 Found 1730 hits with Last Name = 'schiller' and Initial = 'pw'
Found 1730 hits with Last Name = 'schiller' and Initial = 'pw' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008834
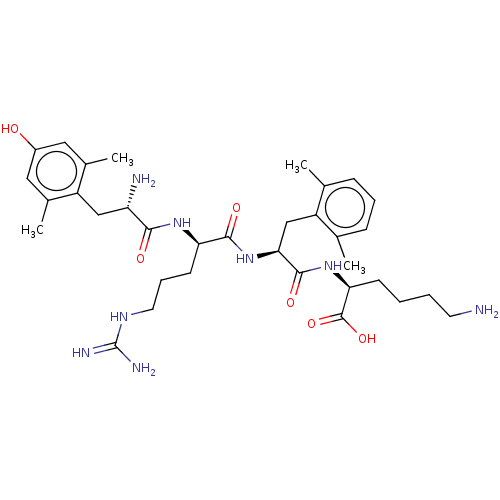
(CHEMBL3236671)Show SMILES Cc1cccc(C)c1C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C34H52N8O6/c1-19-9-7-10-20(2)25(19)18-29(32(46)41-28(33(47)48)11-5-6-13-35)42-31(45)27(12-8-14-39-34(37)38)40-30(44)26(36)17-24-21(3)15-23(43)16-22(24)4/h7,9-10,15-16,26-29,43H,5-6,8,11-14,17-18,35-36H2,1-4H3,(H,40,44)(H,41,46)(H,42,45)(H,47,48)(H4,37,38,39)/t26-,27+,28-,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00935 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50012477
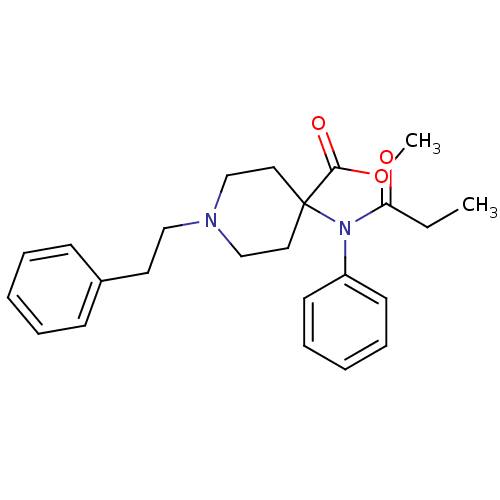
(1-Phenethyl-4-(phenyl-propionyl-amino)-piperidine-...)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CCc2ccccc2)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O3/c1-3-22(27)26(21-12-8-5-9-13-21)24(23(28)29-2)15-18-25(19-16-24)17-14-20-10-6-4-7-11-20/h4-13H,3,14-19H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes |
Bioorg Med Chem 22: 4581-6 (2014)
Article DOI: 10.1016/j.bmc.2014.07.033
BindingDB Entry DOI: 10.7270/Q2P270WT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50198754

(CHEMBL3924888)Show SMILES Cc1cc(O)cc(C)c1C[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CCCCNC(N)=N)NC1=O |r| Show InChI InChI=1S/C33H48N8O5/c1-20-16-23(42)17-21(2)24(20)19-28-32(46)39-26(13-7-9-15-37-33(35)36)29(43)40-27(18-22-10-4-3-5-11-22)31(45)38-25(30(44)41-28)12-6-8-14-34/h3-5,10-11,16-17,25-28,42H,6-9,12-15,18-19,34H2,1-2H3,(H,38,45)(H,39,46)(H,40,43)(H,41,44)(H4,35,36,37)/t25-,26+,27-,28-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0323 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from MOR in rat brain membrane measured after 2 hrs |
J Med Chem 59: 9243-9254 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01200
BindingDB Entry DOI: 10.7270/Q23B623F |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50173133
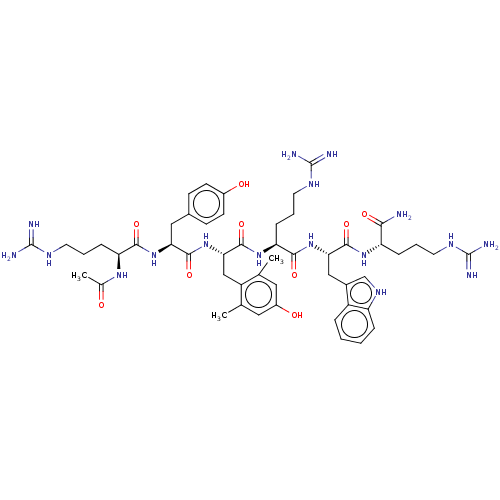
(CHEMBL3810319)Show SMILES CC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCNC(N)=N)C(N)=O |r| Show InChI InChI=1S/C51H73N17O9/c1-27-21-33(71)22-28(2)35(27)25-42(68-46(75)40(23-30-14-16-32(70)17-15-30)66-44(73)38(63-29(3)69)12-7-19-60-50(55)56)48(77)65-39(13-8-20-61-51(57)58)45(74)67-41(24-31-26-62-36-10-5-4-9-34(31)36)47(76)64-37(43(52)72)11-6-18-59-49(53)54/h4-5,9-10,14-17,21-22,26,37-42,62,70-71H,6-8,11-13,18-20,23-25H2,1-3H3,(H2,52,72)(H,63,69)(H,64,76)(H,65,77)(H,66,73)(H,67,74)(H,68,75)(H4,53,54,59)(H4,55,56,60)(H4,57,58,61)/t37-,38-,39-,40-,41-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]OFQ/nociceptin from human nociceptin receptor expressed in CHO cell membrane incubated for 60 mins by liquid scintillation counti... |
J Med Chem 59: 3777-92 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01976
BindingDB Entry DOI: 10.7270/Q2T72KC6 |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50173132

(CHEMBL3808650)Show SMILES CC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccc(F)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCNC(N)=N)C(N)=O |r| Show InChI InChI=1S/C49H68FN17O8/c1-27(68)62-36(10-5-21-59-48(54)55)42(71)65-39(24-29-14-18-32(69)19-15-29)45(74)66-38(23-28-12-16-31(50)17-13-28)44(73)64-37(11-6-22-60-49(56)57)43(72)67-40(25-30-26-61-34-8-3-2-7-33(30)34)46(75)63-35(41(51)70)9-4-20-58-47(52)53/h2-3,7-8,12-19,26,35-40,61,69H,4-6,9-11,20-25H2,1H3,(H2,51,70)(H,62,68)(H,63,75)(H,64,73)(H,65,71)(H,66,74)(H,67,72)(H4,52,53,58)(H4,54,55,59)(H4,56,57,60)/t35-,36-,37-,38-,39-,40-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]OFQ/nociceptin from human nociceptin receptor expressed in CHO cell membrane incubated for 60 mins by liquid scintillation counti... |
J Med Chem 59: 3777-92 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01976
BindingDB Entry DOI: 10.7270/Q2T72KC6 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50090002
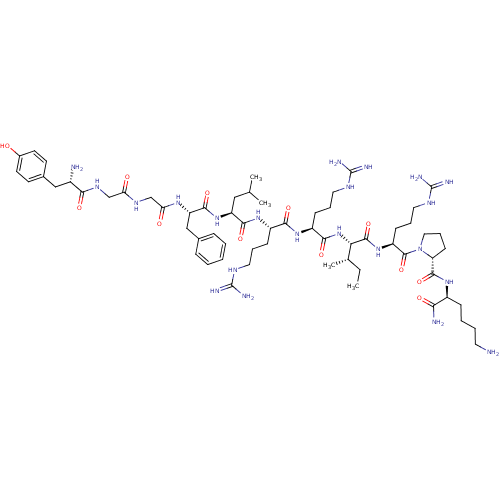
(CHEMBL411003 | Dynorphin A (1-11)-NH2H-Tyr-Gly-Gly...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C63H104N22O12/c1-5-37(4)51(59(96)82-45(20-13-29-75-63(71)72)60(97)85-30-14-21-48(85)58(95)79-42(52(66)89)17-9-10-26-64)84-55(92)44(19-12-28-74-62(69)70)80-54(91)43(18-11-27-73-61(67)68)81-56(93)46(31-36(2)3)83-57(94)47(33-38-15-7-6-8-16-38)78-50(88)35-76-49(87)34-77-53(90)41(65)32-39-22-24-40(86)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,86H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H2,66,89)(H,76,87)(H,77,90)(H,78,88)(H,79,95)(H,80,91)(H,81,93)(H,82,96)(H,83,94)(H,84,92)(H4,67,68,73)(H4,69,70,74)(H4,71,72,75)/t37-,41-,42-,43-,44-,45-,46-,47-,48+,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards human Opioid receptor kappa 1 on CHO cell membranes using [3H]diprenorphine displacement. |
J Med Chem 46: 2104-9 (2003)
Article DOI: 10.1021/jm020125+
BindingDB Entry DOI: 10.7270/Q2NZ88CF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM86416

(Dmt-d-Arg-Phe-A2pr-NH2)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C29H43N9O5/c1-16-11-19(39)12-17(2)20(16)14-21(31)26(41)36-22(9-6-10-35-29(33)34)27(42)37-23(13-18-7-4-3-5-8-18)28(43)38-24(15-30)25(32)40/h3-5,7-8,11-12,21-24,39H,6,9-10,13-15,30-31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H,38,43)(H4,33,34,35)/t21-,22+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0636 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50198760
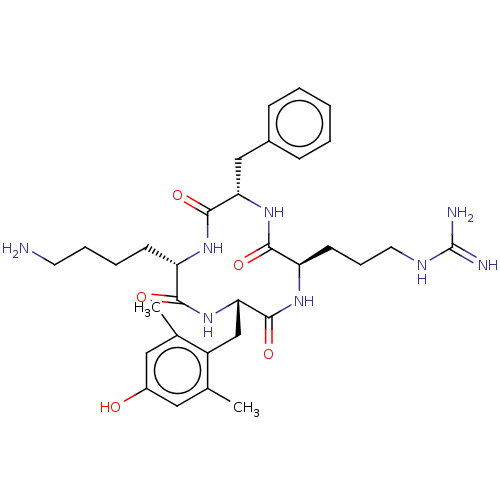
(CHEMBL3897031)Show SMILES Cc1cc(O)cc(C)c1C[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CCCNC(N)=N)NC1=O |r| Show InChI InChI=1S/C32H46N8O5/c1-19-15-22(41)16-20(2)23(19)18-27-31(45)38-25(12-8-14-36-32(34)35)29(43)39-26(17-21-9-4-3-5-10-21)30(44)37-24(28(42)40-27)11-6-7-13-33/h3-5,9-10,15-16,24-27,41H,6-8,11-14,17-18,33H2,1-2H3,(H,37,44)(H,38,45)(H,39,43)(H,40,42)(H4,34,35,36)/t24-,25+,26-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0637 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from MOR in rat brain membrane measured after 2 hrs |
J Med Chem 59: 9243-9254 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01200
BindingDB Entry DOI: 10.7270/Q23B623F |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50090002

(CHEMBL411003 | Dynorphin A (1-11)-NH2H-Tyr-Gly-Gly...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C63H104N22O12/c1-5-37(4)51(59(96)82-45(20-13-29-75-63(71)72)60(97)85-30-14-21-48(85)58(95)79-42(52(66)89)17-9-10-26-64)84-55(92)44(19-12-28-74-62(69)70)80-54(91)43(18-11-27-73-61(67)68)81-56(93)46(31-36(2)3)83-57(94)47(33-38-15-7-6-8-16-38)78-50(88)35-76-49(87)34-77-53(90)41(65)32-39-22-24-40(86)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,86H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H2,66,89)(H,76,87)(H,77,90)(H,78,88)(H,79,95)(H,80,91)(H,81,93)(H,82,96)(H,83,94)(H,84,92)(H4,67,68,73)(H4,69,70,74)(H4,71,72,75)/t37-,41-,42-,43-,44-,45-,46-,47-,48+,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Binding affinity towards cloned human Opioid receptor kappa 1 in CHO cell membranes. |
J Med Chem 43: 2698-702 (2000)
BindingDB Entry DOI: 10.7270/Q25D8SJ1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50198758

(CHEMBL3908315)Show SMILES Cc1cc(O)cc(C)c1C[C@@H]1NC(=O)C(CCCN)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CCCNC(N)=N)NC1=O |r| Show InChI InChI=1S/C31H44N8O5/c1-18-14-21(40)15-19(2)22(18)17-26-30(44)37-24(11-7-13-35-31(33)34)28(42)38-25(16-20-8-4-3-5-9-20)29(43)36-23(10-6-12-32)27(41)39-26/h3-5,8-9,14-15,23-26,40H,6-7,10-13,16-17,32H2,1-2H3,(H,36,43)(H,37,44)(H,38,42)(H,39,41)(H4,33,34,35)/t23?,24-,25+,26+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0745 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from MOR in rat brain membrane measured after 2 hrs |
J Med Chem 59: 9243-9254 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01200
BindingDB Entry DOI: 10.7270/Q23B623F |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070377
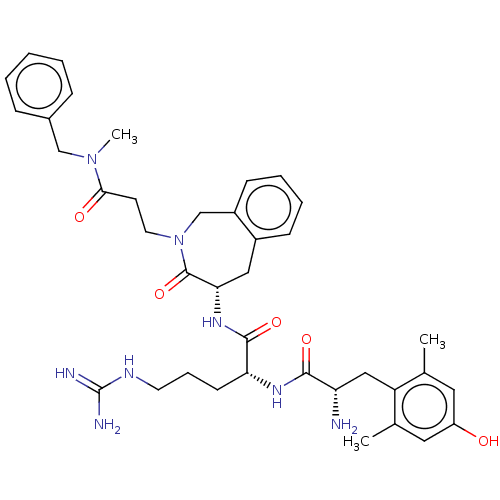
(CHEMBL3408519)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H50N8O5/c1-24-18-29(47)19-25(2)30(24)21-31(39)35(49)43-32(14-9-16-42-38(40)41)36(50)44-33-20-27-12-7-8-13-28(27)23-46(37(33)51)17-15-34(48)45(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,47H,9,14-17,20-23,39H2,1-3H3,(H,43,49)(H,44,50)(H4,40,41,42)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membrane after 2 hrs |
ACS Med Chem Lett 6: 1209-14 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00359
BindingDB Entry DOI: 10.7270/Q2VH5QQJ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070376
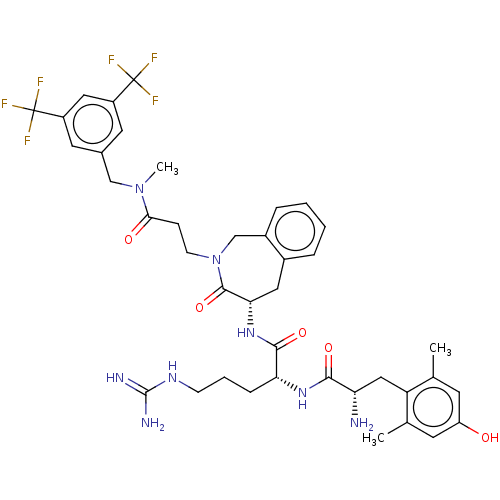
(CHEMBL3408518)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2cc(cc(c2)C(F)(F)F)C(F)(F)F)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C40H48F6N8O5/c1-22-13-29(55)14-23(2)30(22)19-31(47)35(57)51-32(9-6-11-50-38(48)49)36(58)52-33-17-25-7-4-5-8-26(25)21-54(37(33)59)12-10-34(56)53(3)20-24-15-27(39(41,42)43)18-28(16-24)40(44,45)46/h4-5,7-8,13-16,18,31-33,55H,6,9-12,17,19-21,47H2,1-3H3,(H,51,57)(H,52,58)(H4,48,49,50)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070377

(CHEMBL3408519)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H50N8O5/c1-24-18-29(47)19-25(2)30(24)21-31(39)35(49)43-32(14-9-16-42-38(40)41)36(50)44-33-20-27-12-7-8-13-28(27)23-46(37(33)51)17-15-34(48)45(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,47H,9,14-17,20-23,39H2,1-3H3,(H,43,49)(H,44,50)(H4,40,41,42)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50010704
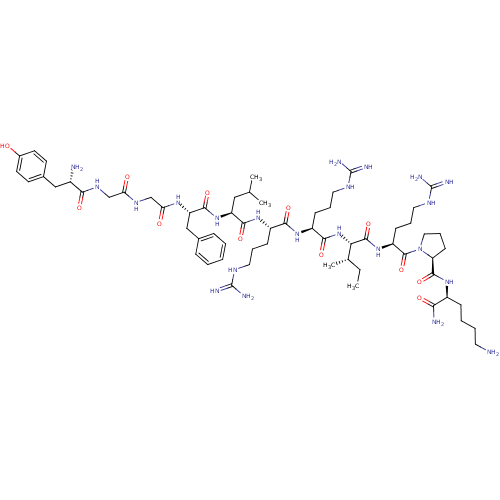
(CHEMBL216640 | Dyn A(1-11)-NH2 | Dynorphin A analo...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C63H104N22O12/c1-5-37(4)51(59(96)82-45(20-13-29-75-63(71)72)60(97)85-30-14-21-48(85)58(95)79-42(52(66)89)17-9-10-26-64)84-55(92)44(19-12-28-74-62(69)70)80-54(91)43(18-11-27-73-61(67)68)81-56(93)46(31-36(2)3)83-57(94)47(33-38-15-7-6-8-16-38)78-50(88)35-76-49(87)34-77-53(90)41(65)32-39-22-24-40(86)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,86H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H2,66,89)(H,76,87)(H,77,90)(H,78,88)(H,79,95)(H,80,91)(H,81,93)(H,82,96)(H,83,94)(H,84,92)(H4,67,68,73)(H4,69,70,74)(H4,71,72,75)/t37-,41-,42-,43-,44-,45-,46-,47-,48-,51-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0869 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Binding affinity was measured on opioid receptor kappa 1 |
J Med Chem 44: 3048-53 (2001)
BindingDB Entry DOI: 10.7270/Q28K79SN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070380
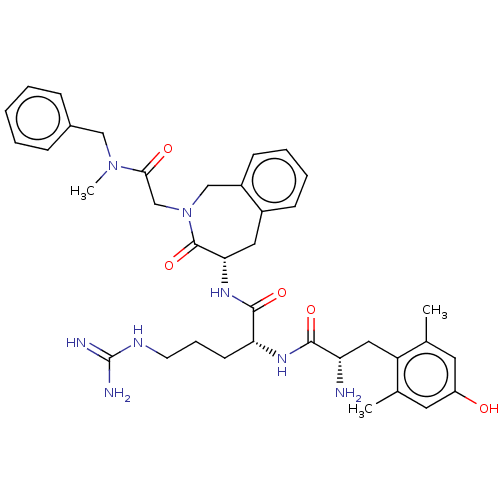
(CHEMBL3408522)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C37H48N8O5/c1-23-16-28(46)17-24(2)29(23)19-30(38)34(48)42-31(14-9-15-41-37(39)40)35(49)43-32-18-26-12-7-8-13-27(26)21-45(36(32)50)22-33(47)44(3)20-25-10-5-4-6-11-25/h4-8,10-13,16-17,30-32,46H,9,14-15,18-22,38H2,1-3H3,(H,42,48)(H,43,49)(H4,39,40,41)/t30-,31+,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008837

(CHEMBL3236674)Show SMILES CCc1cccc(C)c1C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C35H54N8O6/c1-5-23-11-8-10-20(2)26(23)19-30(33(47)42-29(34(48)49)12-6-7-14-36)43-32(46)28(13-9-15-40-35(38)39)41-31(45)27(37)18-25-21(3)16-24(44)17-22(25)4/h8,10-11,16-17,27-30,44H,5-7,9,12-15,18-19,36-37H2,1-4H3,(H,41,45)(H,42,47)(H,43,46)(H,48,49)(H4,38,39,40)/t27-,28+,29-,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0958 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008840
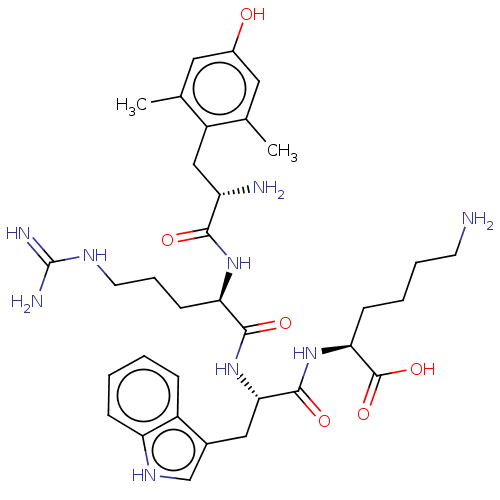
(CHEMBL3236677)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C34H49N9O6/c1-19-14-22(44)15-20(2)24(19)17-25(36)30(45)41-27(11-7-13-39-34(37)38)31(46)43-29(16-21-18-40-26-9-4-3-8-23(21)26)32(47)42-28(33(48)49)10-5-6-12-35/h3-4,8-9,14-15,18,25,27-29,40,44H,5-7,10-13,16-17,35-36H2,1-2H3,(H,41,45)(H,42,47)(H,43,46)(H,48,49)(H4,37,38,39)/t25-,27+,28-,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0991 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM82552
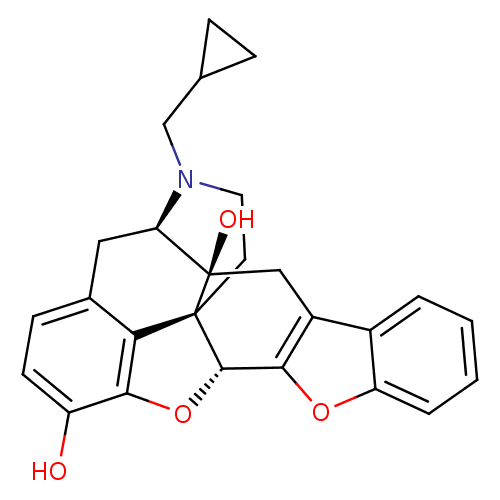
(CAS_111555-58-9 | NTB | naltrindolebenzofuran)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)c1oc2ccccc2c1C[C@@]35O |r| Show InChI InChI=1S/C26H25NO4/c28-18-8-7-15-11-20-26(29)12-17-16-3-1-2-4-19(16)30-22(17)24-25(26,21(15)23(18)31-24)9-10-27(20)13-14-5-6-14/h1-4,7-8,14,20,24,28-29H,5-6,9-13H2/t20-,24+,25+,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
I.G.B.M.C.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 313: 410-21 (2005)
Article DOI: 10.1124/jpet.104.077321
BindingDB Entry DOI: 10.7270/Q2XD107N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070385
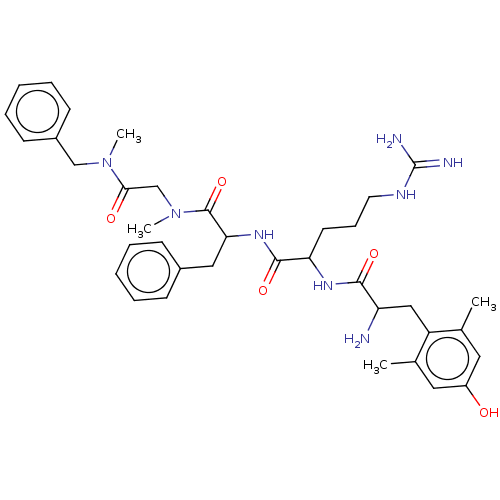
(CHEMBL3408736)Show SMILES CN(CC(=O)N(C)Cc1ccccc1)C(=O)C(Cc1ccccc1)NC(=O)C(CCCNC(N)=N)NC(=O)C(N)Cc1c(C)cc(O)cc1C Show InChI InChI=1S/C37H50N8O5/c1-24-18-28(46)19-25(2)29(24)21-30(38)34(48)42-31(16-11-17-41-37(39)40)35(49)43-32(20-26-12-7-5-8-13-26)36(50)45(4)23-33(47)44(3)22-27-14-9-6-10-15-27/h5-10,12-15,18-19,30-32,46H,11,16-17,20-23,38H2,1-4H3,(H,42,48)(H,43,49)(H4,39,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008838
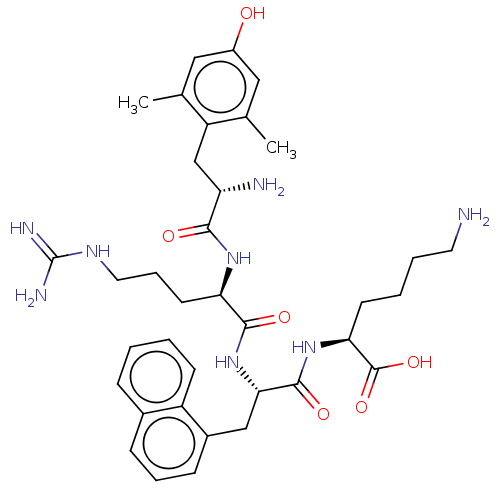
(CHEMBL3236675)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1cccc2ccccc12)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C36H50N8O6/c1-21-17-25(45)18-22(2)27(21)20-28(38)32(46)42-29(14-8-16-41-36(39)40)33(47)44-31(34(48)43-30(35(49)50)13-5-6-15-37)19-24-11-7-10-23-9-3-4-12-26(23)24/h3-4,7,9-12,17-18,28-31,45H,5-6,8,13-16,19-20,37-38H2,1-2H3,(H,42,46)(H,43,48)(H,44,47)(H,49,50)(H4,39,40,41)/t28-,29+,30-,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.109 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50070386
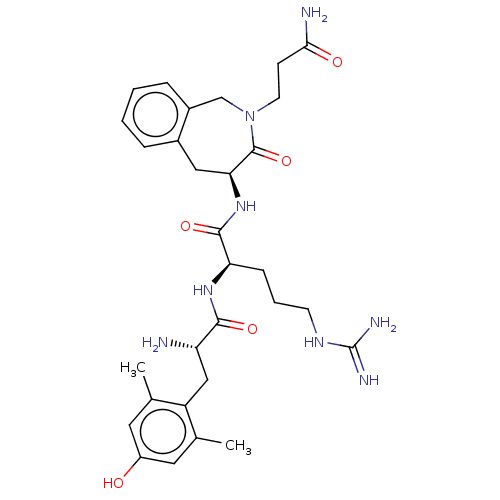
(CHEMBL3408737)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(N)=O)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C30H42N8O5/c1-17-12-21(39)13-18(2)22(17)15-23(31)27(41)36-24(8-5-10-35-30(33)34)28(42)37-25-14-19-6-3-4-7-20(19)16-38(29(25)43)11-9-26(32)40/h3-4,6-7,12-13,23-25,39H,5,8-11,14-16,31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H4,33,34,35)/t23-,24+,25-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from human mu opioid receptor expressed in HEK293 cell membrane incubated for 1 hr by TopCount scintillation counti... |
J Med Chem 59: 3777-92 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01976
BindingDB Entry DOI: 10.7270/Q2T72KC6 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM86667

(HS464)Show SMILES CCO[C@]12Cc3c(nc4ccccc34)C3Oc4c5c(CC1N(CC1CC1)CCC235)ccc4O |r,TLB:22:21:18.19.17:3,16:17:3:21.26.27,THB:2:3:18.19.17:21.26.27| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
I.G.B.M.C.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 313: 410-21 (2005)
Article DOI: 10.1124/jpet.104.077321
BindingDB Entry DOI: 10.7270/Q2XD107N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50010488

(CHEMBL3264361)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](COCc1cn(nn1)[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O)C(N)=O |r| Show InChI InChI=1S/C38H55N11O11/c1-19-11-23(51)12-20(2)24(19)14-25(39)34(56)44-26(9-6-10-43-38(41)42)35(57)45-27(13-21-7-4-3-5-8-21)36(58)46-28(33(40)55)18-59-17-22-15-49(48-47-22)37-32(54)31(53)30(52)29(16-50)60-37/h3-5,7-8,11-12,15,25-32,37,50-54H,6,9-10,13-14,16-18,39H2,1-2H3,(H2,40,55)(H,44,56)(H,45,57)(H,46,58)(H4,41,42,43)/t25-,26+,27-,28-,29+,30-,31-,32+,37+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.112 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes |
ACS Med Chem Lett 5: 352-357 (2014)
Article DOI: 10.1021/ml4004765
BindingDB Entry DOI: 10.7270/Q2VM4DSC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50001157
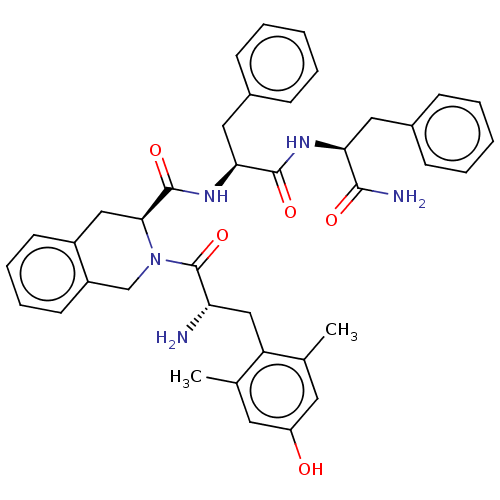
(CHEMBL538700)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H43N5O5/c1-24-17-30(45)18-25(2)31(24)22-32(40)39(49)44-23-29-16-10-9-15-28(29)21-35(44)38(48)43-34(20-27-13-7-4-8-14-27)37(47)42-33(36(41)46)19-26-11-5-3-6-12-26/h3-18,32-35,45H,19-23,40H2,1-2H3,(H2,41,46)(H,42,47)(H,43,48)/t32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.118 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from delta opioid receptor in rat brain membranes |
Bioorg Med Chem Lett 23: 5082-5 (2013)
Article DOI: 10.1016/j.bmcl.2013.07.036
BindingDB Entry DOI: 10.7270/Q2RN3BSK |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50001157

(CHEMBL538700)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H43N5O5/c1-24-17-30(45)18-25(2)31(24)22-32(40)39(49)44-23-29-16-10-9-15-28(29)21-35(44)38(48)43-34(20-27-13-7-4-8-14-27)37(47)42-33(36(41)46)19-26-11-5-3-6-12-26/h3-18,32-35,45H,19-23,40H2,1-2H3,(H2,41,46)(H,42,47)(H,43,48)/t32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Binding affinity for Opioid receptor delta 1 determined by displacing [3H]-DSLET from rat brain membrane binding sites |
J Med Chem 42: 3520-6 (1999)
Article DOI: 10.1021/jm980724+
BindingDB Entry DOI: 10.7270/Q2SJ1JT1 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM86661
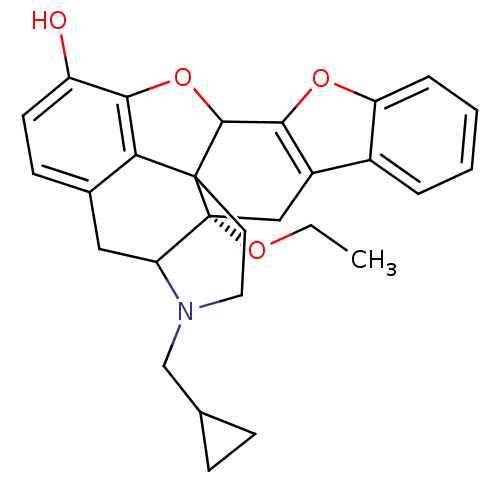
(HS510A)Show SMILES CCO[C@]12Cc3c(oc4ccccc34)C3Oc4c5c(CC1N(CC1CC1)CCC235)ccc4O |r,TLB:22:21:18.19.17:3,16:17:3:21.26.27,THB:2:3:18.19.17:21.26.27| Show InChI InChI=1S/C28H29NO4/c1-2-31-28-14-19-18-5-3-4-6-21(18)32-24(19)26-27(28)11-12-29(15-16-7-8-16)22(28)13-17-9-10-20(30)25(33-26)23(17)27/h3-6,9-10,16,22,26,30H,2,7-8,11-15H2,1H3/t22?,26?,27?,28-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
I.G.B.M.C.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 313: 410-21 (2005)
Article DOI: 10.1124/jpet.104.077321
BindingDB Entry DOI: 10.7270/Q2XD107N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50198755
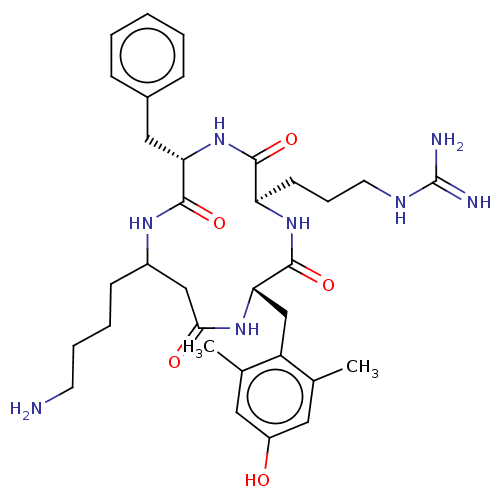
(CHEMBL3979449)Show SMILES Cc1cc(O)cc(C)c1C[C@@H]1NC(=O)CC(CCCCN)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CCCNC(N)=N)NC1=O |r| Show InChI InChI=1S/C33H48N8O5/c1-20-15-24(42)16-21(2)25(20)19-28-32(46)40-26(12-8-14-37-33(35)36)30(44)41-27(17-22-9-4-3-5-10-22)31(45)38-23(11-6-7-13-34)18-29(43)39-28/h3-5,9-10,15-16,23,26-28,42H,6-8,11-14,17-19,34H2,1-2H3,(H,38,45)(H,39,43)(H,40,46)(H,41,44)(H4,35,36,37)/t23?,26-,27+,28+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.124 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from MOR in rat brain membrane measured after 2 hrs |
J Med Chem 59: 9243-9254 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01200
BindingDB Entry DOI: 10.7270/Q23B623F |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070381
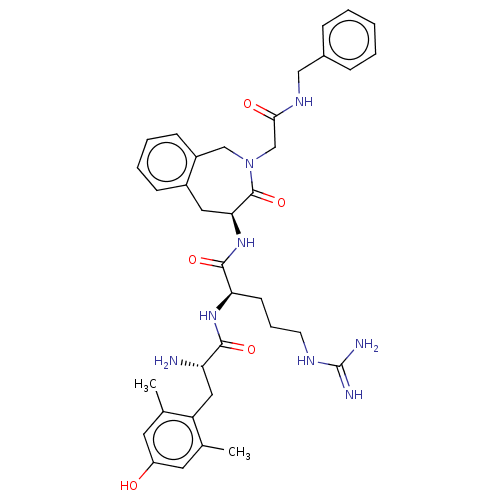
(CHEMBL3408730)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)NCc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C36H46N8O5/c1-22-15-27(45)16-23(2)28(22)18-29(37)33(47)42-30(13-8-14-40-36(38)39)34(48)43-31-17-25-11-6-7-12-26(25)20-44(35(31)49)21-32(46)41-19-24-9-4-3-5-10-24/h3-7,9-12,15-16,29-31,45H,8,13-14,17-21,37H2,1-2H3,(H,41,46)(H,42,47)(H,43,48)(H4,38,39,40)/t29-,30+,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM85731
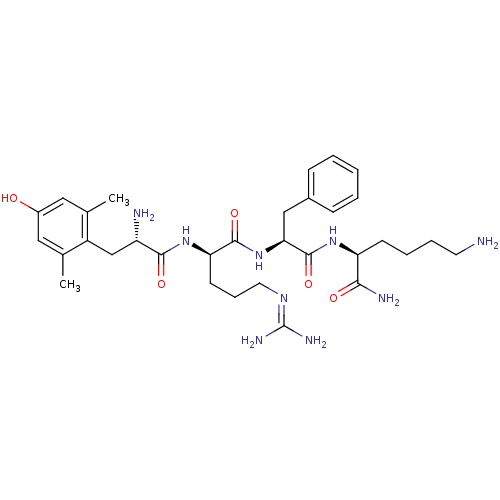
([Dmt1]DALDA)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by PDSP Ki Database
| |
Eur J Med Chem 35: 895-901 (2000)
Article DOI: 10.1016/s0223-5234(00)01171-5
BindingDB Entry DOI: 10.7270/Q23X856N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50010483
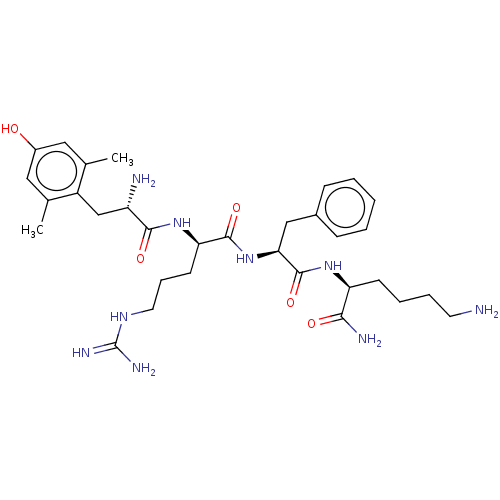
(CHEMBL2181202)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(N)=O |r| Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu-type opioid receptor in rat brain membranes incubated for 2 hrs |
Bioorg Med Chem Lett 26: 3629-31 (2016)
Article DOI: 10.1016/j.bmcl.2016.06.003
BindingDB Entry DOI: 10.7270/Q2ZW1NVQ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM85731

([Dmt1]DALDA)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008841
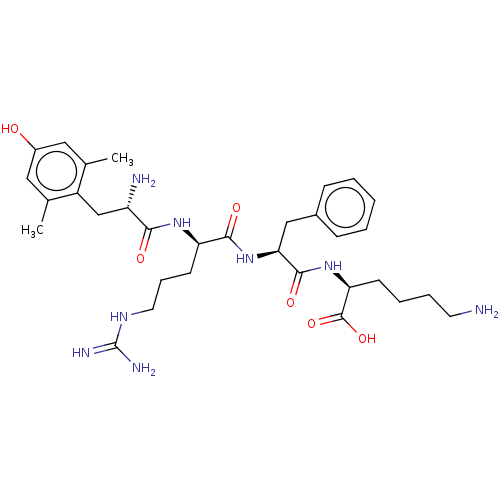
(CHEMBL3236678)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C32H48N8O6/c1-19-15-22(41)16-20(2)23(19)18-24(34)28(42)38-25(12-8-14-37-32(35)36)29(43)40-27(17-21-9-4-3-5-10-21)30(44)39-26(31(45)46)11-6-7-13-33/h3-5,9-10,15-16,24-27,41H,6-8,11-14,17-18,33-34H2,1-2H3,(H,38,42)(H,39,44)(H,40,43)(H,45,46)(H4,35,36,37)/t24-,25+,26-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM85731

([Dmt1]DALDA)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 55: 9549-61 (2012)
Article DOI: 10.1021/jm3008079
BindingDB Entry DOI: 10.7270/Q2TM7C82 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50198757

(CHEMBL363142)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24?,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from MOR in rat brain membrane measured after 2 hrs |
J Med Chem 59: 9243-9254 (2016)
Article DOI: 10.1021/acs.jmedchem.6b01200
BindingDB Entry DOI: 10.7270/Q23B623F |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM85731

([Dmt1]DALDA)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Binding affinity to mu opioid receptor |
J Med Chem 55: 9549-61 (2012)
Article DOI: 10.1021/jm3008079
BindingDB Entry DOI: 10.7270/Q2TM7C82 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50010483

(CHEMBL2181202)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(N)=O |r| Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes |
ACS Med Chem Lett 5: 352-357 (2014)
Article DOI: 10.1021/ml4004765
BindingDB Entry DOI: 10.7270/Q2VM4DSC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM86416

(Dmt-d-Arg-Phe-A2pr-NH2)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C29H43N9O5/c1-16-11-19(39)12-17(2)20(16)14-21(31)26(41)36-22(9-6-10-35-29(33)34)27(42)37-23(13-18-7-4-3-5-8-18)28(43)38-24(15-30)25(32)40/h3-5,7-8,11-12,21-24,39H,6,9-10,13-15,30-31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H,38,43)(H4,33,34,35)/t21-,22+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.145 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50493134

(CHEMBL3038184)Show SMILES Cc1cc(O)cc(C)c1C[C@H](NC(N)=N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C40H45N7O5/c1-24-17-30(48)18-25(2)31(24)22-34(46-40(42)43)39(52)47-23-29-16-10-9-15-28(29)21-35(47)38(51)45-33(20-27-13-7-4-8-14-27)37(50)44-32(36(41)49)19-26-11-5-3-6-12-26/h3-18,32-35,48H,19-23H2,1-2H3,(H2,41,49)(H,44,50)(H,45,51)(H4,42,43,46)/t32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.146 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from delta opioid receptor in rat brain membranes |
Bioorg Med Chem Lett 23: 5082-5 (2013)
Article DOI: 10.1016/j.bmcl.2013.07.036
BindingDB Entry DOI: 10.7270/Q2RN3BSK |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50493138
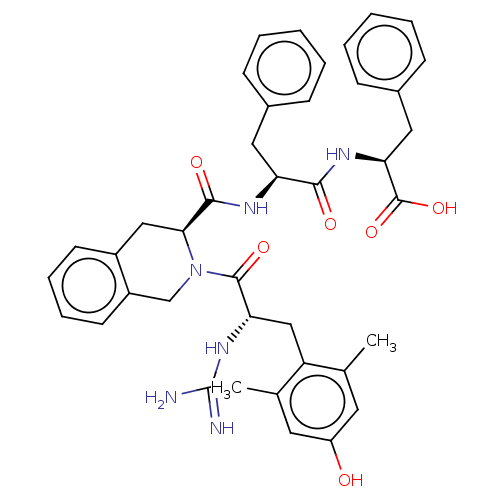
(CHEMBL3038186)Show SMILES Cc1cc(O)cc(C)c1C[C@H](NC(N)=N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(O)=O |r| Show InChI InChI=1S/C40H44N6O6/c1-24-17-30(47)18-25(2)31(24)22-33(45-40(41)42)38(50)46-23-29-16-10-9-15-28(29)21-35(46)37(49)43-32(19-26-11-5-3-6-12-26)36(48)44-34(39(51)52)20-27-13-7-4-8-14-27/h3-18,32-35,47H,19-23H2,1-2H3,(H,43,49)(H,44,48)(H,51,52)(H4,41,42,45)/t32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.146 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from delta opioid receptor in rat brain membranes |
Bioorg Med Chem Lett 23: 5082-5 (2013)
Article DOI: 10.1016/j.bmcl.2013.07.036
BindingDB Entry DOI: 10.7270/Q2RN3BSK |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330
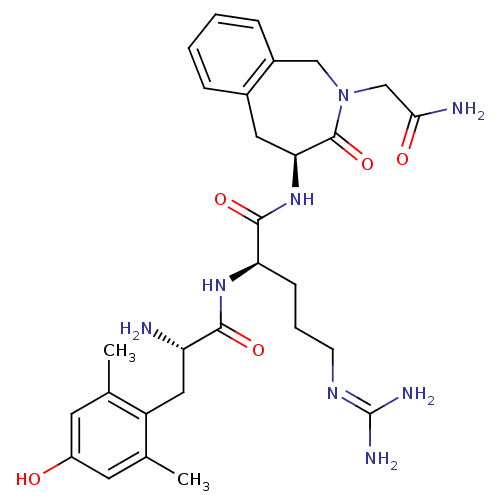
((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 54: 2467-76 (2011)
Article DOI: 10.1021/jm1016285
BindingDB Entry DOI: 10.7270/Q2416XD1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 55: 9549-61 (2012)
Article DOI: 10.1021/jm3008079
BindingDB Entry DOI: 10.7270/Q2TM7C82 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Binding affinity to Mu-type opioid receptor (unknown origin) |
ACS Med Chem Lett 8: 1177-1182 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00347
BindingDB Entry DOI: 10.7270/Q2251MRN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50090003
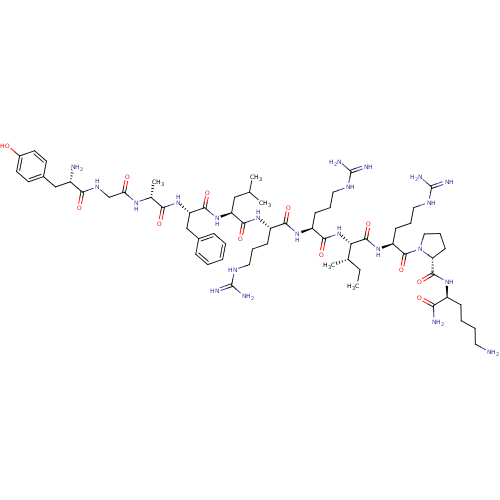
(CHEMBL405190 | Tyr-Gly-[D-Ala]-Phe-Leu-Arg-Arg-Ile...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](C)NC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C64H106N22O12/c1-6-37(4)51(60(97)82-46(21-14-30-76-64(72)73)61(98)86-31-15-22-49(86)59(96)79-43(52(67)89)18-10-11-27-65)85-56(93)45(20-13-29-75-63(70)71)80-55(92)44(19-12-28-74-62(68)69)81-57(94)47(32-36(2)3)84-58(95)48(34-39-16-8-7-9-17-39)83-53(90)38(5)78-50(88)35-77-54(91)42(66)33-40-23-25-41(87)26-24-40/h7-9,16-17,23-26,36-38,42-49,51,87H,6,10-15,18-22,27-35,65-66H2,1-5H3,(H2,67,89)(H,77,91)(H,78,88)(H,79,96)(H,80,92)(H,81,94)(H,82,97)(H,83,90)(H,84,95)(H,85,93)(H4,68,69,74)(H4,70,71,75)(H4,72,73,76)/t37-,38+,42-,43-,44-,45-,46-,47-,48-,49+,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Human Opioid receptor kappa 1 mediated stimulation of [35S]- GTPgammaS binding in CHO cells (Agonist potency). |
J Med Chem 43: 2698-702 (2000)
BindingDB Entry DOI: 10.7270/Q25D8SJ1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM85736
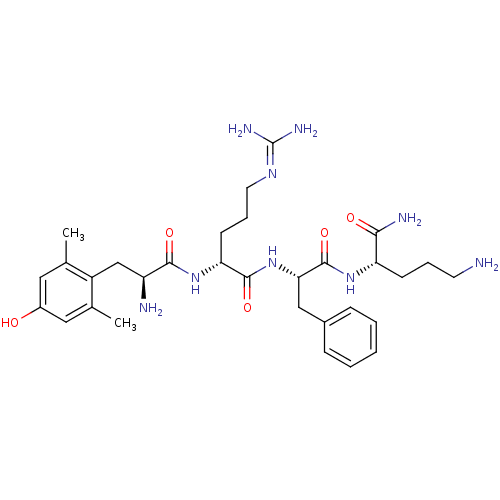
(Dmt-d-Arg-Phe-Orn-NH2 | H-Dmt-D-Arg-Phe-Orn-NH2)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C31H47N9O5/c1-18-14-21(41)15-19(2)22(18)17-23(33)28(43)39-25(11-7-13-37-31(35)36)29(44)40-26(16-20-8-4-3-5-9-20)30(45)38-24(27(34)42)10-6-12-32/h3-5,8-9,14-15,23-26,41H,6-7,10-13,16-17,32-33H2,1-2H3,(H2,34,42)(H,38,45)(H,39,43)(H,40,44)(H4,35,36,37)/t23-,24-,25+,26-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Clinical Research Institute of Montreal
Curated by PDSP Ki Database
| |
Eur J Med Chem 35: 895-901 (2000)
Article DOI: 10.1016/s0223-5234(00)01171-5
BindingDB Entry DOI: 10.7270/Q23X856N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM85736

(Dmt-d-Arg-Phe-Orn-NH2 | H-Dmt-D-Arg-Phe-Orn-NH2)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C31H47N9O5/c1-18-14-21(41)15-19(2)22(18)17-23(33)28(43)39-25(11-7-13-37-31(35)36)29(44)40-26(16-20-8-4-3-5-9-20)30(45)38-24(27(34)42)10-6-12-32/h3-5,8-9,14-15,23-26,41H,6-7,10-13,16-17,32-33H2,1-2H3,(H2,34,42)(H,38,45)(H,39,43)(H,40,44)(H4,35,36,37)/t23-,24-,25+,26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008836
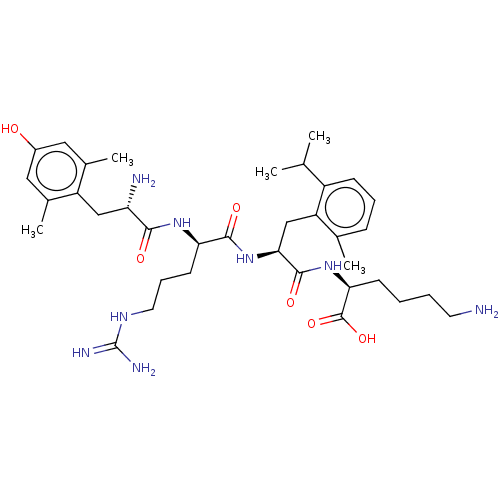
(CHEMBL3236673)Show SMILES CC(C)c1cccc(C)c1C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C36H56N8O6/c1-20(2)25-11-8-10-21(3)27(25)19-31(34(48)43-30(35(49)50)12-6-7-14-37)44-33(47)29(13-9-15-41-36(39)40)42-32(46)28(38)18-26-22(4)16-24(45)17-23(26)5/h8,10-11,16-17,20,28-31,45H,6-7,9,12-15,18-19,37-38H2,1-5H3,(H,42,46)(H,43,48)(H,44,47)(H,49,50)(H4,39,40,41)/t28-,29+,30-,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.155 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50008835
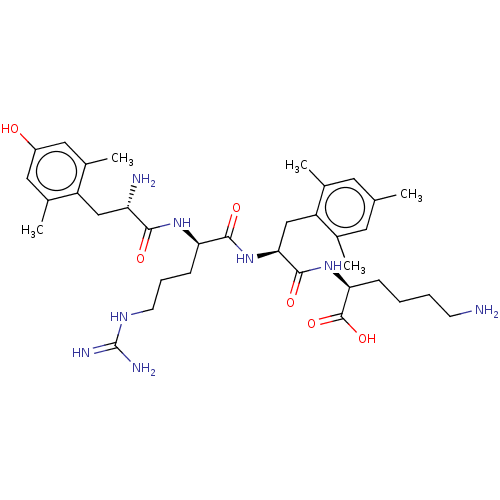
(CHEMBL3236672)Show SMILES Cc1cc(C)c(C[C@H](NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc2c(C)cc(O)cc2C)C(=O)N[C@@H](CCCCN)C(O)=O)c(C)c1 |r| Show InChI InChI=1S/C35H54N8O6/c1-19-13-20(2)26(21(3)14-19)18-30(33(47)42-29(34(48)49)9-6-7-11-36)43-32(46)28(10-8-12-40-35(38)39)41-31(45)27(37)17-25-22(4)15-24(44)16-23(25)5/h13-16,27-30,44H,6-12,17-18,36-37H2,1-5H3,(H,41,45)(H,42,47)(H,43,46)(H,48,49)(H4,38,39,40)/t27-,28+,29-,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
Bioorg Med Chem 22: 2333-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.011
BindingDB Entry DOI: 10.7270/Q20P11JG |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM85736

(Dmt-d-Arg-Phe-Orn-NH2 | H-Dmt-D-Arg-Phe-Orn-NH2)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C31H47N9O5/c1-18-14-21(41)15-19(2)22(18)17-23(33)28(43)39-25(11-7-13-37-31(35)36)29(44)40-26(16-20-8-4-3-5-9-20)30(45)38-24(27(34)42)10-6-12-32/h3-5,8-9,14-15,23-26,41H,6-7,10-13,16-17,32-33H2,1-2H3,(H2,34,42)(H,38,45)(H,39,43)(H,40,44)(H4,35,36,37)/t23-,24-,25+,26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.161 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM85731

([Dmt1]DALDA)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#7])=O Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 307: 947-54 (2003)
Article DOI: 10.1124/jpet.103.054775
BindingDB Entry DOI: 10.7270/Q24748FX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data