 Found 181 hits with Last Name = 'shah' and Initial = 'gp'
Found 181 hits with Last Name = 'shah' and Initial = 'gp' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228930
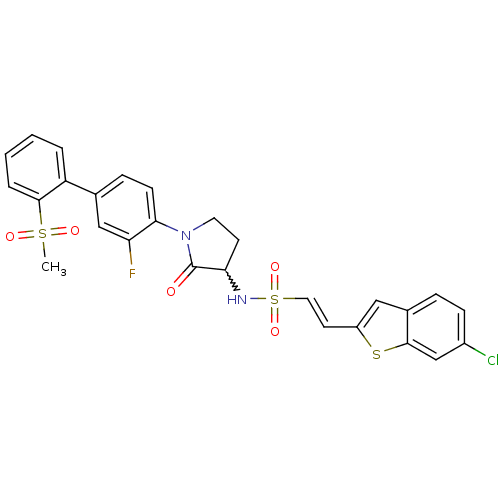
((E)-2-(6-chloro-benzo[b]thiophen-2-yl)-ethenesulfo...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(N2CCC(NS(=O)(=O)\C=C\c3cc4ccc(Cl)cc4s3)C2=O)c(F)c1 |w:17.18| Show InChI InChI=1S/C27H22ClFN2O5S3/c1-38(33,34)26-5-3-2-4-21(26)17-7-9-24(22(29)15-17)31-12-10-23(27(31)32)30-39(35,36)13-11-20-14-18-6-8-19(28)16-25(18)37-20/h2-9,11,13-16,23,30H,10,12H2,1H3/b13-11+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374259
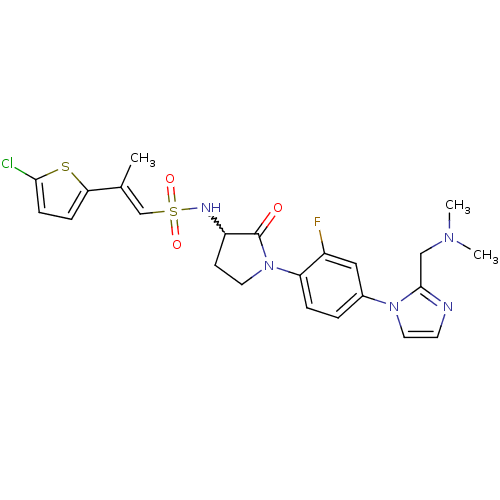
(CHEMBL257741)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CCC(NS(=O)(=O)\C=C(/C)c3ccc(Cl)s3)C2=O)c(F)c1 |w:16.17| Show InChI InChI=1S/C23H25ClFN5O3S2/c1-15(20-6-7-21(24)34-20)14-35(32,33)27-18-8-10-30(23(18)31)19-5-4-16(12-17(19)25)29-11-9-26-22(29)13-28(2)3/h4-7,9,11-12,14,18,27H,8,10,13H2,1-3H3/b15-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228676
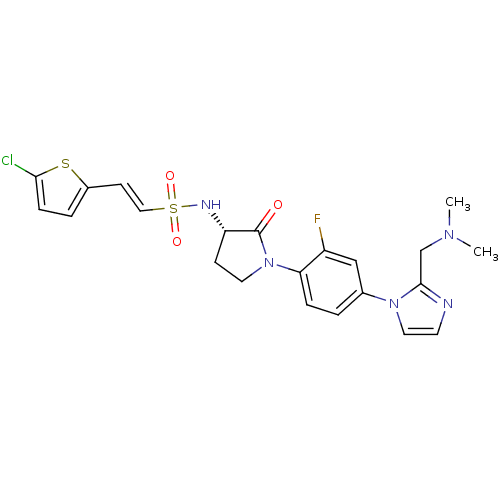
((S)-2-(5-chlorothiophen-2-yl)-N-(1-(4-(2-((dimethy...)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 Show InChI InChI=1S/C22H23ClFN5O3S2/c1-27(2)14-21-25-9-11-28(21)15-3-5-19(17(24)13-15)29-10-7-18(22(29)30)26-34(31,32)12-8-16-4-6-20(23)33-16/h3-6,8-9,11-13,18,26H,7,10,14H2,1-2H3/b12-8+/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228950
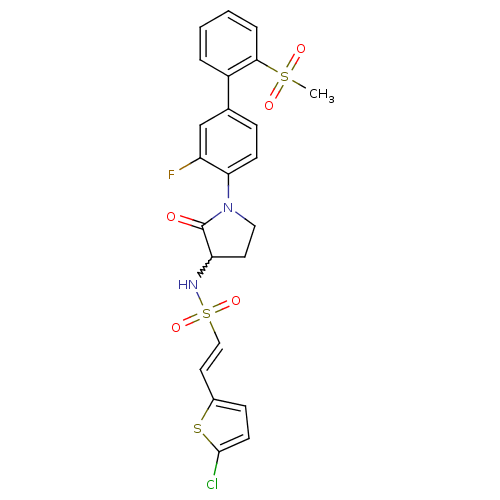
((E)-2-(5-chloro-thiophen-2-yl)-ethenesulfonic acid...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(N2CCC(NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |w:17.18| Show InChI InChI=1S/C23H20ClFN2O5S3/c1-34(29,30)21-5-3-2-4-17(21)15-6-8-20(18(25)14-15)27-12-10-19(23(27)28)26-35(31,32)13-11-16-7-9-22(24)33-16/h2-9,11,13-14,19,26H,10,12H2,1H3/b13-11+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306153
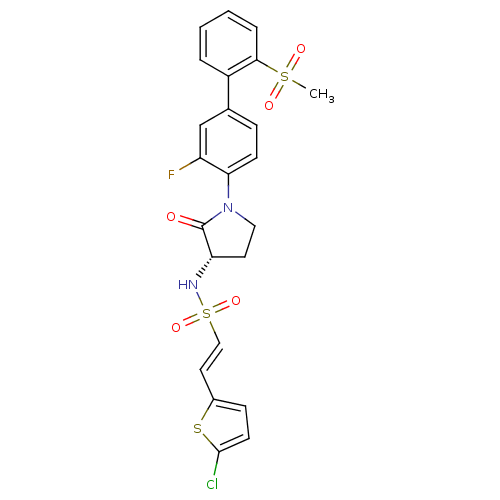
((S)-2-(5-chlorothiophen-2-yl)-N-(1-(3-fluoro-2'-(m...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C23H20ClFN2O5S3/c1-34(29,30)21-5-3-2-4-17(21)15-6-8-20(18(25)14-15)27-12-10-19(23(27)28)26-35(31,32)13-11-16-7-9-22(24)33-16/h2-9,11,13-14,19,26H,10,12H2,1H3/b13-11+/t19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228676

((S)-2-(5-chlorothiophen-2-yl)-N-(1-(4-(2-((dimethy...)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 Show InChI InChI=1S/C22H23ClFN5O3S2/c1-27(2)14-21-25-9-11-28(21)15-3-5-19(17(24)13-15)29-10-7-18(22(29)30)26-34(31,32)12-8-16-4-6-20(23)33-16/h3-6,8-9,11-13,18,26H,7,10,14H2,1-2H3/b12-8+/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374262
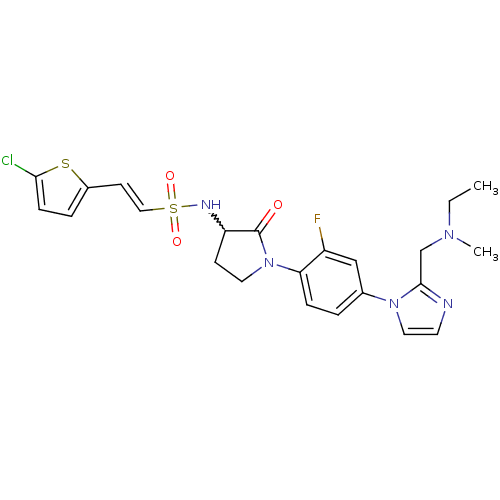
(CHEMBL272779)Show SMILES CCN(C)Cc1nccn1-c1ccc(N2CCC(NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |w:17.18| Show InChI InChI=1S/C23H25ClFN5O3S2/c1-3-28(2)15-22-26-10-12-29(22)16-4-6-20(18(25)14-16)30-11-8-19(23(30)31)27-35(32,33)13-9-17-5-7-21(24)34-17/h4-7,9-10,12-14,19,27H,3,8,11,15H2,1-2H3/b13-9+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228954
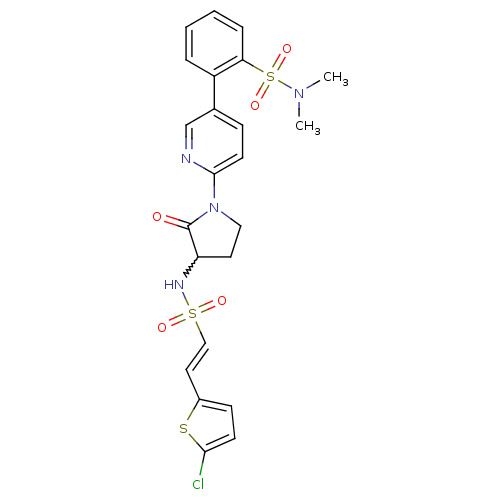
((E)-2-(6-(3-(2-(5-chlorothiophen-2-yl)vinylsulfona...)Show SMILES CN(C)S(=O)(=O)c1ccccc1-c1ccc(nc1)N1CCC(NS(=O)(=O)\C=C\c2ccc(Cl)s2)C1=O |w:21.23| Show InChI InChI=1S/C23H23ClN4O5S3/c1-27(2)36(32,33)20-6-4-3-5-18(20)16-7-10-22(25-15-16)28-13-11-19(23(28)29)26-35(30,31)14-12-17-8-9-21(24)34-17/h3-10,12,14-15,19,26H,11,13H2,1-2H3/b14-12+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374264

(CHEMBL257863)Show SMILES Fc1cc(ccc1N1CCC(NS(=O)(=O)\C=C\c2ccc(Cl)s2)C1=O)-n1ccnc1CN1CCCC1 |w:10.11| Show InChI InChI=1S/C24H25ClFN5O3S2/c25-22-6-4-18(35-22)8-14-36(33,34)28-20-7-12-31(24(20)32)21-5-3-17(15-19(21)26)30-13-9-27-23(30)16-29-10-1-2-11-29/h3-6,8-9,13-15,20,28H,1-2,7,10-12,16H2/b14-8+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374270
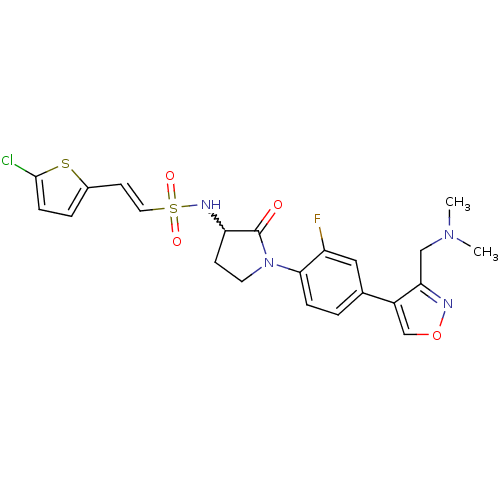
(CHEMBL271015)Show SMILES CN(C)Cc1nocc1-c1ccc(N2CCC(NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |w:16.17| Show InChI InChI=1S/C22H22ClFN4O4S2/c1-27(2)12-19-16(13-32-25-19)14-3-5-20(17(24)11-14)28-9-7-18(22(28)29)26-34(30,31)10-8-15-4-6-21(23)33-15/h3-6,8,10-11,13,18,26H,7,9,12H2,1-2H3/b10-8+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374263
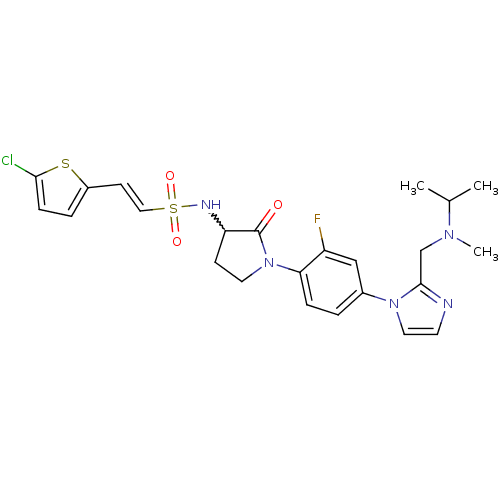
(CHEMBL437097)Show SMILES CC(C)N(C)Cc1nccn1-c1ccc(N2CCC(NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |w:18.19| Show InChI InChI=1S/C24H27ClFN5O3S2/c1-16(2)29(3)15-23-27-10-12-30(23)17-4-6-21(19(26)14-17)31-11-8-20(24(31)32)28-36(33,34)13-9-18-5-7-22(25)35-18/h4-7,9-10,12-14,16,20,28H,8,11,15H2,1-3H3/b13-9+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50328712
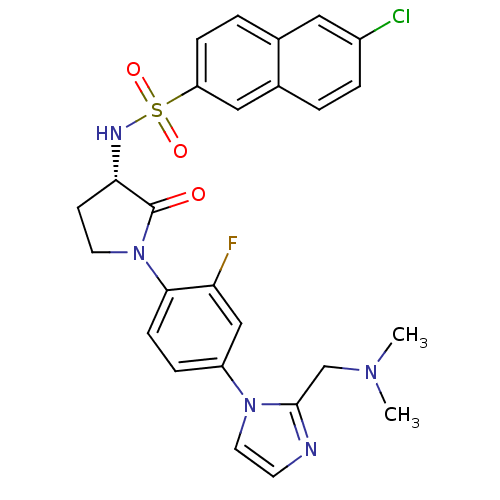
((S)-6-chloro-N-(1-(4-(2-((dimethylamino)methyl)-1H...)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C26H25ClFN5O3S/c1-31(2)16-25-29-10-12-32(25)20-6-8-24(22(28)15-20)33-11-9-23(26(33)34)30-37(35,36)21-7-4-17-13-19(27)5-3-18(17)14-21/h3-8,10,12-15,23,30H,9,11,16H2,1-2H3/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228940
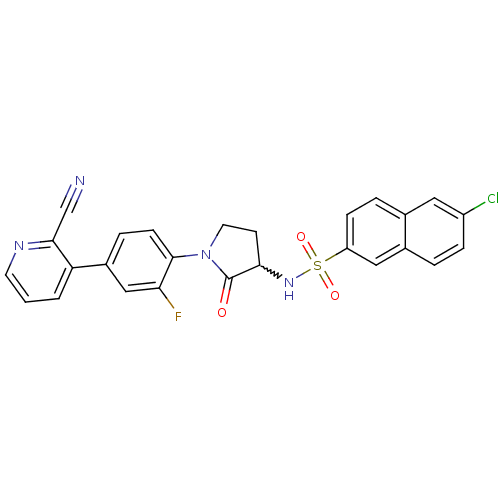
(6-chloro-N-(1-(4-(2-cyanopyridin-3-yl)-2-fluorophe...)Show SMILES Fc1cc(ccc1N1CCC(NS(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O)-c1cccnc1C#N |w:10.11| Show InChI InChI=1S/C26H18ClFN4O3S/c27-19-6-3-17-13-20(7-4-16(17)12-19)36(34,35)31-23-9-11-32(26(23)33)25-8-5-18(14-22(25)28)21-2-1-10-30-24(21)15-29/h1-8,10,12-14,23,31H,9,11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374274

(CHEMBL403111)Show SMILES CN(C)Cc1nccn1-c1ccc(N2CCC(NS(=O)(=O)c3cc4ccc(Cl)cc4s3)C2=O)c(F)c1 |w:16.17| Show InChI InChI=1S/C24H23ClFN5O3S2/c1-29(2)14-22-27-8-10-30(22)17-5-6-20(18(26)13-17)31-9-7-19(24(31)32)28-36(33,34)23-11-15-3-4-16(25)12-21(15)35-23/h3-6,8,10-13,19,28H,7,9,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306138
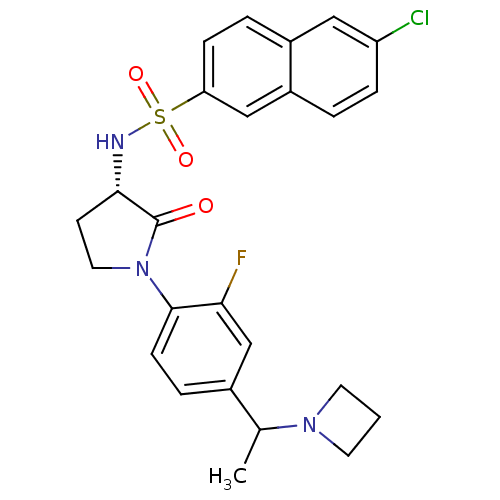
(CHEMBL604887 | N-((3S)-1-(4-(1-(azetidin-1-yl)ethy...)Show SMILES CC(N1CCC1)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C25H25ClFN3O3S/c1-16(29-10-2-11-29)17-5-8-24(22(27)15-17)30-12-9-23(25(30)31)28-34(32,33)21-7-4-18-13-20(26)6-3-19(18)14-21/h3-8,13-16,23,28H,2,9-12H2,1H3/t16?,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306146
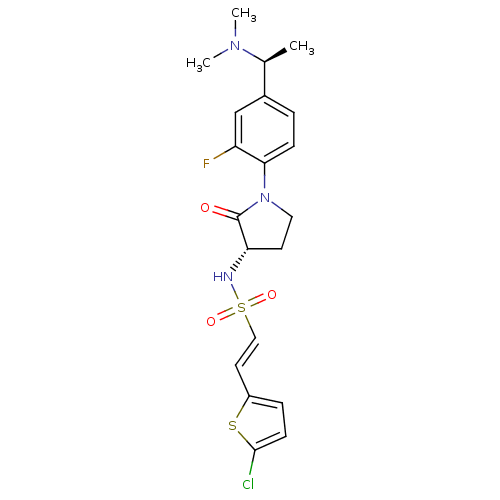
((E)-2-(5-CHLOROTHIOPHEN-2-YL)-N-[(3S)-1-{4-[(1S)-1...)Show SMILES C[C@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C20H23ClFN3O3S2/c1-13(24(2)3)14-4-6-18(16(22)12-14)25-10-8-17(20(25)26)23-30(27,28)11-9-15-5-7-19(21)29-15/h4-7,9,11-13,17,23H,8,10H2,1-3H3/b11-9+/t13-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306142
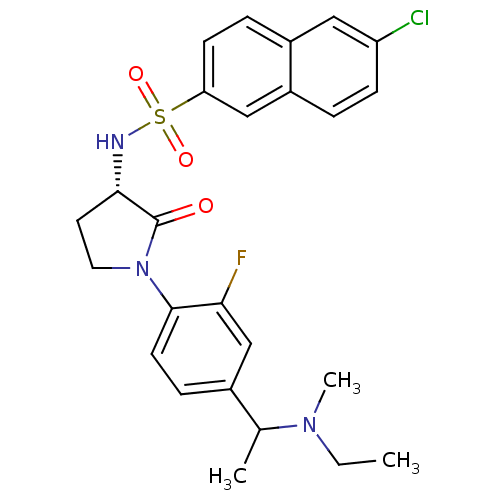
(6-chloro-N-((3S)-1-(4-(1-(ethyl(methyl)amino)ethyl...)Show SMILES CCN(C)C(C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C25H27ClFN3O3S/c1-4-29(3)16(2)17-7-10-24(22(27)15-17)30-12-11-23(25(30)31)28-34(32,33)21-9-6-18-13-20(26)8-5-19(18)14-21/h5-10,13-16,23,28H,4,11-12H2,1-3H3/t16?,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306143
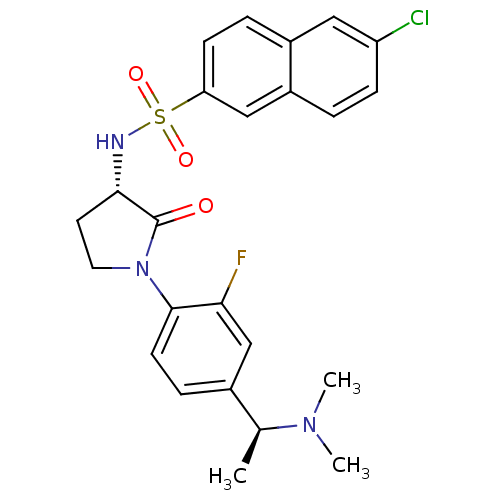
(6-chloro-N-((S)-1-(4-((S)-1-(dimethylamino)ethyl)-...)Show SMILES C[C@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C24H25ClFN3O3S/c1-15(28(2)3)16-6-9-23(21(26)14-16)29-11-10-22(24(29)30)27-33(31,32)20-8-5-17-12-19(25)7-4-18(17)13-20/h4-9,12-15,22,27H,10-11H2,1-3H3/t15-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12539
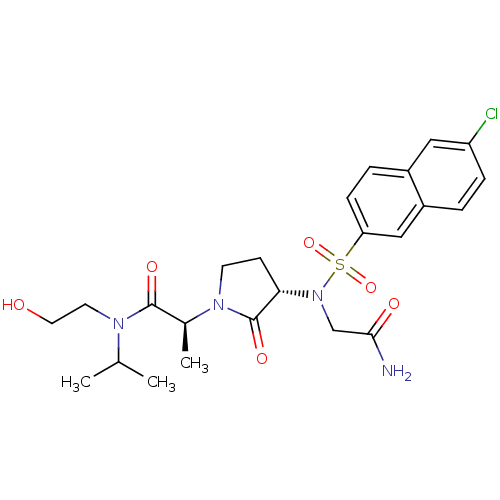
((2S)-2-[(3S)-3-{2-[(6-chloronaphthalene-2-)sulfona...)Show SMILES CC(C)N(CCO)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C24H31ClN4O6S/c1-15(2)27(10-11-30)23(32)16(3)28-9-8-21(24(28)33)29(14-22(26)31)36(34,35)20-7-5-17-12-19(25)6-4-18(17)13-20/h4-7,12-13,15-16,21,30H,8-11,14H2,1-3H3,(H2,26,31)/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12569
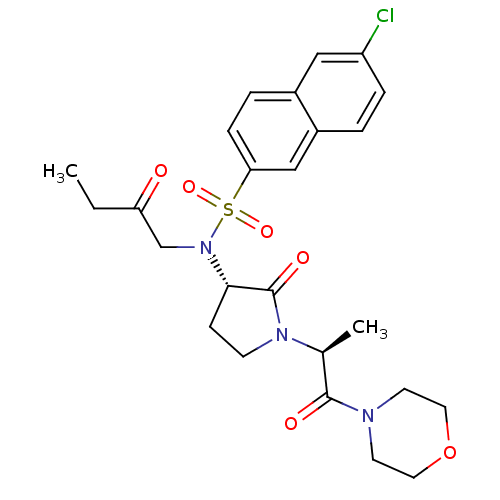
(GTC000006A | N-(6-chloronaphthalen-2-yl)-N'-[(3S)-...)Show SMILES CCC(=O)CN([C@H]1CCN([C@@H](C)C(=O)N2CCOCC2)C1=O)S(=O)(=O)c1ccc2cc(Cl)ccc2c1 |r| Show InChI InChI=1S/C25H30ClN3O6S/c1-3-21(30)16-29(36(33,34)22-7-5-18-14-20(26)6-4-19(18)15-22)23-8-9-28(25(23)32)17(2)24(31)27-10-12-35-13-11-27/h4-7,14-15,17,23H,3,8-13,16H2,1-2H3/t17-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -51.4 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 3784-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.04.053
BindingDB Entry DOI: 10.7270/Q2416V9V |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12528
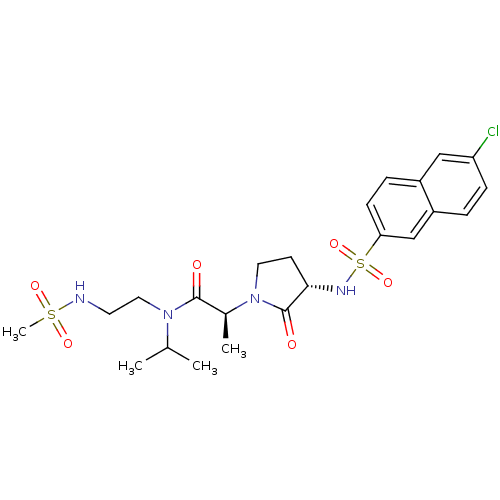
((2S)-2-[(3S)-3-[(6-chloronaphthalene-2-)sulfonamid...)Show SMILES CC(C)N(CCNS(C)(=O)=O)C(=O)[C@H](C)N1CC[C@H](NS(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C23H31ClN4O6S2/c1-15(2)27(12-10-25-35(4,31)32)22(29)16(3)28-11-9-21(23(28)30)26-36(33,34)20-8-6-17-13-19(24)7-5-18(17)14-20/h5-8,13-16,21,25-26H,9-12H2,1-4H3/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12542
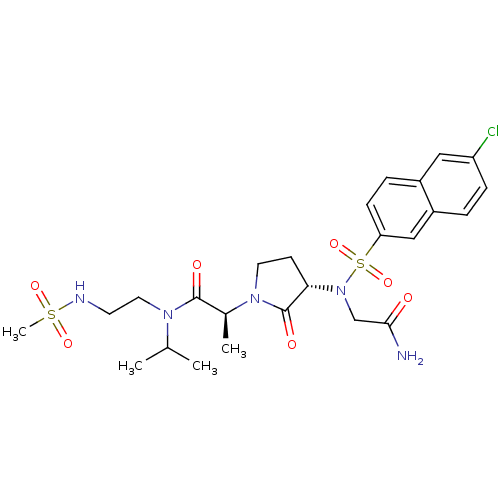
((2S)-2-[(3S)-3-{2-[(6-chloronaphthalene-2-)sulfona...)Show SMILES CC(C)N(CCNS(C)(=O)=O)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C25H34ClN5O7S2/c1-16(2)29(12-10-28-39(4,35)36)24(33)17(3)30-11-9-22(25(30)34)31(15-23(27)32)40(37,38)21-8-6-18-13-20(26)7-5-19(18)14-21/h5-8,13-14,16-17,22,28H,9-12,15H2,1-4H3,(H2,27,32)/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12543

((2S)-2-[(3S)-3-{2-[(6-chloronaphthalene-2-)sulfona...)Show SMILES CC(C)N(CCN1CCCCC1)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C29H40ClN5O5S/c1-20(2)33(16-15-32-12-5-4-6-13-32)28(37)21(3)34-14-11-26(29(34)38)35(19-27(31)36)41(39,40)25-10-8-22-17-24(30)9-7-23(22)18-25/h7-10,17-18,20-21,26H,4-6,11-16,19H2,1-3H3,(H2,31,36)/t21-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306140
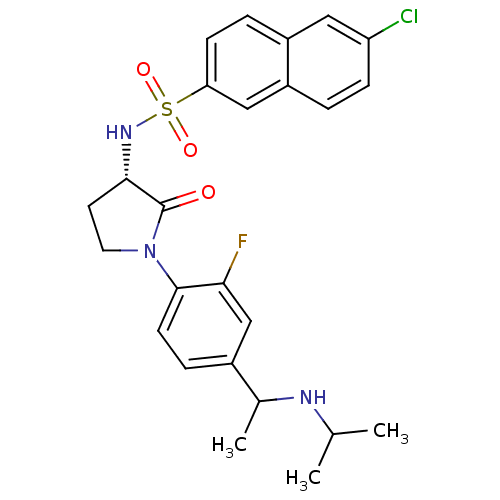
(6-chloro-N-((3S)-1-(2-fluoro-4-(1-(isopropylamino)...)Show SMILES CC(C)NC(C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C25H27ClFN3O3S/c1-15(2)28-16(3)17-6-9-24(22(27)14-17)30-11-10-23(25(30)31)29-34(32,33)21-8-5-18-12-20(26)7-4-19(18)13-21/h4-9,12-16,23,28-29H,10-11H2,1-3H3/t16?,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12544
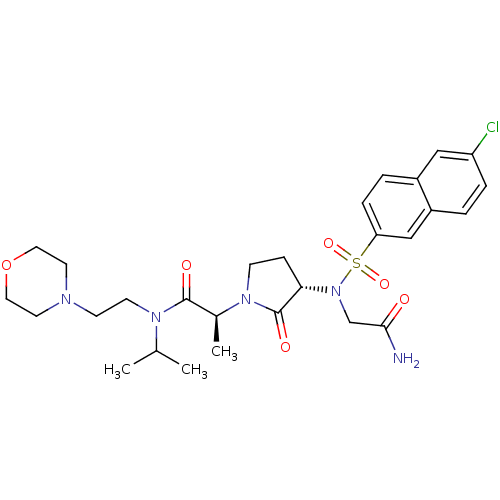
((2S)-2-[(3S)-3-{2-[(6-chloronaphthalene-2-)sulfona...)Show SMILES CC(C)N(CCN1CCOCC1)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C28H38ClN5O6S/c1-19(2)32(11-10-31-12-14-40-15-13-31)27(36)20(3)33-9-8-25(28(33)37)34(18-26(30)35)41(38,39)24-7-5-21-16-23(29)6-4-22(21)17-24/h4-7,16-17,19-20,25H,8-15,18H2,1-3H3,(H2,30,35)/t20-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12545
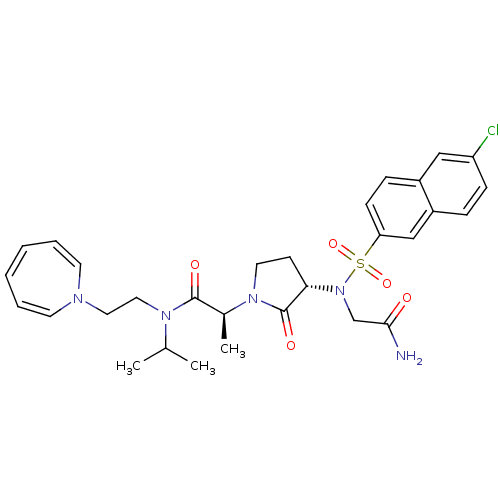
((2S)-N-[2-(1H-azepin-1-yl)ethyl]-2-[(3S)-3-{2-[(6-...)Show SMILES CC(C)N(CCN1C=CC=CC=C1)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r,c:7,9,11| Show InChI InChI=1S/C30H36ClN5O5S/c1-21(2)34(17-16-33-13-6-4-5-7-14-33)29(38)22(3)35-15-12-27(30(35)39)36(20-28(32)37)42(40,41)26-11-9-23-18-25(31)10-8-24(23)19-26/h4-11,13-14,18-19,21-22,27H,12,15-17,20H2,1-3H3,(H2,32,37)/t22-,27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
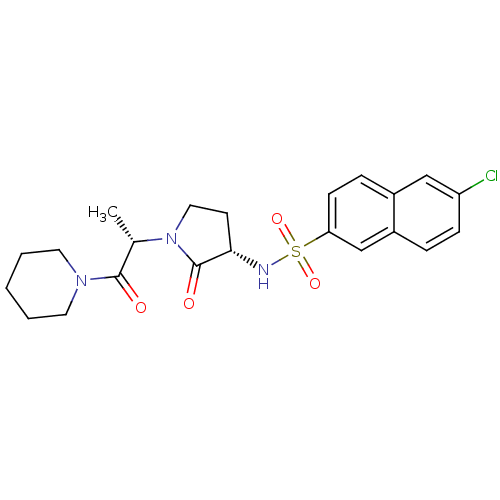
Coagulation factor X
(Homo sapiens (Human)) | BDBM12557
(6-chloro-N-[(3S)-2-oxo-1-[(2S)-1-oxo-1-(piperidin-...)Show SMILES C[C@H](N1CC[C@H](NS(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O)C(=O)N1CCCCC1 |r| Show InChI InChI=1S/C22H26ClN3O4S/c1-15(21(27)25-10-3-2-4-11-25)26-12-9-20(22(26)28)24-31(29,30)19-8-6-16-13-18(23)7-5-17(16)14-19/h5-8,13-15,20,24H,2-4,9-12H2,1H3/t15-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -51.4 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 3784-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.04.053
BindingDB Entry DOI: 10.7270/Q2416V9V |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306146

((E)-2-(5-CHLOROTHIOPHEN-2-YL)-N-[(3S)-1-{4-[(1S)-1...)Show SMILES C[C@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C20H23ClFN3O3S2/c1-13(24(2)3)14-4-6-18(16(22)12-14)25-10-8-17(20(25)26)23-30(27,28)11-9-15-5-7-19(21)29-15/h4-7,9,11-13,17,23H,8,10H2,1-3H3/b11-9+/t13-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12541

((2S)-N-[2-(carbamoylamino)ethyl]-2-[(3S)-3-{2-[(6-...)Show SMILES CC(C)N(CCNC(N)=O)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C25H33ClN6O6S/c1-15(2)30(11-9-29-25(28)36)23(34)16(3)31-10-8-21(24(31)35)32(14-22(27)33)39(37,38)20-7-5-17-12-19(26)6-4-18(17)13-20/h4-7,12-13,15-16,21H,8-11,14H2,1-3H3,(H2,27,33)(H3,28,29,36)/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12540

((2S)-N-(2-aminoethyl)-2-[(3S)-3-{2-[(6-chloronapht...)Show SMILES CC(C)N(CCN)C(=O)[C@H](C)N1CC[C@H](N(CC(N)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C24H32ClN5O5S/c1-15(2)28(11-9-26)23(32)16(3)29-10-8-21(24(29)33)30(14-22(27)31)36(34,35)20-7-5-17-12-19(25)6-4-18(17)13-20/h4-7,12-13,15-16,21H,8-11,14,26H2,1-3H3,(H2,27,31)/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228947
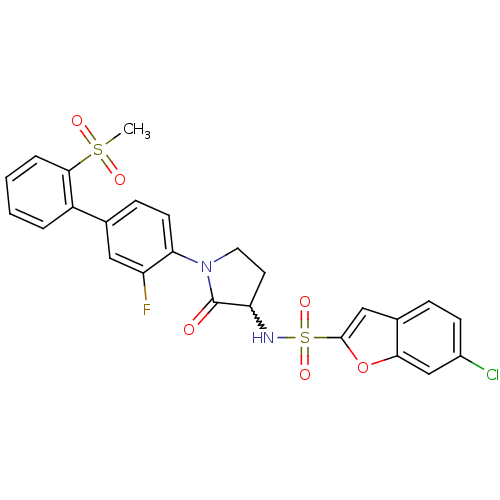
(6-chloro-benzofuran-2-sulfonic acid [1-(3-fluoro-2...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(N2CCC(NS(=O)(=O)c3cc4ccc(Cl)cc4o3)C2=O)c(F)c1 |w:17.18| Show InChI InChI=1S/C25H20ClFN2O6S2/c1-36(31,32)23-5-3-2-4-18(23)15-7-9-21(19(27)12-15)29-11-10-20(25(29)30)28-37(33,34)24-13-16-6-8-17(26)14-22(16)35-24/h2-9,12-14,20,28H,10-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228932
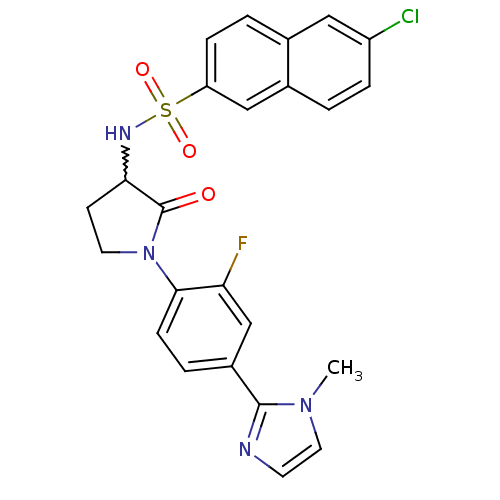
(6-chloro-N-(1-(2-fluoro-4-(1-methyl-1H-imidazol-2-...)Show SMILES Cn1ccnc1-c1ccc(N2CCC(NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |w:13.14| Show InChI InChI=1S/C24H20ClFN4O3S/c1-29-11-9-27-23(29)17-4-7-22(20(26)14-17)30-10-8-21(24(30)31)28-34(32,33)19-6-3-15-12-18(25)5-2-16(15)13-19/h2-7,9,11-14,21,28H,8,10H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50374265
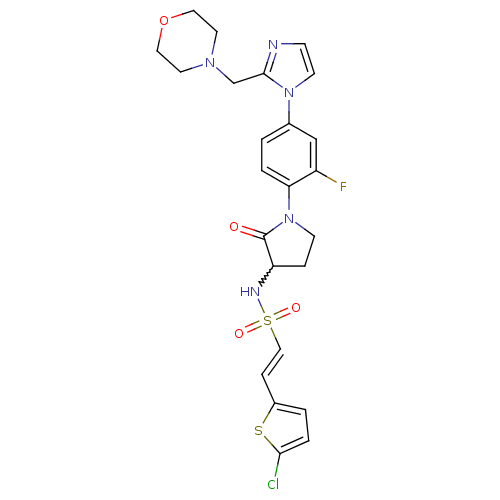
(CHEMBL257862)Show SMILES Fc1cc(ccc1N1CCC(NS(=O)(=O)\C=C\c2ccc(Cl)s2)C1=O)-n1ccnc1CN1CCOCC1 |w:10.11| Show InChI InChI=1S/C24H25ClFN5O4S2/c25-22-4-2-18(36-22)6-14-37(33,34)28-20-5-8-31(24(20)32)21-3-1-17(15-19(21)26)30-9-7-27-23(30)16-29-10-12-35-13-11-29/h1-4,6-7,9,14-15,20,28H,5,8,10-13,16H2/b14-6+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 18: 28-33 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.019
BindingDB Entry DOI: 10.7270/Q29024PS |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306148
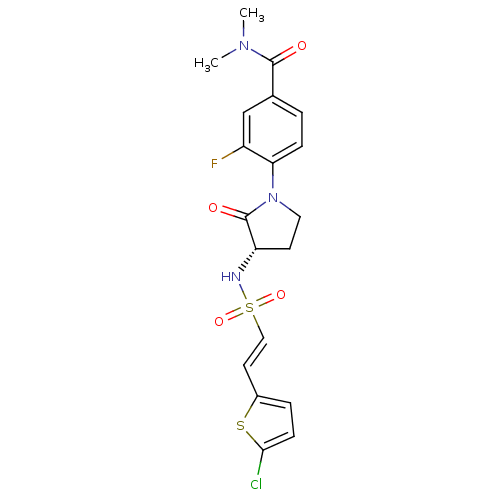
((S)-4-(3-(2-(5-chlorothiophen-2-yl)vinylsulfonamid...)Show SMILES CN(C)C(=O)c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C19H19ClFN3O4S2/c1-23(2)18(25)12-3-5-16(14(21)11-12)24-9-7-15(19(24)26)22-30(27,28)10-8-13-4-6-17(20)29-13/h3-6,8,10-11,15,22H,7,9H2,1-2H3/b10-8+/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228935
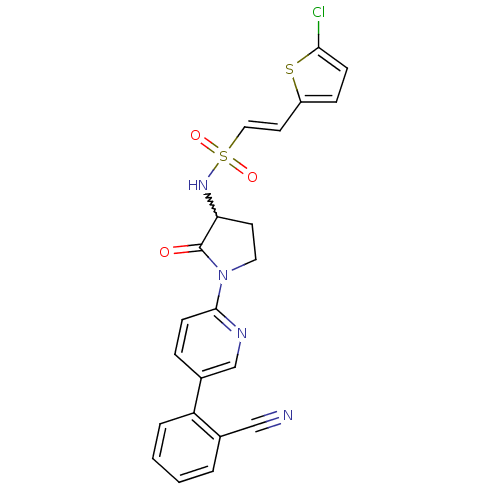
((E)-2-(5-chloro-thiophen-2-yl)-ethenesulfonic acid...)Show SMILES Clc1ccc(\C=C\S(=O)(=O)NC2CCN(C2=O)c2ccc(cn2)-c2ccccc2C#N)s1 |w:11.10| Show InChI InChI=1S/C22H17ClN4O3S2/c23-20-7-6-17(31-20)10-12-32(29,30)26-19-9-11-27(22(19)28)21-8-5-16(14-25-21)18-4-2-1-3-15(18)13-24/h1-8,10,12,14,19,26H,9,11H2/b12-10+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306147
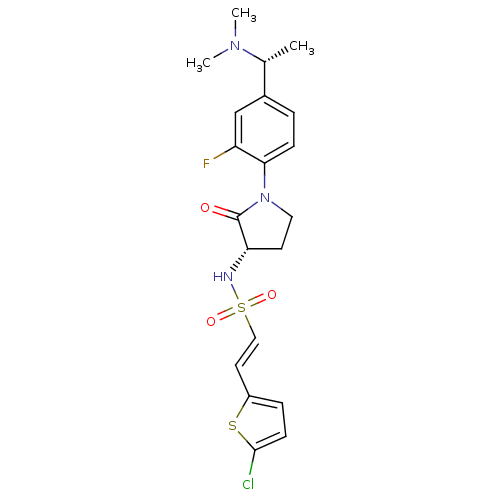
((E)-2-(5-CHLOROTHIOPHEN-2-YL)-N-[(3S)-1-{4-[(1R)-1...)Show SMILES C[C@@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C20H23ClFN3O3S2/c1-13(24(2)3)14-4-6-18(16(22)12-14)25-10-8-17(20(25)26)23-30(27,28)11-9-15-5-7-19(21)29-15/h4-7,9,11-13,17,23H,8,10H2,1-3H3/b11-9+/t13-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306144
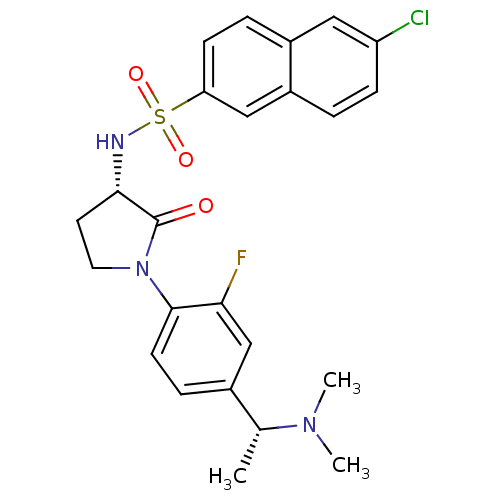
(6-chloro-N-((S)-1-(4-((R)-1-(dimethylamino)ethyl)-...)Show SMILES C[C@@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C24H25ClFN3O3S/c1-15(28(2)3)16-6-9-23(21(26)14-16)29-11-10-22(24(29)30)27-33(31,32)20-8-5-17-12-19(25)7-4-18(17)13-20/h4-9,12-15,22,27H,10-11H2,1-3H3/t15-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306143

(6-chloro-N-((S)-1-(4-((S)-1-(dimethylamino)ethyl)-...)Show SMILES C[C@H](N(C)C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C24H25ClFN3O3S/c1-15(28(2)3)16-6-9-23(21(26)14-16)29-11-10-22(24(29)30)27-33(31,32)20-8-5-17-12-19(25)7-4-18(17)13-20/h4-9,12-15,22,27H,10-11H2,1-3H3/t15-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306141
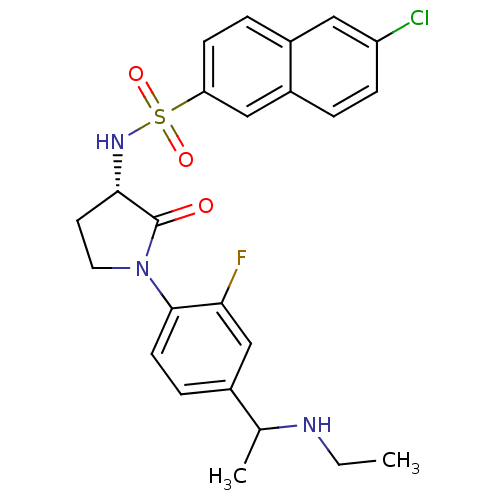
(6-chloro-N-((3S)-1-(4-(1-(ethylamino)ethyl)-2-fluo...)Show SMILES CCNC(C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C24H25ClFN3O3S/c1-3-27-15(2)16-6-9-23(21(26)14-16)29-11-10-22(24(29)30)28-33(31,32)20-8-5-17-12-19(25)7-4-18(17)13-20/h4-9,12-15,22,27-28H,3,10-11H2,1-2H3/t15?,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306137
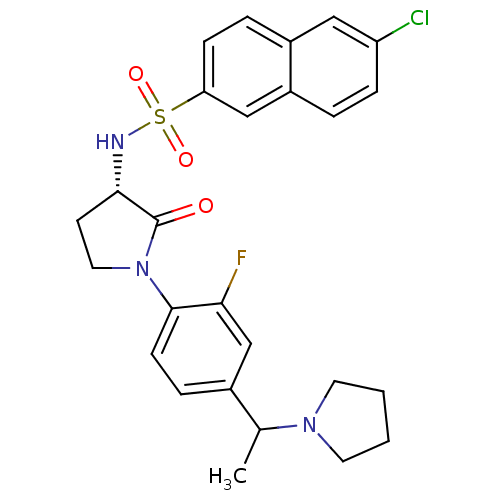
(6-chloro-N-((3S)-1-(2-fluoro-4-(1-(pyrrolidin-1-yl...)Show SMILES CC(N1CCCC1)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C26H27ClFN3O3S/c1-17(30-11-2-3-12-30)18-6-9-25(23(28)16-18)31-13-10-24(26(31)32)29-35(33,34)22-8-5-19-14-21(27)7-4-20(19)15-22/h4-9,14-17,24,29H,2-3,10-13H2,1H3/t17?,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306136
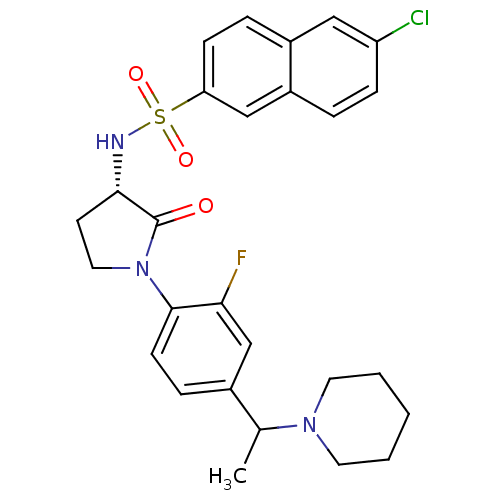
(6-chloro-N-((3S)-1-(2-fluoro-4-(1-(piperidin-1-yl)...)Show SMILES CC(N1CCCCC1)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C27H29ClFN3O3S/c1-18(31-12-3-2-4-13-31)19-7-10-26(24(29)17-19)32-14-11-25(27(32)33)30-36(34,35)23-9-6-20-15-22(28)8-5-21(20)16-23/h5-10,15-18,25,30H,2-4,11-14H2,1H3/t18?,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50306134
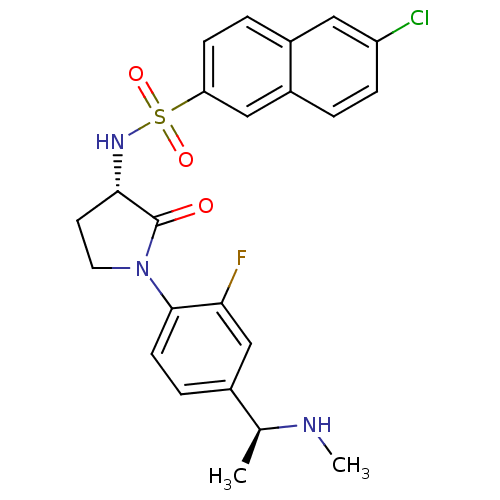
(6-chloro-N-((S)-1-(2-fluoro-4-((S)-1-(methylamino)...)Show SMILES CN[C@@H](C)c1ccc(N2CC[C@H](NS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C2=O)c(F)c1 |r| Show InChI InChI=1S/C23H23ClFN3O3S/c1-14(26-2)15-5-8-22(20(25)13-15)28-10-9-21(23(28)29)27-32(30,31)19-7-4-16-11-18(24)6-3-17(16)12-19/h3-8,11-14,21,26-27H,9-10H2,1-2H3/t14-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a by fluorescence assay |
Bioorg Med Chem Lett 20: 618-22 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.077
BindingDB Entry DOI: 10.7270/Q24M94NN |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50228941
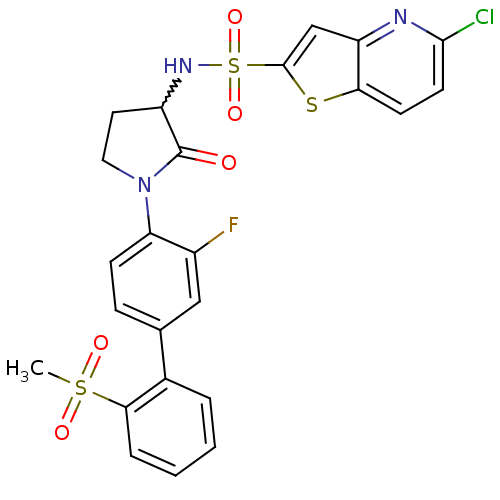
(5-chloro-thieno[3,2-b]pyridine-2-sulfonic acid [1-...)Show SMILES CS(=O)(=O)c1ccccc1-c1ccc(N2CCC(NS(=O)(=O)c3cc4nc(Cl)ccc4s3)C2=O)c(F)c1 |w:17.18| Show InChI InChI=1S/C24H19ClFN3O5S3/c1-36(31,32)21-5-3-2-4-15(21)14-6-7-19(16(26)12-14)29-11-10-17(24(29)30)28-37(33,34)23-13-18-20(35-23)8-9-22(25)27-18/h2-9,12-13,17,28H,10-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human F10a by fluorescence assay |
Bioorg Med Chem Lett 18: 23-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.023
BindingDB Entry DOI: 10.7270/Q2SX6CZF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50206984
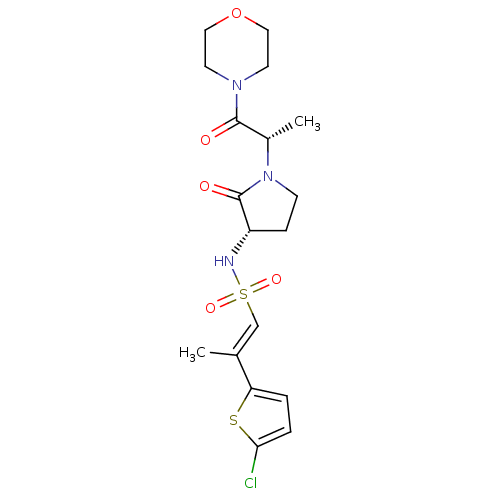
((2R)-2-(5-CHLORO-2-THIENYL)-N-{(3S)-1-[(1S)-1-METH...)Show SMILES C[C@H](N1CC[C@H](NS(=O)(=O)\C=C(/C)c2ccc(Cl)s2)C1=O)C(=O)N1CCOCC1 Show InChI InChI=1S/C18H24ClN3O5S2/c1-12(15-3-4-16(19)28-15)11-29(25,26)20-14-5-6-22(18(14)24)13(2)17(23)21-7-9-27-10-8-21/h3-4,11,13-14,20H,5-10H2,1-2H3/b12-11+/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a by fluorogenic assay |
Bioorg Med Chem Lett 17: 2927-30 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.080
BindingDB Entry DOI: 10.7270/Q22V2FTR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prothrombin
(Homo sapiens (Human)) | BDBM50206984

((2R)-2-(5-CHLORO-2-THIENYL)-N-{(3S)-1-[(1S)-1-METH...)Show SMILES C[C@H](N1CC[C@H](NS(=O)(=O)\C=C(/C)c2ccc(Cl)s2)C1=O)C(=O)N1CCOCC1 Show InChI InChI=1S/C18H24ClN3O5S2/c1-12(15-3-4-16(19)28-15)11-29(25,26)20-14-5-6-22(18(14)24)13(2)17(23)21-7-9-27-10-8-21/h3-4,11,13-14,20H,5-10H2,1-2H3/b12-11+/t13-,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of thrombin by fluorogenic assay |
Bioorg Med Chem Lett 17: 2927-30 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.080
BindingDB Entry DOI: 10.7270/Q22V2FTR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prothrombin
(Homo sapiens (Human)) | BDBM50206984

((2R)-2-(5-CHLORO-2-THIENYL)-N-{(3S)-1-[(1S)-1-METH...)Show SMILES C[C@H](N1CC[C@H](NS(=O)(=O)\C=C(/C)c2ccc(Cl)s2)C1=O)C(=O)N1CCOCC1 Show InChI InChI=1S/C18H24ClN3O5S2/c1-12(15-3-4-16(19)28-15)11-29(25,26)20-14-5-6-22(18(14)24)13(2)17(23)21-7-9-27-10-8-21/h3-4,11,13-14,20H,5-10H2,1-2H3/b12-11+/t13-,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of thrombin by fluorogenic assay |
Bioorg Med Chem Lett 17: 2931-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.02.034
BindingDB Entry DOI: 10.7270/Q2FJ2GFJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17654

(5-(5-chlorothiophen-2-yl)-N-[(3S)-1-[(2S)-1-(morph...)Show SMILES C[C@H](N1CC[C@H](NS(=O)(=O)c2ncc(s2)-c2ccc(Cl)s2)C1=O)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C18H21ClN4O5S3/c1-11(16(24)22-6-8-28-9-7-22)23-5-4-12(17(23)25)21-31(26,27)18-20-10-14(30-18)13-2-3-15(19)29-13/h2-3,10-12,21H,4-9H2,1H3/t11-,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2 | -49.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GSK
| Assay Description
The ability of test compounds to inhibit human fXa in vitro was determined in a fluorescence assay using rhodamime 110, bis-(Boc-L-glycylglycyl-L-arg... |
J Med Chem 50: 1546-57 (2007)
Article DOI: 10.1021/jm060870c
BindingDB Entry DOI: 10.7270/Q2F18X06 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12567
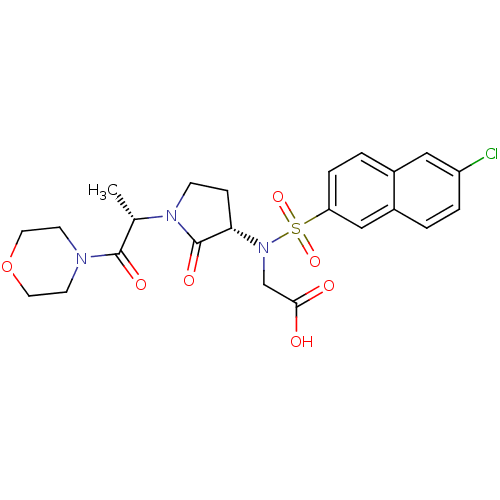
(2-[(6-chloronaphthalene-2-)[(3S)-1-[(2S)-1-(morpho...)Show SMILES C[C@H](N1CC[C@H](N(CC(O)=O)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C23H26ClN3O7S/c1-15(22(30)25-8-10-34-11-9-25)26-7-6-20(23(26)31)27(14-21(28)29)35(32,33)19-5-3-16-12-18(24)4-2-17(16)13-19/h2-5,12-13,15,20H,6-11,14H2,1H3,(H,28,29)/t15-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | -49.7 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 3784-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.04.053
BindingDB Entry DOI: 10.7270/Q2416V9V |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12558
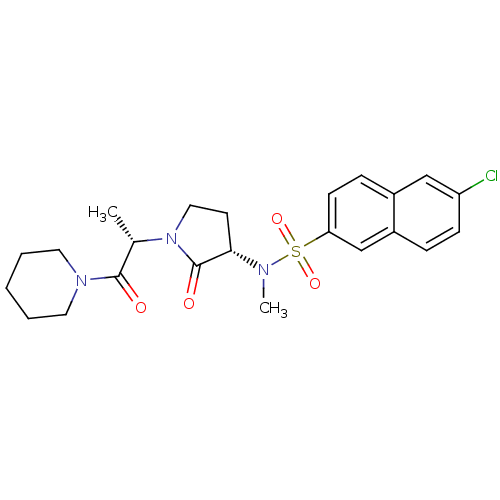
(6-chloro-N-methyl-N-[(3S)-2-oxo-1-[(2S)-1-oxo-1-(p...)Show SMILES C[C@H](N1CC[C@H](N(C)S(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O)C(=O)N1CCCCC1 |r| Show InChI InChI=1S/C23H28ClN3O4S/c1-16(22(28)26-11-4-3-5-12-26)27-13-10-21(23(27)29)25(2)32(30,31)20-9-7-17-14-19(24)8-6-18(17)15-20/h6-9,14-16,21H,3-5,10-13H2,1-2H3/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | -49.7 | n/a | n/a | n/a | n/a | n/a | 7.8 | 25 |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 3784-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.04.053
BindingDB Entry DOI: 10.7270/Q2416V9V |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12535
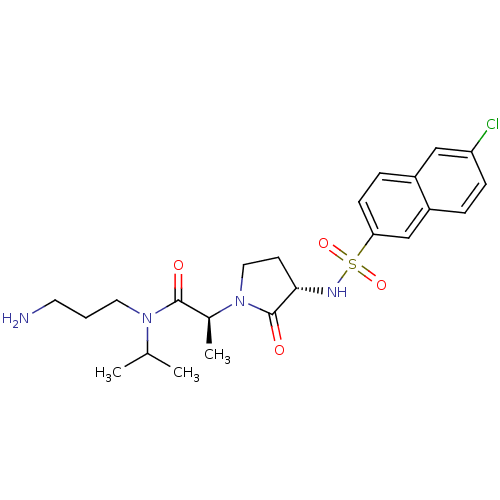
((2S)-N-(3-aminopropyl)-2-[(3S)-3-[(6-chloronaphtha...)Show SMILES CC(C)N(CCCN)C(=O)[C@H](C)N1CC[C@H](NS(=O)(=O)c2ccc3cc(Cl)ccc3c2)C1=O |r| Show InChI InChI=1S/C23H31ClN4O4S/c1-15(2)27(11-4-10-25)22(29)16(3)28-12-9-21(23(28)30)26-33(31,32)20-8-6-17-13-19(24)7-5-18(17)14-20/h5-8,13-16,21,26H,4,9-12,25H2,1-3H3/t16-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The inhibitory effect of test compound for human fXa was determined by using the chromogenic substrate. The hydrolysis rates of chromogenic substrate... |
Bioorg Med Chem Lett 16: 5953-7 (2006)
Article DOI: 10.1016/j.bmcl.2006.09.001
BindingDB Entry DOI: 10.7270/Q2CF9NB8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data