 Found 147 hits with Last Name = 'paradowski' and Initial = 'm'
Found 147 hits with Last Name = 'paradowski' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356880
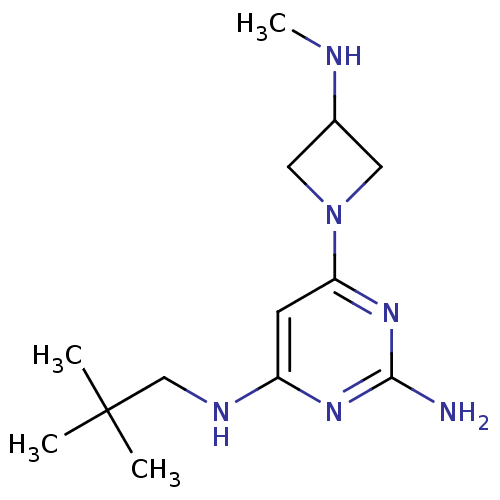
(CHEMBL1915536)Show InChI InChI=1S/C13H24N6/c1-13(2,3)8-16-10-5-11(18-12(14)17-10)19-6-9(7-19)15-4/h5,9,15H,6-8H2,1-4H3,(H3,14,16,17,18) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Alternative oxidase, mitochondrial
(Trypanosoma brucei brucei) | BDBM50459778
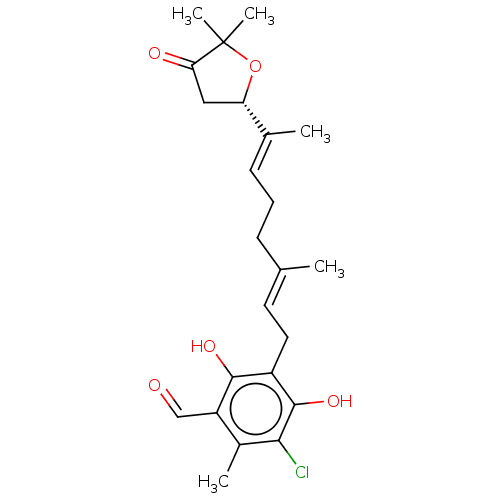
(Ascofuranone | US11565996, Compound Ascochlorin)Show SMILES [H][C@]1(CC(=O)C(C)(C)O1)C(\C)=C\CC\C(C)=C\Cc1c(O)c(Cl)c(C)c(C=O)c1O |r| Show InChI InChI=1S/C23H29ClO5/c1-13(7-6-8-14(2)18-11-19(26)23(4,5)29-18)9-10-16-21(27)17(12-25)15(3)20(24)22(16)28/h8-9,12,18,27-28H,6-7,10-11H2,1-5H3/b13-9+,14-8+/t18-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Sussex
Curated by ChEMBL
| Assay Description
Inhibition of Trypanosoma brucei alternative oxidase expressed in Escherichia coli FN102 using ubiquinol-1 as substrate preincubated for 2 mins follo... |
Eur J Med Chem 141: 676-689 (2017)
Article DOI: 10.1016/j.ejmech.2017.09.067
BindingDB Entry DOI: 10.7270/Q2028V6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356881

(CHEMBL1915537)Show InChI InChI=1S/C14H26N6/c1-14(2,3)5-6-17-11-7-12(19-13(15)18-11)20-8-10(9-20)16-4/h7,10,16H,5-6,8-9H2,1-4H3,(H3,15,17,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356884

(CHEMBL1915540)Show InChI InChI=1S/C13H22N6/c1-15-10-4-5-19(8-10)12-6-11(17-13(14)18-12)16-7-9-2-3-9/h6,9-10,15H,2-5,7-8H2,1H3,(H3,14,16,17,18)/t10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356880

(CHEMBL1915536)Show InChI InChI=1S/C13H24N6/c1-13(2,3)8-16-10-5-11(18-12(14)17-10)19-6-9(7-19)15-4/h5,9,15H,6-8H2,1-4H3,(H3,14,16,17,18) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356884

(CHEMBL1915540)Show InChI InChI=1S/C13H22N6/c1-15-10-4-5-19(8-10)12-6-11(17-13(14)18-12)16-7-9-2-3-9/h6,9-10,15H,2-5,7-8H2,1H3,(H3,14,16,17,18)/t10-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Mus musculus (mouse)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.89 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from mouse H4R expressed in HEK293T cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human histamine H4 receptor |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Rattus norvegicus (rat)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from rat H4R expressed in HEK293T cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362216

(CHEMBL1938977)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H18FN5/c1-20-5-9-7-21(8-10(9)6-20)14(17)15-18-12-3-2-11(16)4-13(12)19-15/h2-4,9-10,17H,5-8H2,1H3,(H,18,19)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362214
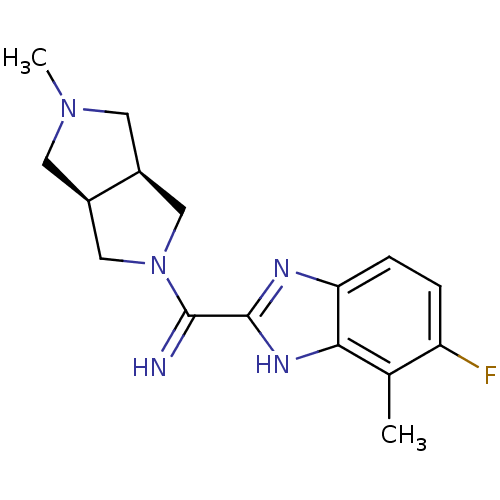
(CHEMBL1938975)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)c(C)c2[nH]1 |r| Show InChI InChI=1S/C16H20FN5/c1-9-12(17)3-4-13-14(9)20-16(19-13)15(18)22-7-10-5-21(2)6-11(10)8-22/h3-4,10-11,18H,5-8H2,1-2H3,(H,19,20)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356881

(CHEMBL1915537)Show InChI InChI=1S/C14H26N6/c1-14(2,3)5-6-17-11-7-12(19-13(15)18-11)20-8-10(9-20)16-4/h7,10,16H,5-6,8-9H2,1-4H3,(H3,15,17,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362218
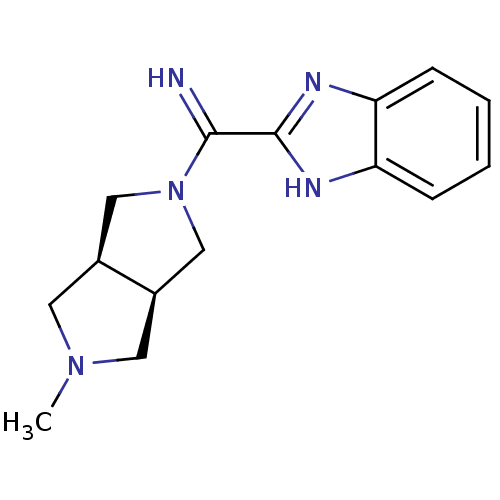
(CHEMBL1938979)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C15H19N5/c1-19-6-10-8-20(9-11(10)7-19)14(16)15-17-12-4-2-3-5-13(12)18-15/h2-5,10-11,16H,6-9H2,1H3,(H,17,18)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM22566

(5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...)Show InChI InChI=1S/C14H16ClN3O/c1-17-4-6-18(7-5-17)14(19)13-9-10-8-11(15)2-3-12(10)16-13/h2-3,8-9,16H,4-7H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362216

(CHEMBL1938977)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H18FN5/c1-20-5-9-7-21(8-10(9)6-20)14(17)15-18-12-3-2-11(16)4-13(12)19-15/h2-4,9-10,17H,5-8H2,1H3,(H,18,19)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 9.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362214

(CHEMBL1938975)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)c(C)c2[nH]1 |r| Show InChI InChI=1S/C16H20FN5/c1-9-12(17)3-4-13-14(9)20-16(19-13)15(18)22-7-10-5-21(2)6-11(10)8-22/h3-4,10-11,18H,5-8H2,1-2H3,(H,19,20)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356888
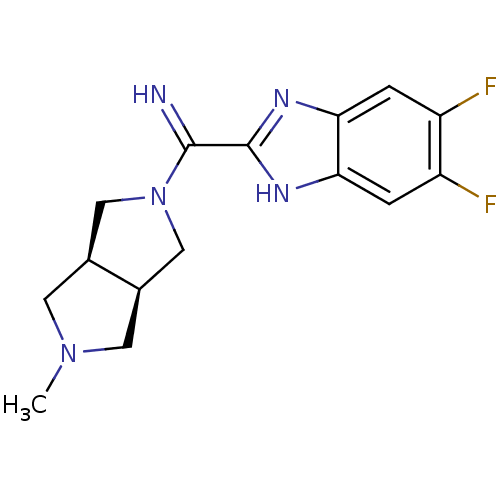
(CHEMBL1915348)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2cc(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-12-2-10(16)11(17)3-13(12)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 9.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356888

(CHEMBL1915348)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2cc(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-12-2-10(16)11(17)3-13(12)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356888

(CHEMBL1915348)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2cc(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-12-2-10(16)11(17)3-13(12)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Rattus norvegicus (rat)) | BDBM50356884

(CHEMBL1915540)Show InChI InChI=1S/C13H22N6/c1-15-10-4-5-19(8-10)12-6-11(17-13(14)18-12)16-7-9-2-3-9/h6,9-10,15H,2-5,7-8H2,1H3,(H3,14,16,17,18)/t10-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 12.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from rat H4R expressed in HEK293T cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362218

(CHEMBL1938979)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C15H19N5/c1-19-6-10-8-20(9-11(10)7-19)14(16)15-17-12-4-2-3-5-13(12)18-15/h2-5,10-11,16H,6-9H2,1H3,(H,17,18)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356882
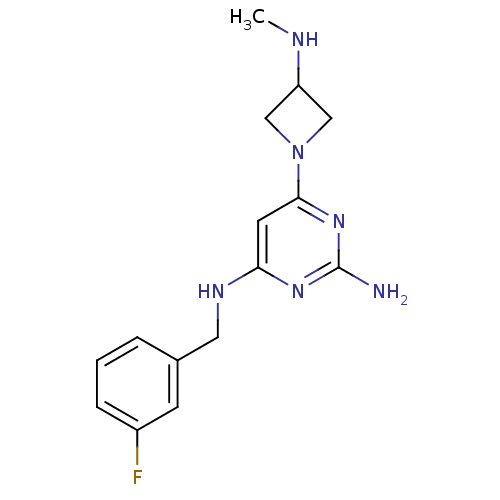
(CHEMBL1915538)Show InChI InChI=1S/C15H19FN6/c1-18-12-8-22(9-12)14-6-13(20-15(17)21-14)19-7-10-3-2-4-11(16)5-10/h2-6,12,18H,7-9H2,1H3,(H3,17,19,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356885
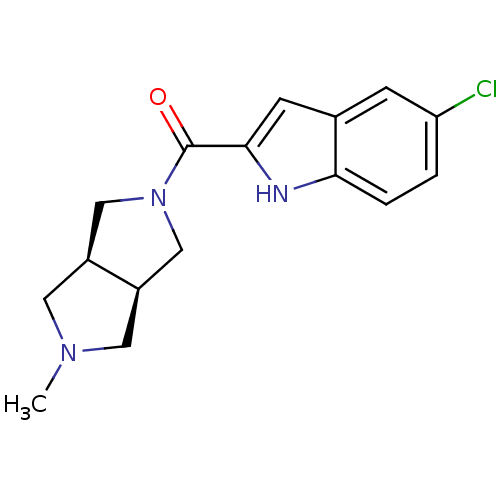
(CHEMBL1915345)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1cc2cc(Cl)ccc2[nH]1 |r| Show InChI InChI=1S/C16H18ClN3O/c1-19-6-11-8-20(9-12(11)7-19)16(21)15-5-10-4-13(17)2-3-14(10)18-15/h2-5,11-12,18H,6-9H2,1H3/t11-,12+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356885

(CHEMBL1915345)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1cc2cc(Cl)ccc2[nH]1 |r| Show InChI InChI=1S/C16H18ClN3O/c1-19-6-11-8-20(9-12(11)7-19)16(21)15-5-10-4-13(17)2-3-14(10)18-15/h2-5,11-12,18H,6-9H2,1H3/t11-,12+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356888

(CHEMBL1915348)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2cc(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-12-2-10(16)11(17)3-13(12)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 17.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356882

(CHEMBL1915538)Show InChI InChI=1S/C15H19FN6/c1-18-12-8-22(9-12)14-6-13(20-15(17)21-14)19-7-10-3-2-4-11(16)5-10/h2-6,12,18H,7-9H2,1H3,(H3,17,19,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362217
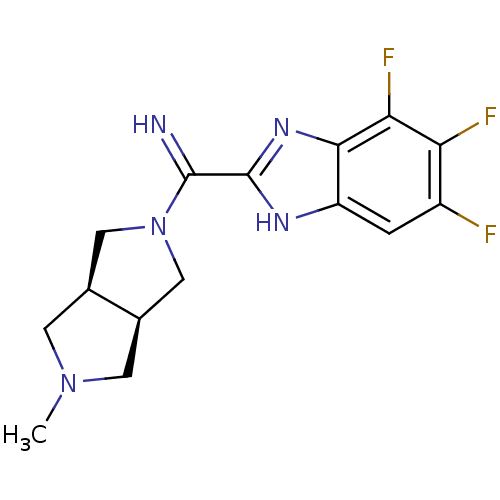
(CHEMBL1938978)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2c(F)c(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H16F3N5/c1-22-3-7-5-23(6-8(7)4-22)14(19)15-20-10-2-9(16)11(17)12(18)13(10)21-15/h2,7-8,19H,3-6H2,1H3,(H,20,21)/t7-,8+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Mus musculus (mouse)) | BDBM50356884

(CHEMBL1915540)Show InChI InChI=1S/C13H22N6/c1-15-10-4-5-19(8-10)12-6-11(17-13(14)18-12)16-7-9-2-3-9/h6,9-10,15H,2-5,7-8H2,1H3,(H3,14,16,17,18)/t10-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 20.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from mouse H4R expressed in HEK293T cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362215
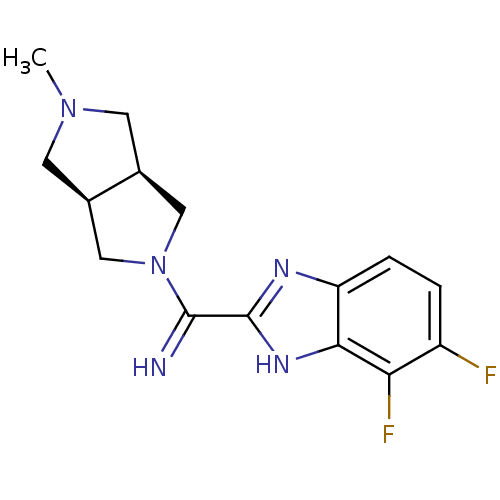
(CHEMBL1938976)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)c(F)c2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-11-3-2-10(16)12(17)13(11)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362213
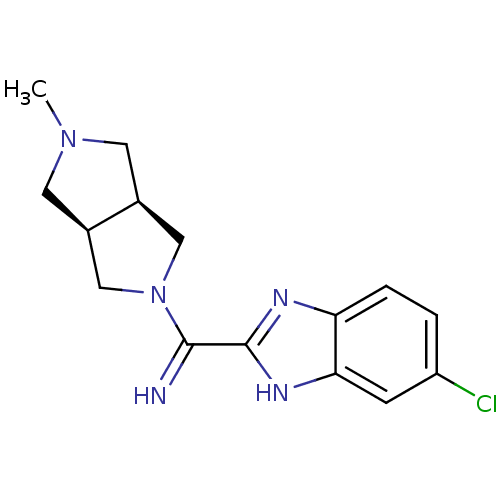
(CHEMBL1938974)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(Cl)cc2[nH]1 |r| Show InChI InChI=1S/C15H18ClN5/c1-20-5-9-7-21(8-10(9)6-20)14(17)15-18-12-3-2-11(16)4-13(12)19-15/h2-4,9-10,17H,5-8H2,1H3,(H,18,19)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356885

(CHEMBL1915345)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1cc2cc(Cl)ccc2[nH]1 |r| Show InChI InChI=1S/C16H18ClN3O/c1-19-6-11-8-20(9-12(11)7-19)16(21)15-5-10-4-13(17)2-3-14(10)18-15/h2-5,11-12,18H,6-9H2,1H3/t11-,12+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356885

(CHEMBL1915345)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1cc2cc(Cl)ccc2[nH]1 |r| Show InChI InChI=1S/C16H18ClN3O/c1-19-6-11-8-20(9-12(11)7-19)16(21)15-5-10-4-13(17)2-3-14(10)18-15/h2-5,11-12,18H,6-9H2,1H3/t11-,12+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356886
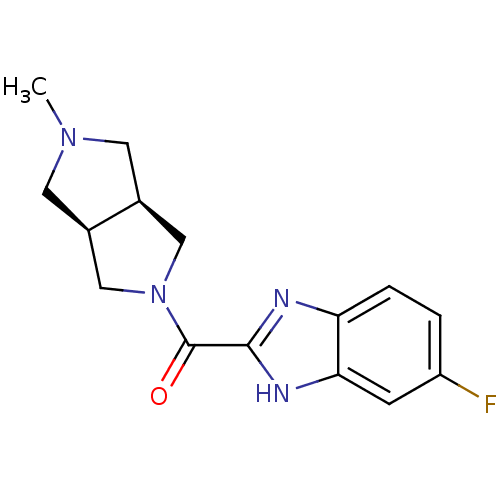
(CHEMBL1915346)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2ccc(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17FN4O/c1-19-5-9-7-20(8-10(9)6-19)15(21)14-17-12-3-2-11(16)4-13(12)18-14/h2-4,9-10H,5-8H2,1H3,(H,17,18)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50179335
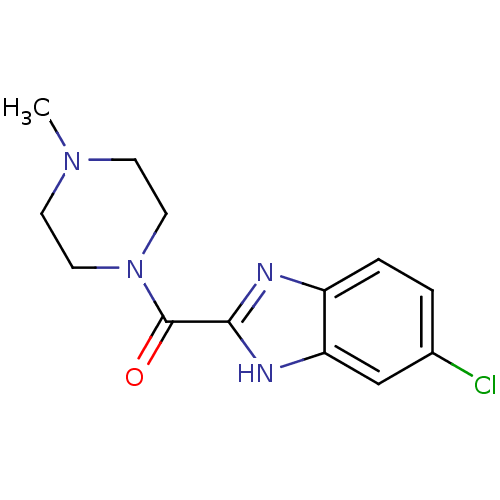
((5-Chloro-1H-benzoimidazol-2-yl)-(4-methyl-piperaz...)Show InChI InChI=1S/C13H15ClN4O/c1-17-4-6-18(7-5-17)13(19)12-15-10-3-2-9(14)8-11(10)16-12/h2-3,8H,4-7H2,1H3,(H,15,16) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 39.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50179335

((5-Chloro-1H-benzoimidazol-2-yl)-(4-methyl-piperaz...)Show InChI InChI=1S/C13H15ClN4O/c1-17-4-6-18(7-5-17)13(19)12-15-10-3-2-9(14)8-11(10)16-12/h2-3,8H,4-7H2,1H3,(H,15,16) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 47.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362215

(CHEMBL1938976)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(F)c(F)c2[nH]1 |r| Show InChI InChI=1S/C15H17F2N5/c1-21-4-8-6-22(7-9(8)5-21)14(18)15-19-11-3-2-10(16)12(17)13(11)20-15/h2-3,8-9,18H,4-7H2,1H3,(H,19,20)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362204
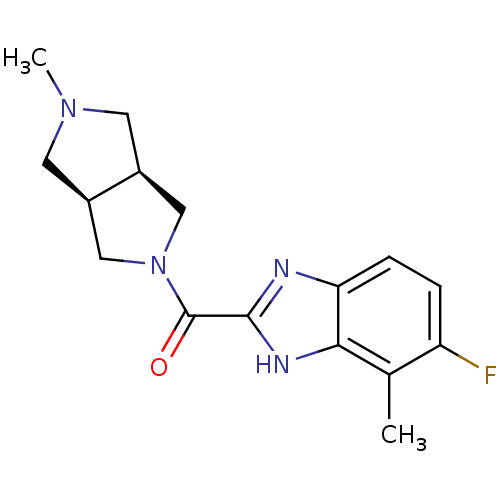
(CHEMBL1938845)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2ccc(F)c(C)c2[nH]1 |r| Show InChI InChI=1S/C16H19FN4O/c1-9-12(17)3-4-13-14(9)19-15(18-13)16(22)21-7-10-5-20(2)6-11(10)8-21/h3-4,10-11H,5-8H2,1-2H3,(H,18,19)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356887
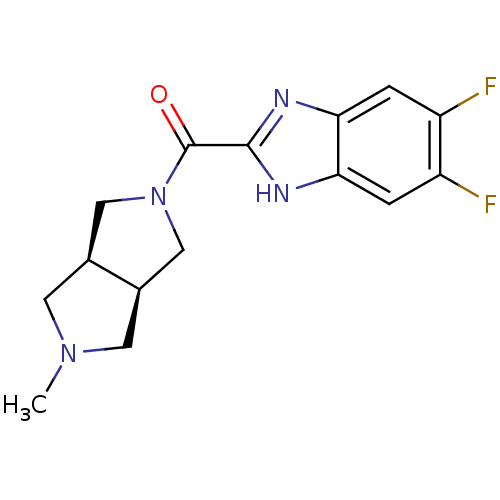
(CHEMBL1915347)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2cc(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H16F2N4O/c1-20-4-8-6-21(7-9(8)5-20)15(22)14-18-12-2-10(16)11(17)3-13(12)19-14/h2-3,8-9H,4-7H2,1H3,(H,18,19)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at full length human H4R expressed in HEK293 cells assessed as reversal of forskolin-induced cAMP production by CRE-beta-lactamas... |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362213

(CHEMBL1938974)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2ccc(Cl)cc2[nH]1 |r| Show InChI InChI=1S/C15H18ClN5/c1-20-5-9-7-21(8-10(9)6-20)14(17)15-18-12-3-2-11(16)4-13(12)19-15/h2-4,9-10,17H,5-8H2,1H3,(H,18,19)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human histamine H4 receptor expressed in HEK293 cells assessed as rev inhibition of forskolin-stimulated cAMP accumulation by ... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362217

(CHEMBL1938978)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=N)c1nc2c(F)c(F)c(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H16F3N5/c1-22-3-7-5-23(6-8(7)4-22)14(19)15-20-10-2-9(16)11(17)12(18)13(10)21-15/h2,7-8,19H,3-6H2,1H3,(H,20,21)/t7-,8+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362205
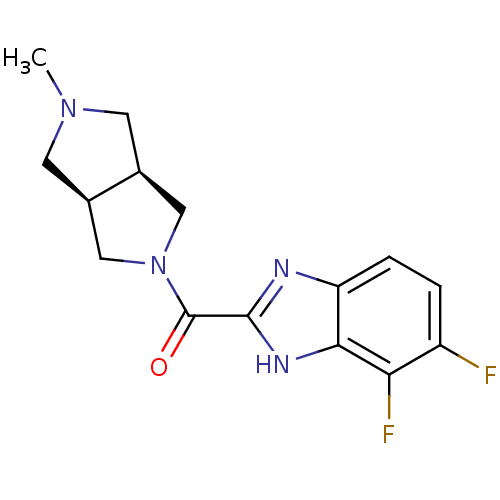
(CHEMBL1938846)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2ccc(F)c(F)c2[nH]1 |r| Show InChI InChI=1S/C15H16F2N4O/c1-20-4-8-6-21(7-9(8)5-20)15(22)14-18-11-3-2-10(16)12(17)13(11)19-14/h2-3,8-9H,4-7H2,1H3,(H,18,19)/t8-,9+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 74 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356879

(CHEMBL1915535)Show InChI InChI=1S/C15H27N5/c1-15(2,3)5-6-16-13-11-14(18-12-17-13)20-9-7-19(4)8-10-20/h11-12H,5-10H2,1-4H3,(H,16,17,18) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356886

(CHEMBL1915346)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2ccc(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17FN4O/c1-19-5-9-7-20(8-10(9)6-19)15(21)14-17-12-3-2-11(16)4-13(12)18-14/h2-4,9-10H,5-8H2,1H3,(H,17,18)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 83 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50362211

(CHEMBL1938972)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1cc2cc(Cl)cnc2[nH]1 |r| Show InChI InChI=1S/C15H17ClN4O/c1-19-5-10-7-20(8-11(10)6-19)15(21)13-3-9-2-12(16)4-17-14(9)18-13/h2-4,10-11H,5-8H2,1H3,(H,17,18)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 83 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]histamine from human recombinant histamine H4 receptor expressed in CHO cells coexpressing Ga15 by radioligand filtration binding... |
Bioorg Med Chem Lett 22: 1156-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.11.098
BindingDB Entry DOI: 10.7270/Q2Q81DJD |
More data for this
Ligand-Target Pair | |
Histamine H4 receptor
(Homo sapiens (Human)) | BDBM50356886

(CHEMBL1915346)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)C(=O)c1nc2ccc(F)cc2[nH]1 |r| Show InChI InChI=1S/C15H17FN4O/c1-19-5-9-7-20(8-10(9)6-19)15(21)14-17-12-3-2-11(16)4-13(12)18-14/h2-4,9-10H,5-8H2,1H3,(H,17,18)/t9-,10+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 83 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-histamine from full length human H4R expressed in HEK293 cells |
Bioorg Med Chem Lett 21: 6596-602 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.125
BindingDB Entry DOI: 10.7270/Q20C4W6C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data