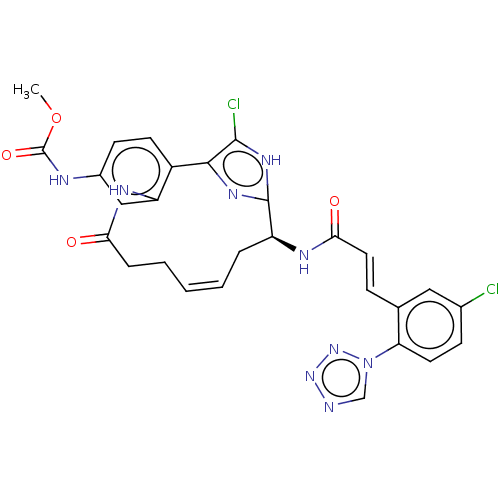
(CHEMBL4060950)Show SMILES COC(=O)Nc1ccc2-c3nc([nH]c3Cl)[C@H](C\C=C\CCC(=O)Nc2c1)NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1 |r,t:18| Show InChI InChI=1S/C28H25Cl2N9O4/c1-43-28(42)32-18-9-10-19-21(14-18)34-23(40)6-4-2-3-5-20(27-35-25(19)26(30)36-27)33-24(41)12-7-16-13-17(29)8-11-22(16)39-15-31-37-38-39/h2-3,7-15,20H,4-6H2,1H3,(H,32,42)(H,33,41)(H,34,40)(H,35,36)/b3-2+,12-7+/t20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a using pyroGlu-Pro-Arg-pNA as substrate after 10 to 120 mins at 37 degC by spectrophotometric method |
J Med Chem 60: 1060-1075 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01460
BindingDB Entry DOI: 10.7270/Q25D8V31 |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50230326

(CHEMBL4060950)Show SMILES COC(=O)Nc1ccc2-c3nc([nH]c3Cl)[C@H](C\C=C\CCC(=O)Nc2c1)NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1 |r,t:18| Show InChI InChI=1S/C28H25Cl2N9O4/c1-43-28(42)32-18-9-10-19-21(14-18)34-23(40)6-4-2-3-5-20(27-35-25(19)26(30)36-27)33-24(41)12-7-16-13-17(29)8-11-22(16)39-15-31-37-38-39/h2-3,7-15,20H,4-6H2,1H3,(H,32,42)(H,33,41)(H,34,40)(H,35,36)/b3-2+,12-7+/t20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human Factor XIa using S-2366 as chromogenic substrate after 60 mins by Lineweaver-Burk plot analysis |
Bioorg Med Chem Lett 27: 3833-3839 (2017)
Article DOI: 10.1016/j.bmcl.2017.06.058
BindingDB Entry DOI: 10.7270/Q2GT5QPS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12693
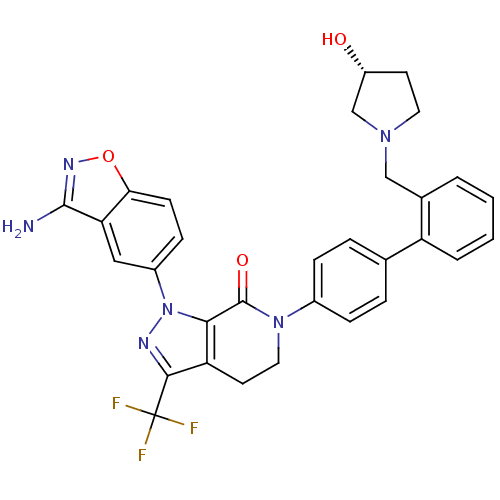
(1-(3-amino-1,2-benzoxazol-5-yl)-6-[4-(2-{[(3R)-3-h...)Show SMILES Nc1noc2ccc(cc12)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)-c1ccccc1CN1CC[C@@H](O)C1)C(F)(F)F |r| Show InChI InChI=1S/C31H27F3N6O3/c32-31(33,34)28-24-12-14-39(30(42)27(24)40(36-28)21-9-10-26-25(15-21)29(35)37-43-26)20-7-5-18(6-8-20)23-4-2-1-3-19(23)16-38-13-11-22(41)17-38/h1-10,15,22,41H,11-14,16-17H2,(H2,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 749-54 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.040
BindingDB Entry DOI: 10.7270/Q2DV1KQS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12693

(1-(3-amino-1,2-benzoxazol-5-yl)-6-[4-(2-{[(3R)-3-h...)Show SMILES Nc1noc2ccc(cc12)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)-c1ccccc1CN1CC[C@@H](O)C1)C(F)(F)F |r| Show InChI InChI=1S/C31H27F3N6O3/c32-31(33,34)28-24-12-14-39(30(42)27(24)40(36-28)21-9-10-26-25(15-21)29(35)37-43-26)20-7-5-18(6-8-20)23-4-2-1-3-19(23)16-38-13-11-22(41)17-38/h1-10,15,22,41H,11-14,16-17H2,(H2,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0300 | -59.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 4141-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.069
BindingDB Entry DOI: 10.7270/Q26T0JW1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12693

(1-(3-amino-1,2-benzoxazol-5-yl)-6-[4-(2-{[(3R)-3-h...)Show SMILES Nc1noc2ccc(cc12)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)-c1ccccc1CN1CC[C@@H](O)C1)C(F)(F)F |r| Show InChI InChI=1S/C31H27F3N6O3/c32-31(33,34)28-24-12-14-39(30(42)27(24)40(36-28)21-9-10-26-25(15-21)29(35)37-43-26)20-7-5-18(6-8-20)23-4-2-1-3-19(23)16-38-13-11-22(41)17-38/h1-10,15,22,41H,11-14,16-17H2,(H2,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0300 | -59.5 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 5584-9 (2006)
Article DOI: 10.1016/j.bmcl.2006.08.027
BindingDB Entry DOI: 10.7270/Q2Z899NQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12751

(1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamo...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C24H22N6O3S/c1-15-13-21(30(29-15)19-6-4-5-17(14-19)23(25)26)24(31)28-18-11-9-16(10-12-18)20-7-2-3-8-22(20)34(27,32)33/h2-14H,1H3,(H3,25,26)(H,28,31)(H2,27,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
In vitro activity against rabbit FXa. |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228913
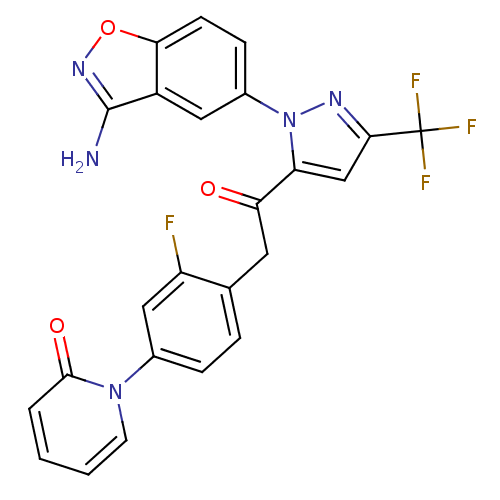
(1-(4-{2-[2-(3-amino-benzo[d]isoxazol-5-yl)-5-trifl...)Show SMILES Nc1noc2ccc(cc12)-n1nc(cc1C(=O)Cc1ccc(cc1F)-n1ccccc1=O)C(F)(F)F Show InChI InChI=1S/C24H15F4N5O3/c25-17-11-14(32-8-2-1-3-22(32)35)5-4-13(17)9-19(34)18-12-21(24(26,27)28)30-33(18)15-6-7-20-16(10-15)23(29)31-36-20/h1-8,10-12H,9H2,(H2,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 749-54 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.040
BindingDB Entry DOI: 10.7270/Q2DV1KQS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243540
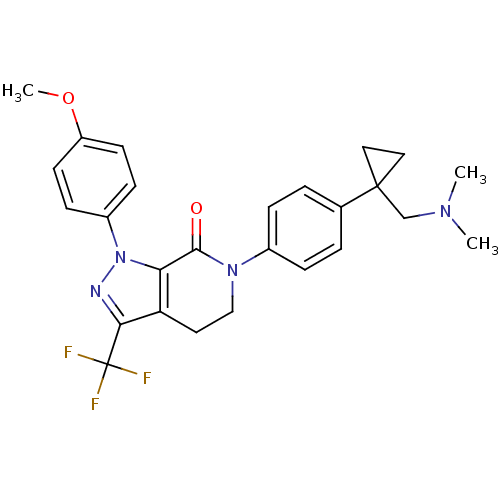
(6-(4-(1-((dimethylamino)methyl)cyclopropyl)phenyl)...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CN(C)C)CC1)C(F)(F)F Show InChI InChI=1S/C26H27F3N4O2/c1-31(2)16-25(13-14-25)17-4-6-18(7-5-17)32-15-12-21-22(24(32)34)33(30-23(21)26(27,28)29)19-8-10-20(35-3)11-9-19/h4-11H,12-16H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 4118-23 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.095
BindingDB Entry DOI: 10.7270/Q20Z7322 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243540

(6-(4-(1-((dimethylamino)methyl)cyclopropyl)phenyl)...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CN(C)C)CC1)C(F)(F)F Show InChI InChI=1S/C26H27F3N4O2/c1-31(2)16-25(13-14-25)17-4-6-18(7-5-17)32-15-12-21-22(24(32)34)33(30-23(21)26(27,28)29)19-8-10-20(35-3)11-9-19/h4-11H,12-16H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12755
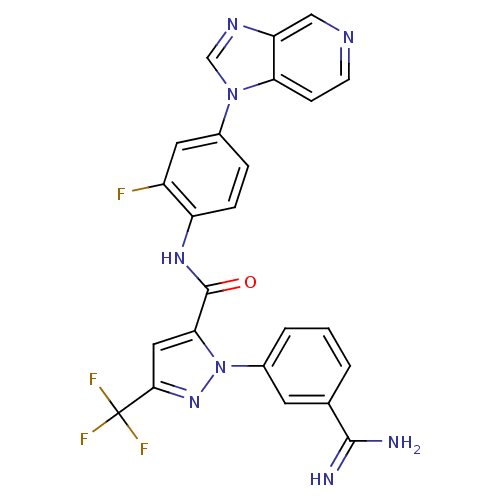
(1-(3-carbamimidoylphenyl)-N-(2-fluoro-4-{1H-imidaz...)Show SMILES NC(=N)c1cccc(c1)-n1nc(cc1C(=O)Nc1ccc(cc1F)-n1cnc2cnccc12)C(F)(F)F Show InChI InChI=1S/C24H16F4N8O/c25-16-9-14(35-12-32-18-11-31-7-6-19(18)35)4-5-17(16)33-23(37)20-10-21(24(26,27)28)34-36(20)15-3-1-2-13(8-15)22(29)30/h1-12H,(H3,29,30)(H,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0360 | -59.0 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 14: 5263-7 (2004)
Article DOI: 10.1016/j.bmcl.2004.08.034
BindingDB Entry DOI: 10.7270/Q2TH8JX0 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50097626
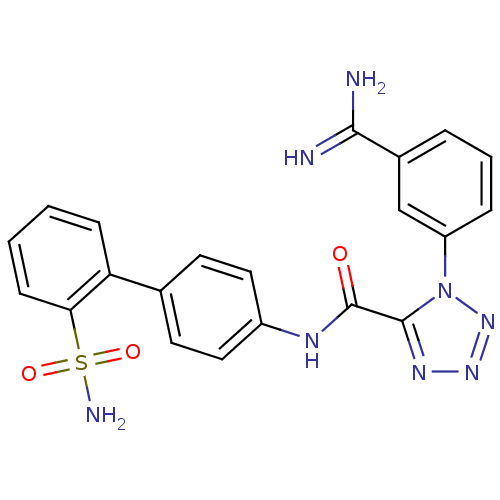
(1-(3-Carbamimidoyl-phenyl)-1H-tetrazole-5-carboxyl...)Show SMILES NC(=N)c1cccc(c1)-n1nnnc1C(=O)Nc1ccc(cc1)-c1ccccc1S(N)(=O)=O Show InChI InChI=1S/C21H18N8O3S/c22-19(23)14-4-3-5-16(12-14)29-20(26-27-28-29)21(30)25-15-10-8-13(9-11-15)17-6-1-2-7-18(17)33(24,31)32/h1-12H,(H3,22,23)(H,25,30)(H2,24,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co.
Curated by ChEMBL
| Assay Description
Inhibition of human purified factor Xa |
Bioorg Med Chem Lett 13: 369-73 (2003)
BindingDB Entry DOI: 10.7270/Q2BG2NC3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374876
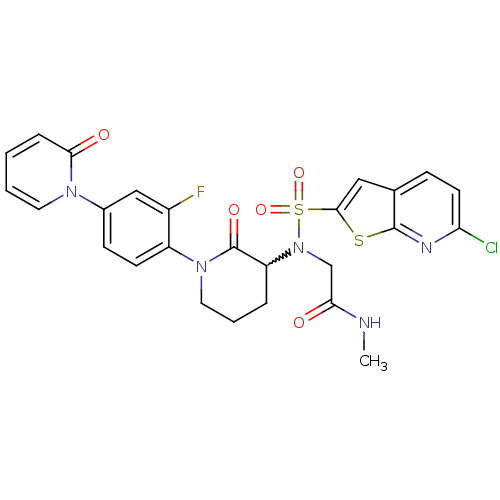
(CHEMBL270034)Show SMILES CNC(=O)CN(C1CCCN(C1=O)c1ccc(cc1F)-n1ccccc1=O)S(=O)(=O)c1cc2ccc(Cl)nc2s1 |w:6.5| Show InChI InChI=1S/C26H23ClFN5O5S2/c1-29-22(34)15-33(40(37,38)24-13-16-7-10-21(27)30-25(16)39-24)20-5-4-12-32(26(20)36)19-9-8-17(14-18(19)28)31-11-3-2-6-23(31)35/h2-3,6-11,13-14,20H,4-5,12,15H2,1H3,(H,29,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 2428-33 (2008)
Article DOI: 10.1016/j.bmcl.2008.02.054
BindingDB Entry DOI: 10.7270/Q2RX9CZ8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50243340
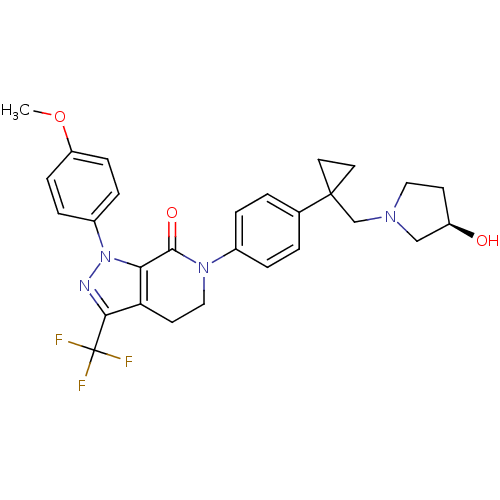
((R)-6-(4-(1-((3-hydroxypyrrolidin-1-yl)methyl)cycl...)Show SMILES COc1ccc(cc1)-n1nc(c2CCN(C(=O)c12)c1ccc(cc1)C1(CN2CC[C@@H](O)C2)CC1)C(F)(F)F |r| Show InChI InChI=1S/C28H29F3N4O3/c1-38-22-8-6-20(7-9-22)35-24-23(25(32-35)28(29,30)31)11-15-34(26(24)37)19-4-2-18(3-5-19)27(12-13-27)17-33-14-10-21(36)16-33/h2-9,21,36H,10-17H2,1H3/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 18: 4118-23 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.095
BindingDB Entry DOI: 10.7270/Q20Z7322 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12681
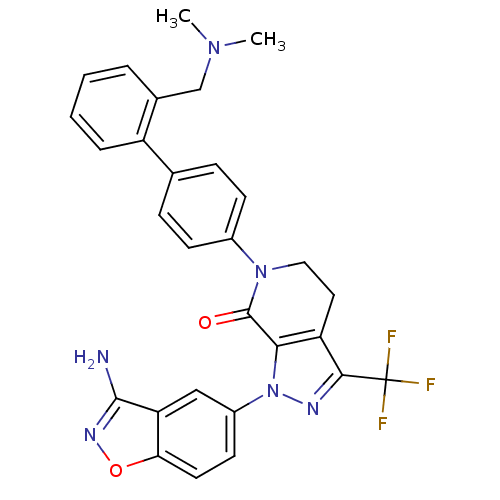
(1-(3-amino-1,2-benzoxazol-5-yl)-6-(4-{2-[(dimethyl...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)N1CCc2c(nn(c2C1=O)-c1ccc2onc(N)c2c1)C(F)(F)F Show InChI InChI=1S/C29H25F3N6O2/c1-36(2)16-18-5-3-4-6-21(18)17-7-9-19(10-8-17)37-14-13-22-25(28(37)39)38(34-26(22)29(30,31)32)20-11-12-24-23(15-20)27(33)35-40-24/h3-12,15H,13-14,16H2,1-2H3,(H2,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | -58.8 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 5584-9 (2006)
Article DOI: 10.1016/j.bmcl.2006.08.027
BindingDB Entry DOI: 10.7270/Q2Z899NQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12681

(1-(3-amino-1,2-benzoxazol-5-yl)-6-(4-{2-[(dimethyl...)Show SMILES CN(C)Cc1ccccc1-c1ccc(cc1)N1CCc2c(nn(c2C1=O)-c1ccc2onc(N)c2c1)C(F)(F)F Show InChI InChI=1S/C29H25F3N6O2/c1-36(2)16-18-5-3-4-6-21(18)17-7-9-19(10-8-17)37-14-13-22-25(28(37)39)38(34-26(22)29(30,31)32)20-11-12-24-23(15-20)27(33)35-40-24/h3-12,15H,13-14,16H2,1-2H3,(H2,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | -58.8 | n/a | n/a | n/a | n/a | n/a | 7.0 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Ki values were obtained from human purified enzyme. All assays were run in microtiter plates. Plates were read for 30 min at 405 nm. Rates were deter... |
Bioorg Med Chem Lett 16: 4141-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.069
BindingDB Entry DOI: 10.7270/Q26T0JW1 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50265826
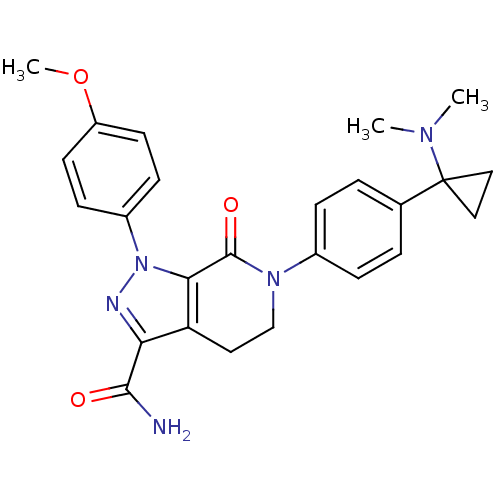
(6-(4-(1-(dimethylamino)cyclopropyl)phenyl)-1-(4-me...)Show SMILES COc1ccc(cc1)-n1nc(C(N)=O)c2CCN(C(=O)c12)c1ccc(cc1)C1(CC1)N(C)C Show InChI InChI=1S/C25H27N5O3/c1-28(2)25(13-14-25)16-4-6-17(7-5-16)29-15-12-20-21(23(26)31)27-30(22(20)24(29)32)18-8-10-19(33-3)11-9-18/h4-11H,12-15H2,1-3H3,(H2,26,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Binding affinity to human coagulation factor 10a |
Bioorg Med Chem Lett 19: 462-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.049
BindingDB Entry DOI: 10.7270/Q2M908HB |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50096094
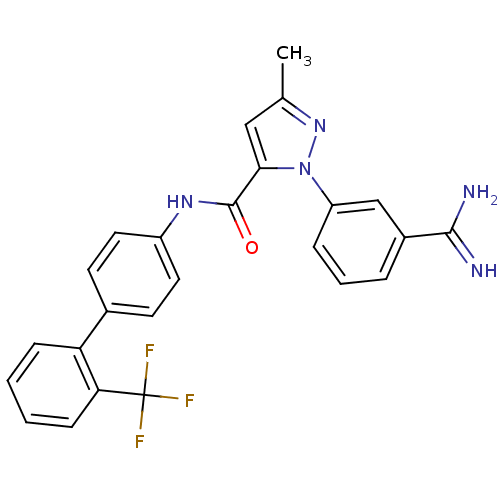
(2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-...)Show SMILES Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2C(F)(F)F)n(n1)-c1cccc(c1)C(N)=N Show InChI InChI=1S/C25H20F3N5O/c1-15-13-22(33(32-15)19-6-4-5-17(14-19)23(29)30)24(34)31-18-11-9-16(10-12-18)20-7-2-3-8-21(20)25(26,27)28/h2-14H,1H3,(H3,29,30)(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 566-78 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5VQK |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data