 Found 5824 hits with Last Name = 'nair' and Initial = 'sk'
Found 5824 hits with Last Name = 'nair' and Initial = 'sk' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM533367
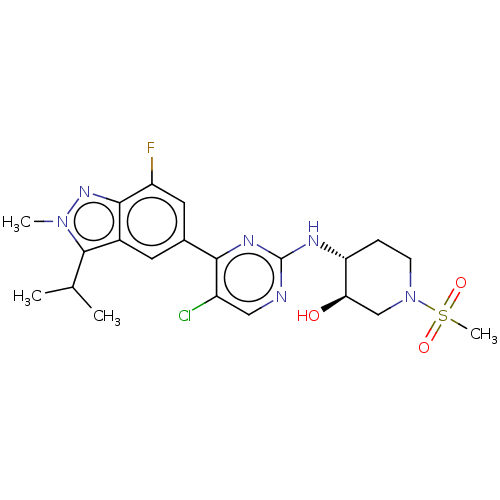
(US11220494, Example A25)Show SMILES CC(C)c1n(C)nc2c(F)cc(cc12)-c1nc(N[C@@H]2CCN(C[C@H]2O)S(C)(=O)=O)ncc1Cl |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The purpose CDK4/Cyclin D1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) in the presence of small molecule inhibitors by us... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2XG9V9M |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM462197
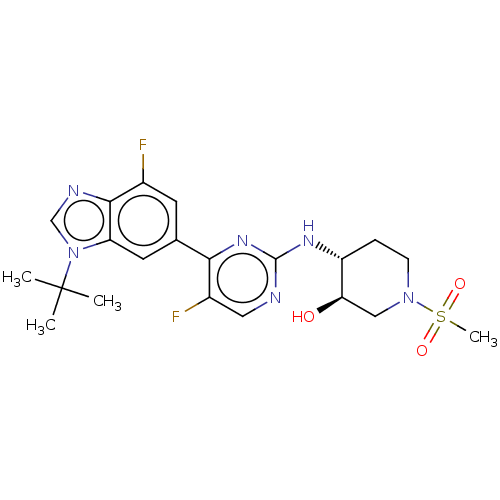
(US10766884, Example F15 | US11220494, Example F15)Show SMILES CC(C)(C)n1cnc2c(F)cc(cc12)-c1nc(N[C@@H]2CCN(C[C@H]2O)S(C)(=O)=O)ncc1F |r| Show InChI InChI=1S/C21H26F2N6O3S/c1-21(2,3)29-11-25-19-13(22)7-12(8-16(19)29)18-14(23)9-24-20(27-18)26-15-5-6-28(10-17(15)30)33(4,31)32/h7-9,11,15,17,30H,5-6,10H2,1-4H3,(H,24,26,27)/t15-,17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The purpose CDK4/Cyclin D1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) in the presence of small molecule inhibitors by us... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2XG9V9M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519706
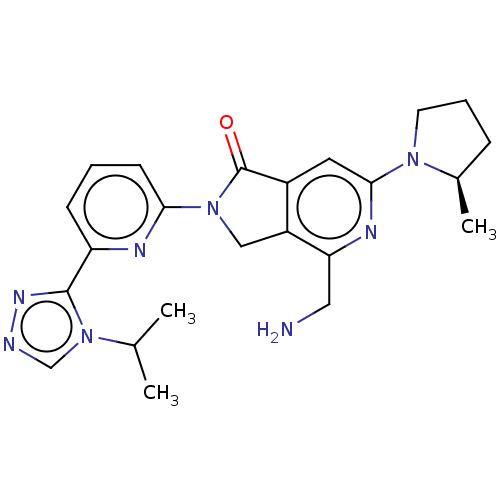
(US11142525, Example 107)Show SMILES CC(C)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CN)N2CCC[C@H]2C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606126
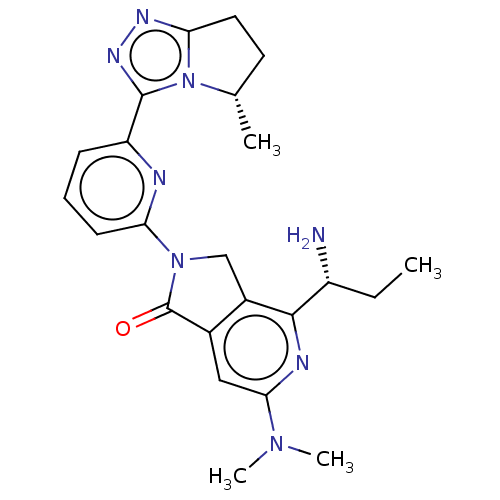
(4-[(1R)-1- aminopropyl]- 6-(dimethyl- amino)-2-{6-...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](C)n12)N(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519601

(US11142525, Example 17)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nncn1[C@@H](C)CC(F)(F)F)N(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519743

(US11142525, Example 144)Show SMILES CCC(CC)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CNC)N2CCC[C@H]2C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606115

(4-(2- aminopropan- 2-yl)-2-{6- [(5S)-5-methyl- 6,7...)Show SMILES C[C@@H]1CCCN1c1cc2C(=O)N(Cc2c(n1)C(C)(C)N)c1cccc(n1)-c1nnc2CC[C@H](C)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606114

(4-[(1S)-1- aminopropyl]- 2-{6-[(5$#958;)-5- ethyl-...)Show SMILES CC[C@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@@H](CC)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606118

(4-[(1R)-1- aminopropyl]- 2-{6-[(5R)-5- (hydroxy- m...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CO)n12)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519590

(US11142525, Example 6 | US11142525, Example 79)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nncn1C(C)C)N(C)C(C)C | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519699

(US11142525, Example 100)Show SMILES CC(C)N(C)c1cc2C(=O)N(Cc2c(CN)n1)c1cccc(n1)N1[C@@H](C)COC1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606123

(4-[(1$#958;)-1- aminoethyl]-2- {6-[(5$#958;)-5- me...)Show SMILES C[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@@H](C)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606098
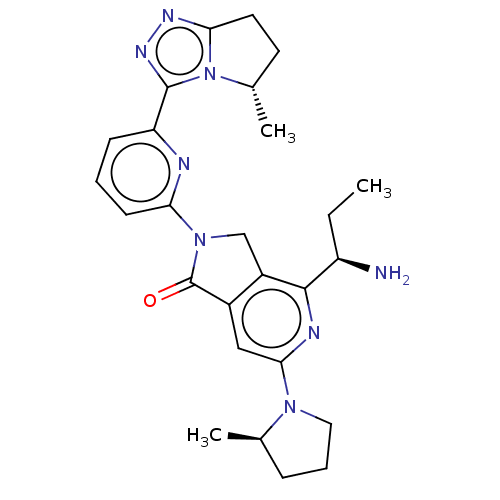
(US11684616, Example 1)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](C)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606099

(US11684616, Example 2)Show SMILES CC[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4[C@@H](C)N)N4CCC[C@H]4C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606100
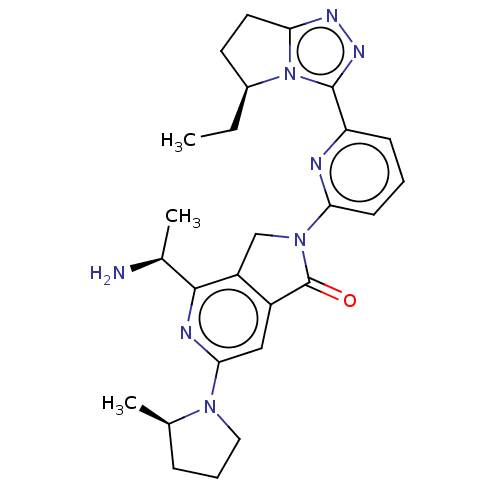
(US11684616, Example 3)Show SMILES CC[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4[C@H](C)N)N4CCC[C@H]4C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606101

(US11684616, Example 4)Show SMILES CC[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4[C@@H](C)N)N(C)C(C)C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606102

(US11684616, Example 5)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](C)n12)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606125

(4-[(1$#958;)-1- aminoethyl]-2- {6-[(5$#958;)-5- me...)Show SMILES C[C@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@@H](C)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606137
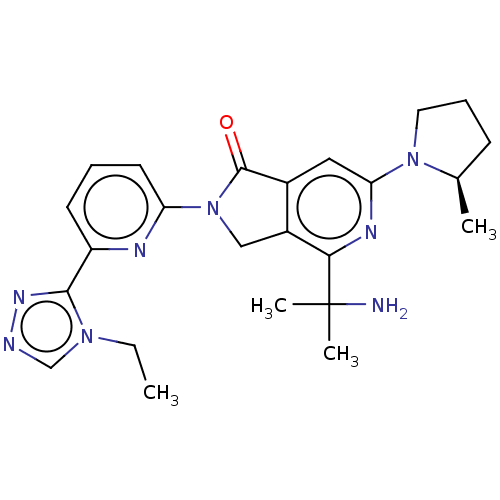
(US11684616, Example 100)Show SMILES CCn1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2C(C)(C)N)N2CCC[C@H]2C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606194

(US11684616, Example 200)Show SMILES CC[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4CNC)N4CCC[C@H]4C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606195

(US11684616, Example 201)Show SMILES CC[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4CNC)N(C)C(C)C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606199

(2-{6-[(5R)-5- (fluoromethyl)- 6,7-dihydro-5H- pyrr...)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CF)n12)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606204

(4-[(methyl- amino)methyl]- 2-{6-[(5S)- 5-methyl-6,...)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](C)n12)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606205

(2-{6-[(5S,7S)- 5,7-dimethyl-6,7- dihydro-5H- pyrro...)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2[C@@H](C)C[C@H](C)n12)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606128
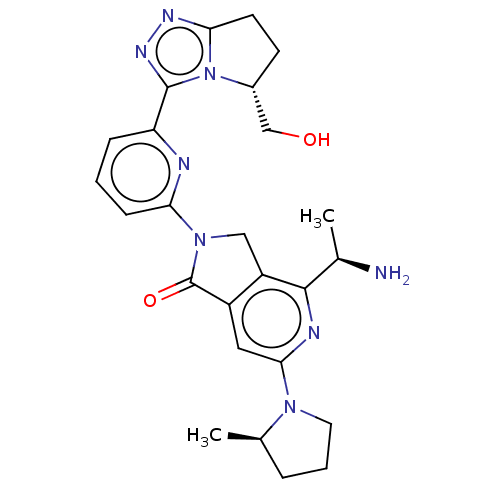
(4-[(1R)-1- aminoethyl]-2- {6-[(5R)-5- (hydroxy- me...)Show SMILES C[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CO)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606129

(4-[(1R)-1- aminopropyl]- 2-{6-[(5R)-5- (hydroxy- m...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CO)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606135
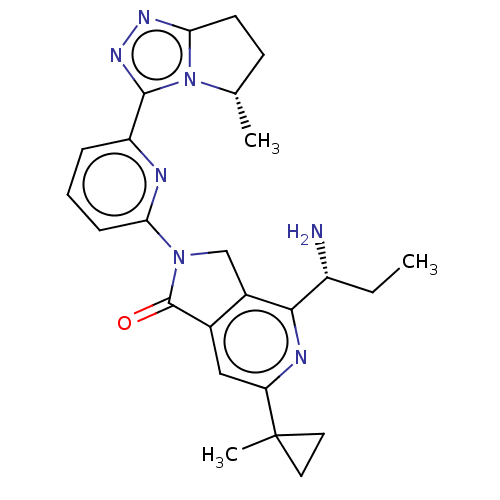
(4-[(1$#958;)-1- aminopropyl]- 6-(1- methylcyclo- p...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](C)n12)C1(C)CC1 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519602

(US11142525, Example 18)Show SMILES CC[C@@H](CC(F)(F)F)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CNC)N(C)C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 6/G1/S-specific cyclin-D1
(Homo sapiens (Human)-Mus musculus (mouse)) | BDBM370149
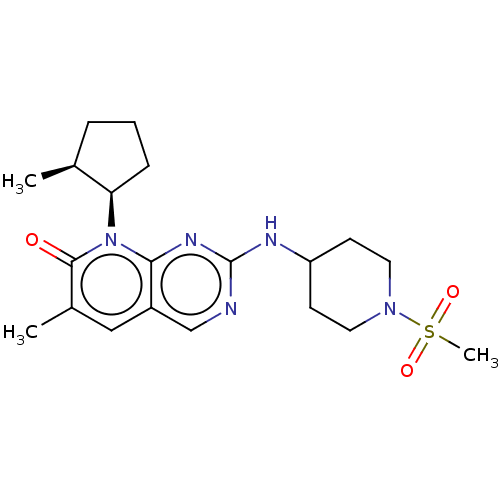
(US10233188, Example 37 | US10800783, Example 37 | ...)Show SMILES C[C@H]1CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C)c1=O |r| Show InChI InChI=1S/C20H29N5O3S/c1-13-5-4-6-17(13)25-18-15(11-14(2)19(25)26)12-21-20(23-18)22-16-7-9-24(10-8-16)29(3,27)28/h11-13,16-17H,4-10H2,1-3H3,(H,21,22,23)/t13-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
US Patent
| Assay Description
The purpose of the CDK6/Cyclin D1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) in the presence of small molecule inhibitor... |
US Patent US10800783 (2020)
BindingDB Entry DOI: 10.7270/Q24X5BVD |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519673

(US11142525, Example 74)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)N1[C@@H](C)COC1=O)N(C)C(C)C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606196

(US11684616, Example 202)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CF)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519653
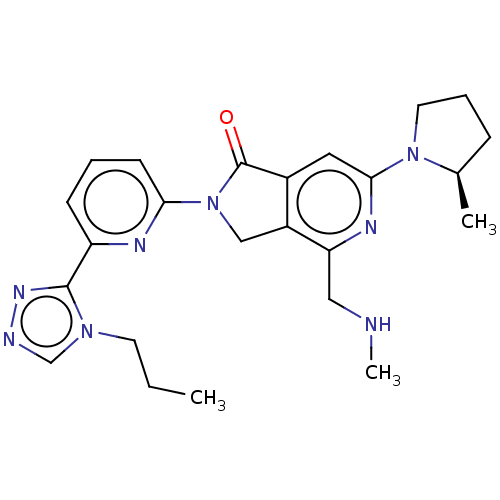
(US11142525, Example 55)Show SMILES CCCn1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CNC)N2CCC[C@H]2C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606130
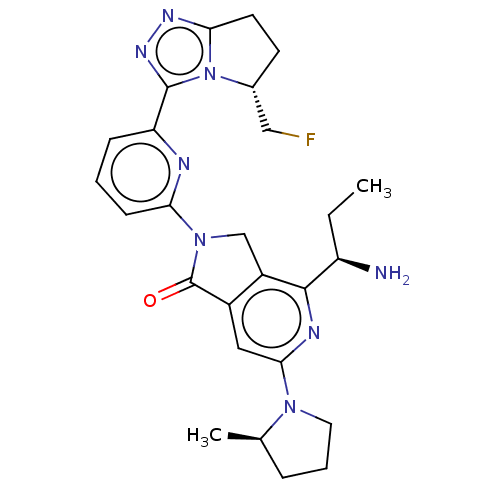
(4-[(1R)-1- aminopropyl]- 2-{6-[(5R)-5- (fluorometh...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CF)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM370149

(US10233188, Example 37 | US10800783, Example 37 | ...)Show SMILES C[C@H]1CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C)c1=O |r| Show InChI InChI=1S/C20H29N5O3S/c1-13-5-4-6-17(13)25-18-15(11-14(2)19(25)26)12-21-20(23-18)22-16-7-9-24(10-8-16)29(3,27)28/h11-13,16-17H,4-10H2,1-3H3,(H,21,22,23)/t13-,17+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
US Patent
| Assay Description
The purpose of the CDK2/Cyclin E1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) of small molecule inhibitors by using a flu... |
US Patent US10800783 (2020)
BindingDB Entry DOI: 10.7270/Q24X5BVD |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM370115
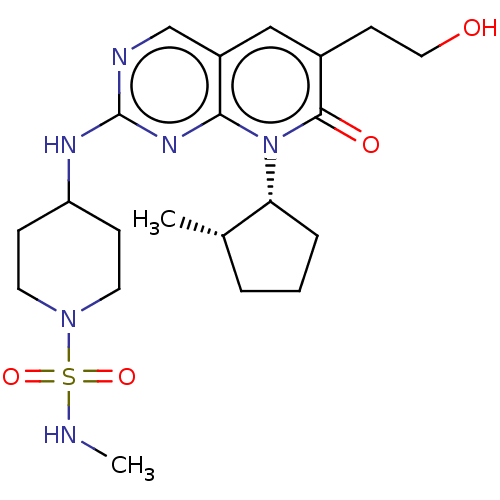
(4-({6-(2-hydroxyethyl)-8-[(1R,2S)-2-methylcyclopen...)Show SMILES CNS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(CCO)c(=O)n([C@@H]3CCC[C@@H]3C)c2n1 |r| Show InChI InChI=1S/C21H32N6O4S/c1-14-4-3-5-18(14)27-19-16(12-15(8-11-28)20(27)29)13-23-21(25-19)24-17-6-9-26(10-7-17)32(30,31)22-2/h12-14,17-18,22,28H,3-11H2,1-2H3,(H,23,24,25)/t14-,18+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The mobility shift assay electrophoretically separates the fluorescently labeled peptides (substrate and phosphorylated product) following the kinase... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2KK9G2V |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM370115

(4-({6-(2-hydroxyethyl)-8-[(1R,2S)-2-methylcyclopen...)Show SMILES CNS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(CCO)c(=O)n([C@@H]3CCC[C@@H]3C)c2n1 |r| Show InChI InChI=1S/C21H32N6O4S/c1-14-4-3-5-18(14)27-19-16(12-15(8-11-28)20(27)29)13-23-21(25-19)24-17-6-9-26(10-7-17)32(30,31)22-2/h12-14,17-18,22,28H,3-11H2,1-2H3,(H,23,24,25)/t14-,18+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
US Patent
| Assay Description
The purpose of the CDK2/Cyclin E1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) of small molecule inhibitors by using a flu... |
US Patent US10800783 (2020)
BindingDB Entry DOI: 10.7270/Q24X5BVD |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519708

(US11142525, Example 109)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nncn1C(C)C)N1CCCC1(C)C | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519746

(US11142525, Example 150)Show SMILES CCC(CC)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CN)N(C)C(C)C)C1=O | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519770
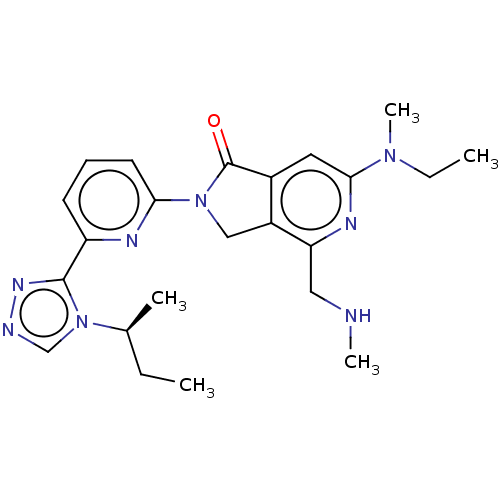
(US11142525, Example 173)Show SMILES CC[C@H](C)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CNC)N(C)CC)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM370149

(US10233188, Example 37 | US10800783, Example 37 | ...)Show SMILES C[C@H]1CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C)c1=O |r| Show InChI InChI=1S/C20H29N5O3S/c1-13-5-4-6-17(13)25-18-15(11-14(2)19(25)26)12-21-20(23-18)22-16-7-9-24(10-8-16)29(3,27)28/h11-13,16-17H,4-10H2,1-3H3,(H,21,22,23)/t13-,17+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The mobility shift assay electrophoretically separates the fluorescently labeled peptides (substrate and phosphorylated product) following the kinase... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2KK9G2V |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 6/G1/S-specific cyclin-D1 [L188C]
(Homo sapiens (Human)) | BDBM370149

(US10233188, Example 37 | US10800783, Example 37 | ...)Show SMILES C[C@H]1CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C)c1=O |r| Show InChI InChI=1S/C20H29N5O3S/c1-13-5-4-6-17(13)25-18-15(11-14(2)19(25)26)12-21-20(23-18)22-16-7-9-24(10-8-16)29(3,27)28/h11-13,16-17H,4-10H2,1-3H3,(H,21,22,23)/t13-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The purpose of the CDK6/Cyclin D1 assay is to evaluate the inhibition (% inhibition, Kiapp and Ki values) in the presence of small molecule inhibitor... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2KK9G2V |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606119
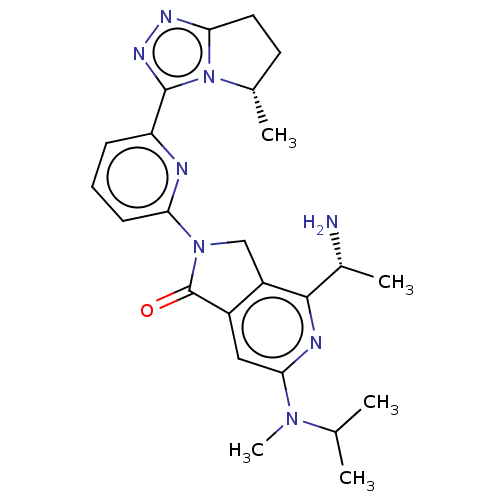
(4-[(1R)-1- aminoethyl]-2- {6-[(5S)-5- methyl-6,7- ...)Show SMILES CC(C)N(C)c1cc2C(=O)N(Cc2c(n1)[C@@H](C)N)c1cccc(n1)-c1nnc2CC[C@H](C)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519599
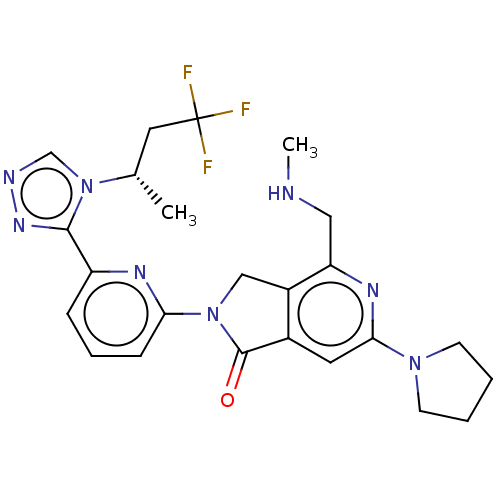
(US11142525, Example 15)Show SMILES CNCc1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nncn1[C@@H](C)CC(F)(F)F)N1CCCC1 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606178

(4-[(1R)-1- aminopropyl]- 2-[6-(4-ethyl- 4H-1,2,4- ...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nncn1CC)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519667

(US11142525, Example 68)Show SMILES C[C@@H]1CCCN1c1cc2C(=O)N(Cc2c(CN)n1)c1cccc(n1)N1[C@@H](CF)COC1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606111

(4-[(1R)-1- aminopropyl]- 2-{6-[(5$#958;)-5- ethyl-...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@@H](CC)n12)N1CCC[C@H]1C |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | <0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606134

(4-[(1$#958;)-1- aminopropyl]- 2-{6-[(5R)-5- (hydro...)Show SMILES CC[C@@H](N)c1nc(cc2C(=O)N(Cc12)c1cccc(n1)-c1nnc2CC[C@H](CO)n12)C1(C)CC1 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM519597
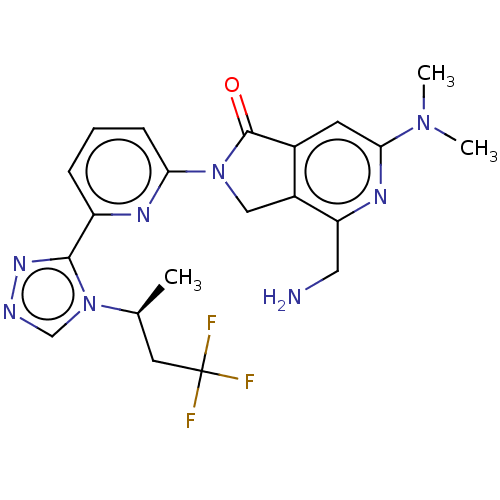
(US11142525, Example 13)Show SMILES C[C@@H](CC(F)(F)F)n1cnnc1-c1cccc(n1)N1Cc2c(cc(nc2CN)N(C)C)C1=O |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
HPK1 enzyme inhibition was measured using a microfluidic mobility shift assay (MSA). The reactions were conducted in 50 μL volumes in 96-well pl... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q21C211B |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM370300
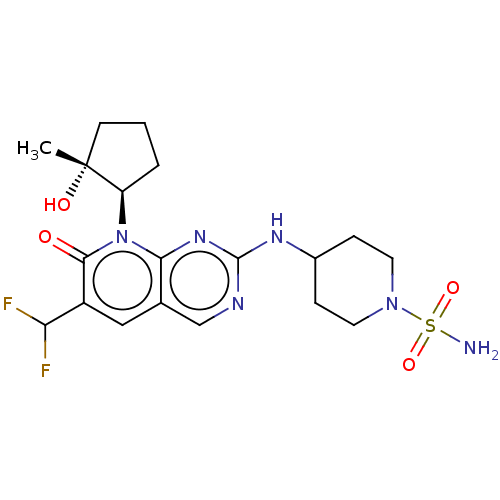
(BDBM467195 | US10233188, Example 187)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(N)(=O)=O)ncc2cc(C(F)F)c1=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| US Patent
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The mobility shift assay electrophoretically separates the fluorescently labeled peptides (substrate and phosphorylated product) following the kinase... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2KK9G2V |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase kinase 1
(Homo sapiens (Human)) | BDBM606120
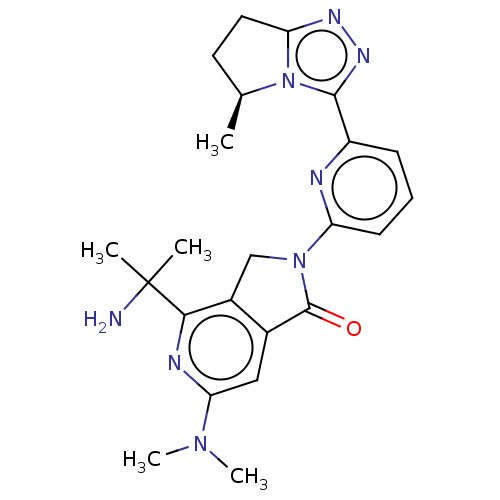
(4-(2- aminopropan- 2-yl)-6- (dimethyl- amino)-2-{6...)Show SMILES C[C@H]1CCc2nnc(-c3cccc(n3)N3Cc4c(cc(nc4C(C)(C)N)N(C)C)C3=O)n12 |r| | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2SF317X |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data