 Found 644 hits with Last Name = 'stone' and Initial = 'dj'
Found 644 hits with Last Name = 'stone' and Initial = 'dj' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154039
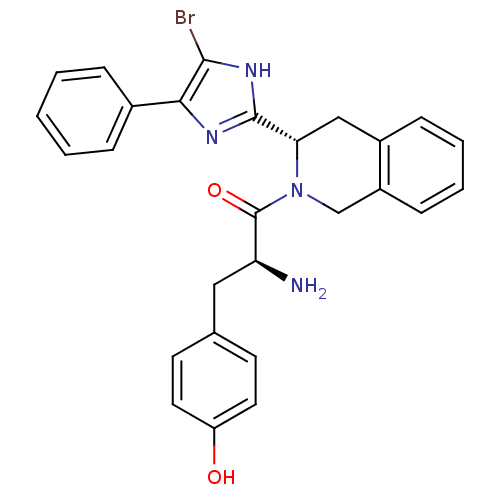
(2-Amino-1-[3-(5-bromo-4-phenyl-1H-imidazol-2-yl)-3...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c(Br)[nH]1)-c1ccccc1 Show InChI InChI=1S/C27H25BrN4O2/c28-25-24(18-6-2-1-3-7-18)30-26(31-25)23-15-19-8-4-5-9-20(19)16-32(23)27(34)22(29)14-17-10-12-21(33)13-11-17/h1-13,22-23,33H,14-16,29H2,(H,30,31)/t22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411339
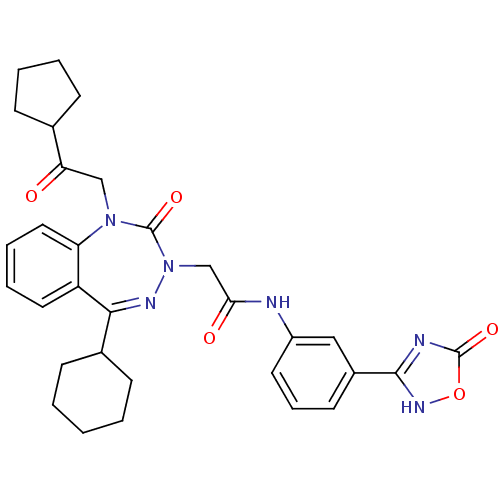
(CHEMBL227276)Show SMILES O=C(CN1N=C(C2CCCCC2)c2ccccc2N(CC(=O)C2CCCC2)C1=O)Nc1cccc(c1)-c1nc(=O)o[nH]1 |t:4| Show InChI InChI=1S/C31H34N6O5/c38-26(20-9-4-5-10-20)18-36-25-16-7-6-15-24(25)28(21-11-2-1-3-12-21)34-37(31(36)41)19-27(39)32-23-14-8-13-22(17-23)29-33-30(40)42-35-29/h6-8,13-17,20-21H,1-5,9-12,18-19H2,(H,32,39)(H,33,35,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50056102
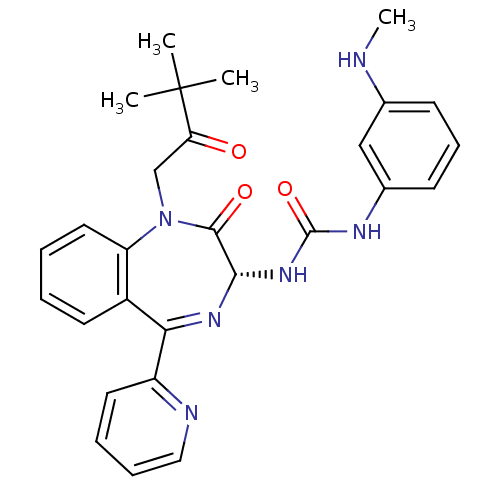
((R)-1-(1-(3,3-dimethyl-2-oxobutyl)-2-oxo-5-(pyridi...)Show SMILES CNc1cccc(NC(=O)N[C@@H]2N=C(c3ccccn3)c3ccccc3N(CC(=O)C(C)(C)C)C2=O)c1 |t:12| Show InChI InChI=1S/C28H30N6O3/c1-28(2,3)23(35)17-34-22-14-6-5-12-20(22)24(21-13-7-8-15-30-21)32-25(26(34)36)33-27(37)31-19-11-9-10-18(16-19)29-4/h5-16,25,29H,17H2,1-4H3,(H2,31,33,37)/t25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50017698
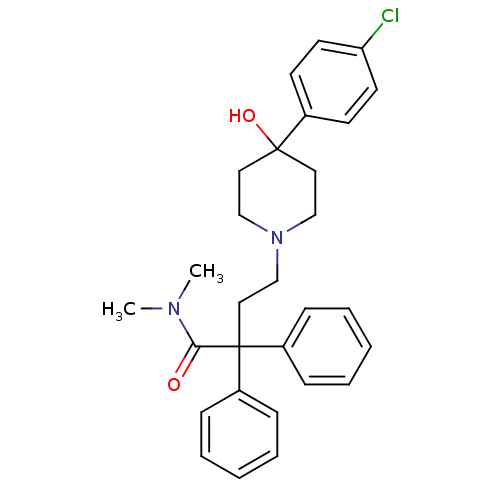
(4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...)Show SMILES CN(C)C(=O)C(CCN1CCC(O)(CC1)c1ccc(Cl)cc1)(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C29H33ClN2O2/c1-31(2)27(33)29(24-9-5-3-6-10-24,25-11-7-4-8-12-25)19-22-32-20-17-28(34,18-21-32)23-13-15-26(30)16-14-23/h3-16,34H,17-22H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for Mu opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411341
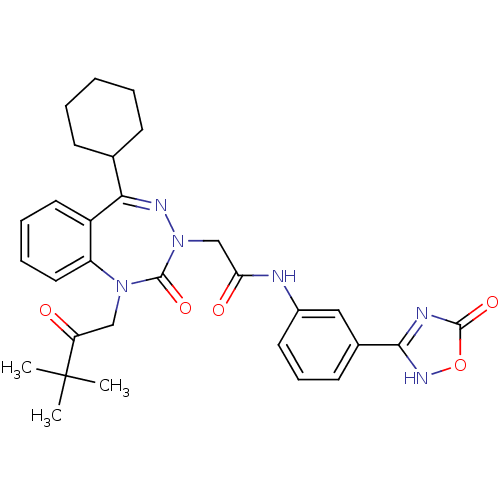
(CHEMBL226583)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2nc(=O)o[nH]2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C30H34N6O5/c1-30(2,3)24(37)17-35-23-15-8-7-14-22(23)26(19-10-5-4-6-11-19)33-36(29(35)40)18-25(38)31-21-13-9-12-20(16-21)27-32-28(39)41-34-27/h7-9,12-16,19H,4-6,10-11,17-18H2,1-3H3,(H,31,38)(H,32,34,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155124
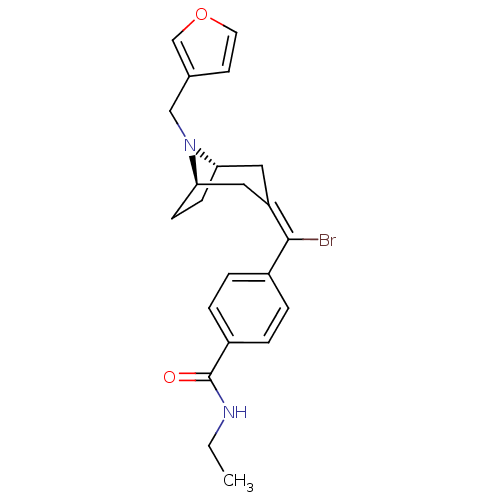
(4-{Bromo-[(1S,5R)-8-furan-3-ylmethyl-8-aza-bicyclo...)Show SMILES CCNC(=O)c1ccc(cc1)C(\Br)=C1\C[C@@H]2CC[C@H](C1)N2Cc1ccoc1 |THB:21:20:13.14.19:17.16| Show InChI InChI=1S/C22H25BrN2O2/c1-2-24-22(26)17-5-3-16(4-6-17)21(23)18-11-19-7-8-20(12-18)25(19)13-15-9-10-27-14-15/h3-6,9-10,14,19-20H,2,7-8,11-13H2,1H3,(H,24,26)/b21-18-/t19-,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002875
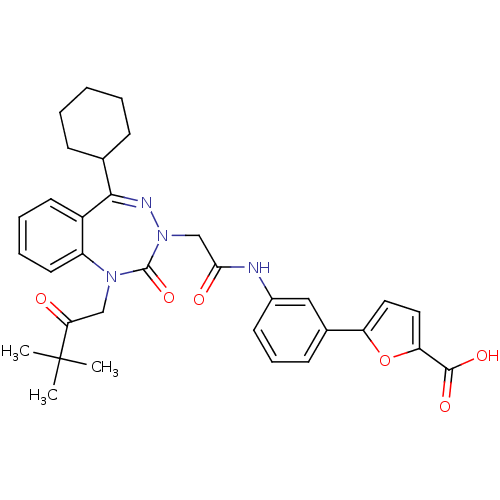
(CHEMBL388144)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2ccc(o2)C(O)=O)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C33H36N4O6/c1-33(2,3)28(38)19-36-25-15-8-7-14-24(25)30(21-10-5-4-6-11-21)35-37(32(36)42)20-29(39)34-23-13-9-12-22(18-23)26-16-17-27(43-26)31(40)41/h7-9,12-18,21H,4-6,10-11,19-20H2,1-3H3,(H,34,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155085
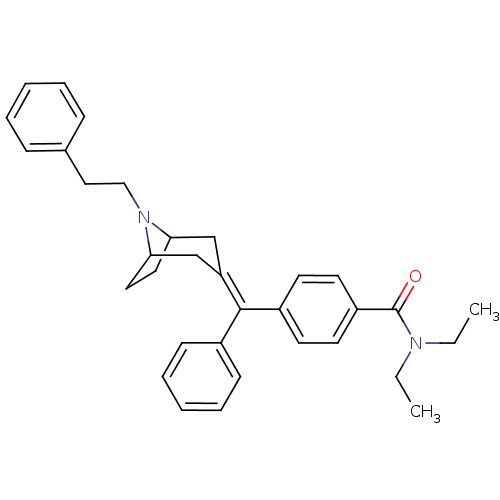
(CHEMBL362331 | N,N-Diethyl-4-[(8-phenethyl-8-aza-b...)Show SMILES [#6]-[#6]-[#7](-[#6]-[#6])-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-[#6]-c1ccccc1)\c1ccccc1 Show InChI InChI=1S/C33H38N2O/c1-3-34(4-2)33(36)28-17-15-27(16-18-28)32(26-13-9-6-10-14-26)29-23-30-19-20-31(24-29)35(30)22-21-25-11-7-5-8-12-25/h5-18,30-31H,3-4,19-24H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411340
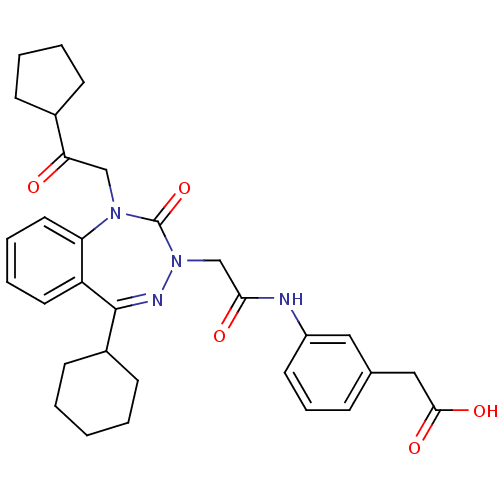
(CHEMBL387948)Show SMILES OC(=O)Cc1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C3CCCC3)C2=O)c1 |t:14| Show InChI InChI=1S/C31H36N4O5/c36-27(22-10-4-5-11-22)19-34-26-16-7-6-15-25(26)30(23-12-2-1-3-13-23)33-35(31(34)40)20-28(37)32-24-14-8-9-21(17-24)18-29(38)39/h6-9,14-17,22-23H,1-5,10-13,18-20H2,(H,32,37)(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154035
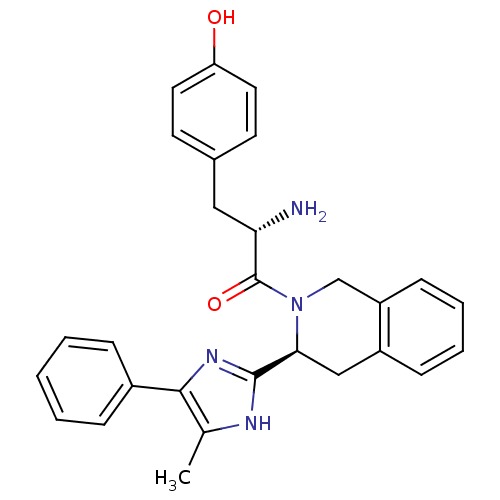
((S)-2-amino-3-(4-hydroxyphenyl)-1-((S)-3-(5-methyl...)Show SMILES Cc1[nH]c(nc1-c1ccccc1)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1ccc(O)cc1 Show InChI InChI=1S/C28H28N4O2/c1-18-26(20-7-3-2-4-8-20)31-27(30-18)25-16-21-9-5-6-10-22(21)17-32(25)28(34)24(29)15-19-11-13-23(33)14-12-19/h2-14,24-25,33H,15-17,29H2,1H3,(H,30,31)/t24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411344
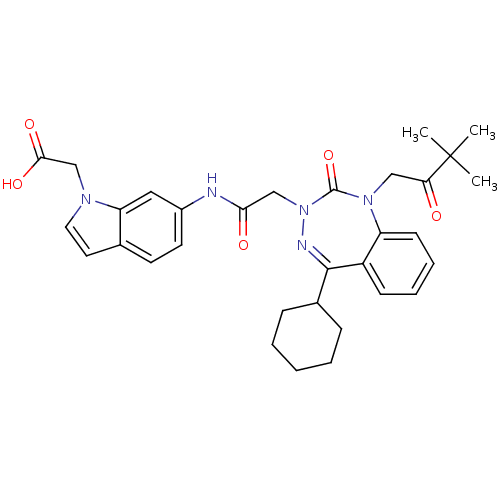
(CHEMBL389711)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2ccc3ccn(CC(O)=O)c3c2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C32H37N5O5/c1-32(2,3)27(38)18-36-25-12-8-7-11-24(25)30(22-9-5-4-6-10-22)34-37(31(36)42)19-28(39)33-23-14-13-21-15-16-35(20-29(40)41)26(21)17-23/h7-8,11-17,22H,4-6,9-10,18-20H2,1-3H3,(H,33,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154035

((S)-2-amino-3-(4-hydroxyphenyl)-1-((S)-3-(5-methyl...)Show SMILES Cc1[nH]c(nc1-c1ccccc1)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1ccc(O)cc1 Show InChI InChI=1S/C28H28N4O2/c1-18-26(20-7-3-2-4-8-20)31-27(30-18)25-16-21-9-5-6-10-22(21)17-32(25)28(34)24(29)15-19-11-13-23(33)14-12-19/h2-14,24-25,33H,15-17,29H2,1H3,(H,30,31)/t24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411342
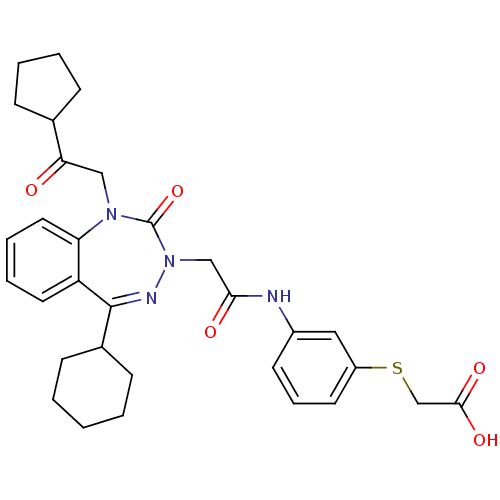
(CHEMBL227333)Show SMILES OC(=O)CSc1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C3CCCC3)C2=O)c1 |t:15| Show InChI InChI=1S/C31H36N4O5S/c36-27(21-9-4-5-10-21)18-34-26-16-7-6-15-25(26)30(22-11-2-1-3-12-22)33-35(31(34)40)19-28(37)32-23-13-8-14-24(17-23)41-20-29(38)39/h6-8,13-17,21-22H,1-5,9-12,18-20H2,(H,32,37)(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411336

(CHEMBL227330)Show SMILES OC(=O)CCc1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C3CCCC3)C2=O)c1 |t:15| Show InChI InChI=1S/C32H38N4O5/c37-28(23-10-4-5-11-23)20-35-27-16-7-6-15-26(27)31(24-12-2-1-3-13-24)34-36(32(35)41)21-29(38)33-25-14-8-9-22(19-25)17-18-30(39)40/h6-9,14-16,19,23-24H,1-5,10-13,17-18,20-21H2,(H,33,38)(H,39,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002880
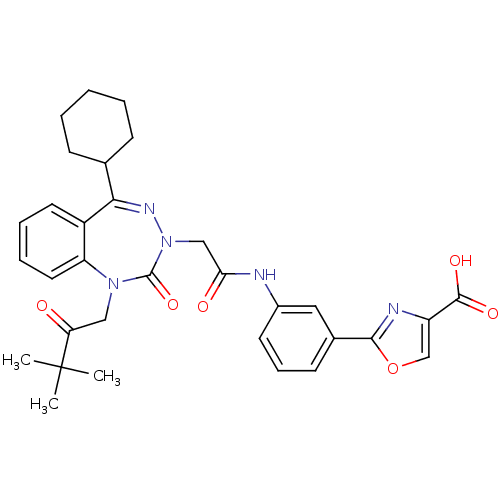
(CHEMBL437736)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2nc(co2)C(O)=O)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C32H35N5O6/c1-32(2,3)26(38)17-36-25-15-8-7-14-23(25)28(20-10-5-4-6-11-20)35-37(31(36)42)18-27(39)33-22-13-9-12-21(16-22)29-34-24(19-43-29)30(40)41/h7-9,12-16,19-20H,4-6,10-11,17-18H2,1-3H3,(H,33,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155068
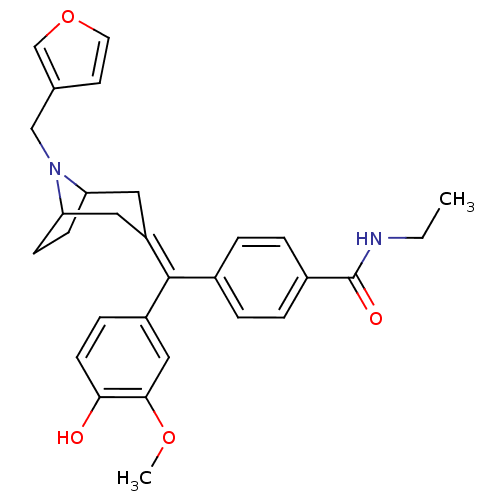
(CHEMBL361720 | N-Ethyl-4-[(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc(-[#8])c(-[#8]-[#6])c1 Show InChI InChI=1S/C29H32N2O4/c1-3-30-29(33)21-6-4-20(5-7-21)28(22-8-11-26(32)27(16-22)34-2)23-14-24-9-10-25(15-23)31(24)17-19-12-13-35-18-19/h4-8,11-13,16,18,24-25,32H,3,9-10,14-15,17H2,1-2H3,(H,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411345
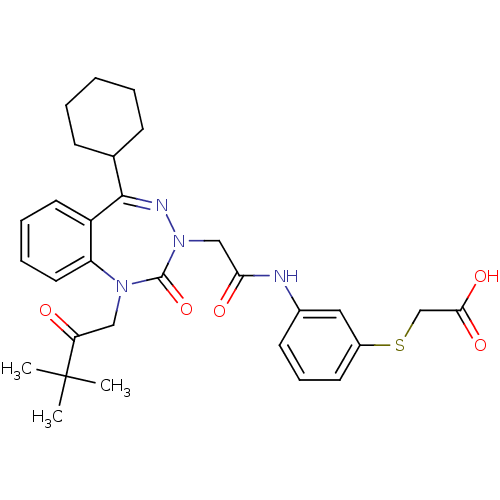
(CHEMBL389639)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(SCC(O)=O)c2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C30H36N4O5S/c1-30(2,3)25(35)17-33-24-15-8-7-14-23(24)28(20-10-5-4-6-11-20)32-34(29(33)39)18-26(36)31-21-12-9-13-22(16-21)40-19-27(37)38/h7-9,12-16,20H,4-6,10-11,17-19H2,1-3H3,(H,31,36)(H,37,38) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002878
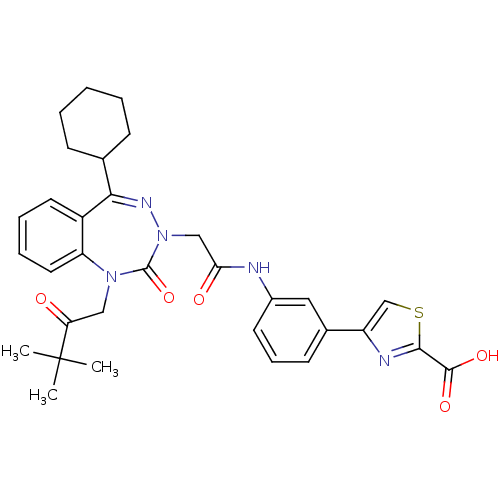
(CHEMBL227275)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2csc(n2)C(O)=O)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C32H35N5O5S/c1-32(2,3)26(38)17-36-25-15-8-7-14-23(25)28(20-10-5-4-6-11-20)35-37(31(36)42)18-27(39)33-22-13-9-12-21(16-22)24-19-43-29(34-24)30(40)41/h7-9,12-16,19-20H,4-6,10-11,17-18H2,1-3H3,(H,33,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002876
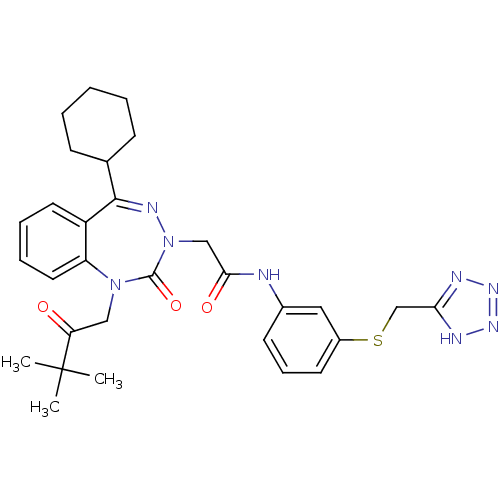
(CHEMBL435143)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(SCc3nn[nH]n3)c2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C30H36N8O3S/c1-30(2,3)25(39)17-37-24-15-8-7-14-23(24)28(20-10-5-4-6-11-20)34-38(29(37)41)18-27(40)31-21-12-9-13-22(16-21)42-19-26-32-35-36-33-26/h7-9,12-16,20H,4-6,10-11,17-19H2,1-3H3,(H,31,40)(H,32,33,35,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50471066
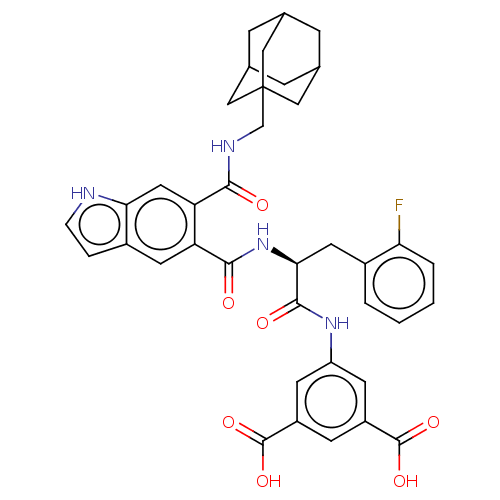
(CHEMBL415936)Show SMILES OC(=O)c1cc(NC(=O)[C@H](Cc2ccccc2F)NC(=O)c2cc3cc[nH]c3cc2C(=O)NCC23CC4CC(CC(C4)C2)C3)cc(c1)C(O)=O |TLB:41:36:43:40.42.39,41:40:43:36.35.37,THB:39:38:35:40.42.41,39:40:35:38.43.37| Show InChI InChI=1S/C38H37FN4O7/c39-30-4-2-1-3-23(30)14-32(35(46)42-27-11-25(36(47)48)10-26(12-27)37(49)50)43-34(45)28-13-24-5-6-40-31(24)15-29(28)33(44)41-19-38-16-20-7-21(17-38)9-22(8-20)18-38/h1-6,10-13,15,20-22,32,40H,7-9,14,16-19H2,(H,41,44)(H,42,46)(H,43,45)(H,47,48)(H,49,50)/t20?,21?,22?,32-,38?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.741 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against Cholecystokinin type B receptor using [125I]BH-CCK-8S as radioligand in mouse cortical membranes |
J Med Chem 39: 1806-15 (1996)
Article DOI: 10.1021/jm9508907
BindingDB Entry DOI: 10.7270/Q2571FR6 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002889
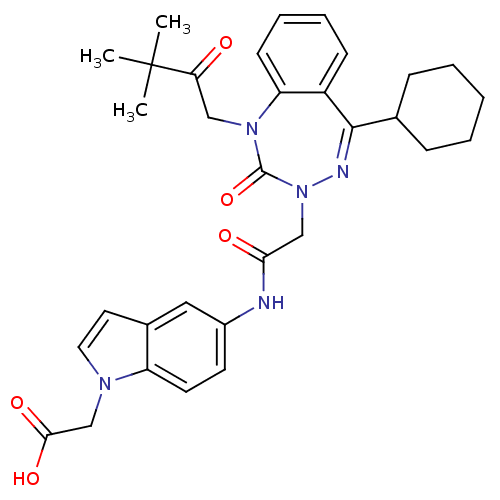
(CHEMBL438843)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2ccc3n(CC(O)=O)ccc3c2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C32H37N5O5/c1-32(2,3)27(38)18-36-26-12-8-7-11-24(26)30(21-9-5-4-6-10-21)34-37(31(36)42)19-28(39)33-23-13-14-25-22(17-23)15-16-35(25)20-29(40)41/h7-8,11-17,21H,4-6,9-10,18-20H2,1-3H3,(H,33,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002902
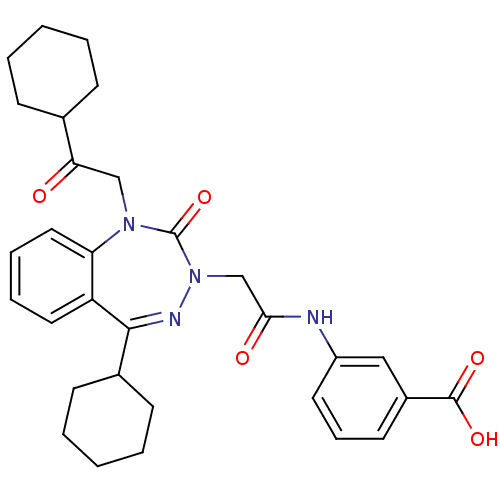
(CHEMBL226622)Show SMILES OC(=O)c1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C3CCCCC3)C2=O)c1 |t:13| Show InChI InChI=1S/C31H36N4O5/c36-27(21-10-3-1-4-11-21)19-34-26-17-8-7-16-25(26)29(22-12-5-2-6-13-22)33-35(31(34)40)20-28(37)32-24-15-9-14-23(18-24)30(38)39/h7-9,14-18,21-22H,1-6,10-13,19-20H2,(H,32,37)(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155120

(4-[(4-Acetylamino-phenyl)-(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc(-[#7]-[#6](-[#6])=O)cc1 Show InChI InChI=1S/C30H33N3O3/c1-3-31-30(35)24-6-4-22(5-7-24)29(23-8-10-26(11-9-23)32-20(2)34)25-16-27-12-13-28(17-25)33(27)18-21-14-15-36-19-21/h4-11,14-15,19,27-28H,3,12-13,16-18H2,1-2H3,(H,31,35)(H,32,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155082
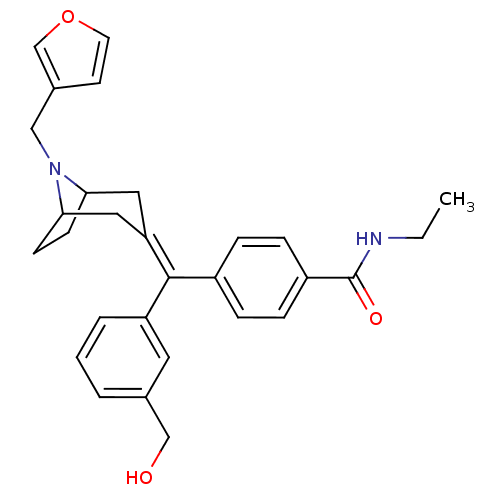
(CHEMBL363287 | N-Ethyl-4-[(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1\[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1cccc(-[#6]-[#8])c1 Show InChI InChI=1S/C29H32N2O3/c1-2-30-29(33)23-8-6-22(7-9-23)28(24-5-3-4-20(14-24)18-32)25-15-26-10-11-27(16-25)31(26)17-21-12-13-34-19-21/h3-9,12-14,19,26-27,32H,2,10-11,15-18H2,1H3,(H,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002888
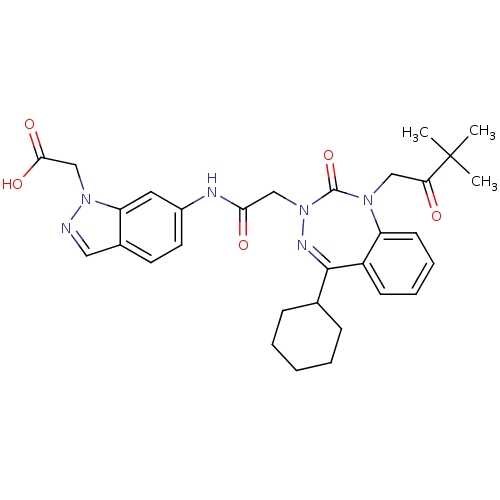
(CHEMBL389713)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2ccc3cnn(CC(O)=O)c3c2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C31H36N6O5/c1-31(2,3)26(38)17-35-24-12-8-7-11-23(24)29(20-9-5-4-6-10-20)34-37(30(35)42)18-27(39)33-22-14-13-21-16-32-36(19-28(40)41)25(21)15-22/h7-8,11-16,20H,4-6,9-10,17-19H2,1-3H3,(H,33,39)(H,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154036

((S)-2-amino-3-(4-hydroxyphenyl)-1-((S)-3-(4-phenyl...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c[nH]1)-c1ccccc1 Show InChI InChI=1S/C27H26N4O2/c28-23(14-18-10-12-22(32)13-11-18)27(33)31-17-21-9-5-4-8-20(21)15-25(31)26-29-16-24(30-26)19-6-2-1-3-7-19/h1-13,16,23,25,32H,14-15,17,28H2,(H,29,30)/t23-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002872
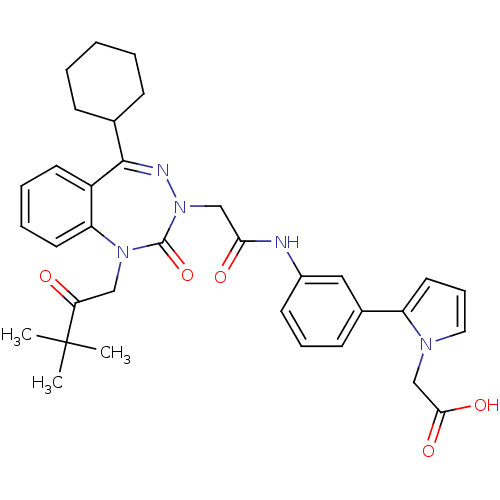
(CHEMBL437930)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2cccn2CC(O)=O)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C34H39N5O5/c1-34(2,3)29(40)20-38-28-16-8-7-15-26(28)32(23-11-5-4-6-12-23)36-39(33(38)44)21-30(41)35-25-14-9-13-24(19-25)27-17-10-18-37(27)22-31(42)43/h7-10,13-19,23H,4-6,11-12,20-22H2,1-3H3,(H,35,41)(H,42,43) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Parathyroid hormone/parathyroid hormone-related peptide receptor
(Homo sapiens (Human)) | BDBM50002926
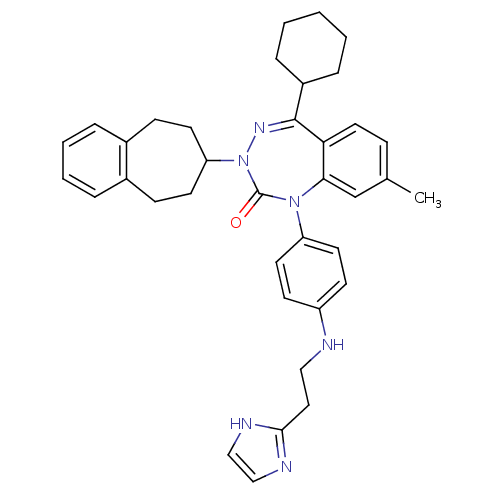
(CHEMBL244325)Show SMILES Cc1ccc2c(c1)N(c1ccc(NCCc3ncc[nH]3)cc1)C(=O)N(N=C2C1CCCCC1)C1CCc2ccccc2CC1 |c:28| Show InChI InChI=1S/C37H42N6O/c1-26-11-20-33-34(25-26)42(31-18-14-30(15-19-31)38-22-21-35-39-23-24-40-35)37(44)43(41-36(33)29-9-3-2-4-10-29)32-16-12-27-7-5-6-8-28(27)13-17-32/h5-8,11,14-15,18-20,23-25,29,32,38H,2-4,9-10,12-13,16-17,21-22H2,1H3,(H,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-[Nle,8,18 Tyr34]-hPTH(1-34) from human recombinant PTH1R expressed in HEK293 cells |
J Med Chem 50: 4789-92 (2007)
Checked by Author
Article DOI: 10.1021/jm0707626
BindingDB Entry DOI: 10.7270/Q2Z039C3 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155069
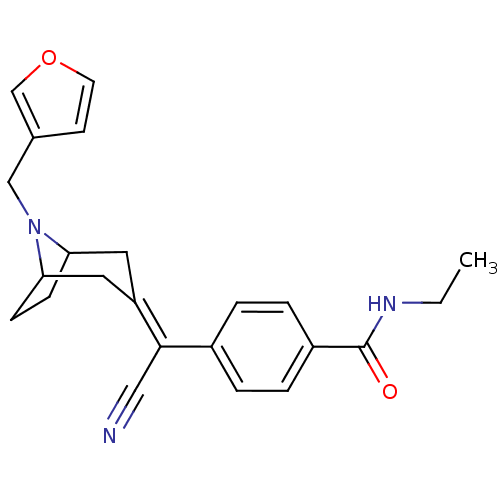
(4-[Cyano-(8-furan-3-ylmethyl-8-aza-bicyclo[3.2.1]o...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](\C#N)=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1 Show InChI InChI=1S/C23H25N3O2/c1-2-25-23(27)18-5-3-17(4-6-18)22(13-24)19-11-20-7-8-21(12-19)26(20)14-16-9-10-28-15-16/h3-6,9-10,15,20-21H,2,7-8,11-12,14H2,1H3,(H,25,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411348
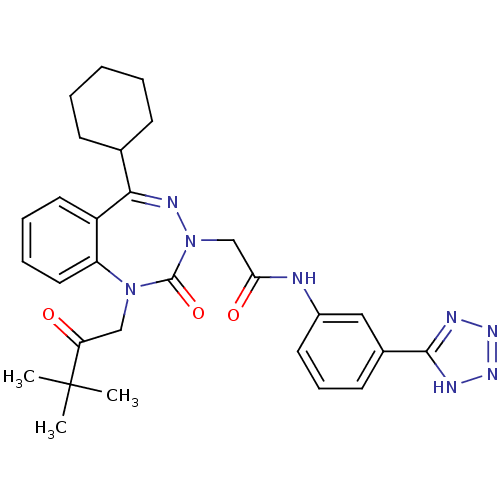
(CHEMBL226533)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2nnn[nH]2)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C29H34N8O3/c1-29(2,3)24(38)17-36-23-15-8-7-14-22(23)26(19-10-5-4-6-11-19)33-37(28(36)40)18-25(39)30-21-13-9-12-20(16-21)27-31-34-35-32-27/h7-9,12-16,19H,4-6,10-11,17-18H2,1-3H3,(H,30,39)(H,31,32,34,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50411347
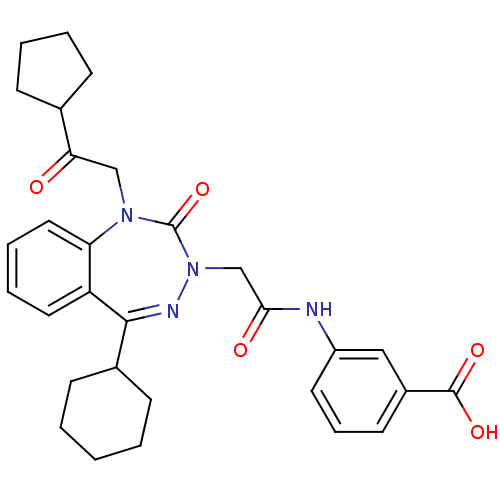
(CHEMBL226620)Show SMILES OC(=O)c1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C3CCCC3)C2=O)c1 |t:13| Show InChI InChI=1S/C30H34N4O5/c35-26(20-9-4-5-10-20)18-33-25-16-7-6-15-24(25)28(21-11-2-1-3-12-21)32-34(30(33)39)19-27(36)31-23-14-8-13-22(17-23)29(37)38/h6-8,13-17,20-21H,1-5,9-12,18-19H2,(H,31,36)(H,37,38) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50213845
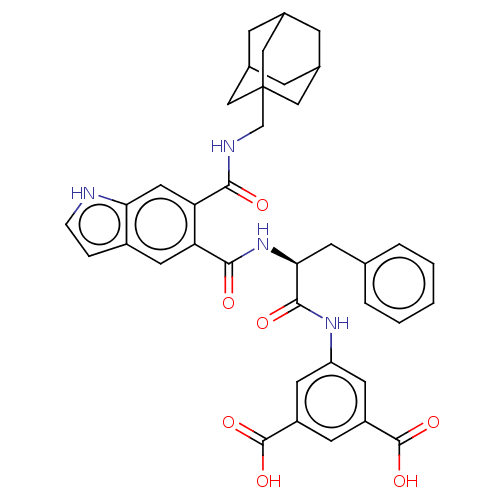
(CHEMBL14557)Show SMILES OC(=O)c1cc(NC(=O)[C@H](Cc2ccccc2)NC(=O)c2cc3cc[nH]c3cc2C(=O)NCC23CC4CC(CC(C4)C2)C3)cc(c1)C(O)=O |TLB:40:35:42:39.41.38,40:39:42:35.34.36,THB:38:37:34:39.41.40,38:39:34:37.42.36| Show InChI InChI=1S/C38H38N4O7/c43-33(40-20-38-17-22-8-23(18-38)10-24(9-22)19-38)30-16-31-25(6-7-39-31)15-29(30)34(44)42-32(11-21-4-2-1-3-5-21)35(45)41-28-13-26(36(46)47)12-27(14-28)37(48)49/h1-7,12-16,22-24,32,39H,8-11,17-20H2,(H,40,43)(H,41,45)(H,42,44)(H,46,47)(H,48,49)/t22?,23?,24?,32-,38?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of binding of [125I]CCK-8S to Cholecystokinin type B receptor in mouse cerebral cortex homogenates |
J Med Chem 43: 3518-29 (2000)
Article DOI: 10.1021/jm000960w
BindingDB Entry DOI: 10.7270/Q2KS6V9G |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002895
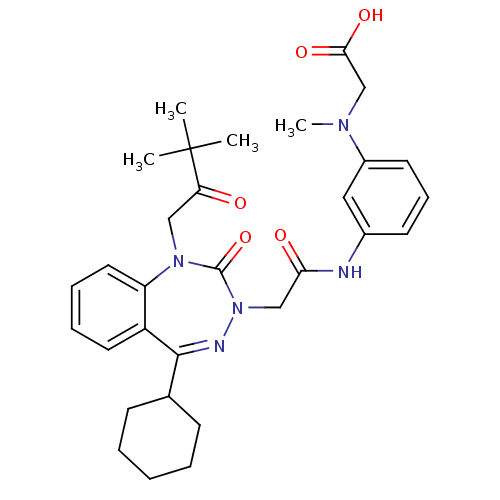
(CHEMBL226726)Show SMILES CN(CC(O)=O)c1cccc(NC(=O)CN2N=C(C3CCCCC3)c3ccccc3N(CC(=O)C(C)(C)C)C2=O)c1 |t:16| Show InChI InChI=1S/C31H39N5O5/c1-31(2,3)26(37)18-35-25-16-9-8-15-24(25)29(21-11-6-5-7-12-21)33-36(30(35)41)19-27(38)32-22-13-10-14-23(17-22)34(4)20-28(39)40/h8-10,13-17,21H,5-7,11-12,18-20H2,1-4H3,(H,32,38)(H,39,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155096
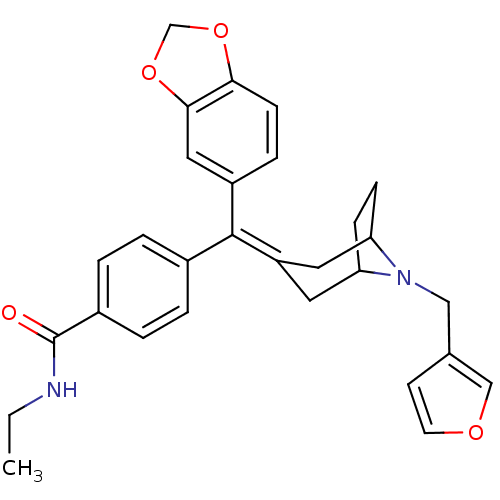
(4-[Benzo[1,3]dioxol-5-yl-(8-furan-3-ylmethyl-8-aza...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1\[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc2-[#8]-[#6]-[#8]-c2c1 Show InChI InChI=1S/C29H30N2O4/c1-2-30-29(32)21-5-3-20(4-6-21)28(22-7-10-26-27(15-22)35-18-34-26)23-13-24-8-9-25(14-23)31(24)16-19-11-12-33-17-19/h3-7,10-12,15,17,24-25H,2,8-9,13-14,16,18H2,1H3,(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50213845

(CHEMBL14557)Show SMILES OC(=O)c1cc(NC(=O)[C@H](Cc2ccccc2)NC(=O)c2cc3cc[nH]c3cc2C(=O)NCC23CC4CC(CC(C4)C2)C3)cc(c1)C(O)=O |TLB:40:35:42:39.41.38,40:39:42:35.34.36,THB:38:37:34:39.41.40,38:39:34:37.42.36| Show InChI InChI=1S/C38H38N4O7/c43-33(40-20-38-17-22-8-23(18-38)10-24(9-22)19-38)30-16-31-25(6-7-39-31)15-29(30)34(44)42-32(11-21-4-2-1-3-5-21)35(45)41-28-13-26(36(46)47)12-27(14-28)37(48)49/h1-7,12-16,22-24,32,39H,8-11,17-20H2,(H,40,43)(H,41,45)(H,42,44)(H,46,47)(H,48,49)/t22?,23?,24?,32-,38?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against Cholecystokinin type B receptor using [125I]BH-CCK-8S as radioligand in mouse cortical membranes |
J Med Chem 39: 1806-15 (1996)
Article DOI: 10.1021/jm9508907
BindingDB Entry DOI: 10.7270/Q2571FR6 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50471075
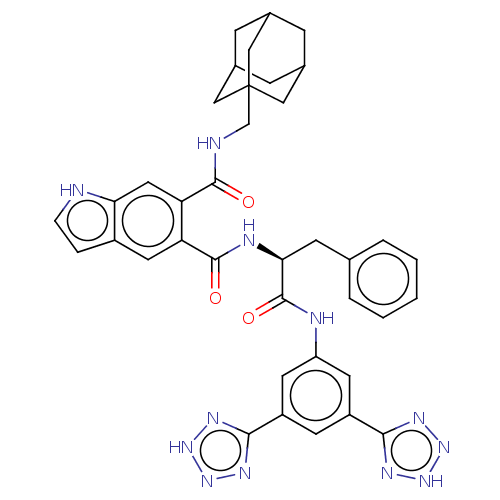
(CHEMBL299540)Show SMILES O=C(Nc1cc(cc(c1)-c1nn[nH]n1)-c1nn[nH]n1)[C@H](Cc1ccccc1)NC(=O)c1cc2cc[nH]c2cc1C(=O)NCC12CC3CC(CC(C3)C1)C2 |TLB:46:47:51:45.44.50,50:49:52:45.44.46,50:45:52:49.51.48,THB:46:45:51:47.52.48| Show InChI InChI=1S/C38H38N12O3/c51-35(40-20-38-17-22-8-23(18-38)10-24(9-22)19-38)30-16-31-25(6-7-39-31)15-29(30)36(52)42-32(11-21-4-2-1-3-5-21)37(53)41-28-13-26(33-43-47-48-44-33)12-27(14-28)34-45-49-50-46-34/h1-7,12-16,22-24,32,39H,8-11,17-20H2,(H,40,51)(H,41,53)(H,42,52)(H,43,44,47,48)(H,45,46,49,50)/t22?,23?,24?,32-,38?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against Cholecystokinin type B receptor using [125I]BH-CCK-8S as radioligand in mouse cortical membranes |
J Med Chem 39: 1806-15 (1996)
Article DOI: 10.1021/jm9508907
BindingDB Entry DOI: 10.7270/Q2571FR6 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50471067
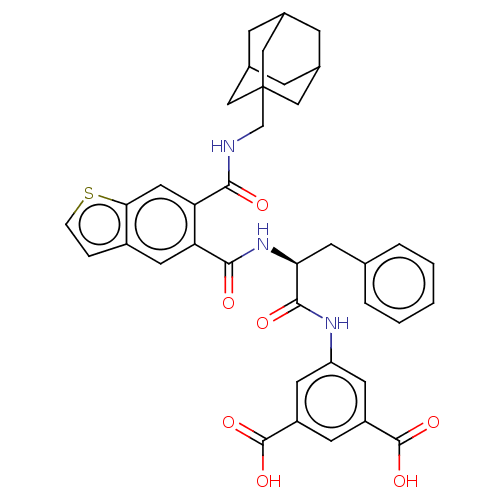
(CHEMBL298521)Show SMILES OC(=O)c1cc(NC(=O)[C@H](Cc2ccccc2)NC(=O)c2cc3ccsc3cc2C(=O)NCC23CC4CC(CC(C4)C2)C3)cc(c1)C(O)=O |TLB:36:37:41:35.34.40,THB:36:35:41:37.42.38,38:37:34:39.41.40,38:39:34:37.42.36| Show InChI InChI=1S/C38H37N3O7S/c42-33(39-20-38-17-22-8-23(18-38)10-24(9-22)19-38)30-16-32-25(6-7-49-32)15-29(30)34(43)41-31(11-21-4-2-1-3-5-21)35(44)40-28-13-26(36(45)46)12-27(14-28)37(47)48/h1-7,12-16,22-24,31H,8-11,17-20H2,(H,39,42)(H,40,44)(H,41,43)(H,45,46)(H,47,48)/t22?,23?,24?,31-,38?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against Cholecystokinin type B receptor using [125I]BH-CCK-8S as radioligand in mouse cortical membranes |
J Med Chem 39: 1806-15 (1996)
Article DOI: 10.1021/jm9508907
BindingDB Entry DOI: 10.7270/Q2571FR6 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002898
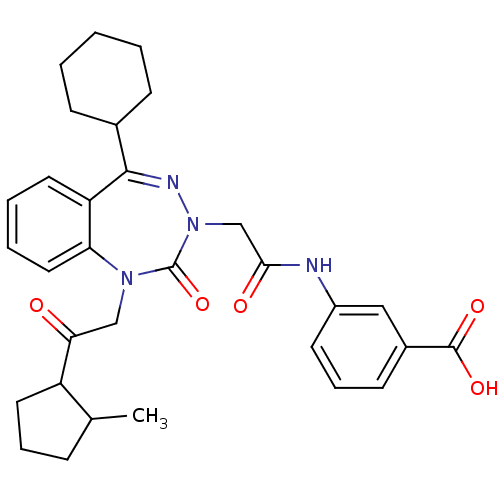
(CHEMBL388068)Show SMILES CC1CCCC1C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)C(O)=O)C1=O)C1CCCCC1 |c:18| Show InChI InChI=1S/C31H36N4O5/c1-20-9-7-15-24(20)27(36)18-34-26-16-6-5-14-25(26)29(21-10-3-2-4-11-21)33-35(31(34)40)19-28(37)32-23-13-8-12-22(17-23)30(38)39/h5-6,8,12-14,16-17,20-21,24H,2-4,7,9-11,15,18-19H2,1H3,(H,32,37)(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155076
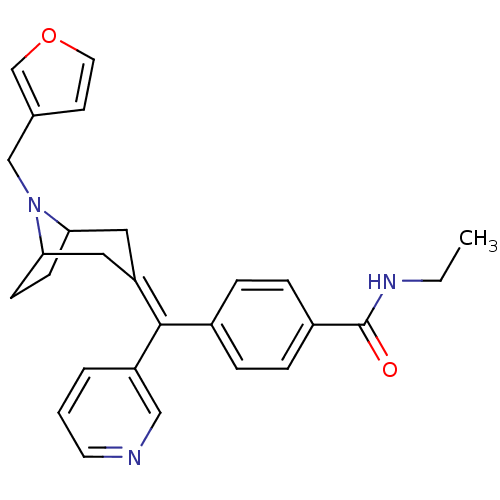
(CHEMBL186530 | N-Ethyl-4-[(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1cccnc1 Show InChI InChI=1S/C27H29N3O2/c1-2-29-27(31)21-7-5-20(6-8-21)26(22-4-3-12-28-16-22)23-14-24-9-10-25(15-23)30(24)17-19-11-13-32-18-19/h3-8,11-13,16,18,24-25H,2,9-10,14-15,17H2,1H3,(H,29,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155119

(4-[(3-Acetylamino-phenyl)-(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1cccc(-[#7]-[#6](-[#6])=O)c1 Show InChI InChI=1S/C30H33N3O3/c1-3-31-30(35)23-9-7-22(8-10-23)29(24-5-4-6-26(15-24)32-20(2)34)25-16-27-11-12-28(17-25)33(27)18-21-13-14-36-19-21/h4-10,13-15,19,27-28H,3,11-12,16-18H2,1-2H3,(H,31,35)(H,32,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155101
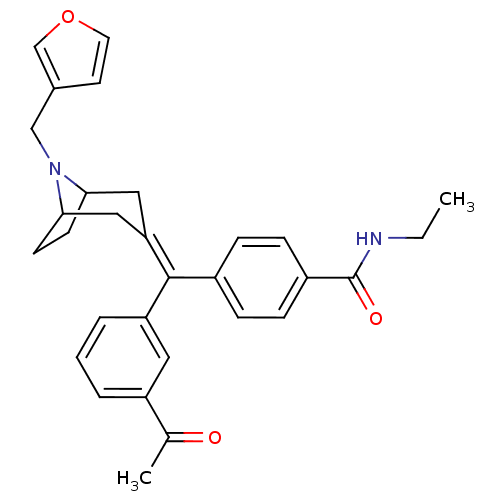
(4-[(3-Acetyl-phenyl)-(8-furan-3-ylmethyl-8-aza-bic...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1cccc(c1)-[#6](-[#6])=O Show InChI InChI=1S/C30H32N2O3/c1-3-31-30(34)23-9-7-22(8-10-23)29(25-6-4-5-24(15-25)20(2)33)26-16-27-11-12-28(17-26)32(27)18-21-13-14-35-19-21/h4-10,13-15,19,27-28H,3,11-12,16-18H2,1-2H3,(H,31,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155089

(4-[Benzo[1,3]dioxol-5-yl-(8-thiophen-3-ylmethyl-8-...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccsc1)\c1ccc2-[#8]-[#6]-[#8]-c2c1 Show InChI InChI=1S/C29H30N2O3S/c1-2-30-29(32)21-5-3-20(4-6-21)28(22-7-10-26-27(15-22)34-18-33-26)23-13-24-8-9-25(14-23)31(24)16-19-11-12-35-17-19/h3-7,10-12,15,17,24-25H,2,8-9,13-14,16,18H2,1H3,(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155112
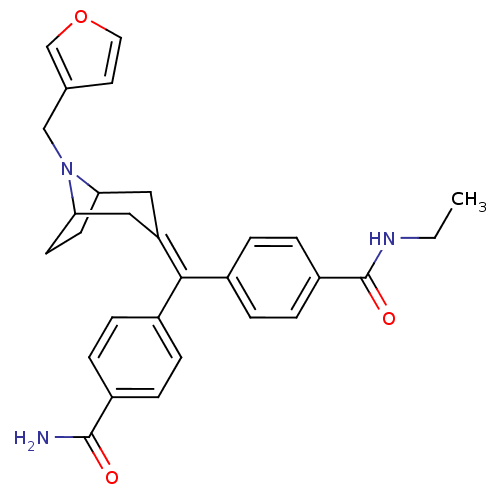
(1N-ethyl-4-{4-carbamoylphenyl[8-(3-furylmethyl)-8-...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc(cc1)-[#6](-[#7])=O Show InChI InChI=1S/C29H31N3O3/c1-2-31-29(34)23-9-5-21(6-10-23)27(20-3-7-22(8-4-20)28(30)33)24-15-25-11-12-26(16-24)32(25)17-19-13-14-35-18-19/h3-10,13-14,18,25-26H,2,11-12,15-17H2,1H3,(H2,30,33)(H,31,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155109
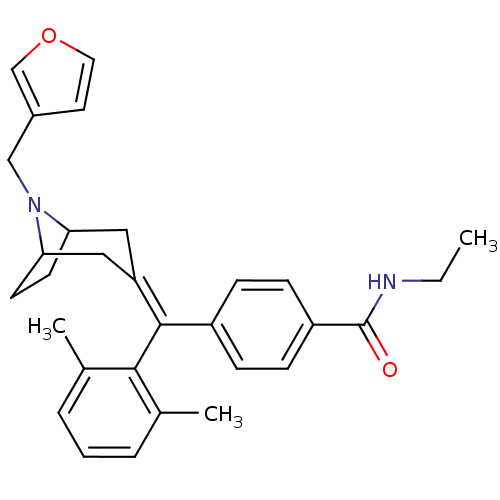
(4-[(2,6-Dimethyl-phenyl)-(8-furan-3-ylmethyl-8-aza...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1\[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1c(-[#6])cccc1-[#6] Show InChI InChI=1S/C30H34N2O2/c1-4-31-30(33)24-10-8-23(9-11-24)29(28-20(2)6-5-7-21(28)3)25-16-26-12-13-27(17-25)32(26)18-22-14-15-34-19-22/h5-11,14-15,19,26-27H,4,12-13,16-18H2,1-3H3,(H,31,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Parathyroid hormone/parathyroid hormone-related peptide receptor
(Homo sapiens (Human)) | BDBM50002913
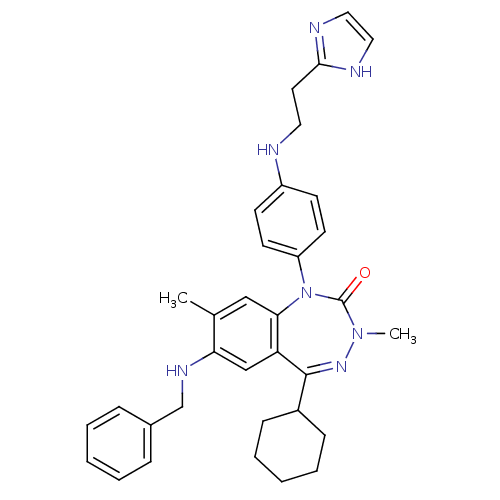
(CHEMBL389783)Show SMILES CN1N=C(C2CCCCC2)c2cc(NCc3ccccc3)c(C)cc2N(c2ccc(NCCc3ncc[nH]3)cc2)C1=O |t:2| Show InChI InChI=1S/C34H39N7O/c1-24-21-31-29(22-30(24)38-23-25-9-5-3-6-10-25)33(26-11-7-4-8-12-26)39-40(2)34(42)41(31)28-15-13-27(14-16-28)35-18-17-32-36-19-20-37-32/h3,5-6,9-10,13-16,19-22,26,35,38H,4,7-8,11-12,17-18,23H2,1-2H3,(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [125I]-[Nle,8,18 Tyr34]-hPTH(1-34) from human recombinant PTH1R expressed in HEK293 cells |
J Med Chem 50: 4789-92 (2007)
Checked by Author
Article DOI: 10.1021/jm0707626
BindingDB Entry DOI: 10.7270/Q2Z039C3 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154041
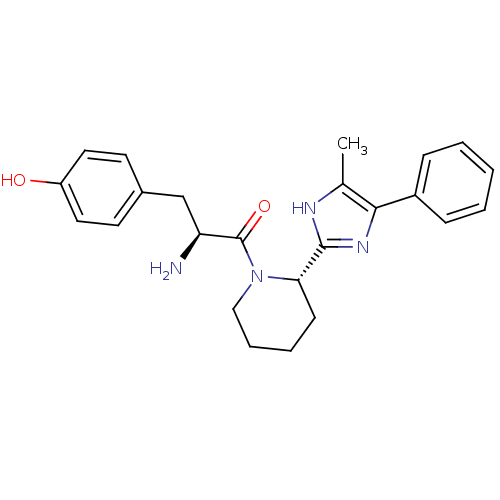
((S)-2-amino-3-(4-hydroxyphenyl)-1-((S)-2-(5-methyl...)Show SMILES Cc1[nH]c(nc1-c1ccccc1)[C@@H]1CCCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1 Show InChI InChI=1S/C24H28N4O2/c1-16-22(18-7-3-2-4-8-18)27-23(26-16)21-9-5-6-14-28(21)24(30)20(25)15-17-10-12-19(29)13-11-17/h2-4,7-8,10-13,20-21,29H,5-6,9,14-15,25H2,1H3,(H,26,27)/t20-,21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for Mu opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155077

(CHEMBL186183 | N-Ethyl-4-[(8-furan-3-ylmethyl-8-az...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc(cc1)-[#7+](-[#8-])=O Show InChI InChI=1S/C28H29N3O4/c1-2-29-28(32)22-5-3-20(4-6-22)27(21-7-9-24(10-8-21)31(33)34)23-15-25-11-12-26(16-23)30(25)17-19-13-14-35-18-19/h3-10,13-14,18,25-26H,2,11-12,15-17H2,1H3,(H,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50002877
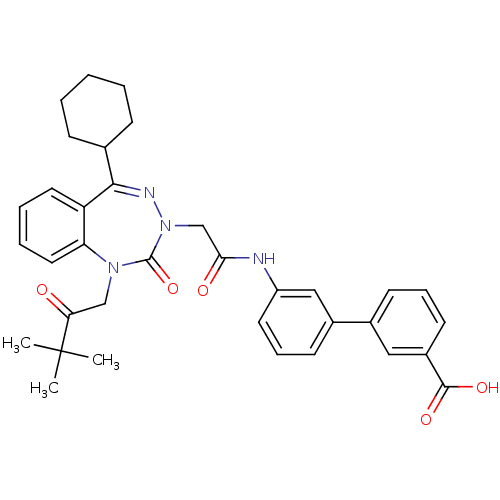
(CHEMBL388143)Show SMILES CC(C)(C)C(=O)CN1c2ccccc2C(=NN(CC(=O)Nc2cccc(c2)-c2cccc(c2)C(O)=O)C1=O)C1CCCCC1 |c:15| Show InChI InChI=1S/C35H38N4O5/c1-35(2,3)30(40)21-38-29-18-8-7-17-28(29)32(23-11-5-4-6-12-23)37-39(34(38)44)22-31(41)36-27-16-10-14-25(20-27)24-13-9-15-26(19-24)33(42)43/h7-10,13-20,23H,4-6,11-12,21-22H2,1-3H3,(H,36,41)(H,42,43) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
Displacement of [3H]BH-CCK-8S from human recombinant CCK2 receptor expressed in NIH3T3 cells |
J Med Chem 50: 3101-12 (2007)
Checked by Author
Article DOI: 10.1021/jm070139l
BindingDB Entry DOI: 10.7270/Q2QJ7JHN |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(MOUSE) | BDBM50471065
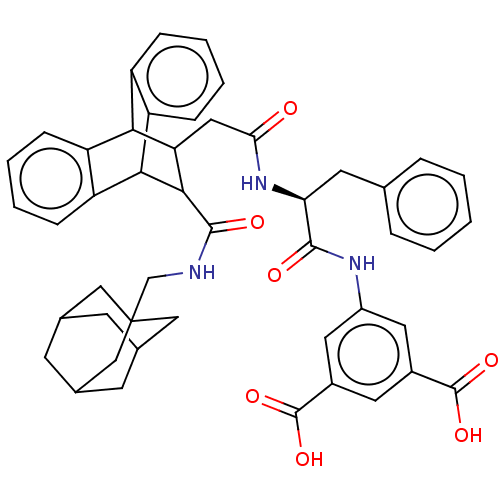
(CHEMBL296167)Show SMILES OC(=O)c1cc(NC(=O)[C@H](Cc2ccccc2)NC(=O)CC2C(C3c4ccccc4C2c2ccccc32)C(=O)NCC23CC4CC(CC(C4)C2)C3)cc(c1)C(O)=O |wD:9.9,TLB:44:45:49:43.42.48,37:22:29.24:36.31,28:29:22.21:36.31,35:36:22.21:29.24,25:24:22.21:36.31,THB:20:21:29.24:36.31,44:43:49:45.50.46,46:45:42:47.49.48,46:47:42:45.50.44,32:31:22.21:29.24,(8.01,-6.63,;9.37,-5.87,;10.71,-6.64,;9.37,-4.31,;8.05,-3.54,;8.05,-1.98,;6.95,-.88,;5.47,-.48,;5.23,1.05,;4.2,-1.35,;4.05,-2.9,;5.21,-3.96,;6.55,-3.18,;7.89,-3.96,;7.89,-5.51,;6.52,-6.29,;5.21,-5.51,;2.83,-.57,;1.51,-1.35,;1.51,-2.9,;1.18,-1.33,;-.3,-.93,;.88,-.85,;.17,-1.58,;-1.64,-1.11,;-1.36,.19,;-2.44,1.1,;-3.8,.71,;-4.09,-.59,;-3,-1.49,;-1.14,-1.86,;-.53,-3.49,;-.89,-4.71,;.13,-5.65,;1.47,-5.34,;1.8,-4.1,;.81,-3.2,;1.28,.65,;.76,2.13,;2.78,.35,;3.8,1.53,;5.02,2.46,;6.24,1.69,;7.4,2.63,;9.16,2.49,;7.89,3.4,;7.47,4.87,;5.82,4.86,;6.97,3.98,;4.6,3.92,;6.64,2.4,;9.39,-1.21,;10.74,-1.98,;10.74,-3.54,;12.08,-1.22,;12.1,.33,;13.42,-2.01,)| Show InChI InChI=1S/C47H47N3O7/c51-39(50-38(17-26-8-2-1-3-9-26)43(52)49-32-19-30(45(54)55)18-31(20-32)46(56)57)21-37-40-33-10-4-6-12-35(33)41(36-13-7-5-11-34(36)40)42(37)44(53)48-25-47-22-27-14-28(23-47)16-29(15-27)24-47/h1-13,18-20,27-29,37-38,40-42H,14-17,21-25H2,(H,48,53)(H,49,52)(H,50,51)(H,54,55)(H,56,57)/t27?,28?,29?,37?,38-,40?,41?,42?,47?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
James Black Foundation
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against Cholecystokinin type B receptor using [125I]BH-CCK-8S as radioligand in mouse cortical membranes |
J Med Chem 39: 1806-15 (1996)
Article DOI: 10.1021/jm9508907
BindingDB Entry DOI: 10.7270/Q2571FR6 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155107
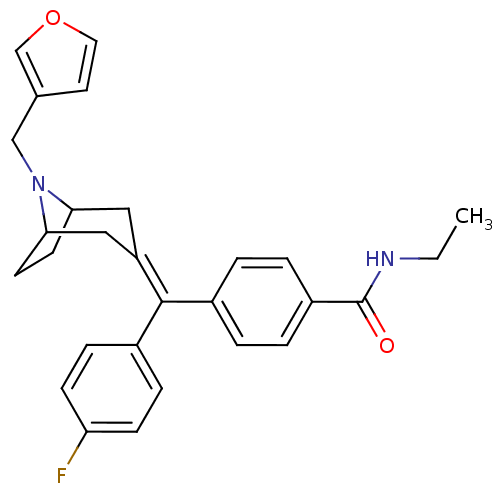
(CHEMBL185229 | N-Ethyl-4-[(4-fluoro-phenyl)-(8-fur...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccoc1)\c1ccc(F)cc1 Show InChI InChI=1S/C28H29FN2O2/c1-2-30-28(32)22-5-3-20(4-6-22)27(21-7-9-24(29)10-8-21)23-15-25-11-12-26(16-23)31(25)17-19-13-14-33-18-19/h3-10,13-14,18,25-26H,2,11-12,15-17H2,1H3,(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data