

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296336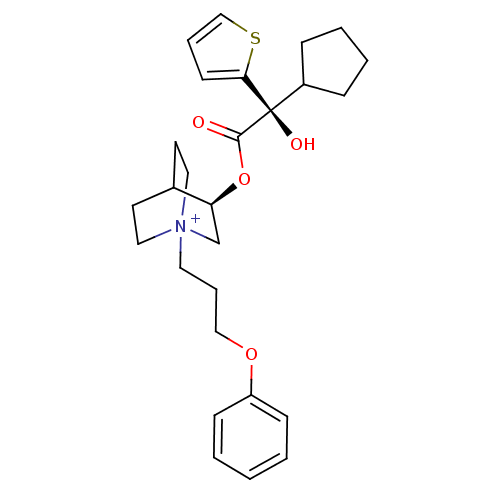 ((3R)-3-{[(2S)-2-Cyclopentyl-2-hydroxy-2-(2-thienyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M2 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296331 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(3-phe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Similars | DrugBank Article PubMed | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M3 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296336 ((3R)-3-{[(2S)-2-Cyclopentyl-2-hydroxy-2-(2-thienyl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M3 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296331 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(3-phe...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M1 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296329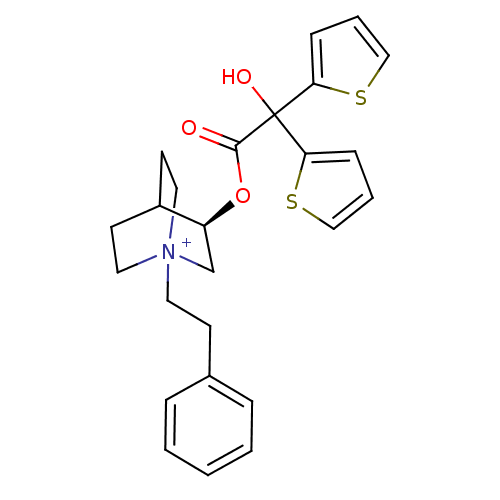 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(2-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M2 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296329 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(2-phe...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M1 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296331 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(3-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M2 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296329 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(2-phe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M3 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296336 ((3R)-3-{[(2S)-2-Cyclopentyl-2-hydroxy-2-(2-thienyl...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M1 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M2 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M1 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50296345 ((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M3 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50378083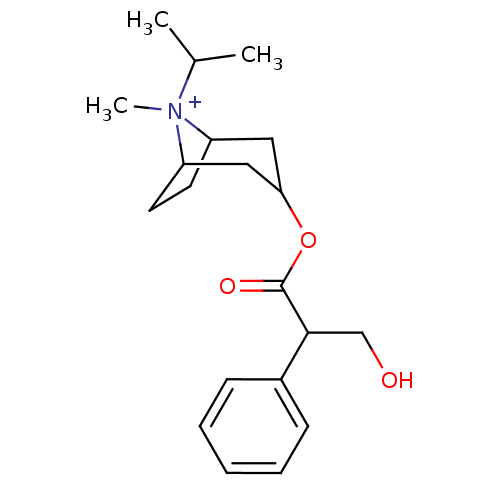 (Atrovent HFA | IPRATROPIUM BROMIDE) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Similars | Article PubMed | 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M3 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Homo sapiens (Human)) | BDBM50378083 (Atrovent HFA | IPRATROPIUM BROMIDE) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Similars | Article PubMed | 1.02 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M2 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50378083 (Atrovent HFA | IPRATROPIUM BROMIDE) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Similars | Article PubMed | 1.51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Binding affinity to muscarinic M1 receptor | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199699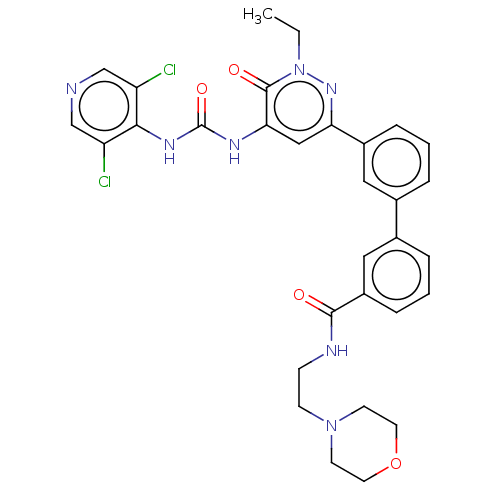 (CHEMBL3986586) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50353707 (CHEMBL1830791) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human PDE4B1 assessed as inhibition of [3H]cAMP hydrolysis to [3H]AMP after 15 mins by scintillation proximity assay | Eur J Med Chem 46: 4946-56 (2011) Article DOI: 10.1016/j.ejmech.2011.07.054 BindingDB Entry DOI: 10.7270/Q2VM4CPM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199727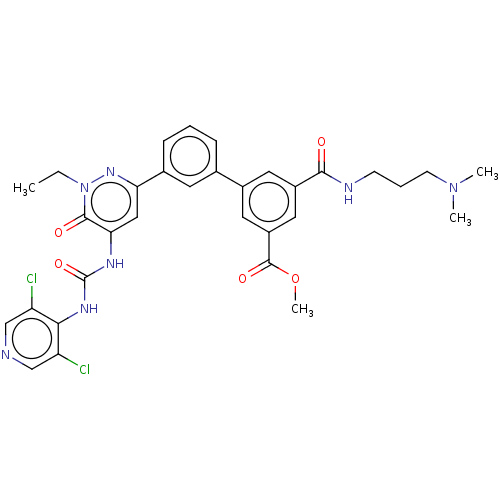 (CHEMBL3913876) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199695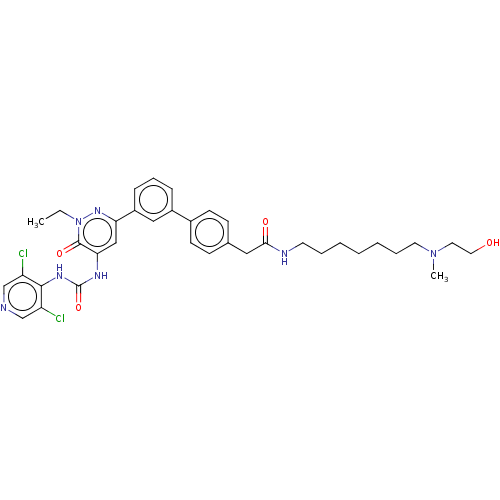 (CHEMBL3957127) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199728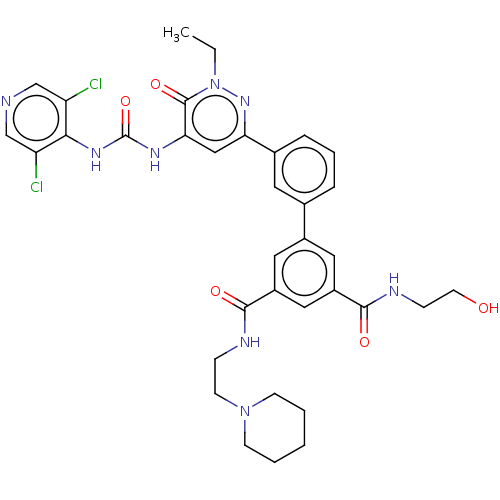 (CHEMBL3919325) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199702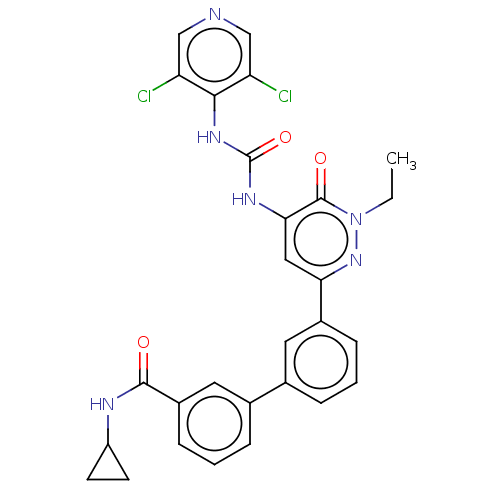 (CHEMBL3952586) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199701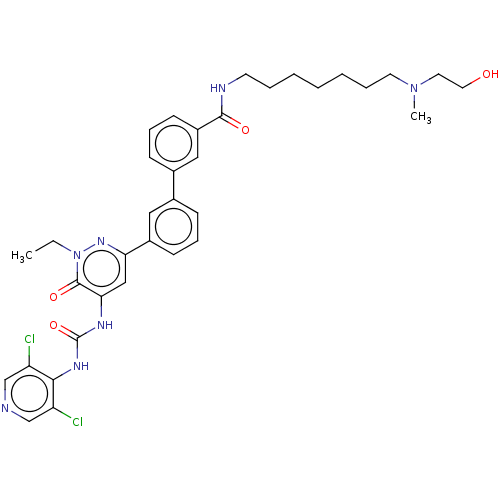 (CHEMBL3968147) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199710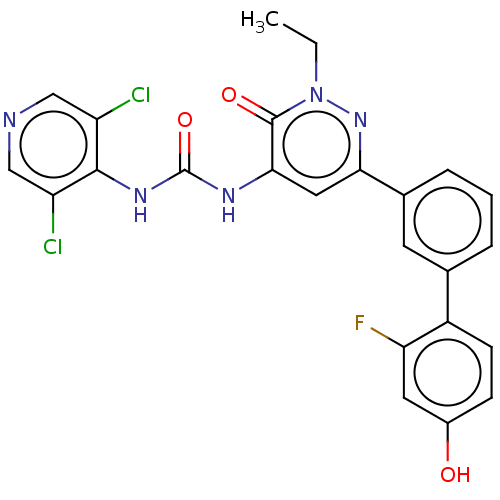 (CHEMBL3974515) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199722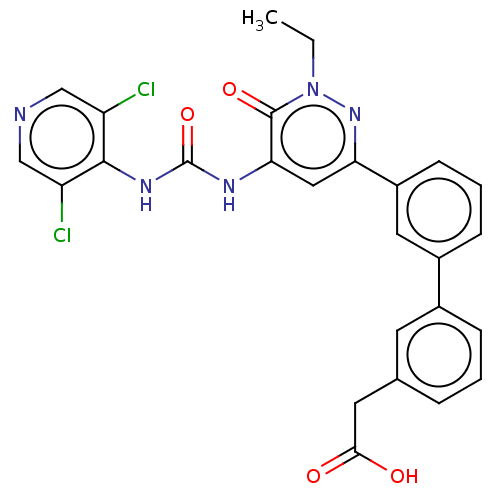 (CHEMBL3950249) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50256195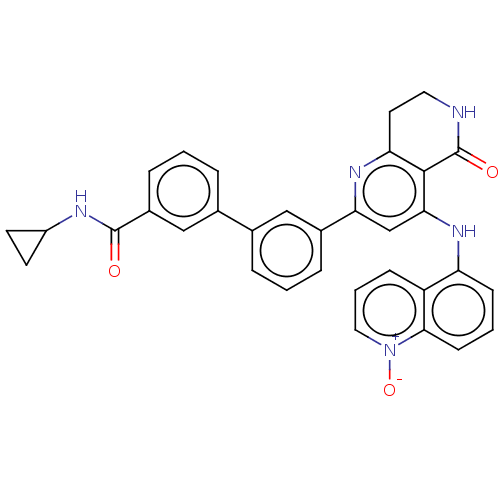 (CHEMBL4095320) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0450 | n/a | n/a | n/a | n/a | n/a | n/a |
Medicinal Chemistry& Screening ,?Pharmacokinetics& Metabolism , and�Experimental Dermatology , Almirall S.A., Centro de Investigaci�n y Desarrollo , Crta. Laure� Mir� 408-410 , Sant Feliu de Curated by ChEMBL | Assay Description Inhibition of platelet-derived growth factor receptor beta phosphorylation in MG63 cells in the absence of plasma | J Med Chem 61: 2472-2489 (2018) Article DOI: 10.1021/acs.jmedchem.7b01751 BindingDB Entry DOI: 10.7270/Q2HQ42BB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199712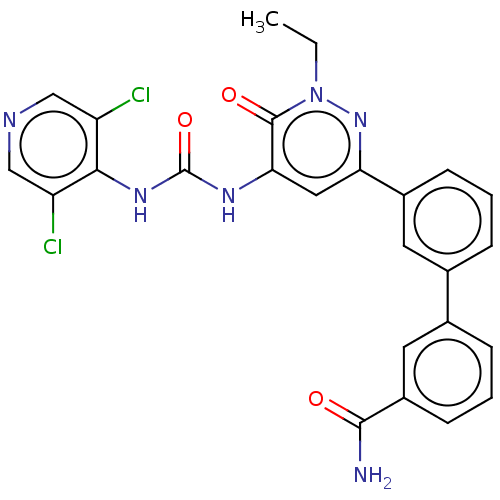 (CHEMBL3958944) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199697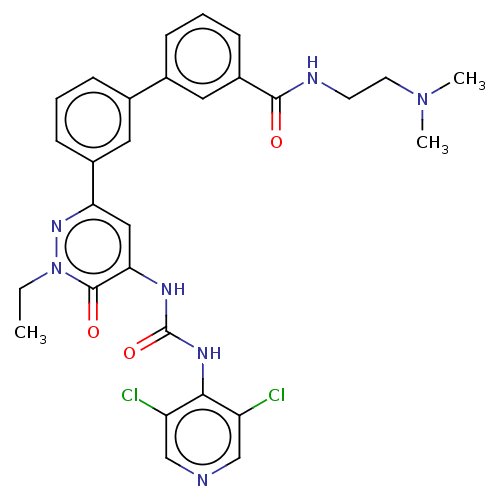 (CHEMBL3980193) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199618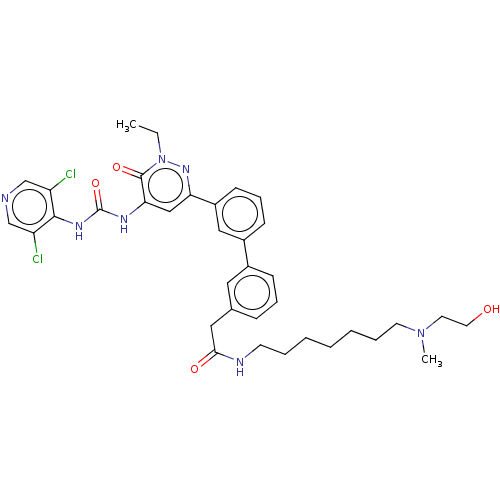 (CHEMBL3896989) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199694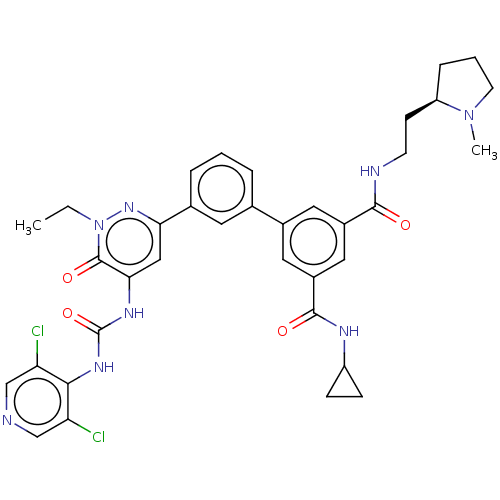 (CHEMBL3945895) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199700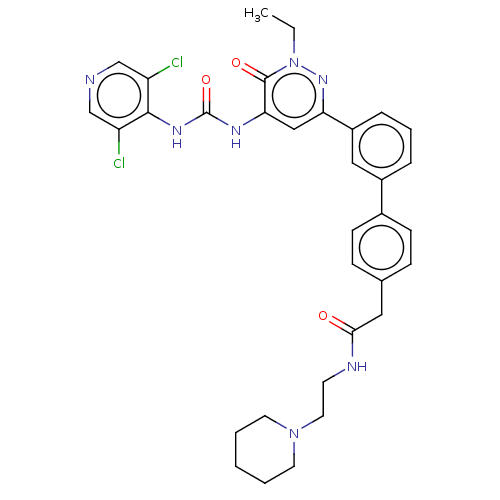 (CHEMBL3905929) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199726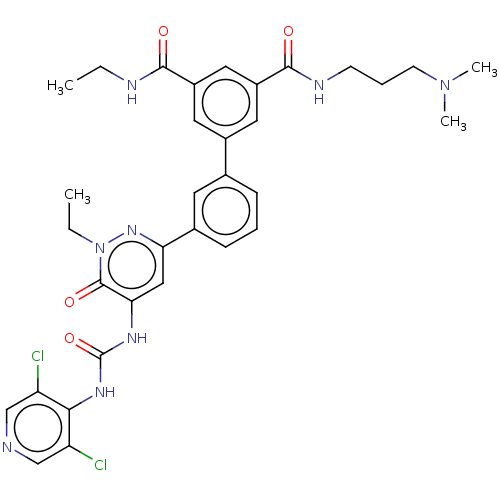 (CHEMBL3941862) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199617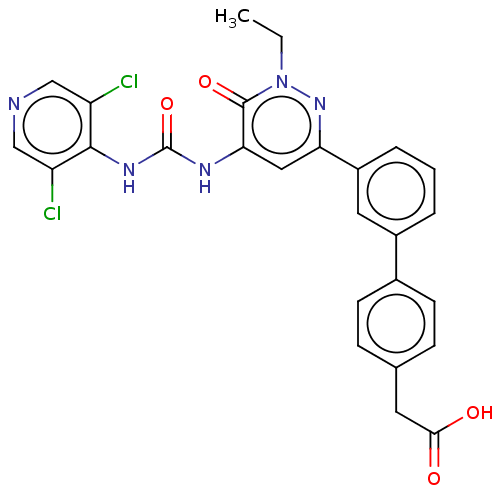 (CHEMBL3892121) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199719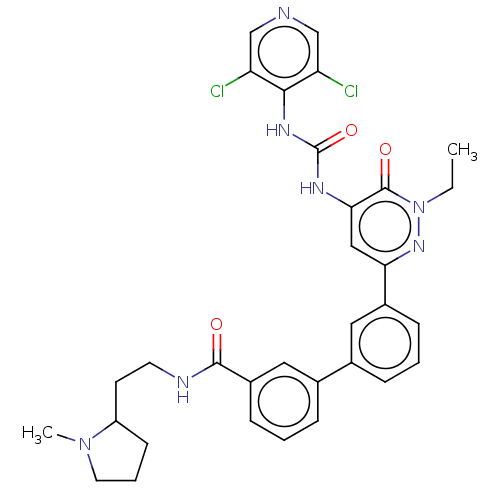 (CHEMBL3918173) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199729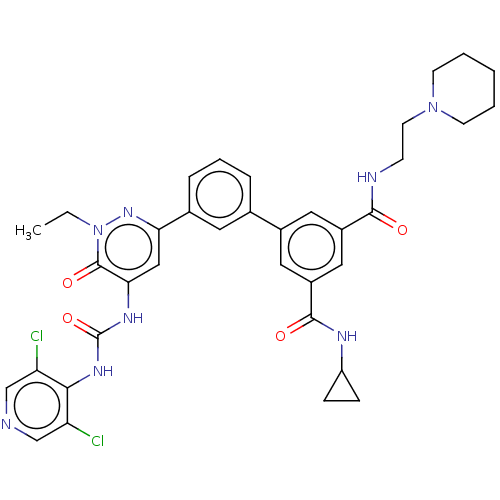 (CHEMBL3922464) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199707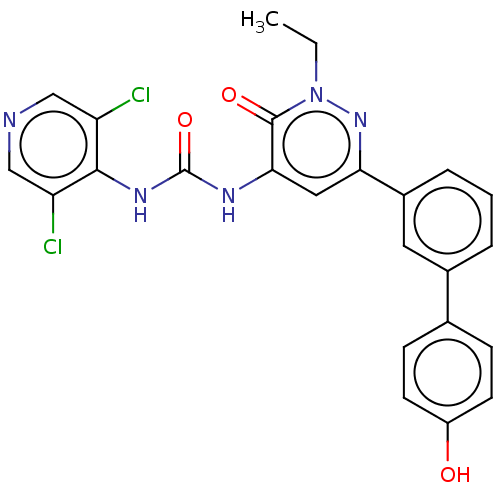 (CHEMBL3889823) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199703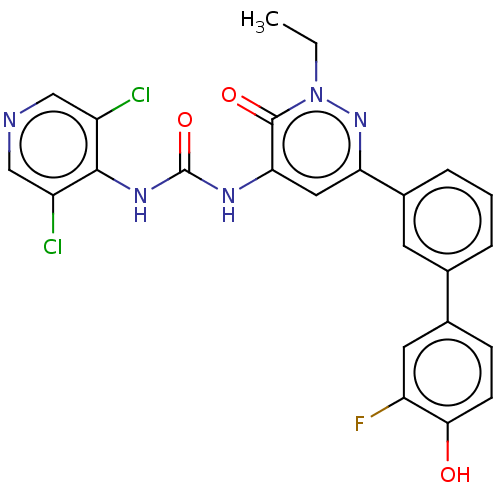 (CHEMBL3920544) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199716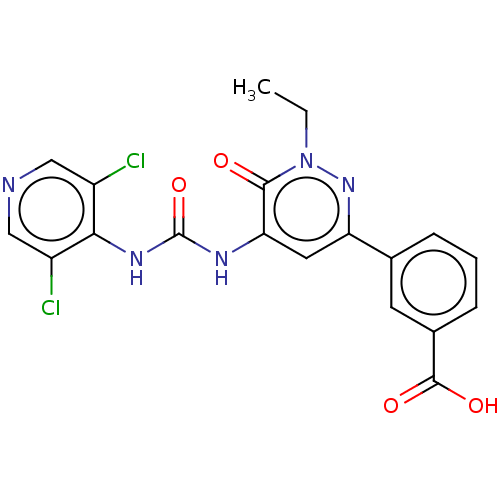 (CHEMBL3963331) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199730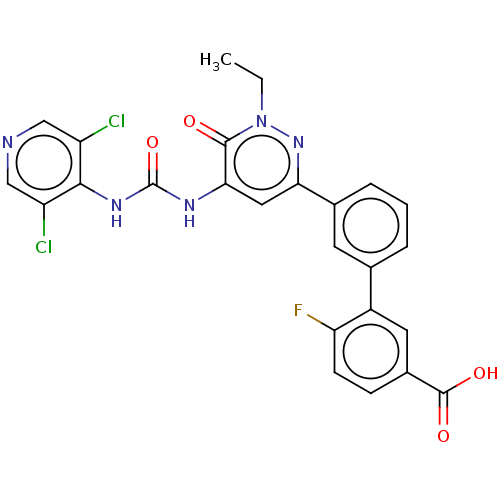 (CHEMBL3931354) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199705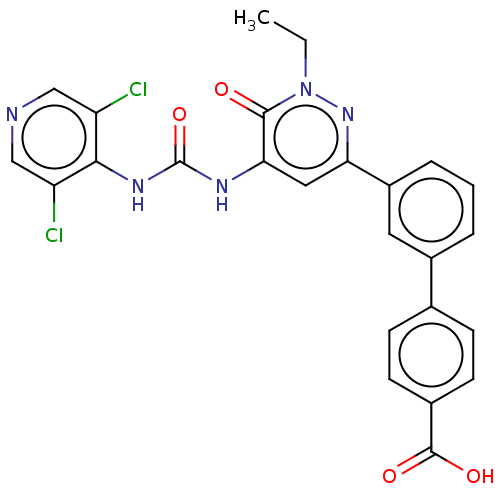 (CHEMBL3984674) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199721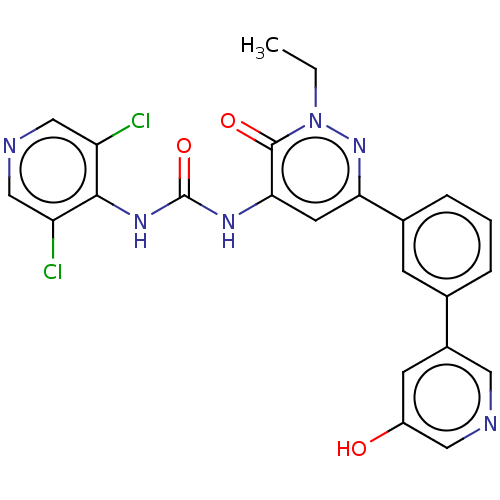 (CHEMBL3903604) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0850 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199692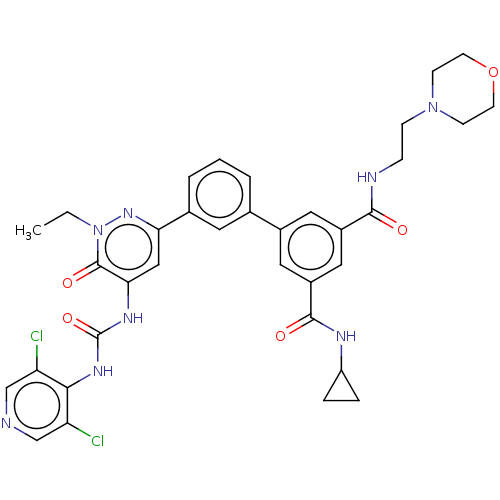 (CHEMBL3955694) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4D (Homo sapiens (Human)) | BDBM50353703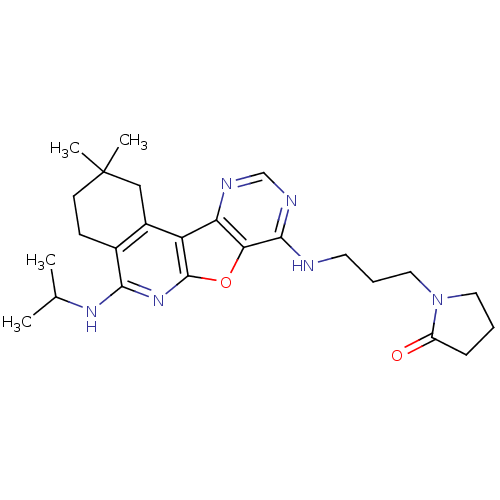 (CHEMBL1830646) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human PDE4D3 assessed as inhibition of [3H]cAMP hydrolysis to [3H]AMP after 15 mins by scintillation proximity assay | Eur J Med Chem 46: 4946-56 (2011) Article DOI: 10.1016/j.ejmech.2011.07.054 BindingDB Entry DOI: 10.7270/Q2VM4CPM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50066861 (BA-679-BR | Spiriva | Tiotropium Bromide | Tiotrop...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Displacement of [3H]NMS from human M1 receptor expressed in CHOK1 cell membranes after 4 hrs by scintillation counting method | Bioorg Med Chem Lett 25: 1736-41 (2015) Article DOI: 10.1016/j.bmcl.2015.02.065 BindingDB Entry DOI: 10.7270/Q21R6S60 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50353703 (CHEMBL1830646) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human PDE4B1 assessed as inhibition of [3H]cAMP hydrolysis to [3H]AMP after 15 mins by scintillation proximity assay | Eur J Med Chem 46: 4946-56 (2011) Article DOI: 10.1016/j.ejmech.2011.07.054 BindingDB Entry DOI: 10.7270/Q2VM4CPM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50256161 (CHEMBL4085121) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Medicinal Chemistry& Screening ,?Pharmacokinetics& Metabolism , and�Experimental Dermatology , Almirall S.A., Centro de Investigaci�n y Desarrollo , Crta. Laure� Mir� 408-410 , Sant Feliu de Curated by ChEMBL | Assay Description Inhibition of platelet derived growth factor receptor beta phosphorylation in MG63 cells in the presence of human plasma | J Med Chem 61: 2472-2489 (2018) Article DOI: 10.1021/acs.jmedchem.7b01751 BindingDB Entry DOI: 10.7270/Q2HQ42BB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50256153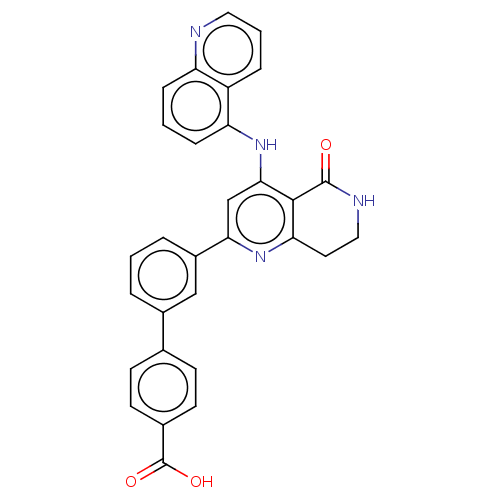 (CHEMBL4072212) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Medicinal Chemistry& Screening ,?Pharmacokinetics& Metabolism , and�Experimental Dermatology , Almirall S.A., Centro de Investigaci�n y Desarrollo , Crta. Laure� Mir� 408-410 , Sant Feliu de Curated by ChEMBL | Assay Description Inhibition of platelet-derived growth factor receptor beta phosphorylation in MG63 cells in the absence of plasma | J Med Chem 61: 2472-2489 (2018) Article DOI: 10.1021/acs.jmedchem.7b01751 BindingDB Entry DOI: 10.7270/Q2HQ42BB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199704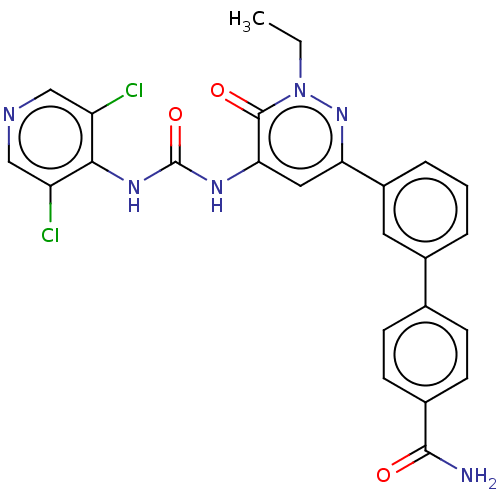 (CHEMBL3925776) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199696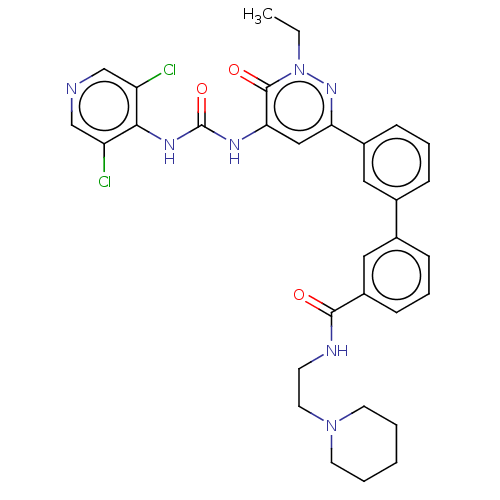 (CHEMBL3941513) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4B (Homo sapiens (Human)) | BDBM50199718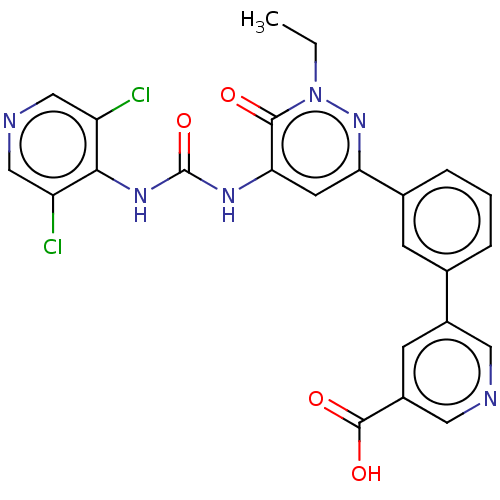 (CHEMBL3935796) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4B1 assessed as reduction in [3H]cAMP hydrolysis to [3H]AMP incubated for 60 mins by PDE-SPA assay | J Med Chem 59: 10479-10497 (2016) Article DOI: 10.1021/acs.jmedchem.6b00829 BindingDB Entry DOI: 10.7270/Q21G0P75 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50296329 ((3R)-3-{[Hydroxy(di-2-thienyl)acetyl]oxy}-1-(2-phe...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer Curated by ChEMBL | Assay Description Displacement of [3H]NMS from human muscarinic M1 receptor expressed in CHOK1 cells by microplate scintillation counting | J Med Chem 52: 5076-92 (2010) Article DOI: 10.1021/jm900132z BindingDB Entry DOI: 10.7270/Q2SX6F5Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 791 total ) | Next | Last >> |