 Found 360 hits with Last Name = 'tsantrizos' and Initial = 'ys'
Found 360 hits with Last Name = 'tsantrizos' and Initial = 'ys' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM25308
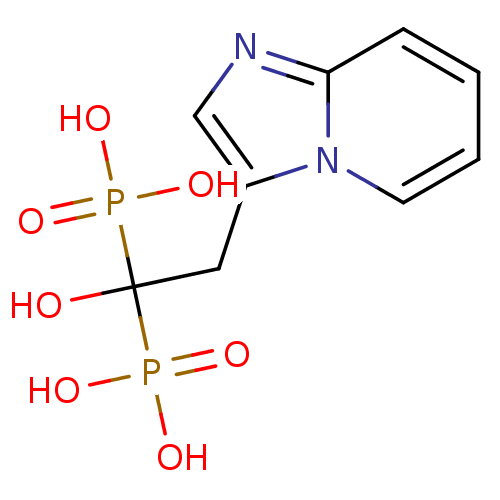
((1-hydroxy-2-{imidazo[1,2-a]pyridin-3-yl}-1-phosph...)Show InChI InChI=1S/C9H12N2O7P2/c12-9(19(13,14)15,20(16,17)18)5-7-6-10-8-3-1-2-4-11(7)8/h1-4,6,12H,5H2,(H2,13,14,15)(H2,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human FPPS |
Bioorg Med Chem Lett 25: 1117-23 (2015)
Article DOI: 10.1016/j.bmcl.2014.12.089
BindingDB Entry DOI: 10.7270/Q25X2BMF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12578
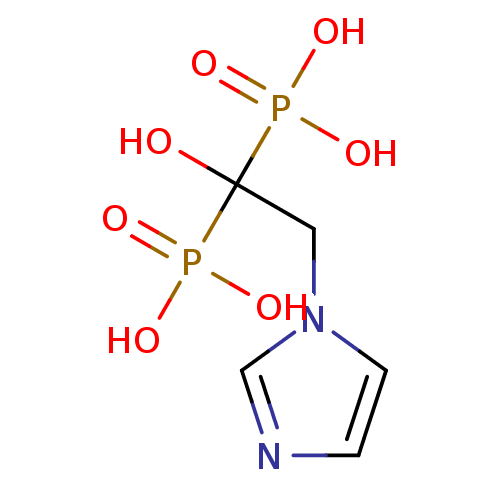
(2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosp...)Show InChI InChI=1S/C5H10N2O7P2/c8-5(15(9,10)11,16(12,13)14)3-7-2-1-6-4-7/h1-2,4,8H,3H2,(H2,9,10,11)(H2,12,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal His-tagged human FPPS (1 to 353 residues) expressed in Escherichia coli BL21 (DE3) pre-incubated for 10 mins before addition... |
J Med Chem 62: 9691-9702 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01104
BindingDB Entry DOI: 10.7270/Q23T9MJV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12578

(2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosp...)Show InChI InChI=1S/C5H10N2O7P2/c8-5(15(9,10)11,16(12,13)14)3-7-2-1-6-4-7/h1-2,4,8H,3H2,(H2,9,10,11)(H2,12,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50098378
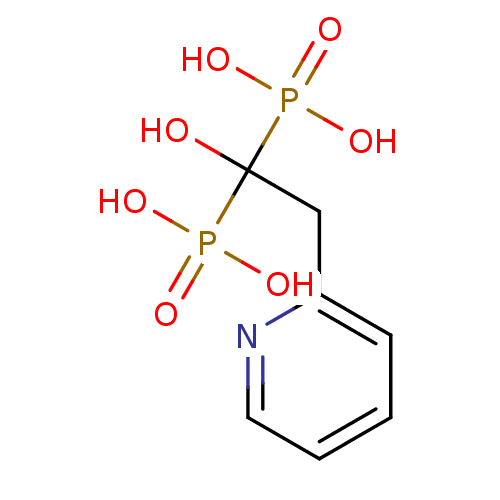
((1-Hydroxy-1-phosphono-2-pyridin-2-yl-ethyl)-phosp...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)5-6-3-1-2-4-8-6/h1-4,9H,5H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FPPS expressed in Escherichia coli BL21 (DE3) preincubated for 10 mins in presence compound relative to control |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50098378

((1-Hydroxy-1-phosphono-2-pyridin-2-yl-ethyl)-phosp...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)5-6-3-1-2-4-8-6/h1-4,9H,5H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human FPPS using [14C]-IPP and FPP as substrates after 10 mins by scintillation counting |
J Med Chem 61: 6904-6917 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00886
BindingDB Entry DOI: 10.7270/Q2CJ8HWF |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12578

(2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosp...)Show InChI InChI=1S/C5H10N2O7P2/c8-5(15(9,10)11,16(12,13)14)3-7-2-1-6-4-7/h1-2,4,8H,3H2,(H2,9,10,11)(H2,12,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human FPPS |
Bioorg Med Chem Lett 25: 1117-23 (2015)
Article DOI: 10.1016/j.bmcl.2014.12.089
BindingDB Entry DOI: 10.7270/Q25X2BMF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12578

(2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosp...)Show InChI InChI=1S/C5H10N2O7P2/c8-5(15(9,10)11,16(12,13)14)3-7-2-1-6-4-7/h1-2,4,8H,3H2,(H2,9,10,11)(H2,12,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [3H]IPP and GPP as substrate incubated for 10 mi... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576
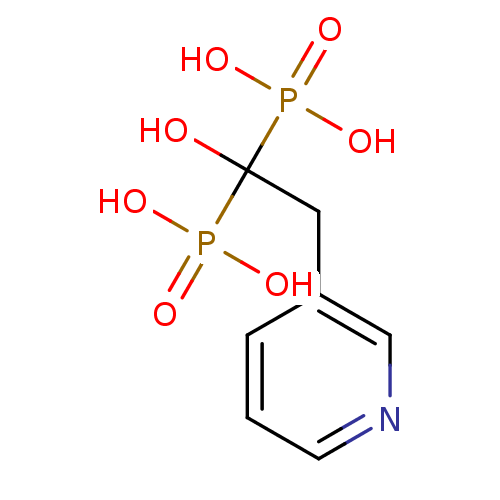
(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576

(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443050
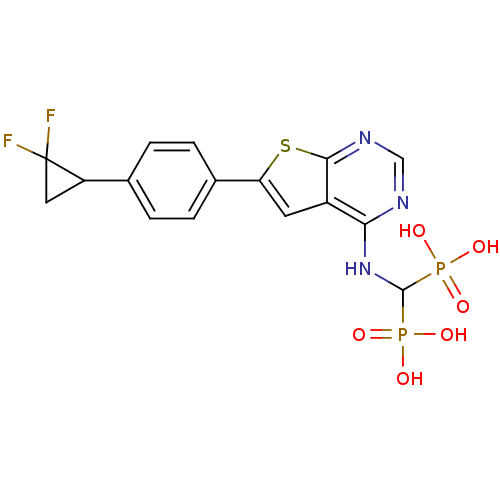
(CHEMBL3087938)Show SMILES OP(O)(=O)C(Nc1ncnc2sc(cc12)-c1ccc(cc1)C1CC1(F)F)P(O)(O)=O Show InChI InChI=1S/C16H15F2N3O6P2S/c17-16(18)6-11(16)8-1-3-9(4-2-8)12-5-10-13(19-7-20-14(10)30-12)21-15(28(22,23)24)29(25,26)27/h1-5,7,11,15H,6H2,(H,19,20,21)(H2,22,23,24)(H2,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576

(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human FPPS |
Bioorg Med Chem Lett 25: 1117-23 (2015)
Article DOI: 10.1016/j.bmcl.2014.12.089
BindingDB Entry DOI: 10.7270/Q25X2BMF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576

(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [14C]IPP and GPP as substrate incubated for 10 m... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576

(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition by scintillation counting analy... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM12576

(Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...)Show InChI InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human His6-tagged recombinant FPPS expressed in Escherichia coli BL21(DE3) using GPP and [3H]IPP as substrate incubated for 5 mins prio... |
Bioorg Med Chem 20: 5583-91 (2012)
Article DOI: 10.1016/j.bmc.2012.07.019
BindingDB Entry DOI: 10.7270/Q2J67J6M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50421091

(CHEMBL2088336)Show SMILES OP(O)(=O)C(Nc1cc(ccn1)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C15H18N2O7P2/c18-25(19,20)15(26(21,22)23)17-14-9-11(7-8-16-14)10-1-3-12(4-2-10)24-13-5-6-13/h1-4,7-9,13,15H,5-6H2,(H,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human His6-tagged recombinant FPPS expressed in Escherichia coli BL21(DE3) using GPP and [3H]IPP as substrate incubated for 5 mins prio... |
Bioorg Med Chem 20: 5583-91 (2012)
Article DOI: 10.1016/j.bmc.2012.07.019
BindingDB Entry DOI: 10.7270/Q2J67J6M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443052
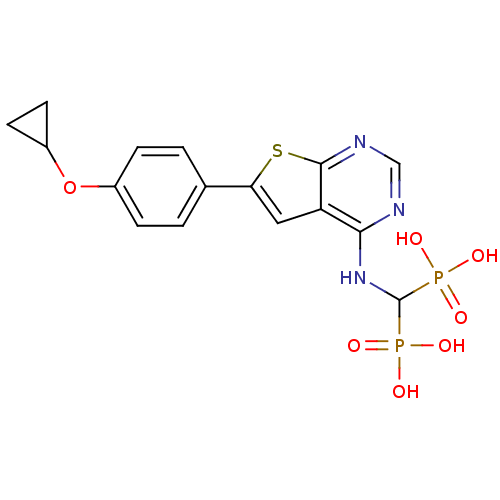
(CHEMBL3087936 | US11279719, Example C-13)Show SMILES OP(O)(=O)C(Nc1ncnc2sc(cc12)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C16H17N3O7P2S/c20-27(21,22)16(28(23,24)25)19-14-12-7-13(29-15(12)18-8-17-14)9-1-3-10(4-2-9)26-11-5-6-11/h1-4,7-8,11,16H,5-6H2,(H,17,18,19)(H2,20,21,22)(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443052

(CHEMBL3087936 | US11279719, Example C-13)Show SMILES OP(O)(=O)C(Nc1ncnc2sc(cc12)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C16H17N3O7P2S/c20-27(21,22)16(28(23,24)25)19-14-12-7-13(29-15(12)18-8-17-14)9-1-3-10(4-2-9)26-11-5-6-11/h1-4,7-8,11,16H,5-6H2,(H,17,18,19)(H2,20,21,22)(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585603

(CHEMBL5087030)Show SMILES Cc1ccc(NC(=O)c2cccc(c2)-c2nc(NC(P(O)(O)=O)P(O)(O)=O)c3ccsc3n2)cc1Br | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443055
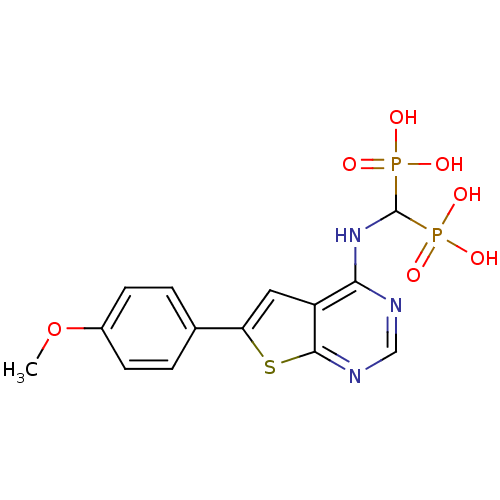
(CHEMBL3087933)Show SMILES COc1ccc(cc1)-c1cc2c(NC(P(O)(O)=O)P(O)(O)=O)ncnc2s1 Show InChI InChI=1S/C14H15N3O7P2S/c1-24-9-4-2-8(3-5-9)11-6-10-12(15-7-16-13(10)27-11)17-14(25(18,19)20)26(21,22)23/h2-7,14H,1H3,(H,15,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50421094
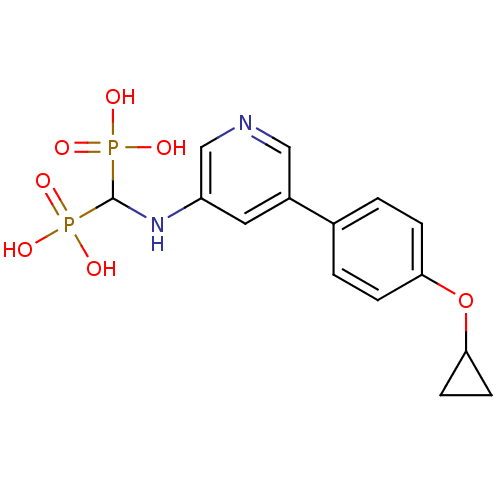
(CHEMBL2088339)Show SMILES OP(O)(=O)C(Nc1cncc(c1)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C15H18N2O7P2/c18-25(19,20)15(26(21,22)23)17-12-7-11(8-16-9-12)10-1-3-13(4-2-10)24-14-5-6-14/h1-4,7-9,14-15,17H,5-6H2,(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition by scintillation counting analy... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50421094

(CHEMBL2088339)Show SMILES OP(O)(=O)C(Nc1cncc(c1)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C15H18N2O7P2/c18-25(19,20)15(26(21,22)23)17-12-7-11(8-16-9-12)10-1-3-13(4-2-10)24-14-5-6-14/h1-4,7-9,14-15,17H,5-6H2,(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human His6-tagged recombinant FPPS expressed in Escherichia coli BL21(DE3) using GPP and [3H]IPP as substrate incubated for 5 mins prio... |
Bioorg Med Chem 20: 5583-91 (2012)
Article DOI: 10.1016/j.bmc.2012.07.019
BindingDB Entry DOI: 10.7270/Q2J67J6M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585613
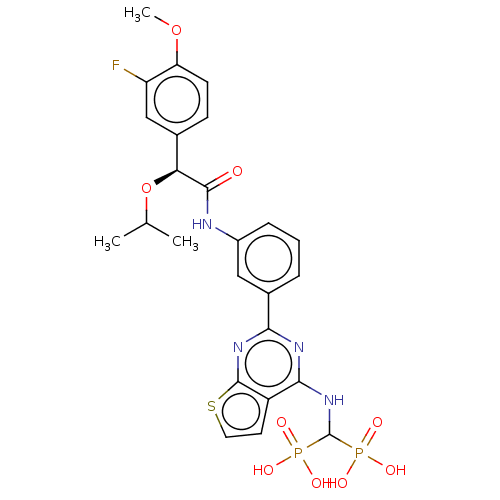
(CHEMBL5088349)Show SMILES COc1ccc(cc1F)[C@H](OC(C)C)C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM544004
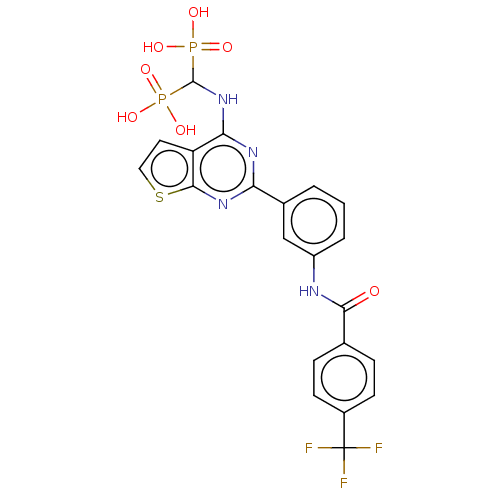
(US11279719, Example I-36)Show SMILES OP(O)(=O)C(Nc1nc(nc2sccc12)-c1cccc(NC(=O)c2ccc(cc2)C(F)(F)F)c1)P(O)(O)=O | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50421094

(CHEMBL2088339)Show SMILES OP(O)(=O)C(Nc1cncc(c1)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C15H18N2O7P2/c18-25(19,20)15(26(21,22)23)17-12-7-11(8-16-9-12)10-1-3-13(4-2-10)24-14-5-6-14/h1-4,7-9,14-15,17H,5-6H2,(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50421094

(CHEMBL2088339)Show SMILES OP(O)(=O)C(Nc1cncc(c1)-c1ccc(OC2CC2)cc1)P(O)(O)=O Show InChI InChI=1S/C15H18N2O7P2/c18-25(19,20)15(26(21,22)23)17-12-7-11(8-16-9-12)10-1-3-13(4-2-10)24-14-5-6-14/h1-4,7-9,14-15,17H,5-6H2,(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50520641

(CHEMBL4455060 | US11279719, Example I-13)Show SMILES OP(O)(=O)C(Nc1nc(nc2sccc12)-c1cccc(NC(=O)c2ccccc2)c1)P(O)(O)=O Show InChI InChI=1S/C20H18N4O7P2S/c25-18(12-5-2-1-3-6-12)21-14-8-4-7-13(11-14)16-22-17(15-9-10-34-19(15)23-16)24-20(32(26,27)28)33(29,30)31/h1-11,20H,(H,21,25)(H,22,23,24)(H2,26,27,28)(H2,29,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585614

(CHEMBL5082193)Show SMILES COc1ccc(cc1F)[C@@H](OC(C)C)C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443054
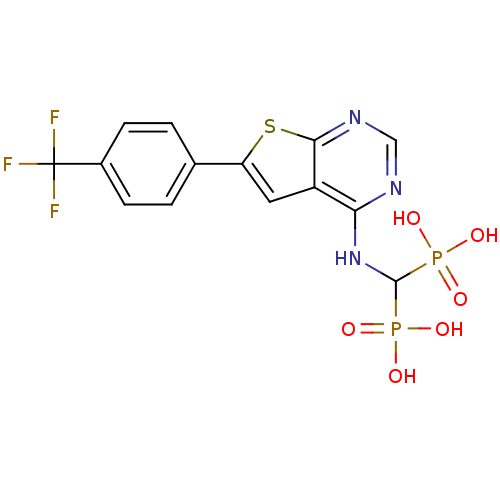
(CHEMBL3087934 | US11279719, Example C-12)Show SMILES OP(O)(=O)C(Nc1ncnc2sc(cc12)-c1ccc(cc1)C(F)(F)F)P(O)(O)=O Show InChI InChI=1S/C14H12F3N3O6P2S/c15-14(16,17)8-3-1-7(2-4-8)10-5-9-11(18-6-19-12(9)29-10)20-13(27(21,22)23)28(24,25)26/h1-6,13H,(H,18,19,20)(H2,21,22,23)(H2,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585601
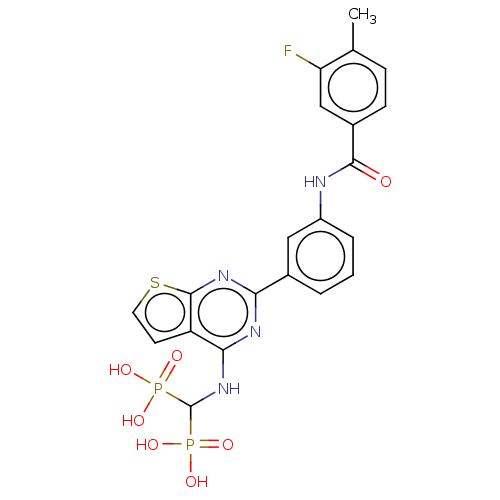
(CHEMBL5077349)Show SMILES Cc1ccc(cc1F)C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585611
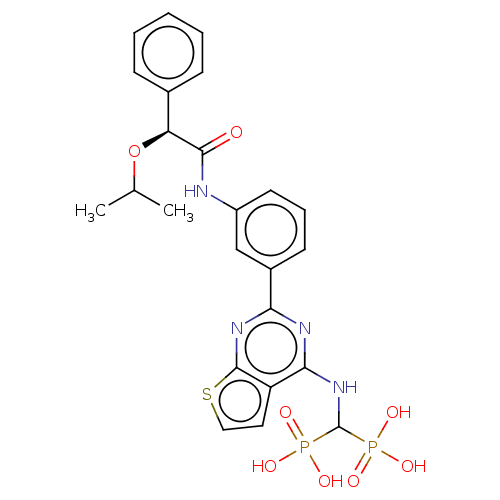
(CHEMBL5077117)Show SMILES CC(C)O[C@H](C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50432306
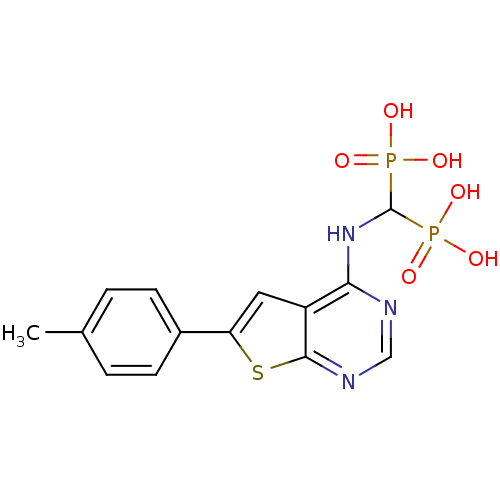
(CHEMBL2347862)Show SMILES Cc1ccc(cc1)-c1cc2c(NC(P(O)(O)=O)P(O)(O)=O)ncnc2s1 Show InChI InChI=1S/C14H15N3O6P2S/c1-8-2-4-9(5-3-8)11-6-10-12(15-7-16-13(10)26-11)17-14(24(18,19)20)25(21,22)23/h2-7,14H,1H3,(H,15,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FPPS using GPP/[3H]IPP as substrate incubated for 10 mins prior to substrate addition measured after 8 mins by scinti... |
J Med Chem 56: 7939-50 (2013)
Article DOI: 10.1021/jm400946f
BindingDB Entry DOI: 10.7270/Q20V8F6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50432306

(CHEMBL2347862)Show SMILES Cc1ccc(cc1)-c1cc2c(NC(P(O)(O)=O)P(O)(O)=O)ncnc2s1 Show InChI InChI=1S/C14H15N3O6P2S/c1-8-2-4-9(5-3-8)11-6-10-12(15-7-16-13(10)26-11)17-14(24(18,19)20)25(21,22)23/h2-7,14H,1H3,(H,15,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50443054

(CHEMBL3087934 | US11279719, Example C-12)Show SMILES OP(O)(=O)C(Nc1ncnc2sc(cc12)-c1ccc(cc1)C(F)(F)F)P(O)(O)=O Show InChI InChI=1S/C14H12F3N3O6P2S/c15-14(16,17)8-3-1-7(2-4-8)10-5-9-11(18-6-19-12(9)29-10)20-13(27(21,22)23)28(24,25)26/h1-6,13H,(H,18,19,20)(H2,21,22,23)(H2,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human FPPS using [14C]-IPP and FPP as substrates after 10 mins by scintillation counting |
J Med Chem 61: 6904-6917 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00886
BindingDB Entry DOI: 10.7270/Q2CJ8HWF |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585612

(CHEMBL5089506)Show SMILES CC(C)O[C@@H](C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1)c1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585606
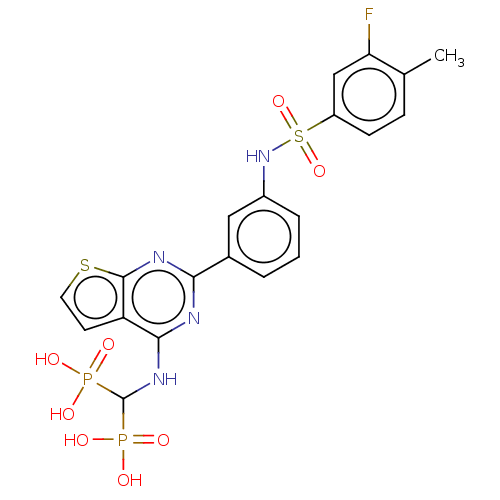
(CHEMBL5085126)Show SMILES Cc1ccc(cc1F)S(=O)(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585604

(CHEMBL5083916)Show SMILES COc1ccc(NS(=O)(=O)c2cccc(c2)-c2nc(NC(P(O)(O)=O)P(O)(O)=O)c3ccsc3n2)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585608
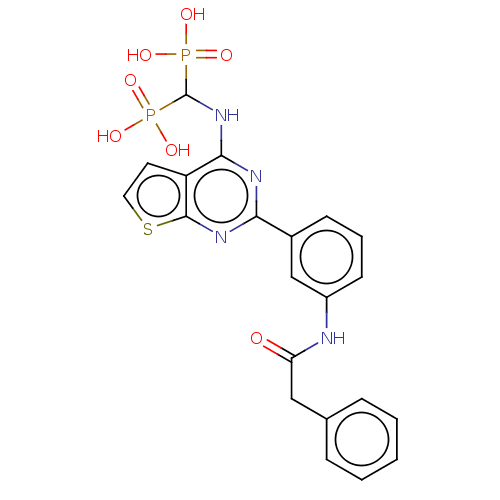
(CHEMBL5075557)Show SMILES OP(O)(=O)C(Nc1nc(nc2sccc12)-c1cccc(NC(=O)Cc2ccccc2)c1)P(O)(O)=O | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585605

(CHEMBL5080782)Show SMILES COc1ccc(cc1F)S(=O)(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50520640
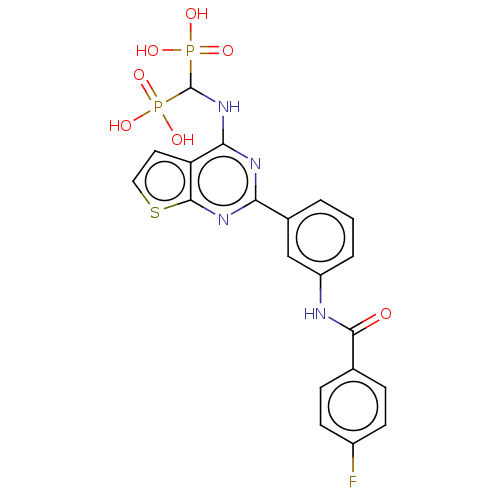
(CHEMBL4472025 | US11279719, Example I-34)Show SMILES OP(O)(=O)C(Nc1nc(nc2sccc12)-c1cccc(NC(=O)c2ccc(F)cc2)c1)P(O)(O)=O Show InChI InChI=1S/C20H17FN4O7P2S/c21-13-6-4-11(5-7-13)18(26)22-14-3-1-2-12(10-14)16-23-17(15-8-9-35-19(15)24-16)25-20(33(27,28)29)34(30,31)32/h1-10,20H,(H,22,26)(H,23,24,25)(H2,27,28,29)(H2,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50520637
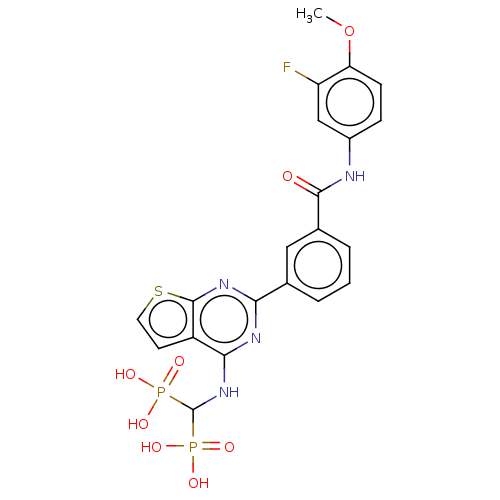
(CHEMBL4577077 | US11279719, Example I-37)Show SMILES COc1ccc(NC(=O)c2cccc(c2)-c2nc(NC(P(O)(O)=O)P(O)(O)=O)c3ccsc3n2)cc1F Show InChI InChI=1S/C21H19FN4O8P2S/c1-34-16-6-5-13(10-15(16)22)23-19(27)12-4-2-3-11(9-12)17-24-18(14-7-8-37-20(14)25-17)26-21(35(28,29)30)36(31,32)33/h2-10,21H,1H3,(H,23,27)(H,24,25,26)(H2,28,29,30)(H2,31,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50386555
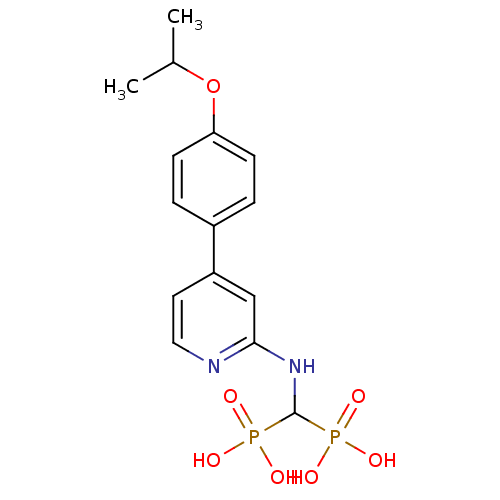
(CHEMBL2048241)Show SMILES CC(C)Oc1ccc(cc1)-c1ccnc(NC(P(O)(O)=O)P(O)(O)=O)c1 Show InChI InChI=1S/C15H20N2O7P2/c1-10(2)24-13-5-3-11(4-6-13)12-7-8-16-14(9-12)17-15(25(18,19)20)26(21,22)23/h3-10,15H,1-2H3,(H,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [14C]IPP and GPP as substrate incubated for 10 m... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50386555

(CHEMBL2048241)Show SMILES CC(C)Oc1ccc(cc1)-c1ccnc(NC(P(O)(O)=O)P(O)(O)=O)c1 Show InChI InChI=1S/C15H20N2O7P2/c1-10(2)24-13-5-3-11(4-6-13)12-7-8-16-14(9-12)17-15(25(18,19)20)26(21,22)23/h3-10,15H,1-2H3,(H,16,17)(H2,18,19,20)(H2,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human His6-tagged recombinant FPPS expressed in Escherichia coli BL21(DE3) using GPP and [3H]IPP as substrate incubated for 5 mins prio... |
Bioorg Med Chem 20: 5583-91 (2012)
Article DOI: 10.1016/j.bmc.2012.07.019
BindingDB Entry DOI: 10.7270/Q2J67J6M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585599
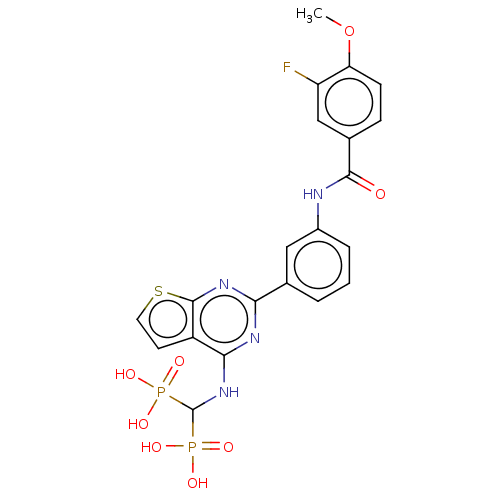
(CHEMBL5088555)Show SMILES COc1ccc(cc1F)C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585602
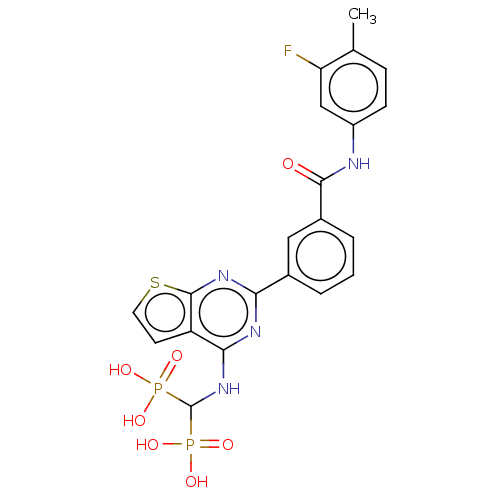
(CHEMBL5075029)Show SMILES Cc1ccc(NC(=O)c2cccc(c2)-c2nc(NC(P(O)(O)=O)P(O)(O)=O)c3ccsc3n2)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50386558
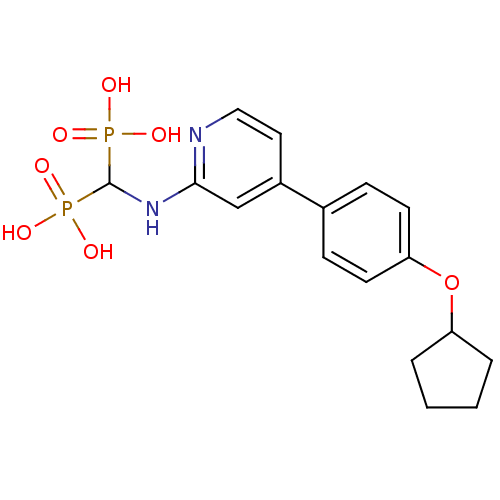
(CHEMBL2048244)Show SMILES OP(O)(=O)C(Nc1cc(ccn1)-c1ccc(OC2CCCC2)cc1)P(O)(O)=O Show InChI InChI=1S/C17H22N2O7P2/c20-27(21,22)17(28(23,24)25)19-16-11-13(9-10-18-16)12-5-7-15(8-6-12)26-14-3-1-2-4-14/h5-11,14,17H,1-4H2,(H,18,19)(H2,20,21,22)(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [14C]IPP and GPP as substrate incubated for 10 m... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50138725

((1-phosphono-2-pyridin-3-yl-ethyl)-phosphonic acid...)Show InChI InChI=1S/C7H11NO6P2/c9-15(10,11)7(16(12,13)14)4-6-2-1-3-8-5-6/h1-3,5,7H,4H2,(H2,9,10,11)(H2,12,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Allosteric inhibition of human recombinant FPPS using GPP and [3H]IPP as substrate incubated with enzyme for 10 mins prior to substrate addition by l... |
J Med Chem 57: 5764-76 (2014)
Article DOI: 10.1021/jm500629e
BindingDB Entry DOI: 10.7270/Q2GH9KJR |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585607
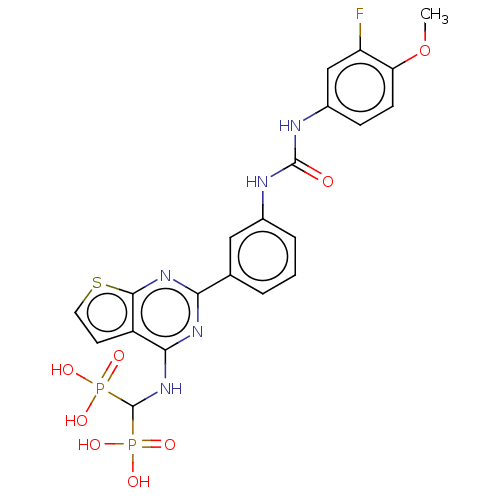
(CHEMBL5080121)Show SMILES COc1ccc(NC(=O)Nc2cccc(c2)-c2nc(NC(P(O)(O)=O)P(O)(O)=O)c3ccsc3n2)cc1F | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50386557

(CHEMBL2048243)Show SMILES Cc1ccc(cc1)-c1ccnc(NC(P(O)(O)=O)P(O)(O)=O)c1 Show InChI InChI=1S/C13H16N2O6P2/c1-9-2-4-10(5-3-9)11-6-7-14-12(8-11)15-13(22(16,17)18)23(19,20)21/h2-8,13H,1H3,(H,14,15)(H2,16,17,18)(H2,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [14C]IPP and GPP as substrate incubated for 10 m... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | |
Farnesyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50386556
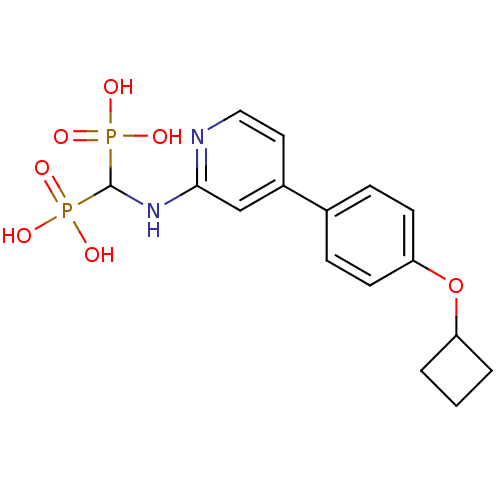
(CHEMBL2048242)Show SMILES OP(O)(=O)C(Nc1cc(ccn1)-c1ccc(OC2CCC2)cc1)P(O)(O)=O Show InChI InChI=1S/C16H20N2O7P2/c19-26(20,21)16(27(22,23)24)18-15-10-12(8-9-17-15)11-4-6-14(7-5-11)25-13-2-1-3-13/h4-10,13,16H,1-3H2,(H,17,18)(H2,19,20,21)(H2,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminal-His6 tagged FPPS expressed in Escherichia coli BL21 using [14C]IPP and GPP as substrate incubated for 10 m... |
J Med Chem 55: 3201-15 (2012)
Article DOI: 10.1021/jm201657x
BindingDB Entry DOI: 10.7270/Q29024V1 |
More data for this
Ligand-Target Pair | |
Geranylgeranyl pyrophosphate synthase
(Homo sapiens (Human)) | BDBM50585600

(CHEMBL5092247)Show SMILES COc1ccc(cc1Br)C(=O)Nc1cccc(c1)-c1nc(NC(P(O)(O)=O)P(O)(O)=O)c2ccsc2n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N- terminal His-tagged /TEV cleavage site fused GGPPS (1 to 300 residues) expressed in Escherichia coli BL21 (DE3) us... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01913
BindingDB Entry DOI: 10.7270/Q28K7F00 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data